Abstract
Feiduqing (FDQ) is a traditional Chinese medicine formula used for many years in the treatment of viral pneumonia (VP). However, the effective components of FDQ and the mechanism by which it affects VP remain unclear. The purpose of this study is to determine the multitarget mechanism of the effect of FDQ on VP through determination and in vivo pharmacodynamics combined with network pharmacology. Firstly, the compound–target–pathway network was constructed by using TCMSP, UniProt, GeneCards, STRING, and DAVID databases through Cytoscape 3.7.0. Secondly, the content of the effective components of the original prescription of FDQ was determined. Finally, the pharmacological activity of FDQ in vivo was verified by an animal model, and the active ingredient composition (AIC), selected by network pharmacology was used for antipyretic, antiinflammatory, antitussive, and expectorant symptoms. Seven compounds of FDQ and 22 potential target genes in the treatment of VP with FDQ were identified by network pharmacology analysis. Kyoto Encyclopedia of genes and genomes enrichment analysis results indicated that the mechanism of FDQ in the treatment of VP was mainly related to pathways in cancer, hepatitis b, tumor necrosis factor (TNF) signaling pathway, Chagas disease, tuberculosis, influenza A, human T-cell leukemia virus, type 1 infection, toxoplasmosis and toll-like receptor signaling pathways, osteoclast differentiation, nonalcoholic fatty liver disease, and leishmaniasis. The results of pharmacodynamic experiments showed that FDQ and AIC possessed antipyretic, cough relieving, and reducing sputum effects. Besides, FDQ and AIC could also significantly reduce the content of prostaglandin E2, TNF-α, cyclic adenosine monophosphate, interleukin-1β, and myeloperoxidase in vivo, while increasing the content of interleukin-10 in vivo. The active ingredients of FDQ prescriptions could be accurately screened by network pharmacological analysis, as they clarified the mechanism of FDQ in the treatment of VP. The research results provided potential ideas and methods for the screening and purification of active ingredients in traditional Chinese medicine prescriptions.
Introduction
Viral pneumonia (VP) is an inflammation of the lungs caused by viral upper respiratory tract infection, which spreads downward and has symptoms of headache, fever, cough, and expectoration. Feiduqing (FDQ) is a prescription for clinical experience in the treatment of VP. It is composed of 3 herbal components: Oroxylum indicum (L.) Vent (Muhudie [MHD]), Gardenia jasminoides Ellis (Zhizi [ZZ]), and Sophora flavescens Ait (Kushen [KS]) in a weight ratio of 1:3:2. It has the effects of heat-clearing, detoxifying, and moisturizing lungs. FDQ also plays a positive role in relieving fever, cough, and irritability caused by VP. Modern pharmacological research of MHD 1 has studied its obvious inhibitory effect on inflammation. ZZ has the functions of heat-clearing, fire-purging, blood-cooling, and detoxifying.2,3 Furthermore, ZZ contains a large amount of cyclic olefin ether terpenoids, organic acids, flavonoids, and other ingredients with antiinflammatory, antipyretic, analgesic, and antioxidative effects. KS has functions of heat-clearing, dampness-drying, bacteriostasis, and insect killing. In addition, because it contains a variety of active ingredients, it shows obvious antiinflammatory effects. 4
Traditional Chinese medicine (TCM) refers to drugs used to prevent, treat, and diagnose diseases under the guidance of TCM theory. Chinese medicine is highly valued worldwide for its clinical efficacy and safety. TCM has significant antibacterial and other pharmacological effects.5,6 Through the methods of multicomponents, multitargets, and multipathways, the action mechanism of TCM is described systematically based on its therapeutic function. 7 At present, the technological route and quality control of TCMs are mostly based on chemical components, but the correlation of these with clinical safety and effectiveness is not yet clear, which may affect the efficacy of the drugs to a certain extent. 8 Hopkins 9 proposed the concept of “Network pharmacology” in 2007. With the emergence of multicomponent and multitarget modern drug development models, the advantages of TCM in the treatment of complex diseases have become increasingly prominent. The efficacy of TCM is the result of the interaction and mutual adjustment of the active ingredient group formed by multiple active ingredients in TCM and multiple disease-related targets. It is called active ingredient omics.10,11 Network pharmacology is an emerging discipline based on the theory of systems biology. It conducts network analysis of biological systems, builds a “drug–target–disease” network relationship, and then select specific signal nodes to design multitarget drug molecules. 12 In this study, network pharmacology analysis was used to identify the possible mechanism of FDQ in VP treatment. The antipyretic, antitussive, and phlegm-reducing effects of FDQ and AIC suggested by the network analysis were further verified by in vivo biological experiments.
Materials and Methods
Screening of Chemical Ingredients in FDQ
The main chemical constituents of O indicum (L.) Vent (MHD), Gardenia jasminoides Ellis (ZZ), and S flavescens Ait (KS) were searched for using the TCM systems pharmacology database and analysis platform (TCMSP; http://lsp.nwu.edu.cn/tcmsp.php) and literature analysis in different platform databases. Higher oral bioavailability (OB) values generally indicate that the bioactive molecules of the drug have drug-likeness (DL). FDQ shows positive effects on the treatment of VP, mainly for oral administration. Therefore, the OB values ≥30% and DL values ≥0.18 were selected to screen the active ingredients of the 3 Chinese medicines in the TCMSP database.
Collection of Compound-Related Targets and VP-Related Targets
