Abstract
From the leaves of Aralia chinensis, 3 oleanane-type triterpene glycosides have been isolated, including 1 new glycoside, 3β,23-dihydroxyolean-12-ene-28-oic acid 3-O-β-
Introduction
The genus Aralia contains about 70 species used medicinally in Asia and the Americas. 1 Some of these species have been used in traditional medicine, such as A. armata, 2,3 A. elata, 4 and A. chinensis. 5 Phytochemical study of Aralia plants has led to the isolation of oleanane-type triterpenoid saponins, diterpenoids, phenolics, and acetylenic lipids. 1
Aralia chinensis L. is endemic to China and Vietnam and has been used as a traditional medicine to cure rheumatism, hepatitis, nephritis, and diabetes.
6
Twenty-seven oleanane-type saponins have been isolated from the root bark of A. chinensis.
7,8
However, there has been no report on the chemical composition of this plant growing in Vietnam. As part of our continuing research to find bioactive components from the genus Aralia,
9
we report herein the identification of 1 new (
Results and Discussion
Compounds
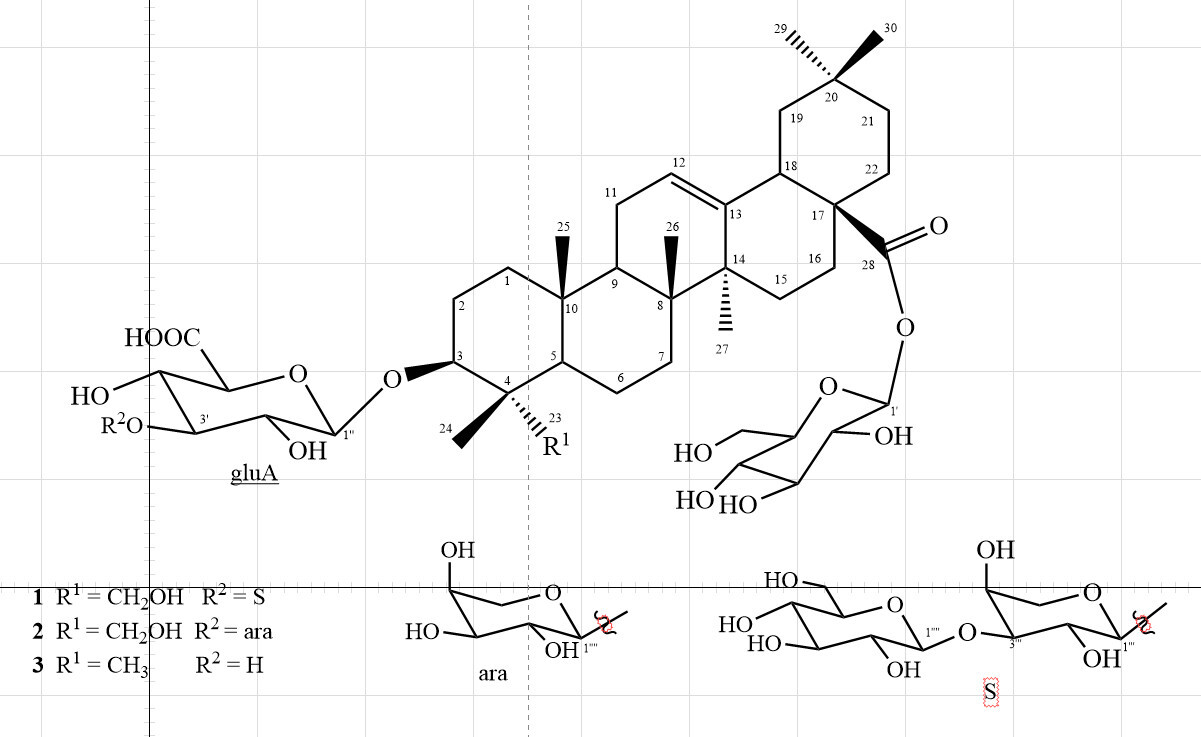
Chemical structures of compounds 1
1H and 13C NMR Spectroscopic Data for Compounds

Abbreviations: Abbreviations: NMR, nuclear magnetic resonance; NOESY, nuclear Overhauser effect spectroscopy.
NMR data were assigned by heteronuclear single quantum correlation, heteronuclear multiple quantum correlation, 1H-1H correlation spectroscopy, nuclear Overhauser effect spectroscopy spectra.
As with compounds

The 1H-1H correlation spectroscopy (COSY) and key heteronuclear multiple quantum correlation (HMBC) and nuclear Overhauser effect spectroscopy (NOESY) correlation of compound 1.
Compounds
Cytotoxic Effects of Compounds 1

