Abstract
Zanthoxylum leprieurii Guill. and Perr. (Rutaceae) stem bark is used locally in Uganda for treating tuberculosis (TB) and cough-related infections. Lupeol (
Introduction
Tuberculosis (TB) is a cause of ill-health for about 10 million people globally each year and is ranked among the leading 10 causes of death globally, as well as the main cause of death arising from a sole infectious agent, and ranked above human immunodeficiency virus/acquired immunodeficiency syndrome. Currently, Africa has the most significant burden of TB globally, with a prevalence of 226 cases per 100,000 individuals in relation to the global average of 130 cases per 100,000 individuals. In Uganda, the average TB prevalence is 200 cases per 100,000 people, which puts this country among the high TB-laden countries in the world. 1
The rise of drug-resistant TB strains remains a public health danger and is among the primary reasons contributing to the increase of global incidence of TB. 1 In 2019, World Health Organization (WHO) reported that there were about 500,000 new cases of TB which had developed resistance to rifampicin, and, of those, 78% had developed multidrug resistance (MDR-TB). 1 Consequently, this necessitates exploring any existing avenues to develop novel and highly efficacious drugs for the treatment of TB.
Natural products have proved to be templates for novel drug development. They have several exciting biological activities and thus, they have been accorded significant consideration as possible sources of drugs for TB. 2 One of the plants that needed to be evaluated for anti-TB agents is Z. leprieurii since its stem bark is being used in Uganda by the local communities to manage TB and other infections related to cough. 3
Consequently, this study aimed to determine the constituents of the chloroform/methanol (1:1, v/v) extract of Z. leprieurii stem bark and evaluate the in vitro anti-TB activities of the extract and the compounds isolated from it against both the susceptible (H37Rv) and MDR-TB strains.
Results and Discussion
Structures of Isolated Compounds
The spectroscopic data of the isolated compounds are presented in the supplemental material. Compounds

Structures of compounds
The chloroform/methanol (1:1) extract afforded a triterpene, a lignan, a coumarin, alkaloids, and a flavanone, as compared to a previous report where a methanol extract of Z. leprieurii stem bark afforded only acridone alkaloids. 18 A chloroform/methanol (1:1, v/v) mixture, which is of medium polarity, was chosen for extraction because it has been reported that organic extractions made using such solvents are expected to be composed of phytochemicals like alkaloids, polyphenols, and terpenoids, having activity against microbes.19–21 Furthermore, such a solvent mixture can extract lipophilic constituents of the plant, which can easily permeate through the M. tuberculosis cell wall, inhibiting its growth.
Antimycobacterial Activity
We examined the in vitro antimycobacterial activities of the crude extract and the pure compounds isolated from it (
In the preliminary sensitivity tests against both the susceptible (H37Rv) and MDR-TB strains, the results were based on the following classification: nonsensitive with a diameter < 8 mm; sensitive when diameter was from 9 to 14 mm; very sensitive when diameter ranged from 15 to 19 mm, and diameter > 20 mm was classified as extremely sensitive. 22 Accordingly, the crude extract with zones of inhibition (diameter, 30 and 26 mm) at 50 mg/mL against the susceptible (H37Rv) TB strain and MDR-TB strain, respectively, was considered extremely sensitive against both strains (rifampicin 38.0 ± 0.0 mm against H37Rv strain; 0.00 against MDR strain).
