Abstract
The 2 new oleanane-type triterpene glycosides, 23-hydroxyoleanolic acid-[28-O-β-D-glucopyranosyl]-3-O-{β-D-glucopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→3)]-β-D-galactopyranoside}, (
Introduction
Aralia armata (Wall.) Seem. (Araliaceae) is a common herbal in Vietnam that has been used in traditional medicine to cure hepatitis, arthritis, stomachache, malaria, and snake rolling. 1 Phytochemical study of A armata led to the isolation of oleanane-type triterpene glycosides as the main components of the leaves 2 and roots. 3 From the leaves of this plant growing in Vietnam, 14 oleanane-type triterpene glycosides have been isolated and some of them exhibited cytotoxic activity against some cancer cell lines.4-6 However, no chemical and bioactive studies have been made on the roots of Vietnamese A armata. Continuing our search for bioactive compounds from A armata, herein, we report the isolation and structural elucidation of 2 previously undescribed oleanane-type triterpene saponins (named as armatosides C and D) from the roots of this plant. The cytotoxic activity of these compounds on some cell lines was also evaluated.
Results and Discussion
Compound

Chemical structures of compounds
NMR Spectroscopic Data for the Sugar Moieties of

Measured at 100 MHz.
Measured at 400 MHz.
Abbreviation: NMR, nuclear magnetic resonance.
Compound

