Abstract
Objective
The leaves of Alstonia boonei and aerial parts of Ipomoea cairica are used for treatment of microbial infections among other ailments in African traditional medicine. The aim of this study was to investigate the antimicrobial phytochemicals in A. boonei leaves and Ipomoea cairica aerial parts to validate their traditional use in Ugandan herbal medicine.
Methods
The plant materials were separately extracted using a dichloromethane/methanol (1:1) solvent system and subjected to repeated chromatographic separation to isolate pure compounds. The chemical structures of the isolated compounds were determined through 1H NMR, 13C NMR and 2D NMR (COSY, HSQC and HMBC). The antibacterial activity of the extracts and pure compounds were assessed using the agar well diffusion method.
Results
Chromatographic fractionation of the extracts yielded trans-fagaramide and a pentacyclic lupane-type triterpenoid, lupeol, from A. boonei, and friedelin from I. cairica. Trans-fagaramide was identified for the first time in the Alstonia genus while friedelin was identified for the first time in I. cairica. The isolated compounds demonstrated antibacterial activity, with trans-fagaramide showing a minimum inhibitory concentration (MIC) of 125 μg/mL against Pseudomonas aeruginosa and 250 μg/mL against Staphylococcus aureus, Salmonella typhi and Escherichia coli. Friedelin exhibited a MIC of 125 μg/mL against Escherichia coli and 250 μg/mL against Pseudomonas aeruginosa, Staphylococcus aureus and Salmonella typhi.
Conclusion
The antibacterial activities observed in this study support the traditional use of A. boonei and I. cairica by indigenous communities in Uganda for treating microbial infections.
Introduction
Despite the significant progress made in the treatment of emerging and known microbial infections, pathogenic diseases have remained among the major causes of morbidity and mortality. For example, it accounted for at least 13.5% (13.7 million) deaths reported globally in 2019. 1 The underlying causes of this disease burden is partly due to continuous development of resistance to the current-use conventional drugs. In addition, infringement on the pristine environment and domestication of wildlife have spiraled human–animal interactions, opening new windows for transmission of zoonoses and unique pathogens to humans. 2 A case in point is the COVID-19 pandemic that has, by and large, been considered the worst infectious disease witnessed in the twenty-first century. 3
The burden of pathogenic diseases is disproportionately high in developing countries and this has prompted communities to embrace traditional medicine for healthcare.4–6 In Africa, traditional medicine has long been a cornerstone of healthcare for communities where plant-based remedies are frequently used to treat a wide range of ailments, including microbial infections. The preference for medicinal plants over conventional medicine is largely driven by cultural perspicacity and beliefs, both of which varies across countries.7–9 In East Africa, the use of herbs for primary healthcare has been widely documented. 10 In Uganda, the use of herbal remedies is an integral part of the healthcare systems.11,12 This study focused on two species (Alstonia boonei and Ipomoea cairica) used in the treatment of microbial infections in Uganda.
Alstonia boonei De Wild. (Apocynaceae) is a deciduous tree locally known as mubajangalabi (Luganda), nsiwa (Lusoga) and mujwa (Runyoro) in Uganda. 13 It is used in the treatment of malaria and microbial infections. 14 In other parts of Africa (where it is called pattern wood or stoolwood), this species is used in the treatment of (cerebral) malaria, snake envenomation, insomnia, arrow poison, hypertension, microbial infections, cancer, painful micturition, diarrhoea and inflammations.15–17 In some African countries, A. boonei is a sacred tree that is cherished and therefore not usually eaten. 18 The stem bark, leaves and roots of this species has antiplasmodial,19–23 analgesic, anti-inflammatory, 24 antipyretic, 25 antimicrobial, 26 antidiarrheal, 27 anticancer, 28 antioxidant, anti-ulcer, 29 hepatoprotective and neuroprotective activities.16,30 Plants are known to produce a vast array of secondary metabolites that serve protective roles against pathogens and environmental stressors. These bioactive compounds, including alkaloids, flavonoids, terpenoids, and phenolics, are often responsible for the antimicrobial properties observed in medicinal plants. Although the antimicrobial and antidiarrheal activities of A. boonei has been studied,26,27 complete characterization of the compounds associated with its bioactivities is far from accomplished. Marini-Bettolo et al 31 first isolated boonein, a monoterpenoid δ-lactone from the bark of A. boonei. Omitola 27 reported the presence of boonein and β-amyrin in the leaf extracts while Olanlokun et al found an antimalarial compound: tetrahydro-4-((E)-7-hydroxy-10-methoxy-6,14-dimethyl-15-m-tolylpentadec-13-enyl) pyran-2-one in the stem bark extract of this species. 32 Kiganda 33 found that A. boonei stem and root barks had cycloeucalenol, lupeol, phenanthridine-6(5H)-one, lupeol acetate, stigmasterol, lichexanthone, β-sitosterol and echitamine. Acylated anthocyanins and flavonoids have also been characterized in this species. 34 Recently, methanolic stem bark extract of A. boonei was found to contain loganic acid, along with two new compounds: secoxyloganin and sweroside. 35
