Abstract
In this study, jujube polysaccharide (JP) was extracted from Ziziphus jujuba cv. Jinsixiaozao and sulfated JP (SJP) was prepared. The optimum preparation conditions were as follows: reaction temperature 75°C, reaction time 1 h, ratio of chlorosulfonic acid-to-pyridine (VCSA/VPyr) 1. The degree of substitution of SJP was 0.664 ± 0.014. JP and SJP were typical heteropolysaccharides, which were composed of rhamnose, arabinose, xylose, mannose, glucose, and galactose, but the molar ratio of monosaccharides was different. Fourier transform infrared spectra showed that JP was sulfated successfully. Compared with JP, the molecular weight of SJP increased to 3.17 × 105 Da, its water solubility increased significantly, and its viscosity decreased significantly. When the microstructure of SJP was examined, it was found that the surface of the polysaccharides became loose and porous after sulfation. SJP had a higher hydroxyl radical scavenging activity than the unsulfated polysaccharide. Moreover, sulfation enhanced the antibacterial activity of the polysaccharides against Escherichia coli and Bacillus subtilis. Therefore, sulfation is an effective way to improve the biological activity of the polysaccharide, and SJP can be used as a potential antioxidant and antimicrobial agent in the field of food and medicine.
Keywords
Introduction
Polysaccharides are a class of high molecular weight (Mw) compounds that are widely found in animals, plants, and microorganisms. Polysaccharides have rich functions such as antioxidation, antitumor, hypoglycemic, and immune regulation, and have received more and more attention. 1 In recent years, there has been great interest in the biological activity of polysaccharides and their derivatives, especially sulfated derivatives. Sulfation refers to the introduction of sulfuric acid groups on the hydroxyl groups of polysaccharides to change their physical and chemical properties and increase or produce new biological activities. Compared with natural polysaccharides, they have broad or stronger biological activities, such as anticoagulation, 2 antioxidation, 3 immune regulation, 4 antitumor, 5 antiviral, 6 antibacterial, 7 and anti-inflammatory. 8
The key to obtaining sulfated polysaccharides with high biological activity is to adopt suitable sulfation methods. Up to now, these include chlorosulfonic acid–pyridine (CSA–Pyr) method, concentrated sulfuric acid method, and sulfur trioxide-pyridine method. Considering the high degree of substitution (DS) and yield of the prepared sulfated polysaccharides, the CSA–Pyr method is still the most commonly used one for preparing sulfated polysaccharides. 9 In order to obtain highly active sulfated polysaccharides, orthogonal test methods and response surface methods (RSM) are usually used to determine the optimal sulfation conditions. 10
Chinese jujube (Ziziphus jujuba Mill.) belongs to the Rhamnaceae family, which is mainly distributed in subtropical and tropical regions. 11 Jujube, which has been cultivated for about 4000 years in China, is mainly distributed in the Yellow River Basin, Northwest, and East regions. China is the only exporter of jujube fruits. At present, the planting area of jujube trees in China is about 2 million hectares, and the annual output is more than 8 million tons. 12 Jujube is rich in a variety of biologically active ingredients, such as polysaccharides, flavonoids, triterpene acids, saponins, phenolic acids, and vitamin C (Vc). 13 Polysaccharide is one of the most abundant ingredients in jujube fruit. Jujube polysaccharides (JPs) have immune regulation, antioxidant, antitumor, hypoglycemic, and gastrointestinal protection effects. 14 Therefore, as a potential biologically active molecule, JP has received extensive attention. However, the limited solubility of JP limits its application. Our study attempts to increase its solubility through chemical modification and to explore its biological activity. So far, there have been few reports on chemical modification, especially sulfation, and the characteristics and activities of the sulfated derivatives of jujube. In this study, JP was extracted with water and precipitated with ethanol to produce the raw material, which was then sulfated. RSM was used to optimize the preparation conditions of sulfated JP (SJP). SJP was characterized and its activity was studied to explore the effects of sulfation on its antioxidant and antibacterial activity. This work provides a theoretical basis for further development and utilization of JP resources.
Results and Discussion
Single-Factor Experiment Analysis
The reaction temperature was considered to be a key factor in the preparation of SJP. In order to investigate the effect of reaction temperature on DS of SJP, the preparation was carried out at different reaction temperatures (50°C, 60°C, 70°C, 80°C, and 90°C). Other preparation parameters were: reaction time 2 h, and VCSA/VPyr 1:2. As shown in Figure 1A, DS increased and then decreased with the increase of reaction temperature. The results showed that higher reaction temperatures have a negative influence on DS of SJP. This may be due to the degradation of JP at high temperatures. Therefore, in the RSM optimization experiment, 70°C was chosen as the center point of the reaction temperature.