The target with a known chemical composition in FDQ has been screened by the target prediction function in the TCMSP database. The predicted targets of the compounds were screened and searched in the UniProt database (https://www.uniprot.org/). Uniprot (https://www.genecards.org/) database was used to import the names of proteins and limit the “species” to “Homo sapiens.” All retrieved protein targets were corrected to Uniprot ID and gene names corresponding to FDQ target proteins were obtained. VP-related targets were collected from GeneCards (https://www.genecards.org/). 13
Construction and Analysis of the Protein–Protein Interaction (PPI) Network
The screened and disease targets were uploaded to Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny/index.html) to obtain the intersection of the active compounds and targets of VP genes, as the active ingredients of FDQ in the treatment of VP. To explain further the mechanism of FDQ active ingredient targets and disease targets at the protein level, the potential target genes of FDQ for the treatment of VP were introduced into the protein interaction analysis platform STRING11.0 (https://string-db.org/) to obtain the relationship of protein interaction. Then, the “species” was set as “human,” and the PPI network map was obtained. PPIs were predicted through STRING11.0, including direct and indirect interactions between proteins, and the key points were highlighted and assigned to the information about each protein interaction. The higher the score, the more confident the target protein interactions were. Then, the results exported from the STRING11.0 were imported into the software of Cytoscape 3.7.0 for visual analysis and network analysis. Finally, the PPI network map was obtained from the comprehensive score reflected by color, degree value, size, and edge thickness.
Network Construction and Analysis
To observe further the biological function of targets, 49 compound targets and VP targets were imported into the DAVID v6.8 (https://david.ncifcrf.gov/) database for pathway enrichment analysis. The identifier was set to “OFFICIAL GENE SYMBOL,” the list type was set to “Gene List,” the species to “Human,” and the P-value <.05; the data were classified according to the P-value, and the pathways were sorted from high to low. The Kyoto Encyclopedia of genes and genomes (KEGG) pathway information of FDQ involved in the pathological process of VP was screened out. The KEGG pathway information with significant differences was screened out using the Omicshare database (http://www.omicshare.com) to draw an advanced bubble chart. Then, GraphPad Prism 6.0 was used to draw histograms.
According to the above target prediction results of FDQ for the treatment of VP, the FDQ component–target–pathway network model was constructed by the “merging” function of Cytoscape software. Compounds, targets, and pathways were represented by nodes in the network.
Preparation and Chemical Analysis of FDQ Extract
Materials
BS-6KH electronic scale was purchased from Shanghai Yousheng Weighing Apparatus Co., Ltd; DZTW electronic thermostat electric heating jacket was purchased from Tianjin Industrial Laboratory Instrument Co., Ltd; FA2004 Analytical Electronic Balance was purchased from Shanghai Liangping Instrumentation Co., Ltd; and Waters 2695 High-Performance Liquid Chromatograph was purchased from Waters.
The 3 herbs in FDQ were purchased from Beijing Tong Ren Tang Chinese Medicine Co. Ltd, quercetin (≥98% Lot no. 100081-299406) from China Institute for the Control of Pharmaceutical and Biological Products, matrine (≥98% Lot no. 519-02-8) from Chengdu Phytochemical Pure Biotechnology Co., Ltd, and kaempferol (≥98% Lot no. wkq19011609), aloe-emodin (≥98% Lot no. wkq19040302), ellagic acid (≥98% Lot no. wkq19010702), oroxylin a (≥98% Lot no. wkq19022608), and formononetin (≥98% Lot no. wkq19013002) from Sichuan Weikeqi Pharmaceutical Technology Co., Ltd.
Determination of Potential Active Constituents of FDQ
FDQ was extracted by traditional techniques to determine the content ratio of the active ingredients in the original prescription.
The 3 herbs in FDQ were mixed in a ratio of 1:3:2 (w/w/w). KS and MHD were extracted 3 times (1.5 h each time) by adding 10 times the amount of 70% ethanol. ZZ was extracted 3 times (1 h each time) by adding 12 times the amount of water. The water extract of ZZ was concentrated to a density of 1.20, and then it was precipitated with 85% ethanol for 24 h. After standing for 24 h, the precipitate was collected by filtration. To determine the content of each index and prepare FDQ ethanol (FDQA) extract, the ethanol extracts of KS and MHD were combined with the ZZ ethanol precipitated solution after water extraction. Then, the FDQ extract was filtered and concentrated by rotary evaporation. Finally, the extract was dried under reduced pressure and stored in a desiccator before use. The 3 Chinese medicinal materials were extracted by refluxing 3 times according to the prescription plus 12 times the amount of water for 1 h each time. The combined extract was concentrated by rotary evaporation and dried under reduced pressure to obtain FDQ water extract (FDQW).
For quercetin analysis, 10 µL of FDQA solution was prefiltered through a 0.45 μm Millipore filter and injected into a Supercell ODS-B C18 column (250 mm × 4.6 mm, 5 μm). Linear gradient elution with acetonitrile (B) and 0.2% phosphoric acid (C) was carried out. The program was set as follows: time (min)/B (v/v):C (v/v); T0.01/15:85, T10/23:77, T15/30:70, T20/50:50, and T22/15:85. The flow rate was 1.0 mL/min and the column temperature was kept constant at 35 °C. The detector was ultraviolet absorption (UV) and the detection was performed at 360 nm. For kaempferol analysis, the mobile phase was methanol (A) and 0.1% acetic acid (C). The composition of the eluent was 30% methanol in the first 72 min, 30%-100% methanol from 72 to 76 min, 100% methanol from 76 to 78 min, 100%-30% methanol from 78 to 80 min, 30% methanol from 80 to 85 min. The column temperature was maintained at 30 °C and the detection wavelength was 270 nm. The other analytical conditions were the same as the above quercetin analysis. For matrine analysis, the mobile phase was acetonitrile (B) and 0.05 mol/L potassium dihydrogen phosphate (C) (containing triethylamine 2.0 mL/L). The gradient elution procedure was as follows: time (min)/B (v/v):C (v/v); T0.01/6:94, T12/10:90, and T45/10:90. The column temperature was 30 °C, and the detection wavelength was set at 220 nm. Other conditions were the same as above. For aloe-emodin analysis, the mobile phase was the same as for kaempferol. The gradient elution procedure was as follows: time (min)/A (v/v):C (v/v); T0.01/72:28, T6.5/92:8, T22.5/72:28, and T30/72:28. The detection wavelength was 270 nm. Other conditions were the same as for the determination of kaempferol. For formononetin analysis, a linear gradient elution of acetonitrile (B) and 0.01% phosphoric acid (C) was used. The program was as follows: time (min)/B (v/v):C (v/v); T0.01/30:70, T12/33:67, T13/40:60, and T25/40:60. Except for the detection wavelength of 248 nm, the other conditions were the same as those for kaempferol content determination.