On the other hand, Ipomoea cairica (L.) Sweet is an invasive vine in family Convolvulaceae. 36 It is a twining perennial herb that persists for more than two years. With showy white to lavender-colored flowers (hence the name five-fingered morning glory), Ipomoea cairica (I. cairica hereafter) is native to tropical Africa and Asia. 34 In Uganda, it is known as akarandarugo, and its leaves and stems are used in the treatment of microbial infections, 37 cervical cancer, uterine diseases and colic pain. 38 In Kenya, powdered leaves and roots are applied topically for breast, cervical and skin cancer treatment. 39 In Brazilian traditional medicine, I. cairica is used in the treatment of rheumatism and inflammations. 40 To date, the antinociceptive, 40 larvicidal,41–43 anticancer 44 and antidiabetic 45 activities of I. cairica extracts and some coumarins (scopoletin, umbelliferone), lignans (arctigenin, matairesinol, (+)-pinoresinol and trachelogenin) and acylated anthocyanins in it has been reported. 34 This species is also rich in pentasaccharide resin glycosides (cairicosides A-F), mainly in its aerial parts.44–46
Research into the chemical composition and biological activities of compounds from A. boonei and I. cairica are still inadequate to understand the compounds associated with their uses claimed in traditional medicine. Such studies are crucial for understanding the pharmacological basis of their traditional uses and for potentially developing novel plant-derived antimicrobial agents that could be used to counter the increasing antimicrobial resistance crisis. 26 In continuity of our search for bioactive natural products with antimicrobial activities in Uganda,47–50 the objective of the present study was to isolate and characterize the phytochemicals in A. boonei leaves and I. cairica aerial parts and to assess their antibacterial activities. By identifying and testing these compounds against common bacterial pathogens, this study aimed to provide scientific validation for the traditional use of these plants in treating microbial infections and to contribute to the growing body of knowledge on plant-based antibacterial agents.
Materials and Methods
Plant Organs and Extraction of Phytochemicals
Leaves of A. boonei and aerial parts of I. cairica were sampled from Nakawuka village, Wakiso District and Nakawa Division, Kampala District, Uganda, respectively. Both samples were identified and authenticated by a taxonomist at Makerere University Herbarium where voucher samples (IG005 and IG006) were deposited. The samples were dried under shade for 3 weeks.
The pulverized samples (1 kg of A. boonei leaf powder and 620 g of I. cairica aerial parts) were exhaustively extracted by cold percolation at room temperature using a mixture of dichloromethane and methanol (DCM/MeOH, 1:1, v/v) for 48 h, with occasional stirring. The choice of this solvent system is because DCM and MeOH have different polarities and their mixture is able to solubilize both non-polar and polar compounds. Thus, we targeted this solvent system because it would extract a wide variety of phytochemicals, including alkaloids, flavonoids, terpenoids, and steroids. The extracts obtained were then filtered, and the filtrates were concentrated by rotary evaporation at 40 °C. The crude extracts obtained were stored at 4 °C until further analysis.
Isolation and Spectroscopic Analysis of Compounds
Chromatographic separation was done using 15 g and 100% of n-hexane (to remove fats and oils for two days) and then serially with solvent systems of increasing polarity at 5% increments upto 100% ethyl acetate and methanol. The fractions obtained were concentrated on a rotary evaporator and spotted on analytical TLC plates (TLC sheets ALUGRAM® Xtra SIL G/UV254, Macherey-Nagel GmbH & Co. KG, Germany). Fractions with similar TLC profiles were combined. 51 For each eluent system, two litre solvent volumes were used and 250 ml fractions were collected. The fractions were concentrated and repeatedly fractionated using DCM/MeOH (1:1) as the eluent system to obtain the pure compounds. 52
The structure of each compound isolated was established from NMR data obtained on a Bruker AV-500 spectrometer (MC-Murry) ie, 1D (1H and 13C) and 2D (H-H COSY, HSQC and HMBC) spectra of the samples dissolved in deuterated chloroform. The spectra in FID format were processed using MestReNova (version 8.1.1). NMR results with residual chloroform peaks were used as the references. The results were compared with published spectroscopic data.