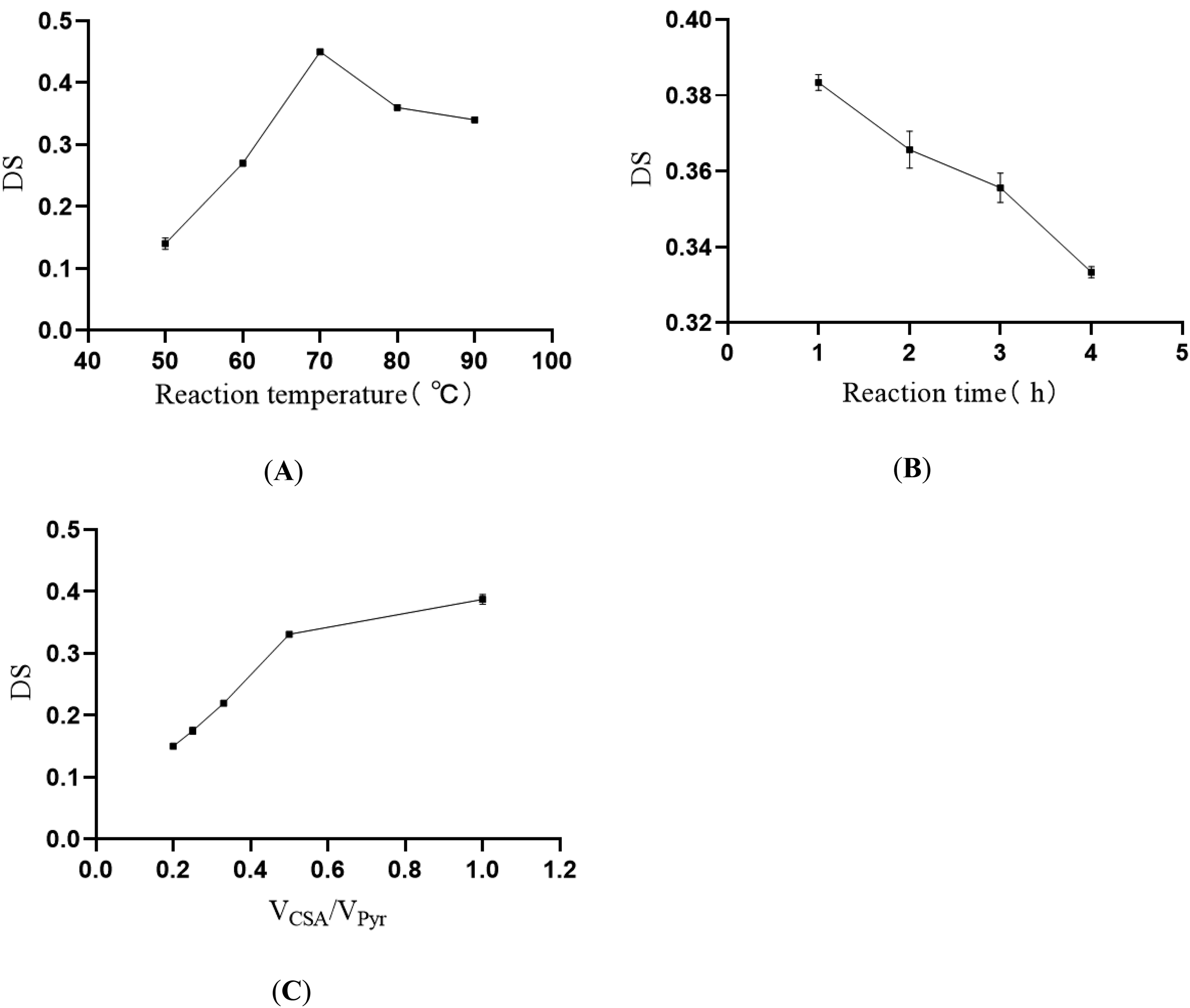
The effect of reaction temperature, reaction time, and VCSA/VPyr on the DS of SJP. (A) Reaction temperature, (B) reaction time, and (C) VCSA/VPyr.
The reaction time was another factor affecting the DS of SJP. The reaction time was set to 1, 2, 3, and 4 h to determine the effect of reaction time on DS. The reaction temperature was set at 70°C, and VCSA/VPyr at 1:2. As shown in Figure 1B, with the increase in reaction time, DS showed a downward trend, which may be related to the degradation of JP. With the increase in reaction time, acid hydrolysis of chlorosulfonic acid (CSA) occurred, which was consistent with previous research results. 15 Therefore, 1 h was chosen as the central time point of the RSM experiment.
The effect of VCSA/VPyr on DS of SJP is shown in Figure 1C. The reaction temperature was 70°C and the reaction time was 1 h. DS increased with the increase in the ratio of CSA to Pyr. When the ratio was 1, DS reached its maximum. This may be related to the complete reaction of CSA when the ratio of CSA-to-pyridine (Pyr) was increased, and excessive Pyr may affect the substitution reaction. In this process, Pyr was mainly used as a catalyst. Therefore, in the RSM experiment, 1 was chosen as the center of VCSA/VPyr.
RSM Analysis
As shown in Table 1, the experiment was run 17 times to optimize 3 separate preparation parameters. The results showed that the DS of SJP ranged from 0.1390 to 0.6768. The experimental data were analyzed by multiple regression with Design Expert 11.0 software, and the response variables and test variables were related by the following second-order polynomial equation:
Experimental Scheme and Results.

Note: CSA, chlorosulfonic acid; Pyr, pyridine; DS, degree of substitution.
The significance of model coefficients was measured by F and P values. Generally, the larger the value of F, the smaller the value of P, and the more meaningful the model and corresponding variables will be. 16 It is shown in Table 2 that the P value of the model was less than .05 and the F value was 5.30, which indicated that the model was significant. In addition, we found that the P values of 2 quadratic terms (B2 and C2) were lower than .01, which indicated that they had a significant influence on DS. The deterministic coefficient (R2) was the ratio of variability in data, which can be explained by the model. When the independent variables in the model have no statistical influence, Radj2 should be smaller than R2. Actually, Radj2 played a more important role than R2 in evaluating the fitting degree of the model. In our experiment, we found that Radj2 was .7074, which was very close to R2 (.8720).
Variance Analysis.

Note: CSA, chlorosulfonic acid; Pyr, pyridine; R2 = .8720; Radj2 = .7074.