The 1H-1H COSY and key heteronuclear multiple bond correlation (HMBC) correlations of compounds
NMR Spectroscopic Data for the Aglycone Moieties of
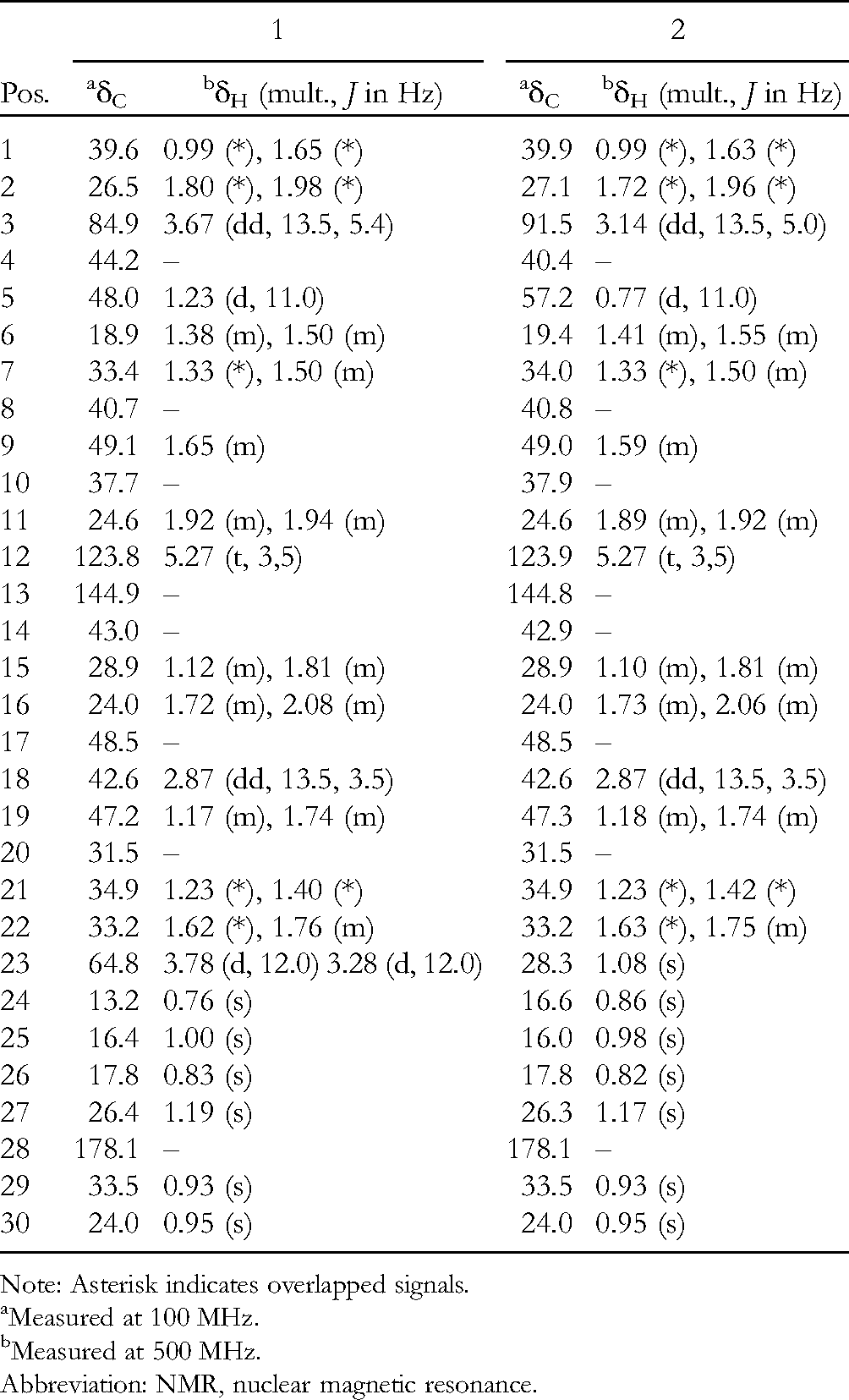
Note: Asterisk indicates overlapped signals.
Measured at 100 MHz.
Measured at 500 MHz.
Abbreviation: NMR, nuclear magnetic resonance.
Compounds
The Cytotoxic Effects of Compounds
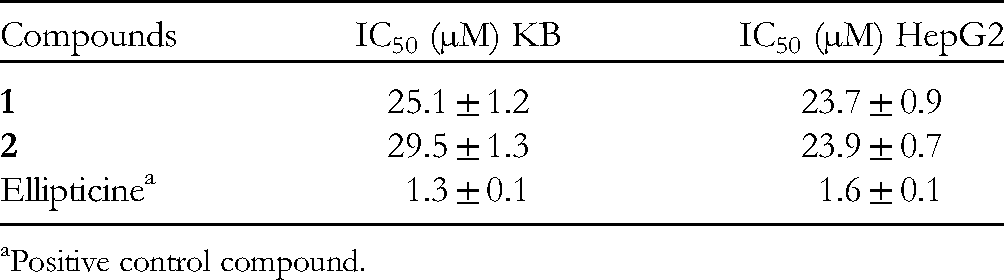
Positive control compound.
Experimental
General
HR-ESI-MS was carried out on an Agilent 6530 Accurate Mass Q-TOF LC/MS. The QTOF instrument was set at 2 GHz in extended dynamic range resolution mode, a negative ESI capillary voltage of 3500 V, fragment or voltage of 175 V, MS scan range of m/z 100 to 1700, and MS acquisition rate of 1.0 spectra per second. The NMR spectra were recorded on a Bruker 500 MHz spectrometer and optical rotation on a Jasco P2000 polarimeter. Column chromatography was performed using silica gel, reverse phase C-18, and diaion HP-20 resins as stationary phase. HPLC was carried out using an AGILENT 1100 HPLC system. For thin layer chromatography, precoated silica gel 60 F254 and RP-18 F254S plates were used. The compounds were visualized by spraying with a solution of H2SO4 5% in ethanol, followed by heating with a heat gun.
Plant Material
A armata roots were collected in Danang province, Vietnam in January 2021. The plant was identified by Dr Nguyen The Cuong at the Institute of Ecology and Biological Resources, VAST. A voucher specimen (coded: NCCT-P71R) was deposited at the Institute of Marine Biochemistry, VAST.
Extraction and Isolation
The dried roots of A armata (5.0 kg) were cut into small species, powdered and then ultrasonically extracted with MeOH, 3 times (each 12 L of MeOH for 60 min). After filtration, the solvent was removed in vacuo to give 135 g of methanol extract. This was suspended in water and successively partitioned with dichloromethane and ethyl acetate (EtOAc) to give organic soluble fractions and a water layer. The water layer was chromatographed on a diaion (HP-20) column washing with water to remove salts and oligosaccharides. Saponins were stepwise eluted by methanol/water (25%, 50%, 75%, and 100% volume of methanol) to give 4 fractions AA1-AA4. Fraction AA2 (12.0 g) was chromatographed on a silica gel column, eluting with dichloromethane/methanol (1/0-0/1, v/v) to give 4 fractions AA2A-AA2D. Fraction AA2C (3.0 g) was chromatographed on a reverse phase C18 column, eluting with methanol/water (3/5, v/v) to give 4 fractions AA2C1-AA2C4. Fraction AA2C2 was further chromatographed by HPLC (J'sphere H-80 column, 250 mm length × 20 mm ID), eluting with 16% acetonitrile in water, with a flow rate of 2 mL/min to give compounds
23-Hydroxyoleanolic Acid-[28-O-β-D-Glucopyranosyl]-
3-O-{β-D-Glucopyranosyl-(1→2)-[β-D-Glucopyranosyl-(1→3)]-β-D-Galactopyranoside} (Aramatoside C, 1 )
White amorphous powder,
Oleanolic Acid-[28-O-β-D-Glucopyranosyl]-3-O-{β-D-Glucopyranosyl-(1→2)-[β-D-Glucopyranosyl-(1→3)]-β-D-Galactopyranoside} (Aramatoside D, 2)
White amorphous powder,
Acid Hydrolysis of Compounds 1 and 2
Compounds
Cytotoxic Assay
The cytotoxic assays are the same as described in our previous work5,6 (see Supplemental Material).
Conclusions
The 2 previously undescribed oleanane-type triterpene glycosides, oleanolic acid-[28-O-β-D-glucopyranosyl]-3-O-{β-D-glucopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→3)]-β-D-galactopyranoside} (
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Funds for Science and Technology of the University of Danang, University of Science and Education (grant number T2020-TĐ-01) and Vingroup Joint Stock Company and supported by the Domestic Master/ PhD Scholarship Programme of Vingroup Innovation Foundation (VINIF), Vingroup Big Data Institute (VINBIGDATA) for Nguyen Thi Hong Chuong (grant number: VINIF.2020.TS.55).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
