Abstract
The inhibition of acetylcholinesterase (AChE) is still considered a strategy for the treatment of Alzheimer’s disease. The aim of this study was the search for potential drugs from natural sources which can inhibit AChE. The methanol extract of fresh flowers of Tabernaemontana pandacaqui was partitioned with n-hexane and ethyl acetate. All extracts were evaluated for AChE inhibitory activity. The ethyl acetate fraction, which showed the strongest AChE inhibitory activity, was fractionated using various chromatographic techniques, leading to the isolation of 6 compounds (
Recently, the strategy for the treatment of Alzheimer’s disease involves the inhibition of acetylcholinesterase (AChE). 1 -4 Some potent AChE drugs derived from natural sources, such as galanthamine and huperzine A, have been used for the treatment of Alzheimer’s disease, but these drugs also show many side effects in patients. 5,6 Thus, there is a need to search for compounds with anti-AChE activity with a minimum of these undesirable effects. Plants belonging to the genus Tabernaemontana (Apocynaceae), such as Tabernaemontana pandacaqui, have a worldwide distribution, including parts of the America, Africa, Asia, and Australia. 7 These plants are used in traditional medicine and for other purposes for the treatment of sore throat, hypertension, inflammatory, and abdominal pain. 8 -10 Many constituents of Tabernaemontana species have already been identified and monoterpene indole alkaloids have been shown to be present in several species. 8 However, the chemical constituents and AChE inhibitory activity of T. pandacaqui flowers have not been studied before. We now report the AChE inhibitory constituents from T. pandacaqui flower, as well as their structure-AChE inhibitory relationship.
The methanol extract of T. pandacaqui flowers {TPF(M)} was fractionated successively with n-hexane and ethyl acetate to afford n-hexane (TPF(M)-H), ethyl acetate (TPF(M)-EA), and aqueous residue fractions (TPF(M)-W). The modified Ellman’s assay was used to evaluate the AChE inhibitory activity of these fractions. The ethyl acetate fraction showed the strongest AChE inhibitory activity, with an inhibition at 5 mg/mL of 35.4% ± 0.14% (Figure 1). The ethyl acetate fraction was then subjected to silica gel 60 (Merck) quick column chromatography (QCC) using a gradient system of n-hexane-ethyl acetate and ethyl acetate-methanol to give 7 fractions (A1-A7). Fraction A2 was purified by column chromatography using a gradient system of n-hexane-ethyl acetate to give

Anti-acetylcholinesterase activity of methanol extract and other fractions.
A number of flavonoid constituents have been reported to be AChE inhibitors, such as quercitrin, tiliroside, 3-methoxyquercetin, and quercetin from Agrimonia pilosa,
20
and catechin and epigallocatechin gallate from Camellia sinensis.
21
Therefore, a major constituent, astragalin (
A major flavonoid glucoside constituent, kaemferol-3-O-β-
Acetylcholinesterase Inhibitory Activity of Astragalin and Its Derivatives.

Data followed by different letters indicate statistically significant differences (P < 0.05).
aAll values are mean ± SD based on 3 replicates.
bStandard drug.
Based on the structure-activity relationship analysis, it was found that the effect of a free hydroxyl group at position 3 in the flavonoid moiety on AChE was observed. The isolated 3-O-β-
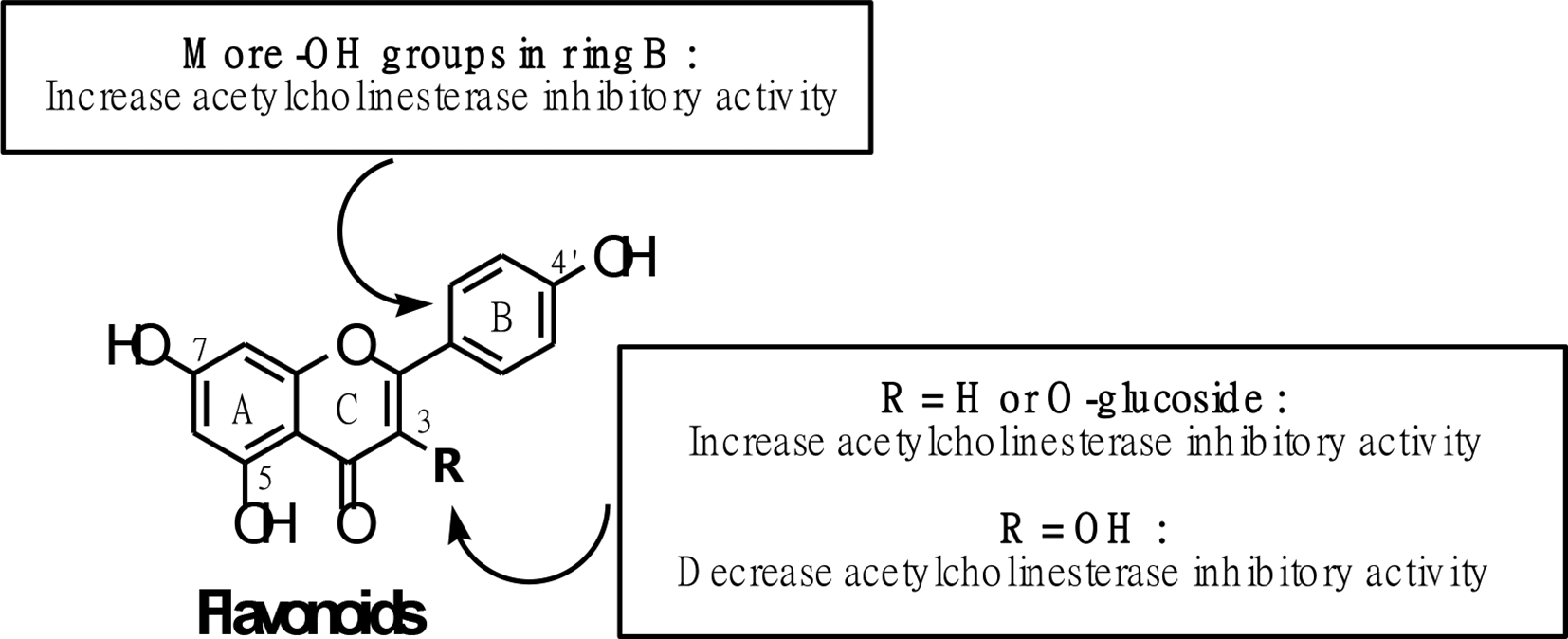
Structure-activity relationship of isolated flavonoid and its derivatives.