Quercetin, kaempferol, matrine, aloe-emodin, and formononetin were mixed as the active ingredient composition (AIC) according to the ratio of the above measurement results.
Pharmacological Verification on Network Analysis
Materials
Codeine phosphate (CP) (Lot no. 20180433) was supplied by Qinghai Pharmaceutical Factory Co., Ltd, bromhexine hydrochloride (BH) by Zhejiang Wanbang Pharmaceutical Co., Ltd, aspirin effervescent tablets (Lot no. 1903009) by Astra Zeneca Pharmaceutical Co., Ltd, emergency branch syrup (EBS) (Lot no. 18110256) by Taiji Group Chongqing Fuling Pharmaceutical Factory Co., Ltd, and Lianhua Qingwen Capsule (LHQW-C) (Lot no. A1811006) by Shijiazhuang Yiling Pharmaceutical Co., Ltd. All other reagents were of analytical grade or higher. Prostaglandin E2 (PGE2), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-10 (IL-10) Elisa kit, and cyclic adenosine monophosphate (cAMP) and myeloperoxidase (MPO) kits were purchased from Jiangsu Meimian industrial Co., Ltd.
Animals
Sprague–Dawley rats (180-200 g, 5-6 weeks old) and specific-pathogen-free mice (18-20 g, 4 weeks old) were supplied by Chengdu Dashuo Technology Co., Ltd, and maintained according to the Guidelines for the Care and Use of Laboratory Animals. All animal research and experimental protocols were approved by the Institutional Animal Experiment Committee of Chengdu University (SCXK [Sichuan Province, China] 2015-030).
Animal Treatment and Tissue Preparation
Four days before the experiment, the rats and mice were provided water and food freely under laboratory conditions.
Antipyretic effect: Ten grams of chloral hydrate was dissolved in 100 mL of physiological saline to obtain 10% chloral hydrate solution. Lipopolysaccharide (LPS) solution (10 mg of LPS in 1 L of physiological saline) was prepared under sterile conditions.
Before grouping, the anal temperature was measured at 9:00 and 16:00 for 3 consecutive days, and the average of 6 consecutive body temperatures was taken as the basal body temperature (T0). Rats with a body temperature of >38 °C or a temperature difference of >0.5 °C were excluded. Rats were randomly divided into control group (10 mL/kg), aspirin group (330 mg/kg), LHQW-C group (420.0 mg/kg), FDQW group (460.0 mg/kg), FDQA group (336.2 mg/kg), and AIC group (19.10 mg/kg). Rats in each group were continuously administered for 7 days at the above doses. The control group was administrated orally with water at the same time. LHQW-C, FDQW, FDQA, and AIC suspensions were prepared by dissolving crude drugs and 0.6% sodium carboxymethylcellulose in purified water. Thirty minutes after the last dose, rats were injected subcutaneously with 10 μg/100 g of LPS. After injection, the anal temperature of rats was measured at 0, 1.0, 2.0, 3.0, and 4.0 h. The changes in body temperature were recorded (T = T1–T2). T is the body temperature change value, T1 is the body temperature after heating, and T2 is the body temperature before administration. After measuring all the anal temperatures, the rats were executed, and their abdominal aorta blood and liver were taken. The activities of PGE2, TNF-α, IL-1β, and cAMP in rat serum and the activity of MPO in liver homogenate were measured.
Antitussive effect: The screened mice were randomly divided into 6 groups: control group, CP group (12 mg/kg), EBS group (20 mL/kg), FDQW group (920.0 mg/kg), FDQA group (672.4 mg/kg), and AIC group (38.2 mg/kg). An ammonia cough test was performed about 1 h after administration, and the cough latency of the mice and the number of coughs within 2 min were recorded. 14 Blood from the mice was collected from the orbit. IL-10 activity of serum was measured according to the manufacturer's recommendations using an Elisa kit.
Expectoration effects: Mice were randomly divided into control group (10 mL/kg), BH group (4.8 mg/kg), EBS group (20 mL/kg), FDQW group (920.0 mg/kg), FDQA group (672.4 mg/kg), and AIC group (38.2 mg/kg). Mice were sacrificed after 7 days’ administration and the trachea was removed. The phenol red secretory volume of the windpipe was observed. 15
Statistics
Statistical analysis was performed using SPSS 20.0 (IBM). The statistical results of each group were compared with each other using analysis of variance, and the statistical significance was set to a P-value of <.05.
Results
Screening of Chemical Constituents of FDQ
Thirty-two active compounds were identified in FDQ after eliminating the duplicates from the TCMSP database (Table 1).
Compounds and Molecular Details.
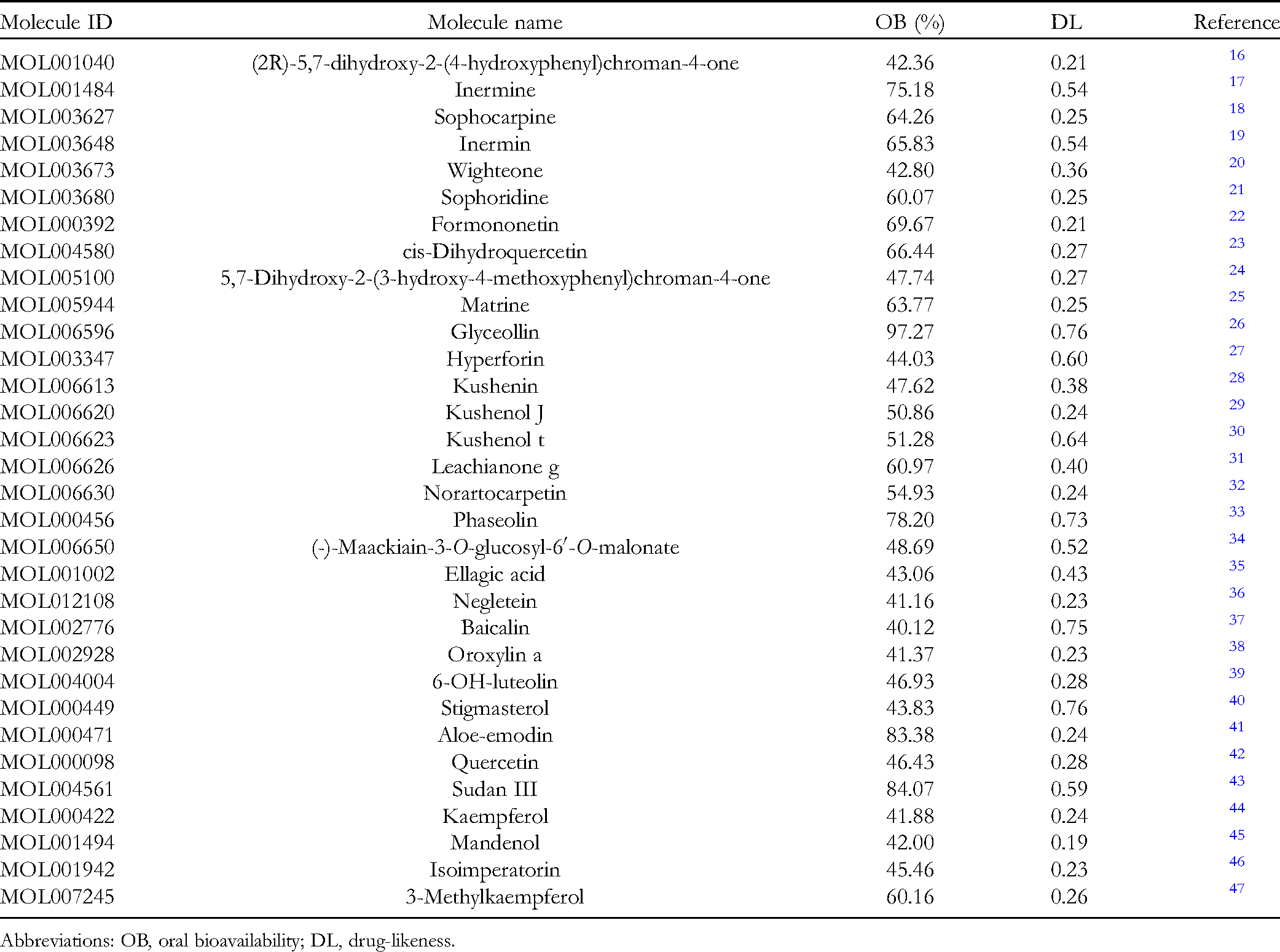
Abbreviations: OB, oral bioavailability; DL, drug-likeness.
Construction of Target Protein Interaction Network for FDQ in the Treatment of VP
As shown in Figure 1, there are 49 targets between 209 compound-related targets and 300 VP-related targets. The 49 shared targets of the PPI network map generated by the STRING database possessed high confidence scores, indicating a strong correlation between these targets. The PPI network of these 47 shared targets is depicted in Figure 2. The potential target information is shown in Table 2. Topology analysis indicated that TNF, IL-6, RELA, JUN, mitogen-activated protein kinase 1 (MAPK1), IL-1β, C-X-C motif chemokine-8 (CXCL8), TP53, AKT1, and C-C motif chemokine 2 (CCL2) were the top 10 shared targets based on degree centrality.
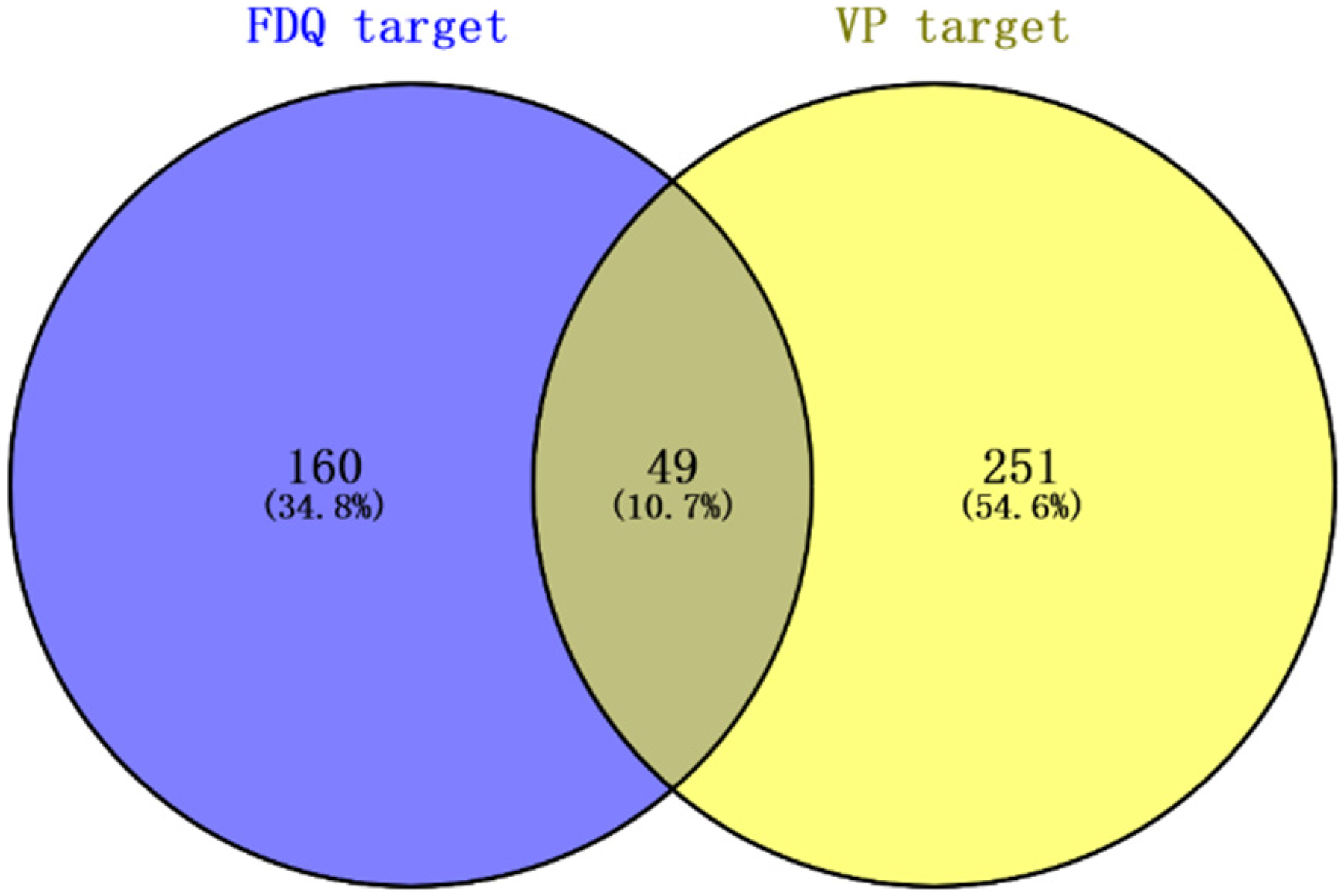
Distribution of FDQ potential targets and VP targets.