The MIC values for both the susceptible (H37Rv) and MDR-TB strains are shown in Table 1. The activity of the extract against M. tuberculosis was classified using the following criteria: significant for MIC (μg/mL) < 100, moderate for MIC (μg/mL) 100 to 625, and weak or low for MIC (μg/mL) > 625. 23 Meanwhile, the activity for a compound was classified as follows: significant for MIC (μg/mL) < 10, moderate for MIC (μg/mL) 10 to 100, and weak or low when MIC (μg/mL) > 100. 23
Mean MIC Values of the Crude Extract and Compounds

From the antimycobacterial test results (Table 1), the activity (MIC 586 μg/mL) recorded for the crude extract was considered remarkable because it was moderately active against the susceptible strain of M. tuberculosis. This result justified the isolation of compounds from the crude extract to establish those responsible for the activity. Also, an earlier study by Bunalema et al had indicated a significant antimycobacterial activity of a Z. leprieurii stem bark methanol extract. Furthermore, previous studies on extracts from some species of Zanthoxylum, such as Z. capense, Z. macrophylla, and Z. xanthoxyloides, showed antimycobacterial activity.24,25
Considering the isolated compounds, the activity registered for compound
Generally, the antimycobacterial activity detected in the isolated compounds could be attributed to their lipophilic nature, which allows them to penetrate easily into the mycobacterial cell wall to hamper their growth. 27 According to Brennan and Nikaido, 28 M. tuberculosis is armed with lipid and waxed cell walls, making penetration very hard for most antibiotics and, hence, a hard-to-treat organism. They are instead vulnerable to compounds that are less polar and having lipophilic properties. 2 The lipophilicity of a drug molecule may enhance its ability to penetrate various biomembranes, subsequently improving its permeability into microbial cells. 29
For compound
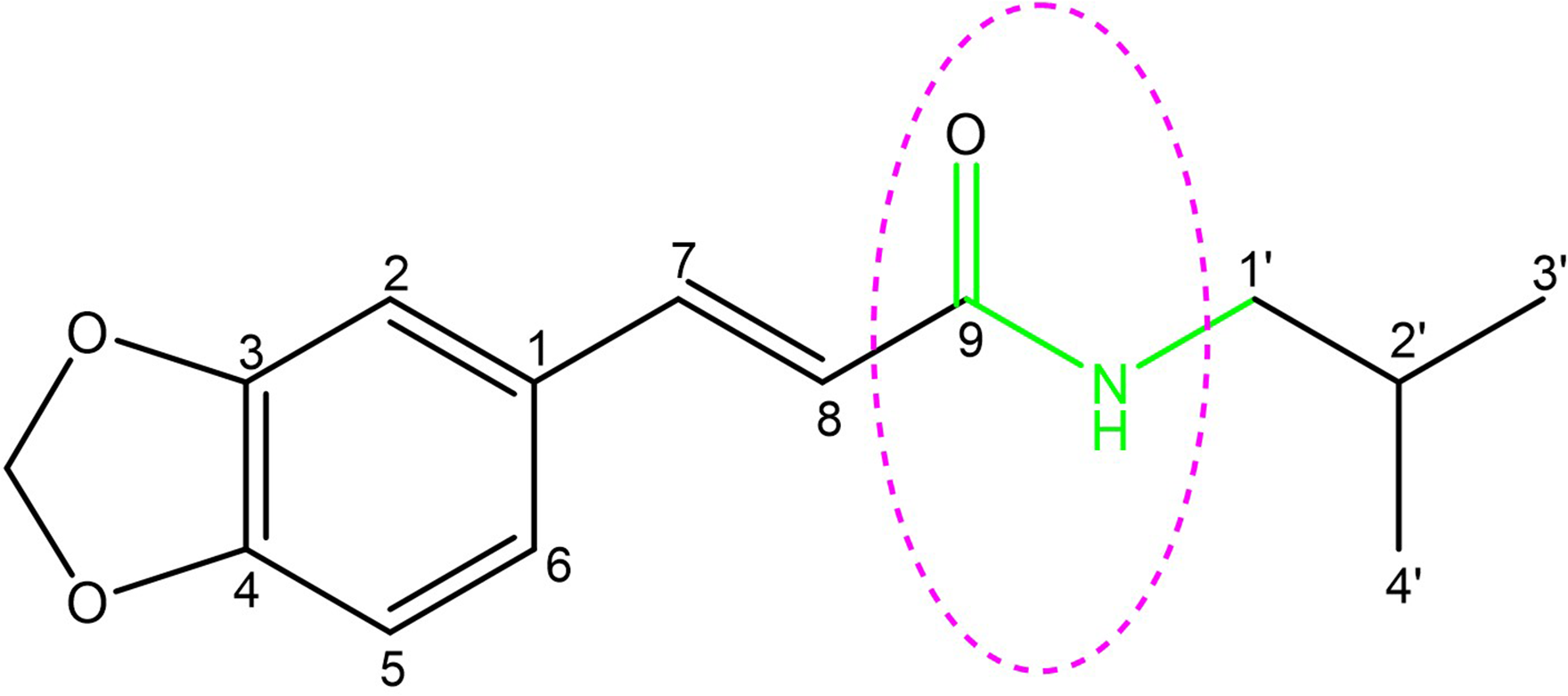
The structure of trans-fagaramide showing a pharmacophore that could be responsible for its anti-TB activity.
It is worth noting that the secondary amide linker pharmacophore exists in the structures of rifampicin, isoniazid, and pyrazinamide, 3 of the 4 drugs used as first-line treatments for TB. In addition, amikacin and capreomycin, which are among the 4 second-line medicines for treating TB, contain this pharmacophore in their structures. In addition, a potent clinical drug candidate, Q203, which has shown excellent activity against the multidrug-resistant (MDR) and extensively drug-resistant strains of TB, contains the secondary amide linker in its structure.