The inhibition of AChE is still considered an important strategy for the treatment of Alzheimer’s disease. The aim of this study was to search for new drugs from natural sources. Tabernaemontana pandacaqui flowers were found to contain constituents which have AChE inhibitory activity. In particular, the flavonoid glucoside astragalin (
Experimental
General Experimental Procedures
All the organic solvents and chemicals used in this study were of analytical grade. 1H and 13C NMR spectra were recorded on a Bruker AVANCE 400 spectrometer operating at 400 and 100 MHz, respectively. Unless indicated otherwise, column chromatography was carried out using Merck silica gel 60 (finer than 0.063 mm) and Pharmacia Sephadex LH-20. For Thin layer chromatography (TLC), Merck precoated silica gel 60 F254 plates were used. Reversed phase column chromatography was performed on Merck silica gel 60 RP-18. Compounds on TLC were detected under UV light and spraying with anisaldehyde-H2SO4 reagent followed by heating.
Plant Material
The flowers of T. pandacaqui (Apocynaceae) were purchased from a local market in Saensuk district, Chonburi province, Thailand during the month of May 2014. A voucher specimen has been deposited at the Faculty of Science, Burapha University.
Preparation of the Extract and Fractionation
The fresh powdered flowers of T. pandacaqui (2.0 kg) were immersed in methanol at room temperature and the extracts filtered. The solvent was then removed under reduced pressure at 50°C to give the dry methanol extract (150 g), which was dissolved in methanol-water (4:1) and extracted successively with equal volumes of n-hexane and ethyl acetate. Each fraction was then concentrated under reduced pressure at 50°C to yield the hexane, ethyl acetate, and residual fractions. The methanol extract and its fractions were freeze-dried and then refrigerated until further use.
Isolation of Active Compound From Tabernaemontana pandacaqui Flowers
The ethyl acetate fraction of TPF(M) (7.02 g) was subjected to silica gel 60 (Merck) QCC using a gradient system of hexane-ethyl acetate and ethyl acetate-methanol to give 7 fractions (A1-A7). Fraction A2 (309.1 mg) was purified by column chromatography using a gradient system of n-hexane-ethyl acetate to give
Acetylcholinesterase Inhibitory Activity
Acetylcholinesterase inhibition was determined spectrophotometrically using acetylthiocholine iodide (ATCI) as substrate, by modifying the method of Ellman. 22 Briefly, in a 96-well plate, 150 µL of 10 mM phosphate buffer (pH 8.0), 20 µL of a solution of AChE (4.0 U/mL in 10 mM phosphate buffer, pH 8.0), and 10 µL of the test compound solution dissolved in dimethyl sulfoxide (DMSO) were mixed and incubated at room temperature for 15 minutes. The reaction was started by adding 20 µL of either a solution of 5 mM 5,5′-dithiobis-(2-nitrobenzoic acid) or DTNB in 10 mM phosphate buffer (pH 8.0), containing 0.1% bovine serum albumin and 5 mM ATCI in 10 mM phosphate buffer, pH 8.0 (3:1). The hydrolysis of acetylthiocholine was determined by monitoring the formation of the yellow 5-thio-2-nitrobenzoate anion as the product of the reaction with DTNB and thiocholines, catalyzed by enzymes at the wavelength of 405 nm using an EPOCH-2 microplate reader spectrophotometer and the absorbance was measured after 5 minutes of incubation at room temperature. Galanthamine was used as a reference standard. The percentage of inhibition was calculated by comparing the rate of enzymatic hydrolysis of ATCI for the sample to that of the blank (DMSO in buffer), which was calculated by the following equation: inhibition (%) = {[(A – B) – (C – D)]/(A – B)} × 100, where A is the activity of the enzyme without the inhibitor; B is the control of A without the inhibitor and enzyme; and C and D are the activities of the inhibitors with and without AChE, respectively. All experiments were carried out in triplicate.
Statistical Analysis
All analyses in this study were performed in 3 replicates. Minitab software version 18 was used for statistical analyses. Tukey’s tests were used to determine the variations between the means. Differences at 5% (P < 0.05) level were considered as significant.
Footnotes
Acknowledgments
The authors wish to thank the Center of Excellence for Innovation in Chemistry (PERCH-CIC), the Research Unit in Synthetic Compounds and Synthetic Analogues from Natural Product for Drug Discovery (RSND, Grant no. 14/2562), and Department of Chemistry, Faculty of Science, Burapha University for providing research facilities. Special thanks to Associate Professor Dr Surachai Nimgirawath for his comments and grammatical suggestions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Research Grant of Burapha University though the National Council of Thailand (Grant nos. 80/2558 and 73/2559).