*P < .05 significant; **P < .01 highly significant.
As shown in Figure 2, three-dimensional response surface plots and two-dimensional contour plots are graphical representations of regression equations. The figure shows the interaction between 2 variables and the relationship between the response value of each variable and the experimental level. As shown in Figure 2A and D, when VCSA/VPyr (Figure 2C) was fixed at 0, the comprehensive influence of reaction temperature (Figure 2A) and reaction time (Figure 2B) on DS is shown. DS increased at first and then decreased with the increase in reaction temperature and reaction time. Similar trends were found in the reaction temperature and VCSA/VPyr, and reaction time and VCSA/VPyr interaction (Figure 2B and E, C and F). As can be seen from the P values of the 3 factors in Table 2, the reaction temperature has the greatest influence on the DS of SJP, followed by VCSA/VPyr, and the reaction time was the last.
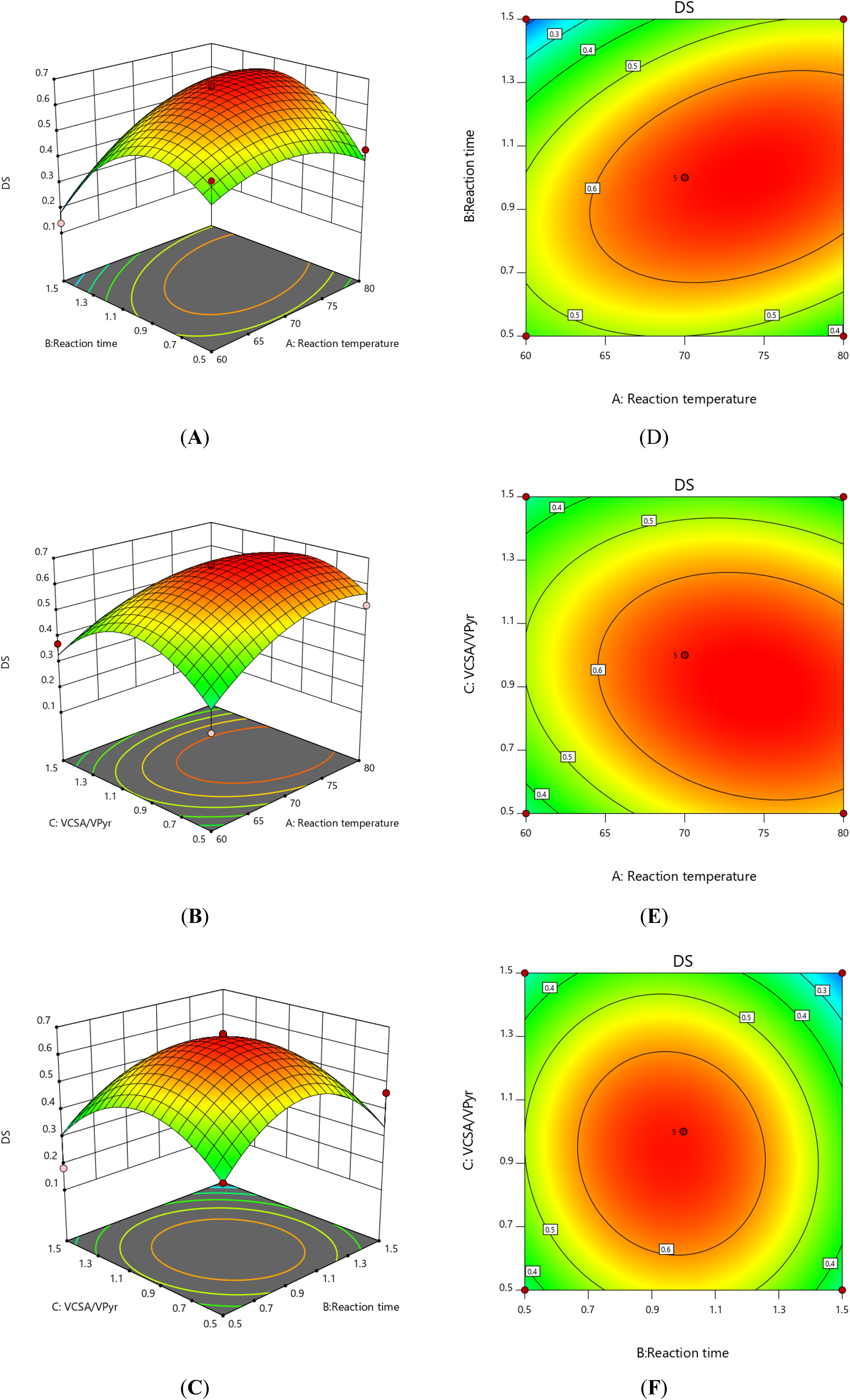
Three factors affect the DS of SJP. (A, D) Reaction temperature and reaction time; (B, E) reaction temperature and VCSA/VPyr; and (C, F) reaction time and VCSA/VPyr.
By using Design-Expert 11.0 software, the optimum preparation conditions of SJP were obtained as follows: reaction temperature 74.5°C, reaction time 1.02 h, and VCSA/VPyr 0.90, under the optimal conditions; the maximum predicted value of DS was 0.685. Considering the practical operability of the preparation process, the optimized conditions were as follows: reaction temperature was 75°C, extraction time was 1 h, and VCSA/VPyr was 1. In addition, 5 parallel experiments were carried out under the improved conditions to verify the findings. As a result, the actual value of DS was 0.664 ± 0.014, which was not significantly different from the predicted value of 0.685. The results showed that the model designed for this experiment was effective for DS prediction.