35
This relationship strongly supports the present study's findings regarding the significant activity of compound
Biological activities of the isolated compounds have been reported in many previous studies. Compound
Generally, medicinal plants are presumed to be harmless, although some of them possess toxic properties. Consequently, studies are normally conducted to establish their toxicity profiles to confirm that any biological activities observed from the plant extracts do not result from general metabolic toxic effects. Previous studies indicate that compounds
Conclusion
This is the first report of the isolation from the stem bark of Z. leprieurii of lupeol (
Experimental
General Experimental Procedures
Chromatography columns were packed with silica gel 60 (Merck; 70-230 mesh) and Sephadex LH-20 (Amersham Pharmacia Biotech AB) as stationary phases. Nuclear magnetic resonance spectra were measured from a Bruker Avance 500 MHz Spectrometer (Bruker Biospin). High-resolution mass spectra were obtained on an ESI-Q-TOF Micro, 6200 Series Spectrometer (Agilent Technologies).
Plant Material
Zanthoxylum leprieurii (known locally as “Munyenye” in Luganda) stem bark was collected from Mpanga Forest Reserve in Mpigi district, Uganda (0.206032N, 32.300608E) in October 2018. Botanical identification was carried out by a taxonomist from Makerere University Herbarium and a voucher specimen No. OB 15 (with Plant Accession No. 50967) was deposited at the same Herbarium.
Extraction and Isolation
The air-dried and powdered stem bark (1000 g) was soaked in a chloroform/methanol (1:1, v/v) mixture for 24 h with occasional shaking. The extraction was repeated twice on the same sample to maximize extraction. The extract was filtered carefully, and the crude extract (94.25 g) was obtained upon concentration.
The extract (50 g) was loaded onto a silica gel packed column and eluted first with petroleum ether/ethyl acetate solvent mixture (100:0, 95:5, 90:10, 85:15, 80:20, 70:30, 60:40, 50:50, 30:70, 10:90, 0:100), followed by ethyl acetate/methanol solvent mixture (90:10, 80:20, 60:40). One hundred forty fractions (each 200 mL) were collected and combined in accordance to their TLC profiles to obtain eleven (11) main fractions labelled as AB, 2C, 2D, 2E, 2F, 2G, 2H, 2I, 2J, 2K, and 2L.
Fractions AB-2J were subjected separately to repetitive column chromatography employing silica gel as the stationary phase and using gradient elution to obtain the following pure compounds:
Antimycobacterial Activity
Mycobacterium tuberculosis strains and preparation of inoculums. A fully susceptible laboratory strain (H37Rv) and a known MDR TB (375) strain from a WHO Proficiency Testing panel were used as experimental mycobacterial strains. Both were obtained from the College of American Pathologists (CAP) ISO 15189 accredited Mycobacteriology Laboratory (BSL-3), College of Health Sciences, Makerere University.
Preserved strains of H37Rv and MDR TB were revived on Middlebrook 7H10 agar before the susceptibility tests.58,59 Cells scraped from freshly growing colonies (3 weeks old) were introduced into normal saline (10 mL). A bacterial suspension equivalent to 0.5 McFarland standard (1.5 × 108 CFU) was prepared using a Sensititre Nephelometer by either adding more cells or diluting with more normal saline.59,60.
Preparation of Plant Extracts and Compounds/Drugs. The dried crude extract (1000 mg) was dissolved in dimethyl sulfoxide (DMSO) (10 mL) to give a concentration of 100 mg/mL, and the extract was later sterilized using 0.2 μm single-use filters before use. A stock solution of rifampicin (1000 μg/mL) was diluted serially, and a working solution (100 μg/mL) was obtained.
Preparation of Bio Discs. Bio discs (diameter, 6 mm) were punched from a filter paper (Whatman No.1). They were sterilized at 121°C for 15 min and then impregnated with the extract/drug (20 μL) prepared earlier. The discs were dried in a hood for 12 h before use.