The network of the shared targets between FDQ potential targets and VP targets. The color of nodes is proportional to the degree centrality by topology analysis.
Potential Target Information.

Abbreviations: AP-1, activator protein 1; BAX, bcl-2-associated X protein.
Bioinformatics Analysis for FDQ in the Treatment of VP
Gene ontology (GO) and KEGG enrichment analysis were conducted on the corresponding targets of active components of FDQ using the DAVID database, and a threshold value of P < .05 was set. The most important biological processes (BPs) or pathways were screened and plotted using GraphPad Prism. GO enrichment analysis refers to a directed acyclic graph composed of the number of proteins or genes at a certain functional level, including 3 branches of BPs, cellular components (CCs), and molecular functions (MFs). Cytokine-mediated signaling pathway, response to cytokine, and cellular response to cytokine stimulus ranked first in Figure 3 (BP). Extracellular space, extracellular region part, and extracellular region ranked first in Figure 4 (CC). Cytokine receptor binding, cytokine activity, and signaling receptor binding are the top ones in Figure 5 (MF). Besides, the KEGG pathway enrichment analysis showed that these 49 key targets contributed to 198 pathways (P-value <.05). The top 20 pathways closely related to the mechanism of FDQ in the treatment of VP are shown in Figures 6 and 7.

Enriched genes for BP as the potential targets of main active ingredients of FDQ to treat VP.
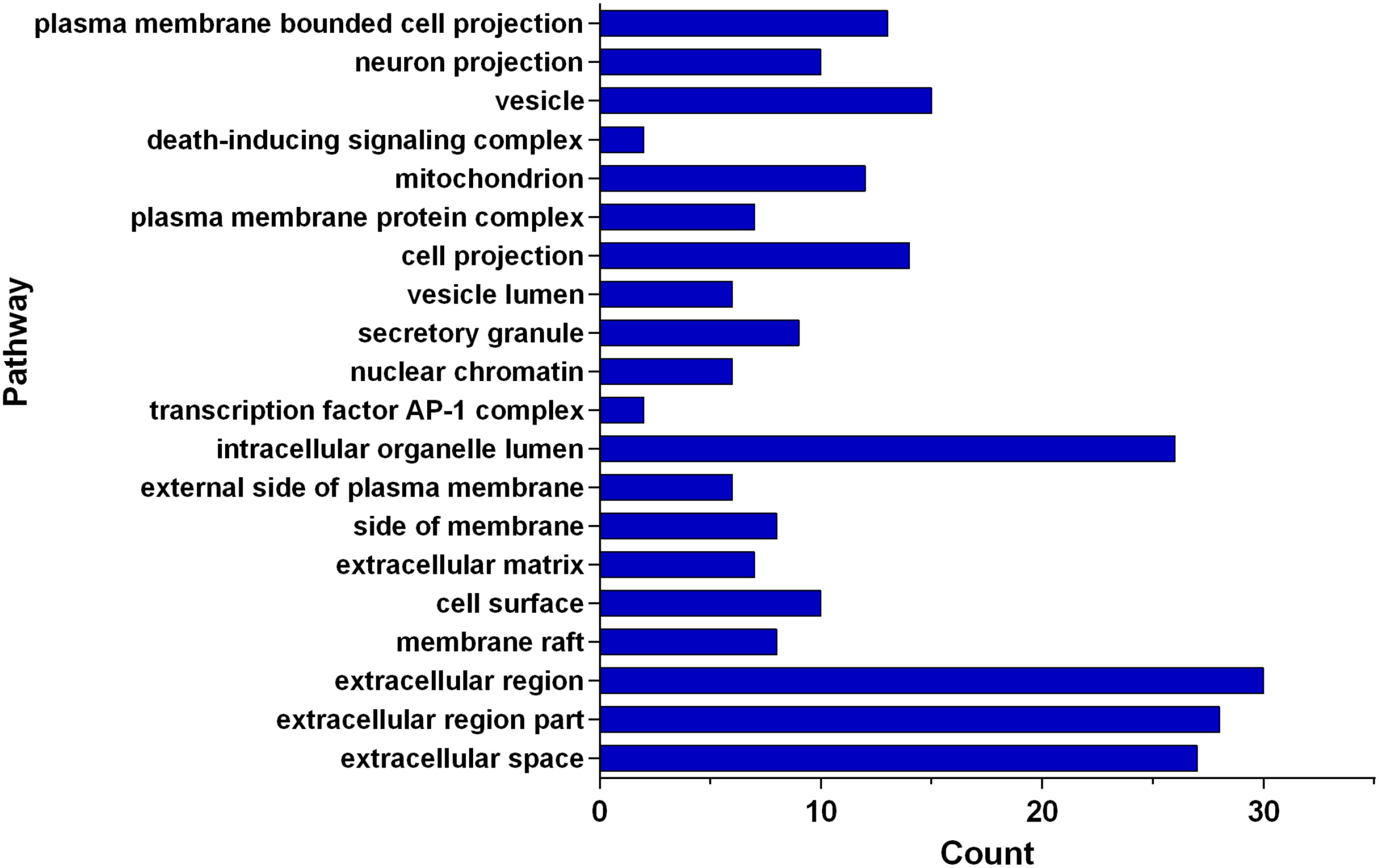
Enriched genes for CC as the potential targets of main active ingredients of FDQ to treat VP.
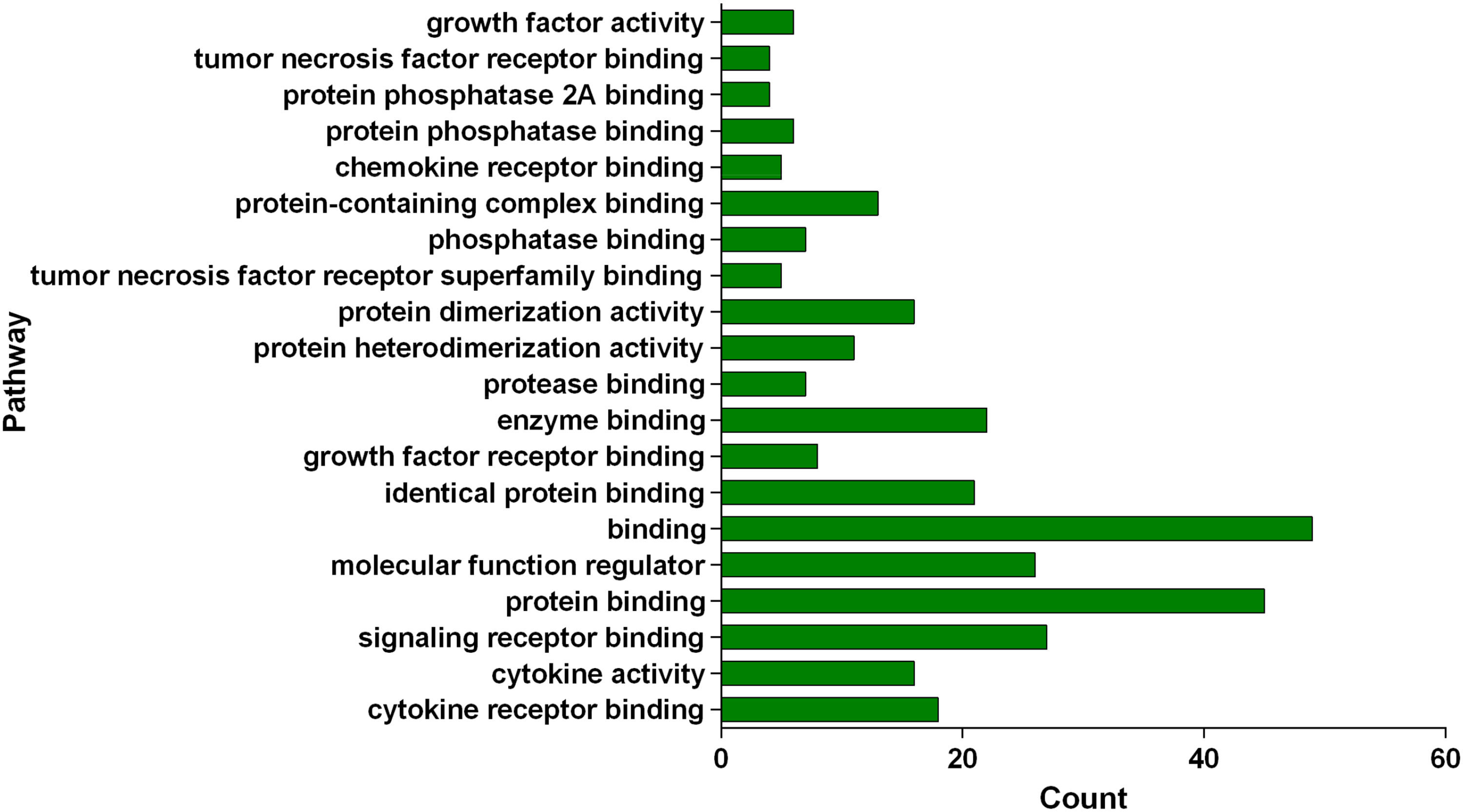
Enriched genes for MF as the potential targets of main active ingredients of FDQ to treat VP.
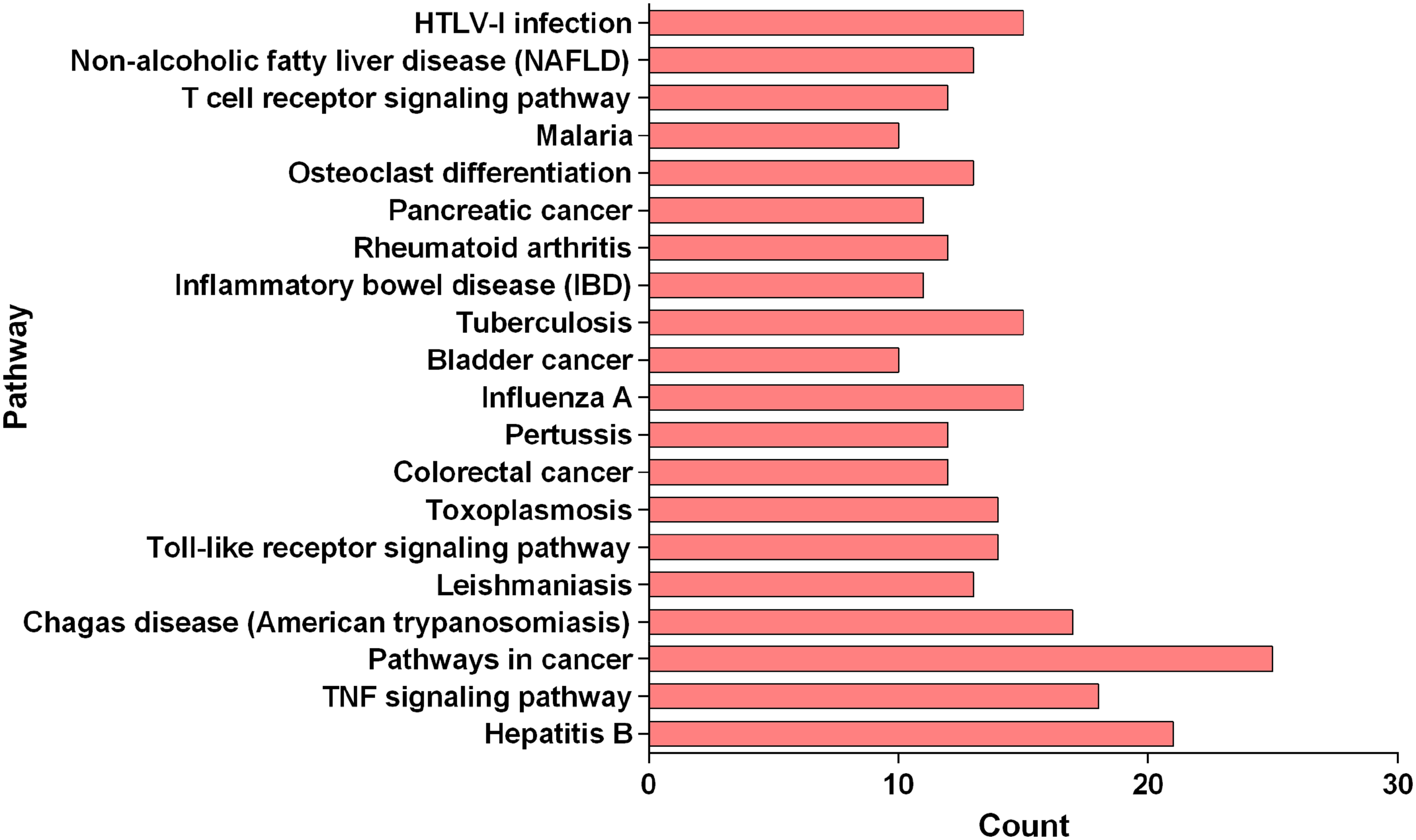
Enriched gene for KEGG as the potential targets of main active ingredients of FDQ to treat VP.