Characterization of SJP
Physicochemical Properties
The chemical composition of JP and SJP is shown in Table 3. The contents of total carbohydrate and uronic acid in JP were 75.4 ± 1.1% and 39.8 ± 0.6%, respectively. The contents of total carbohydrate and uronic acid in SJP decreased to 63.4 ± 1.3% and 36.7 ± 1.0%, respectively. The change in physical and chemical properties may be related to the degradation of polysaccharides under acidic conditions, which was consistent with the reported results. 17
Physicochemical Properties of JP and its Sulfated Derivatives.

Note: JP, jujube polysaccharide; SJP, sulfated jujube polysaccharide; Mw, molecular weight; Rha, rhamnose; Ara, arabinose; Xyl, xylose; Man, mannose; Glc, glucose; Gal, galactose.
Water Solubility Analysis
As shown in Table 3, the water solubility of JP increased from 50.8 ± 0.93 g/L to 93.3 ± 0.96 g/L after sulfation. This indicated that sulfation significantly improved the water solubility of the natural polysaccharides, which was beneficial for further study on their biological activities. Chaidedgumjorn et al 18 found that sulfation not only improved the water solubility of polysaccharides, but also changed their structure and physical and chemical properties, resulting in changes in biological activity. Tao et al 19 found that water solubility and the introduction of sulfuric acid groups are the main factors that enhance antitumor activity.
Viscosity Analysis
As shown in Table 3, the viscosity of JP decreased significantly from 498.7 ± 3.8 mPa·s to 45.3 ± 2.5 mPa·s. This may be because the introduction of sulfate partially blocked the cross-linking between polysaccharide molecules, which reduced the degree of polymerization of the polysaccharide, thus reducing its viscosity in solution.
Mw Analysis
The Mw of JP and SJP were determined by high-performance liquid chromatography (HPLC); the standard curve and HPLC curve of JP and SJP are shown in Figure 3. It can be seen in Table 3 that, compared with natural polysaccharides, the Mw of SJP was obviously increased, which was attributed to the introduction of sulfate groups, which increased the Mw.

Standard curve and HPLC curve of JP and SJP. (A) Standard curve, (B) JP and SJP.
Monosaccharide Composition Analysis
As shown in Table 3, JP and SJP were typical heteropolysaccharides, which were composed of rhamnose, arabinose, xylose, mannose, glucose, and galactose, and had different molar ratios. The main monosaccharides of JP and SJP were rhamnose, arabinose, and xylose. After sulfation, the monosaccharide composition of the polysaccharides did not change, and the ratio of Ara, Man, Glc, and Gal in SJP increased, while the ratio of Xyl decreased. These results suggest that sulfation in this study may have led to changes in the monosaccharide composition of the polysaccharides, but this change did not lead to changes in the backbone of the polysaccharides. 20
Fourier-Transform Infrared Analysis
Fourier-transform infrared (FT-IR) spectra of JP and SJP are shown in Figure 4. Two samples had the typical signal peaks of polysaccharides at 3420, 3273, 2930, 1620, and 1099 cm−1. 21 The absorption peak at 1260 cm−1 was the asymmetric tensile vibration of S = O, which indicated the existence of substituted sulfate. The absorption peak at 812 cm−1 was the tensile vibration of C–O–S. 22 The results showed that the sulfation of JP was successful.

Infrared spectra of JP and SJP.
Scanning Electron Microscopy Analysis
Figure 5 shows the magnified scanning electron microscopy (SEM) images of 500 ×, 1000 ×, and 5000 × of JP and SJP. The results showed that JP was flaky with a compact structure and smooth surface (5000 ×), while SJP was fragmentary with a rough surface that was loose and porous. The differences in the surface morphology of the polysaccharides indicated that sulfation had changed the surface morphology of JP, which may be related to the increase in its solubility.

SEM images of JP and SJP. (A-C) JP × 500, × 1000, × 5000, respectively and (D-F) SJP × 500, × 1000, × 5000, respectively.