Antibacterial Activity Assessment
The antibacterial assay followed the agar well diffusion method. Briefly, the bacterial strains were American Type Culture Collection (ATCC) of Escherichia coli (E. coli), Staphylococcus aureus (S. aureus), Salmonella typhi (S. typhi) and Pseudomonas aeruginosa (P. aeruginosa). The choice of the bacteria was informed by the bacterial diseases for which the plants are traditionally used to treat, the WHO priority list of bacteria as well as the availability of the test strains. 49 The bacteria were all obtained from Department of Biological Sciences, Kyambogo University, Kampala, Uganda where the assays were performed.
The bacterial cultures were maintained on nutrient agar slants for a day after incubation at 37 °C. They were stored at 4 °C as stock cultures for antibacterial assays. Using sterile 6-mm glass cork borers, wells were carefully made on the agar plates without distorting the media. Tetracycline (0.5 mg/mL) and dimethyl sulfoxide (DMSO) were used as the positive and negative controls, respectively. In the bioassays, exactly 50 μL of the prepared extracts (0.5 mg/mL in DMSO) and the controls were pipetted into the wells, left on the bench for 60 min and thereafter incubated at 37 °C for 24 h. The antibacterial activities were determined by measuring the inhibition zone diameter (ZOI).
Utilizing the broth-dilution method, bacteria with the highest sensitivity to the extracts and isolated compounds (
Results
Isolated Compounds from A. boonei Leaf Extracts
Two compounds: trans-fagaramide (
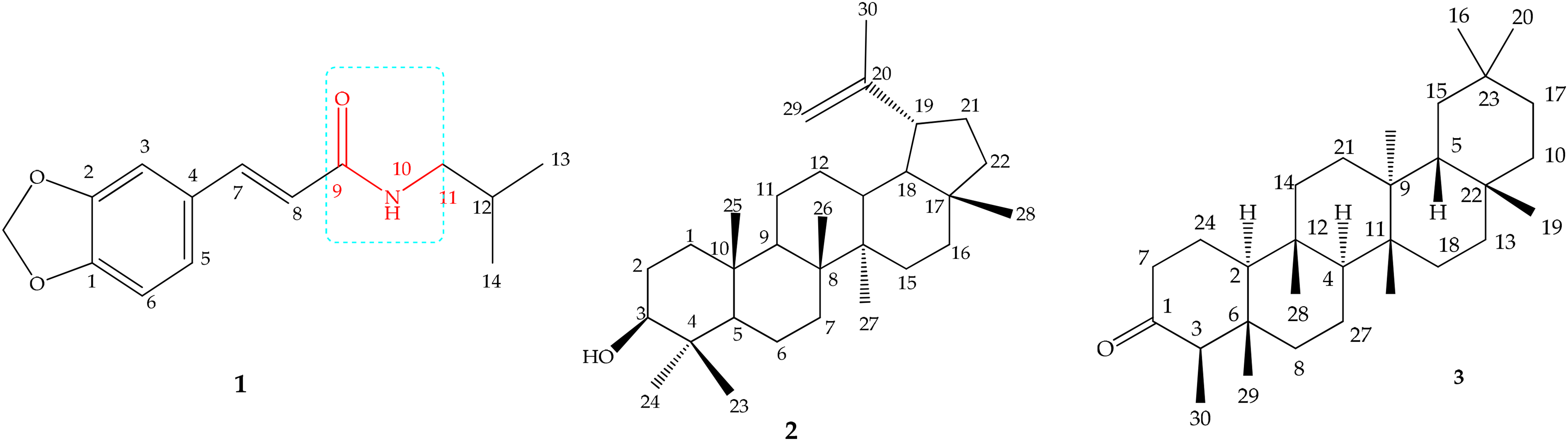
Structure of bioactive compounds characterized in dichloromethane/methanol extracts of A. boonei leaves (
Compound
The confirmation of the structure was accomplished through 2D NMR experiments (COSY, HSQC and HMBC) (Figures S10–S12). The H-H COSY spectrum of the compound exhibited some cross peaks such as between δ 2.38, H-19 and one methylene proton δ1.33, H-21 and between methine proton δ3.18, H-3 and methylene s (δ 1.57, H-2). In the HMBC spectrum, the methine proton at δ3.18 (H-3) showed cross peaks with a methyl carbon δ28.1, C-23) by J2 correlation and a methyl carbon δ 18.4, C-6) by J3 correlation. The methine proton at δ 2.38 (H-19) showed cross peaks with two methylene carbon δ29.9 (C-21) and δ109.5 (C-29), a methine carbon 48.4 (C-18), a methyl carbon δ 19.4 (C-30)] and a quaternary carbon δ 150.6 (C-20). The pair of broad singlets of proton at δ 4.57 and 4.69 showed cross peaks with a methylene carbon δ 48.1(C-19), δ150.6 (C-20) and δ 109.5 (C-29)] by J3 correlation.