KEGG enrichment analysis of FDQ to treat VP.
The component–target–pathway network of FDQ treating VP was constructed by using the “merge” function of Cytoscape. As shown in Figure 8, the main active ingredients of FDQ were distributed in different pathways and coordinated with each other to adjust the effects of FDQ in the treatment of VP.

The component-target-pathway network construction for 20 pathways and 37 compounds in Feiduqing (FDQ) and 41 candidate protein targets. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article).
As shown in Figure 8, there were 137 nodes and 572 edges in this interaction network. Red rectangle nodes represent active ingredients, light green circles represent targets, and yellow arrows represent signal pathways. Degree, average shortest path length (ASPL), betweenness centrality (BC), and closeness centrality (CC) were ordered to determine the key nodes (Table 3). In the network, the components, targets, and signal pathways of which degree was greater or equal to median (component median was 1, target median was 10, and pathway median was 13) are shown in Table 3. Specifically, according to the degree of topological analysis and BC, quercetin, kaempferol, matrine, aloe-emodin, ellagic acid, oroxylin a, and formononetin are predicted to be important active compounds, indicating their critical roles in FDQ. In addition, prostaglandin-endoperoxide synthase 2 (PTGS2), IL-6, TNF, RELA, JUN, AKT1, and TP53 represented the crucial targets of FDQ based on degree and BC. Furthermore, most of these target proteins were enriched in pathways in cancer, hepatitis B, TNF signaling pathway, Chagas disease, tuberculosis, influenza A, human T-cell leukemia virus, type I infection, toxoplasmosis, and toll-like receptor signaling pathway.
Key Node Related Information.