Antioxidant Activity of JP and SJP
2,2-Diphenyl-1-picrylhydrazyl Radical Scavenging
2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical assay is one of the most commonly used methods to determine the antioxidant activity of polysaccharides in vitro. Therefore, the scavenging ability of JP and SJP on the DPPH radical was evaluated and compared with Vc. Figure 6A shows the DPPH radical scavenging activities of JP, SJP, and Vc. The DPPH radical scavenging ability of the sample was positively correlated with the concentration of the sample. When the concentration was in the range of 1 to 3 mg/mL, the scavenging rate of JP on the DPPH radical increased significantly with the increase in concentration. When the concentration was in the range of 4 to 5 mg/mL, the scavenging rate of the DPPH radical by the 2 polysaccharides did not change obviously with the increase of concentration. When the sample concentration was 5 mg/mL, the DPPH scavenging activities of JP, SJP, and Vc were 83.5%, 82.7%, and 96.8%, respectively. The scavenging activity of JP and SJP on DPPH free radicals was lower than that of Vc, but JP showed stronger scavenging activity. Sulfated Cyclocarya paliurus polysaccharides (S-CP1-1 and S-CP1-4) have lower DPPH free radical scavenging activity than natural polysaccharides (CP) 20 ; this was consistent with the results of this study. As shown in Table 3, the content of uronic acid in JP (39.84%) was higher than that in SJP (36.68%). It can be seen that the higher the content of uronic acid in polysaccharides, the stronger is their antioxidant activity, which was consistent with previous reports. 23
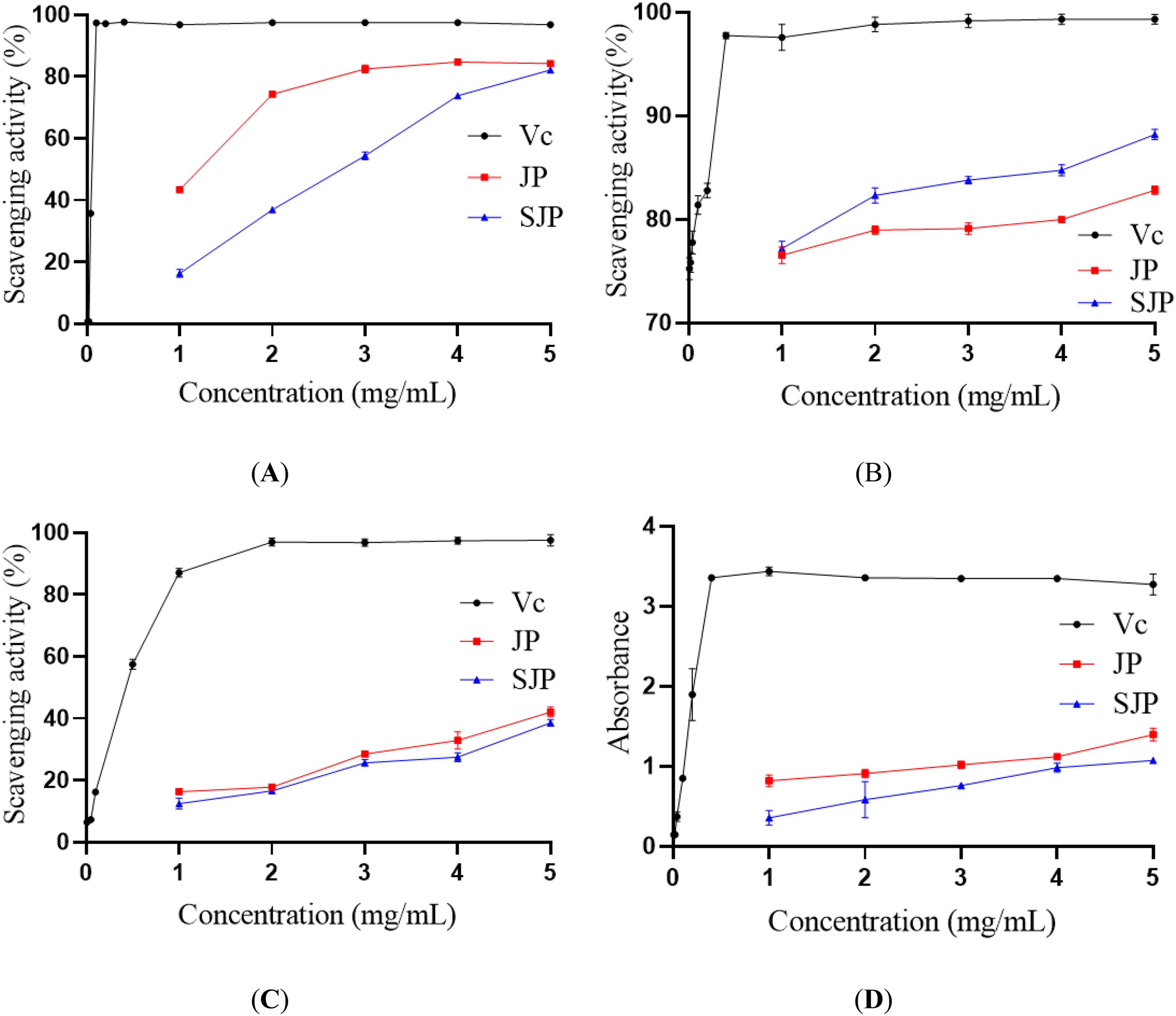
Antioxidant and free radical scavenging activities of JP and SJP. (A) DPPH radical assay, (B) hydroxyl radical assay, (C) superoxide anion radical assay, and (D) reducing power assay.
Hydroxyl Radical Scavenging
Hydroxyl is a strong oxidant, which easily reacts with biological macromolecules, such as nucleic acids, proteins, and lipids, resulting in cell damage. 24 The production of reactive oxygen species is promoted, which results in severe oxidative damage to different tissues. The hydroxyl radical scavenging activities of JP and SJP are shown in Figure 6B. This shows that the scavenging activities of JP and SJP on hydroxyl radicals increase with an increase in concentration, which indicates that the concentration of polysaccharides has a dose–dependent relationship with scavenging activities. When the concentration was in the range of 2 to 5 mg/mL, the scavenging activity of SJP on the hydroxyl radical was significantly higher than that of JP. When the concentration was 5 mg/mL, the hydroxyl radical scavenging activities of JP, SJP, and Vc were 82.8%, 88.2%, and 99.3%, respectively. The results show that the introduction of a sulfate group could significantly improve the scavenging ability of JPs on hydroxyl radicals, which was conducive to the exertion of its antioxidant activity. Sulfation improved the scavenging activity of Cyclocarya paliurus polysaccharide (S-CP1-8) on hydroxyl free radicals. 20 The results show that sulfate groups play an important role in the scavenging activity of hydroxyl free radicals, and the scavenging effect of polysaccharides on hydroxyl free radicals may be related to their hydroxyl groups. 25 This may be attributed to the combination of sulfate and hydroxyl groups to form a stable group, which inhibits its reaction with biological macromolecules and inhibits the production of reactive oxygen species, thus preventing damage to cells.