Abbreviation: Abbreviation: IC50, half-maximal inhibitory concentration.
aPositive control compound.
Experimental
General
The NMR spectra were recorded on a Bruker 500 MHz spectrometer and HR-ESI-MS on an Agilent 6530 Accurate Mass Q-TOF LC/MS. The QTOF instrument was set at 2 GHz extended dynamic range resolution mode, negative ESI capillary voltage of 3500 V, fragmentor voltage of 175 V, MS scan ranging from m/z 100-1700, and an MS acquisition rate of 1.0 spectra/s. Optical rotation was measured on a Jasco P2000 polarimeter. Column chromatography was performed using silica gel, reverse phase C-18, and Diaion HP-20 resins as a stationary phase. HPLC was carried out using an AGILENT 1100 HPLC system. For thin-layer chromatography, precoated silica gel 60 F254 and RP-18 F254S plates were used. The compounds were visualized by spraying with a 5% solution of sulphuric acid in ethanol, followed by heating with a heat gun.
Plant Material
Aralia chinensis L. leaves were collected at Cao Loc, Lang Son, Vietnam, in May 2018, and identified by botanist Nguyen. The Cuong at the Institute of Ecology and Biological Resources, VAST. A voucher specimen (coded: NCCT-P71B) was deposited at the Institute of Marine Biochemistry, VAST.
Extraction and Isolation
Dried A. chinensis leaves (4.5 kg) were ultrasonically extracted with methanol (MeOH), 3 times (each 10 L of MeOH for 60 minutes). After filtration, the solvent was removed in vacuo to give 230 g of a dark methanolic residue. This crude extract was suspended in water and successively partitioned with dichloromethane and ethyl acetate to give organic soluble fractions and a water layer. The water layer was poured onto a Diaion (HP-20) column and washed with water to remove salts and oligosaccharides. Saponins were stepwise eluted by MeOH/water (25%, 50%, 75%, and 100% vol of MeOH) to give 4 fractions (ACW1-ACW4). Fraction ACW3 (12.5 g) was chromatographed on a silica gel column, eluting with dichloromethane/MeOH (8/1, v/v) to give 4 fractions, ACW3A-ACW3D. Fraction ACW3B (4.2 g) was further chromatographed on a reverse phase C18 column, eluting with MeOH/water (3/1, v/v) to give 4 fractions (ACW3B1- ACW3B4). Fraction ACW3B3 (76 mg) was subjected to HPLC (J’sphere H-80 column, length 250 mm × 20 mm ID, eluting with 18% acetonitrile in water, a flow rate of 3 mL/min) to yield compound
3β,23-Dihydroxyolean-12-ene-28-oic acid 3-O-β-d -glucopyranosyl-(1→3)-α-l -arabinopyranosyl-(1→3)-β-d -glucuronopyranoside 28-O-β-d -glucopyranosyl ester (araliachinoside A, 1 )
White amorphous powder.
3β,23-Dihydroxyolean-12-ene-28-oic acid 3-O-α-l -arabinopyranosyl-(1→3)-β-d -glucuronopyranoside 28-O-β-d -glucopyronosyl ester (2 )
White amorphous powder.
3β-Hydroxyolean-12-ene-28-oic acid 3-O-β-d -glucuronopyranoside 28-O-β-d -glucopyronosyl ester (3)
White amorphous powder.
Cytotoxic Assay
Two human cancer cell lines (epidermoid carcinoma cell line KB-ATCC CCL-17 and hepatoma carcinoma cell line HepG2-ATCCHB-8065), obtained from the American Type Culture Collection (USA) ATCC, were used for cytotoxic evaluation. The cells were grown in Roswell Park Memorial Institute Medium 1640 supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 µg/mL streptomycin at 37 °C in a humidified atmosphere (95% air and 5% carbon dioxide). The exponentially growing cells were used throughout the experiments. The inhibitory effects of the compounds on the growth of the human cancer cell lines were determined by measuring the metabolic activity using an MTT assay. Briefly, human cancer cell lines (1 × 105 cells/mL) were treated for 3 days with a series of concentrations of the compounds (in dimethylsulfoxide): 0.125, 0.5, 2.0, 8.0, 32.0, and 128.0 µg/mL. After incubation, 0.1 mg MTT solution (50 µL of a 2 mg/mL solution) was added to each well, and the cells were then incubated at 37 °C for 4 hours. The plates were centrifuged at 1000 rpm for 10 minutes at room temperature, and the media were then carefully aspirated. Dimethylsulfoxide (150 µL) was added to each well to dissolve the formazan crystals. The plates were read immediately at 540 nm on a microplate reader (TECAN GENIOUS). All the experiments were performed 3 times, and the mean absorbance values were calculated. The results are expressed as the percentage of inhibition that produced a reduction in the absorbance by the treatment of the compounds compared with the untreated controls. A dose-response curve was generated, and the IC50 was determined for each compound as well as each cell line.
Conclusions
Three oleanane-type triterpene glycosides, including a new one, 3β,23-dihydroxyolean-12-ene-28-oic acid 3-O-β-
Supplemental Material
Online supplementary file 1 - Supplemental material for Araliachinoside A: A New Triterpene Glycoside From Aralia chinensis Leaves
Supplemental material, Online supplementary file 1, for Araliachinoside A: A New Triterpene Glycoside From Aralia chinensis Leaves by Pham Hai Yen, Nguyen Thi Cuc, Phan Thi Thanh Huong, Nguyen Xuan Nhiem, Nguyen Thi Hoai, Ho Duc Viet, Bui Huu Tai, Nguyen Van Tuyen, Chau Van Minh and Phan Van Kiem in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research is funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 104.01-2017.08.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