Procedure for Susceptibility Testing. Middlebrook 7H10 agar was used as the culture medium. 60 The medium (20 mL) was poured into Petri dishes (diameter, 90 mm) with 4 quadrants such that each contained 5 mL of the medium. Then M. tuberculosis strain inoculums (100 μL) at 0.5 McFarland concentration were inoculated using a sterile single-use plastic loop on the solidified medium such that each quadrant contained 25 μL. An extract impregnated disc with a concentration of 50 mg/mL was placed in the first quadrant. A rifampicin impregnated disc containing 100 μg/mL was placed in the second quadrant. A blank disc and another disc impregnated with DMSO were placed in the third and fourth quadrants, respectively. The Petri dishes were left in the hood for 12 h before sealing them with micropore (carbon dioxide-permeable tape) to permit diffusion of the extract and drugs. After that, they were subjected to incubation in a carbon dioxide incubator running at 37°C for 21 days. Zones of inhibition (mm) that surrounded the discs were measured using a ruler to determine the sensitivity of M. tuberculosis strains to the extract and drugs.
Determination of MIC for Crude Extract. MIC was determined using the macro broth dilution method by diluting the extract serially in Middlebrook 7H9 medium.61,62 The medium (5 mL) was dispensed into a 15 mL Falcon tube before adding an equal volume of the extract at a 50 mg/mL concentration. A 20-fold dilution was made by mixing the extract with the medium 6 times before pipetting off 5 mL to the next tube containing 7H9 medium (5 mL) until a final concentration (0.1 μg/mL) was obtained. The extract was inoculated with 0.5 mL of 0.5 McFarland of H37Rv and MDR strains. The tubes were left in the incubator shaker at 37°C for 14 days. The MIC was the lowest concentration having no visible growth, as indicated by clog formation of M. tuberculosis cells on the macro broth tube walls. The tests were made in duplicates. DMSO was used as a negative control.
Determination of MIC for Isolated Compounds. The activities of the compounds against susceptible (H37Rv) and MDR TB strains were tested following the microplate Alamar blue assay. 63 Sterile distilled water (200 μL) was added to all outer perimeter wells of sterile 96-well plates to minimize evaporation. The remaining wells received 100 μL of 7H9 broth. Double concentration drug solutions (100 µL) were added to each of the wells in rows B to G in column 2, and 2-fold serial dilutions were made through column 10 using a multichannel pipette. All the wells in columns 2 to 11 were inoculated with 100 μL of M. tuberculosis, bringing the final volume to 200 μL per well. The plates were incubated at 37°C for 21 days. Freshly prepared Alamar blue reagent (30 µL) was later added to one of the control wells and incubated at 37°C for 24 h. Observation of a color change indicated that there was growth. The dye was later added to all the wells and further incubated for 24 h. The MIC of the isolated compounds was then determined. Standard drugs: rifampicin and isoniazid were used as positive controls. The tests were prepared in triplicates for each of the strains used. The bioassays were performed in a Mycobacteriology Laboratory (BSL-3).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211035851 - Supplemental material for Antimycobacterial Activity of the Extract and Isolated Compounds From the Stem Bark of Zanthoxylum leprieurii Guill. and Perr.
Supplemental material, sj-docx-1-npx-10.1177_1934578X211035851 for Antimycobacterial Activity of the Extract and Isolated Compounds From the Stem Bark of Zanthoxylum leprieurii Guill. and Perr. by Benson Oloya, Jane Namukobe, Matthias Heydenreich, Willy Ssengooba, Bernd Schmidt and Robert Byamukama in Natural Product Communications
Footnotes
Acknowledgments
The authors thank the following sincerely: Mr Peter Nelson Sekandi from the Department of Chemistry, College of Natural Sciences, Makerere University and Mr George Kwesiga from the Institute of Chemistry, University of Potsdam, Germany, for their technical support during the isolation of the compounds; Ms Angela Krtitschka and Ms Sylvia Fürstenberg both from the Institute of Chemistry, University of Potsdam, Germany for carrying out spectroscopic analyses of the compounds; and Mr Kevin Komakech from the Mycobacteriology Laboratory (BSL-3) at the College of Health Sciences, Makerere University, for carrying out the antimycobacterial assay.
Also, the authors acknowledge valuable support to Benson Oloya through the attendance of a Summer School on “Separation and structure elucidation of phytochemicals” for 2 months hosted at Egerton University, Kenya, by the Phytochem Project. The project was funded by the DAAD and the German Ministry of Education and Research (BMBF) under the “Partnership for Sustainable Solution in Sub-Saharan Africa,” Grant no. SSA2015 to 33 to 074.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutscher Akademischer Austauschdienst (DAAD) In-Country Scholarship, Uganda 2016 (through a DAAD PhD Scholarship and a DAAD Short Term Research Travel Grant to Benson Oloya, Personal no. 91560246).
The authors gratefully acknowledge the DAAD for funding this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