Superoxide Radical Assay
Superoxide free radicals are a kind of initial free radicals produced by the mitochondrial electron transport system, which can induce the production of hydroxyl free radicals and lipid peroxidation products, which are harmful to biological molecules (such as enzymes, proteins, and DNA). 26 Figure 6C shows the scavenging effects of JP and SJP on superoxide radicals. Both JP and SJP showed different levels of scavenging activity against superoxide radicals, and they are concentration-dependent. When the concentration was in the range of 1 to 5 mg/mL, the scavenging ability of JP for the superoxide radical was stronger than that of SJP, but the difference was not significant (P > .05). The results indicated that the introduction of sulfate groups did not improve the ability of JPs to scavenge superoxide radicals. After garlic polysaccharide was modified by sulfation, its superoxide-free radical scavenging activity was lower than that of the natural polysaccharide. 27 This may be due to the fact that the introduction of sulfate groups reduces the sugar content of polysaccharides; similar results have been obtained from previous studies. 20
Reducing Power Assay
The reducing power of JP and SJP are shown in Figure 6D. The reducing power of JP and SJP increased with increases in their concentration. However, JP showed higher reducing power than SJP. The reducing power is usually related to the reducing agent, which exerts its antioxidant activity by providing hydrogen atoms. 28 The hydroxyl group in JP was replaced by a sulfuric acid group, which reduced the reducing power of JP. The results show that sulfation was not conducive to an improvement in the reducing power of JP. The reducing power of SJP was less than that of JP, which indicates that the introduction of sulfate groups weakens the reducing power of the polysaccharides. 29
Antibacterial Activity of JP and SJP
Polysaccharides could become a barrier to inhibit the growth of bacteria by preventing the entry of nutrients. Polysaccharides could change the permeability of the cell wall and cell membrane of bacteria, 30 leading to a rapid increase of water-soluble intracellular protein or inhibit the growth of bacteria. 31 For the same kind of bacteria, the barrier effect may increase with the increase of polysaccharide concentration. 32 However, the antibacterial mechanism of JP and SJP is still unclear, and further research is needed.
The size of inhibition zone reflects the antibacterial activity of polysaccharides to the tested strains. There was no significant difference in the antibacterial activity of different concentrations of JP against E coli, while the antibacterial activity of different concentrations of SJP from high to low was: 4% > 2% > 1%. At the same concentration, the antibacterial activity of SJP was stronger than that of JP. The results showed that the antibacterial activity of SJP against E coli was in accordance with the dose–response relationship, which was consistent with the results previously reported. 33 Therefore, sulfation could improve the antibacterial activity of JP against E coli.
There was no obvious difference in the antibacterial activity of JP at different concentrations against B subtilis, while the antibacterial activity of SJP with different concentrations was in the following order: 4% > 2% > 1%. At the same concentration, SJP had stronger antibacterial activity against B subtilis than JP. The results showed that the antibacterial activity of SJP against B subtilis was in accordance with a dose-effect relationship, and that the antibacterial activity of JP against B. subtilis could be improved by sulfation. Previous studies have shown that sulfated polysaccharide from Pleurotus eryngii has a stronger antibacterial effect than natural polysaccharide, while sulfated exopolysaccharide from Streptococcus thermophiles ASCC 1275 has stronger antibacterial effect than natural polysaccharide. 31 Li et al 34 reported that the sulfated derivatives showed the best antibacterial activity against 4 kinds of bacteria compared with the natural polysaccharides from “peony seed dreg.”
In our study, sulfation improved the antibacterial activity of JP against E coli and B subtilis. The antimicrobial activity of SJPs may be related to their water solubility and viscosity, which may further affect its antimicrobial activity. Therefore, sulfation can be used as an effective way to improve the antibacterial activity of JPs.
Conclusions
In this study, the structural characteristics, antioxidant and antibacterial activities of SJPs were studied. After sulfation, the polysaccharides have the same monosaccharide composition, but different molar ratios. Sulfation can increase the Mw and solubility of natural polysaccharides, and reduce their viscosity. The surface structure becomes loose and hollow, which may be related to the increase in solubility. Sulfation can also significantly improve the hydroxyl radical scavenging ability of polysaccharides, and the antibacterial activity against E coli and B subtilis is also improved. Therefore, SJP can be used as a potential antioxidant and antibacterial agent. Sulfation of polysaccharides is of great significance for screening polysaccharides with high antioxidant activity. It can also improve the water solubility of polysaccharides and expand their applications in food and medicine.
Materials and Methods
Materials and Reagents
Ziziphus jujuba cv. Jinsixiaozao was purchased from Cang County, Hebei, China in 2019.
Monosaccharide standards (D-glucose, D-galactose, D-arabinose, D-xylose, D-mannose, and L-rhamnose) and trifluoroacetic acid (TFA) were purchased from Sigma Company (Sigma), different Mw dextrans (T-10, T-40, T-70, T-500, and T-2000) from Beijing Solarbo Technology Co., Ltd, and CSA and pyridine (Pyr) from Sigma (Sigma). Ultrapure water was obtained from a Milli-Q water purification system (Millipore). All reagents were of analytical grade.
E coli strain OP50 and B subtilis CMCC(B) 63501 were purchased from Nantong Kaiheng Biotechnology Development Co., Ltd.