Abbreviations: ASPL, average shortest path length; BC, betweenness centrality; CC, closeness centrality; PTGS2, prostaglandin-endoperoxide synthase 2; IL6, interleukin 6; TNF, tumor necrosis factor; MAPK1, mitogen activated protein kinase 1; CXCL8, C-X-C motif chemokine-8; VEGFA, vascular endothelial growth factor A; IL4, interleukin 4; CCL2, C-C motif chemokine 2; ICAM1, intercellular adhesion molecule 1; MAPK8, mitogen activated protein kinase 8; IL10, interleukin 10; IL2, interleukin 2; STAT1, Signal transducer and activator of transcription 1; IFNG, interferon gamma; EGFR, epidermal growth factor receptor; HTLV-I; human T-cell leukemia virus, type I; NAFLD, non-alcoholic fatty liver disease.
Determination of Potential Active Constituents of FDQ
The extraction yield of FDQA was 18.7% (w/w) and of FDQW was 25.5% (w/w). The contents of quercetin, kaempferol, matrine, aloe-emodin, and formononetin in the FDQ extract were 0.41%, 0.01%, 1.0%, 0.03%, and 0.07% (w/w), respectively.
Pharmacological Verification
Antipyretic Effect
Compared with the control group, the aspirin FDQW groups could significantly inhibit the fever caused by the LPS solution in rats, and the values for each group were lower than that of the control group at 1 h. At the second hour, the body temperature of the rats was significantly suppressed in all groups except the FDQA group. The body temperature of the rats was significantly inhibited in all groups at 3 and 4 h compared with the control group. The results are shown in Figure 9 and Table 4. In summary, LPS-induced fever in the rats was effectively suppressed in the AIC group, the effect being comparable to that in the FDQW and FDQA groups. As depicted in Figure 10 and Table 5, compared with the control group, the levels of PGE2, TNF-α, cAMP, IL-1β, and MPO in the serum of fever rats in the LHQW-C, FDQW, FDQA, and AIC group decreased to varying degrees (P < .05 and P < .01), and all of them showed obvious pharmacological effects.

Changes in body temperature in all groups.

PGE2, TNF-α, cAMP, IL-1β, and MPO activity. (A) The effect on the level of PGE2 in serum of rats. (B) The effect on the level of TNF-α in serum of rats. (C) The effect on the level of cAMP in serum of rats. (D) The effect on the level of IL-1β in serum of rats. (E) The effect on the level of MPO in serum of rats.
Effect of FDQ on LPS-Induced Fever in Rats (x ± s, n = 10).

Abbreviations: FDQ, Feiduqing; LPS, lipopolysaccharide; LHQW-C, Lianhua Qingwen Capsule; FDQW, FDQ water extract; FDQA, FDQ ethanol; AIC, active ingredient composition.
*P < .05 compared with control group.
**P < .01 compared with control group. ***P < .001 compared with control group.
Effects of Integration Processing of FDQ Water Extract, Ethanol Extract, and AIC on PGE2, TNF-α, cAMP, IL-1β and MPO Levels of LPS Induced Fever Rats.
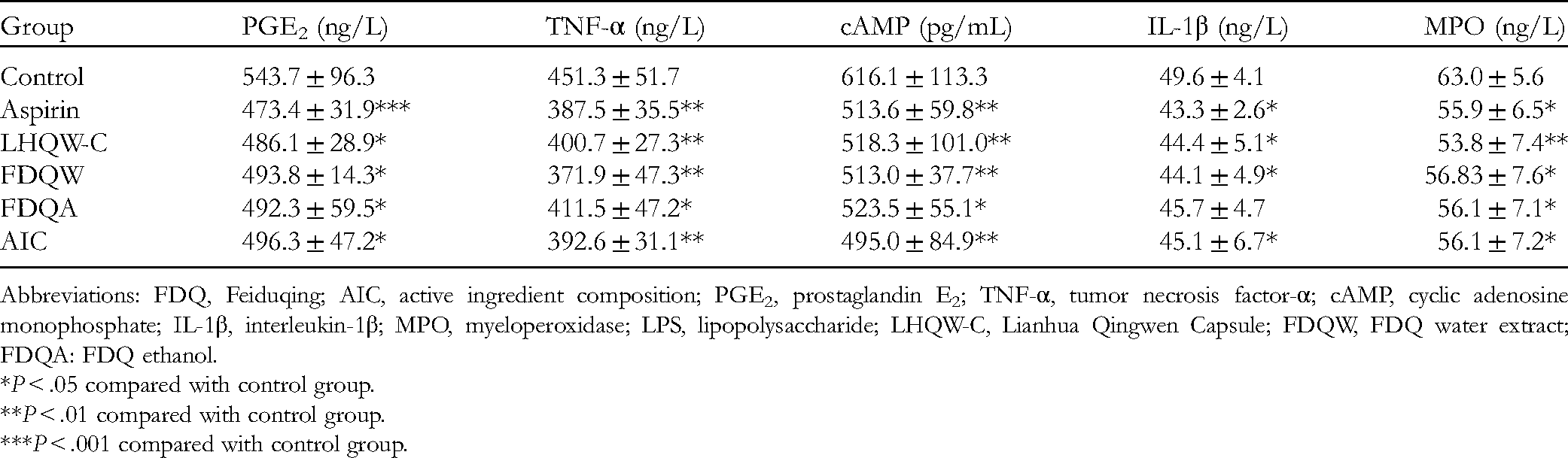
Abbreviations: FDQ, Feiduqing; AIC, active ingredient composition; PGE2, prostaglandin E2; TNF-α, tumor necrosis factor-α; cAMP, cyclic adenosine monophosphate; IL-1β, interleukin-1β; MPO, myeloperoxidase; LPS, lipopolysaccharide; LHQW-C, Lianhua Qingwen Capsule; FDQW, FDQ water extract; FDQA: FDQ ethanol. *P < .05 compared with control group. **P < .01 compared with control group. ***P < .001 compared with control group.
Antitussive Effect
As shown in Figure 11 and Table 6, compared with the control group, the cough latency of the mice was significantly prolonged in the EBS, FDQW, and AIC groups (P < .01). Also, certain effects were observed in the CP and FDQA groups (P < .05). The number of coughs in mice was significantly decreased in the FDQW, FDQA, AIC, and EBS groups. The activity of IL-10 in the serum of mice in each group was significantly increased compared with the control group (Figure 12 and Table 7). Therefore, the cough latency induced by ammonia in mice was prolonged, the number of coughs was reduced, and the activity of IL-10 in the serum of mice was enhanced after gavage with FDQW, FDQA, or AIC.