Isolated Compound from I. cairica Aerial Part Extract
One compound (
The 1H NMR spectrum of
Antibacterial Activity of the Crude Extracts and Isolated Compounds
The antimicrobial activity of crude extracts and isolated compounds (
Upon testing for MIC, compounds
Minimum Inhibitory Concentrations of the Extracts (mg/mL) and Compounds (μg/mL) from A. boonei and I. cairica.

Discussion
Chromatographic fractionation of the extracts followed by detailed spectroscopic analysis in this study yielded trans-fagaramide (
Similarly, the analysis and comparison of spectral data of
The antibacterial activity of extracts and compounds
Assessment of MIC of
Although lupeol (
On the other hand, friedelin (
Taken together, these reports suggests that the isolated compounds could be responsible for the claimed antibacterial effect of these compounds. Such inhibitory activities may also be related to the interaction of the compounds with the bacterial membrane proteins or lipids leading to bacterial membrane stabilization and disruption, inhibition of membrane-bound enzymes, modulation of immune response and lipid raft disruption. For example, trans-fagaramide being an amide compound with potential amphipathic properties may insert itself into the bacterial membrane. This can disrupt the membrane's lipid bilayer, increasing its permeability and leading to leakage of cellular contents. The disruption of membrane integrity can cause osmotic imbalance, leading to cell death. 86
The observed differences in ZOI and MIC in the antimicrobial assays could be due to factors such as physicochemical properties of the compounds (solubility, molecular size, stability and lipophilicity), diffusion rate in the agar medium, microbial strain differences and compound interactions (in the case of extracts). 87 The higher MIC values obtained suggests that higher doses of the plant extracts are needed to effectively treat microbial infections using the plant parts. 49
Conclusion
This study was undertaken to verify the traditional claims of using A. boonei leaves and I. cairica aerial parts in ethnomedicinal treatment of microbial infections in Uganda. Isolation and characterization of compounds from their DCM/MeOH extracts afforded antibacterial compounds (trans-fagaramide and lupeol from A. boonei, and friedelin from I. cairica). These results lend credence to the use of these species in herbal treatment of bacterial diseases in Uganda. In vivo studies using animal models to evaluate the efficacy and safety of the extracts or isolated compounds from A. boonei and I. cairica could provide stronger evidence of their pharmacological effects, dosage safety, and potential toxicity, complementing the in vitro antibacterial results. Further studies should consider examining the polyphenolic content and antioxidant potential of the extracts and isolated compounds.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241286425 - Supplemental material for Antibacterial Properties of Phytochemicals Isolated from Leaves of Alstonia boonei and Aerial Parts of Ipomoea cairica
Supplemental material, sj-docx-1-npx-10.1177_1934578X241286425 for Antibacterial Properties of Phytochemicals Isolated from Leaves of Alstonia boonei and Aerial Parts of Ipomoea cairica by Ivan Gumula, Christine Kyarimpa, Sarah Kiwanuka Nanyonga, George Kwesiga, George Busulwa, Boniface Opio, Mathias Heydenreich and Timothy Omara in Natural Product Communications
Footnotes
Acknowledgments
We acknowledge the laboratory support of John Chrysostom Opedun and Fred Mutai during the isolation of compounds and antibacterial assays, respectively.
Data Availability
Data supporting the conclusions of this study are available within this article and its supplementary files.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