Fractionation and Purification of JP
For the fractionation and purification of JP a published method was used, 20 with slight improvement. In short, the jujube was washed and pitted, dried at 80°C and crushed. The obtained jujube powder was soaked in 3 times its volume of 95% ethanol, and the ethanol was replaced every 24 h. This was repeated 3 times to remove fat-soluble impurities. The defatted material was mixed with distilled water in a ratio of 1:20, heated in a boiling water bath for 2 h, and filtered. Distilled water was added to the residue and extracted again. This was repeated 3 times, stirring intermittently during the extraction process. The filtrates were combined, concentrated by evaporation, and 4 times the volume of 95% ethanol was added. The resulting precipitate was washed 3 times with 80% ethanol. The fractions were deproteinized 5 times by the Sevage method, and then purified by the cut-off ultrafiltration method, with an Mw cut-off (MW) of 8000 to 14,000 Da (XL, Pilli-con®, Millipore) to remove small molecular impurities. The collected polysaccharides were freeze-dried and coded as JP hot water extract fraction (JP).
Preparation of SJP
SJP was obtained by the CSA–Pyr method. 35 In an ice-water bath, CSA was dropped into anhydrous Pyr in different proportions, and the reaction was stirred at room temperature for 30 min to prepare a sulfation reagent. JP (500 mg) was suspended in precooled anhydrous formamide (100 mL), and the prepared sulfation reagent (20 mL) was added dropwise. Under constant magnetic stirring, the sulfation reaction was kept at 70°C and the reaction time was 1 h. After the reaction, the mixture was cooled to room temperature, and the pH of the reaction system was adjusted to 7.0 with 4 M NaOH solution. Then, the polysaccharide solution was dialyzed in a dialysis bag to remove Pyr and small molecules; the dialysate was concentrated by rotary evaporation, and the sulfated derivative was obtained by vacuum freeze-drying and labeled SJP.
Experimental Design
Single-Factor Experimental Design
The DS of sulfated polysaccharides is mainly affected by reaction temperature, reaction time, and VCSA/VPyr. 36 In this study, these 3 factors were selected for a single-factor experimental design. In short, the reaction temperature was set from 50°C to 90°C, the reaction time from 1 to 4 h, and the VCSA/VPyr from 1:1 to 1:5. In the process of optimizing the experimental factors, one factor was changed, while the other factors remained the same in each experiment.
Experimental Design for Optimization
According to the results of a single-factor test, factor A (reaction temperature), factor B (reaction time), and factor C (VCSA/VPyr) were used for response surface analysis. In the response surface analysis of Design-Expert 11.0, the Box–Behnken design was selected for 3 factors and 3 levels of experimental design. The encoded and actual values are shown in Table 4; DS was the response value.
Experimental Factors and Levels in Response Surface Analysis.

Note: CSA, chlorosulfonic acid; Pyr, pyridine.
Characterization of JP and SJP
Determination of the Contents of Carbohydrate, Uronic Acid, and DS
The phenol–sulfuric acid method was used to determine the carbohydrate content, with D-glucose as the standard, 37 and the meta-hydroxybiphenyl method for uronic acid content, with D-glucuronic acid as the standard. 38
The BaCl2-gelatin method was used to determine the DS of SJP.
20
In short, K2SO4 was used as a standard product to establish the standard curve. A 5 mg SJP was dissolved in 4 mL HCl (1 M) and heated at 105°C for 12 h. After drying with nitrogen, the product was dissolved in 1 mL of water, the sample solution fully mixed with 1 mL HCl (1 M) and 0.5 mL BaCl2-gelatin (5%), and reacted at 25°C for 20 min. The absorbance was measured at 360 nm; ultrapure water was the blank. In this case, the calculation formula of DS was as follows:
Water Solubility Analysis
Natural polysaccharides are poorly soluble in water, which affects their biological activity.
39
The water solubility of JP and SJP was determined according to previous studies.
40
A 100 mg sample was added to 1 mL ultrapure water, which was shaken at room temperature for 12 h, and then centrifuged at 12,000 rpm for 20 min. The undissolved residue was separated from the supernatant and freeze-dried. The solubility of the sample was calculated according to the following formula:
Viscosity Measurements
The viscosity of JP and SJP solutions was measured with a digital viscometer (NDJ-5S). During the process, the temperature of the samples was maintained at 30°C, the speed was controlled at 60 rpm, and the viscosity value was recorded in mPa·s. 41
Determination of Mw
The Mw of JP and SJP was determined using HPLC and an ELSD-1260 (Agilent Technologies) evaporative light detector. A Shodex KS-805 (300 mm × 8.0 mm, Japan SHOWA DENKO KK) gel filtration chromatography column was used. The chromatographic conditions were as follows: polysaccharide concentration 0.5 mg/mL; the solution was filtered through a 0.22 μm syringe membrane filter; mobile phase was ultrapure water (pH 7.0), flow rate was 1 mL/min, injection volume 20 μL, and column temperature 35°C. A series of dextran standards (T-2000, T-500, T-70, T-40, and T-10) were used to establish a standard curve. According to the retention time of JP and SJP, the Mw of polysaccharides was calculated. 42
Determination of Monosaccharide Composition
The monosaccharide composition of JP and SJP was determined by gas chromatography (GC7890A, Agilent). The instrument was equipped with a TG-5MS capillary column (30 m × 0.25 mm × 0.25 μm) and a flame ionization detector. A 20 mg of polysaccharide was hydrolyzed with 2 mL of TFA (4 M) at 110°C for 4 h, and then methanol was added and evaporated repeatedly to remove TFA. Then 20 mg hydroxylamine hydrochloride and 1 mL Pyr were added and reacted in a 90°C water bath for 30 min. After cooling to room temperature, acetic anhydride (1 mL) was added, and the reaction was continued for 30 min at 90°C. The reaction product was directly analyzed by GC.