Latent period and cough times of mice. (A) The latent period of mice in different groups. (B) The cough times of mice in different groups.

Interleukin-10 (IL-10) concentration in mouse serum of each group.
Effects of Ammonia on Cough in Mice.

Abbreviations: CP, codeine phosphate; EBS, emergency branch syrup; FDQW, FDQ water extract; FDQA, FDQ ethanol; AIC, active ingredient composition. *P < .05 compared with control group. **P < .01 compared with control group. ***P < .001 compared with control group.
IL-10 Results for Each Group.

Abbreviations: IL-10, interleukin-10; BH, bromhexine hydrochloride; EBS, emergency branch syrup; FDQW, FDQ water extract; FDQA, FDQ ethanol; AIC, active ingredient composition. *P < .05 compared with control group. **P < .01 compared with control group. ***P < .001 compared with control group.
Expectoration Effects
The results showed that the phenol red excretion of mice was significantly increased in the FDQW, FDQA, and AIC groups compared with the control group (Table 8 and Figure 13). In summary, the AIC screened by network pharmacology possessed the effect of reducing sputum.

Phenol red concentration in mice trachea.
The Phenol Red Concentration in Each Group.

Abbreviations: CP, codeine phosphate; EBS, emergency branch syrup; FDQW, FDQ water extract; FDQA, FDQ ethanol; AIC, active ingredient composition. *P < .05 compared with control group. **P < .01 compared with control group. ***P < .001 compared with control group.
Discussion
FDQ is a combined formula of MHD, ZZ, and KS, which is widely used in the treatment of VP. The quality standard of FDQ's original medicinal materials have been studied and the FDQ extraction process through anti-virus test in vitro, anti-inflammatory, antipyretic, and analgesic effects in vivo have been preliminarily screened in the previous stage. Besides, the extraction, concentration, drying, and molding process research have also been optimized by our research group.48–52 In addition, we screened 6 active ingredients for VP in FDQ through internet pharmacology, including quercetin, kaempferol, matrine, aloe-emodin, and formononetin, and then the observed effects of these ingredients on animals were compared with FDQ water and ethanol extracts.
In this study, the traditional efficacy of FDQ was selected to observe the relevant indicators of antipyretic, antiinflammatory, antitussive, and phlegm-resolving effects. Among them, PGE2 is the most important medium related to thermoregulation. 53 Similarly, it was discovered that cAMP becomes one of the currently validated central heating media. IL-1β is an important cytokine involved in a variety of pyrogenic fevers. 54 TNF-α is an endogenous pyrogen, which can significantly increase the central mediator of PGE2, and then induce fever in the hypothalamus. Furthermore, it can directly stimulate the thermoregulatory center or indirectly induce macrophages to secrete a large amount of IL-1β, causing fever. 55 MPO remains relatively constant in individual inflammatory cells. Activated neutrophils and macrophages will be released when inflammation occurs.56,57 Therefore, the degree of tissue inflammatory cell infiltration can be determined indirectly by MPO content. 58 In this study, it was found that in the in vivo antipyretic experiment, the contents of PGE2, TNF-α, cAMP, IL-1β, and MPO in the control group were significantly increased, while those of FDQW, FDQA, and AIC groups were significantly reduced when the body temperature decreases. Therefore, it could be speculated that FDQ and AIC can relieve heat through the center, and exert the antipyretic effect by inhibiting the peripheral liver tissue inflammation. IL-10 is an important antiinflammatory factor, which plays a role by inhibiting the release of inflammatory factors from monocytes and various immune cells. From the results of the antitussive experiment, we found that the amount of IL-10 in FDQ and AIC increased significantly, indicating that both FDQ and AIC can inhibit inflammation to a certain extent. The effective ingredients of TCM are not easy to determine because of their complex composition. To ensure the effectiveness of the medicine, the dosage form of the decoction is commonly used. The effective ingredients should be taken together with other noneffective ingredients, resulting in a much larger dosage. Therefore, it cannot be developed into modern dosage forms due to the limitation of drug loading. Network pharmacology is a new discipline based on the basic theories of systems biology which conducts a comprehensive analysis of biological systems and further obtains the specific node with multiple targets. The network analysis based on a wide range of databases enables us to form a preliminary understanding of the mechanism of drugs. The main effective ingredients of FDQ, such as quercetin, kaempferol, matrine, aloe-emodin, and formononetin were screened by network pharmacology. According to the content of the original medicinal materials, the main pharmacodynamics and mechanism of the original prescription were compared by the method of “composition reduction.” The results showed that AIC was equivalent to FDQW and FDQA, which showed that network pharmacology can accurately screen the effective ingredients. The results provided a potential idea and method for the screening and purification of the effective ingredients of TCM. However, our research only predicted and validated 1 disease at present. In the future, other pathogenic mechanisms related to FDQ should be further studied, such as quantitative polymerase chain reaction, Western blot to confirm the pathogenic mechanisms and targets discovered by network pharmacology.
Conclusion
In this study, the main active ingredients in FDQ in the treatment of VP were screened by a network pharmacology method and the drug–disease–target interrelationships and mechanisms were confirmed. The pharmacological activities of FDQ and AIC were verified in vivo. The results showed that pharmacological activity was consistent between the components screened by network pharmacology and FDQ, and contents of PGE2, TNF-α, cAMP, IL-1β, and MPO in rats were suppressed while the content of antiinflammatory factor IL-10 was increased.
Footnotes
Author Contributions
TL contributed to the design of this study and the revision of the manuscript. QL and YLX conducted the experiments. BWL and HY drafted the main manuscript. HBM and ADY performed data analysis. YZH drew the picture in the manuscript. All the authors participated in the interpretation of the results. All the authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Science and Technology Program (No. 2020YJ0477), the National Key R&D Program of China (2017YFC1701900), and the Technology Innovation R&D Project of Chengdu Science and Technology Bureau in 2021 (2021-YF05-00080-SN).
Ethical Approval
This study was approved by the Administration Committee of Experimental Animals, Sichuan Province, China.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care Guidelines of Chengdu University, China and approved by the Administration Committee of Experimental Animals, Sichuan Province, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