FT-IR Spectral Analysis
Infrared spectroscopy is a method to analyze the primary structure of polysaccharides. 43 The absorption peaks of JP and SJP were measured using a FT-IR spectrophotometer (Spectrum 65, PerkinElmer). 20 The samples were mixed with KBr particles in the ratio of 1:100, ground evenly, and analyzed by tablet pressing. The frequency resolution was 1 cm−1, 32 scans were made, and the frequency range was 4000 to 400 cm−1.
SEM Analysis
The polysaccharide samples were fixed on the holder, sputtered with gold, and the surface morphology of JP and SJP was characterized by SEM (Model VEGA3 LMH, TESCAN).
Antioxidant Activity
DPPH Radical Scavenging Activity
The scavenging activity of JP and SJP for the DPPH free radical was determined according to the method of Brand-Williams et al,
44
but with slight modification. The samples were dissolved in deionized water to prepare solutions of different concentrations (1, 2, 3, 4, and 5 mg/mL). A 2 mL sample solution was mixed with 2.0 mL DPPH-ethanol solution (0.04 mg/mL). The mixed solution was left in the dark at 37°C for 30 min, and the absorbance of the sample was determined at 517 nm. Distilled water was used as a negative control and Vc as a positive control. The scavenging activity of the DPPH free radical was calculated according to the following formula:
Hydroxyl Radical Scavenging Activity
The scavenging activity of JP and SJP for the hydroxyl radical was determined using a reported method, with slight modification.
45
A 1 mL salicylic acid-ethanol (4.5 mM), 1 mL FeSO4 (4.5 mM), and 1 mL H2O2 (4.5 mM) were added successively and reacted in a water bath at 37°C for 30 min. The absorbance of the reaction solution was determined at 510 nm. Distilled water was used as the blank control and Vc as the positive control. The scavenging activity of the hydroxyl radical was calculated according to the following formula: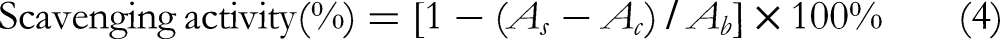
Superoxide Radical Scavenging Activity
The scavenging activities of JP and SJP for the superoxide radical were measured according to a reported method, with slight modification.
46
A 3 mL of 0.05 mol/L Tris-HCl buffer (pH 8.2) was added to 0.5 mL of sample solution of different concentrations (1, 2, 3, 4, and 5 mg/mL), and after reacting at 30°C for 20 min, 3 mL (7 mM) of prewarmed gallic acid solution was added. After reaction for 4 min, 1 mL of concentrated hydrochloric acid (HCl) was added to terminate the reaction. The absorbance of the sample was determined at 320 nm. Tris-HCl buffer was used as blank and Vc as a positive control. The scavenging activity for the superoxide radical was calculated according to the following formula: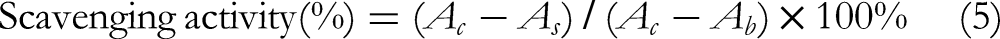
Reducing Power
According to a reported method, 47 the reducing ability of JP and SJP was measured. In short, 2.5 mL phosphate buffer (pH 6.6), 2.5 mL K3Fe (CN)6 solution (1%), and 1.0 mL sample solution of different concentrations (1, 2, 3, 4, and 5 mg/mL) were mixed and reacted in a water bath at 50°C for 20 min. After cooling to room temperature, 2.5 mL trichloroacetic acid solution (10%) was added to terminate the reaction and centrifuged at 3000 rpm for 10 min. Supernatant (2.5 mL), 2.5 mL distilled water, and 0.5 mL FeCl3 solution (1 mg/mL) were mixed and reacted for 10 min, then the absorbance was measured at 700 nm. The absorbance value of the sample indicates its reducing power.
Antibacterial Activity
The in vitro antibacterial activities of JP and SJP against 2 kinds of bacteria were analyzed by agar diffusion. In this study, 48 the antibacterial activities of JP and SJP were determined by an agar diffusion method, but slightly modified. Sample solutions of different concentrations (1%, 2%, and 4%) were prepared with ultrapure water, and then sterilized with a 0.45 μm microporous filter. Bacteria were cultured overnight and their concentration adjusted to 1 × 106 CFU/mL using a blood cell counter (Shanghai Qiujing Biochemical Reagent Instrument Co., Ltd). Bacterial suspension (300 mL) was evenly smeared on the surface of an aseptic nutrient agar plate with a sterilized coater. The filter paper with different concentrations of polysaccharides in a diameter of 8.0 mm was placed on the inoculated agar medium with 7 pieces per plate, with a gap between the filter paper. The filter paper was soaked in sterile water, and gently pressed onto the solid medium under aseptic conditions. The plate was inverted in a 37°C constant temperature incubator for 48 h.
Statistical Analysis
Data are expressed as means ± standard deviation. The RSM data were statistically analyzed by design-expert 11.0. Other statistical analyses were performed using GraphPad Prism 8.0 software (GraphPad Software Inc.). One-way analysis of variance was used for statistical comparison, followed by Tukey test; P < .05 or P < .01 was considered statistically significant.
Footnotes
Acknowledgments
This study was supported by the Natural Science Foundation of Hebei Province and the Key R&D projects in Hebei Province, Funding Project for Introducing Overseas Students in Hebei Province.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key R&D projects in Hebei Province (grant no. 20327123D), Natural Science Foundation of Hebei Province (grant no. C2019204366) and Funding Project for Introducing Overseas Students in Hebei Province.

