Abstract
Yi-Shen-Hua-Shi (YSHS) Granule is a well-known patented herbal drug for treating chronic glomerulonephritis (CGN) in China. Its chemical compositions and anti-CGN components are not fully understood. This study aimed to establish the chemical profile and to identify the anti-CGN components of YSHS Granule. For these purposes, a HPLC-Q-TOF-MS/MS method was developed. A total of 105 peaks were detected in the mass spectrum of the Granule. Of these, 99 compounds were tentatively identified as terpenoids, flavonoids, coumarins, alkaloids, phenols, and other types of compounds, and 15 were further identified with reference substances. To screen bioactive compounds, a cell membrane immobilized chromatography (CMIC) method was used. Lipopolysaccharide (LPS)-challenged rat glomerular mesangial cells (HBZY-1) were incubated with YSHS Granule (100 μg/mL), and the binding components to the cell membrane were extracted and analyzed using the established HPLC-Q-TOF-MS/MS method. Seven potential bioactive components that bound to HBZY-1 cell membranes were detected and identified as calycosin-7-O-β-D-glucoside, 6-gingerol, naringin, ginsenoside Re, poncirin, liquiritigenin, and isoliquiritigenin. The outcomes of the present study provide a chemical basis for clinical use of the Granule in managing CGN, and provide quality control markers for the Granule’s production and use.
Keywords
Chronic glomerulonephritis (CGN), the most common form of glomerular disease, accounts for about 20% of chronic kidney disease (CKD) cases worldwide. 1 It is pathologically characterized by varying degrees of glomerular injury, and can lead to many complications, including metabolic acidosis, anemia, mineral disorders, and cardiovascular diseases. The pivotal clinical feature of CGN is the presence of urinary abnormalities such as albuminuria or proteinuria. High serum creatinine and blood urea nitrogen levels are also found in CGN patients. 2 If not cured at an early stage, CGN may progress to end-stage renal disease which is fatal and requires dialysis or renal replacement therapy. Nowadays, there is no good chemotherapeutic agent for managing this complex condition. 3 The herbal drugs of traditional Chinese medicine (TCM) have advantages in treating complex diseases because of their multi-component and multi-target nature; hence, it is reasonable to expect that TCM herbs could be valuable in the treatment of CGN.
Yi-Shen-Hua-Shi (YSHS) Granule is a China National Medical Products Administration-approved herbal drug. It is manufactured by the Guangzhou Consun Pharmaceuticals Co., Ltd. YSHS Granule was derived from a TCM formula, Sheng-Yang-Yi-Wei decoction, that was first documented in the TCM classic Nei-Wai-Shang-Bian-Huo-Lun written in 1247 AD. The Granule is composed of 16 herbs, namely: Ginseng Radix et Rhizoma (GRR), Astragali Radix (ASR), Atractylodis Macrocephalae Rhizoma (AMR), Poria (POR), Alismatis Rhizoma (ALR), Pinelliae Rhizoma Praeparatum Cum Alumine (PRP), Notopterygii Rhizoma et Radix (NRR), Angelicae Pubescentis Radix (APR), Saposhnikoviae Radix (SAR), Bupleuri Radix (BUR), Coptidis Rhizoma (COR), Paeoniae Radix Alba (PRA), Citri Reticulatae Pericarpium (CRP), Glycyrrhizae Radix et Rhizoma Praeparata Cum Melle (GRP), Zingiberis Rhizoma Recens (ZRR) and Jujubae Fructus (JUF). 4,5
Clinical data have demonstrated that YSHS Granule is able to improve inflammation and oxidative stress conditions of CKD patients. 6 A randomized controlled clinical study showed that YSHS Granule combined with losartan potassium has better efficacy and fewer adverse reactions than losartan potassium monotherapy in treating CGN. 7 Hu et al. found that the combination of YSHS Granule and sulodexide injection significantly delays the progression of diabetic nephropathy in patients. 4 Liu et al. demonstrated that YSHS Granule in combination with calcitriol significantly improves renal functions of diabetic nephropathy patients. 8 In a previous study, we found that YSHS Granule has therapeutic effects in a rat model of CGN, and inhibition of the TGFbeta/Smad signaling pathway is one of the mechanisms of action of the Granule. 5
Although the pharmacological effects and clinical efficacy of YSHS Granule have been recognized, its chemical compositions and anti-CGN components are not fully known, which impedes upgrading its quality control methods and restricts understanding the chemical basis of its clinical use.
High performance liquid chromatography coupled with electrospray ionization tandem quadrupole time-of-fight mass spectrometry (HPLC-Q-TOF/MS) is a powerful technique for online structural elucidation of herbal medicines, owing to its characteristics of high resolution, accurate mass measurement and remarkable sensitivity. 9 In this study, we aimed to establish the chemical profile and to identify the anti-CGN compounds of YSHS Granule using this method.
Modern pharmacological studies have demonstrated that binding to specific receptors or channels on the cell membrane is the first step of drug action. 10 Cell membrane immobilized chromatography (CMIC) has been applied to screen active compounds from herbal drugs. 11,12 In this study, CMIC was used to screen and identify the components of YSHS Granule that bind to HBZY-1 cell membranes after the cells had been treated with YSHS Granule.
Materials and Methods
Herbal Materials and Reagents
YSHS Granule was provided by Guangzhou Consun Pharmaceuticals Co., Ltd. (Batch no. 20180201). Acetonitrile was of LC/MS grade (Fisher Scientific, Pittsburgh, PA, USA). Formic acid was of HPLC grade (Sigma-Aldrich, St. Louis, MO, USA). Ultra-pure water was prepared using a Milli-Q Plus water purification system (Millipore, Billerica, MA, USA). Other reagents used for sample preparation were of analytical grade. Reference samples of caffeic acid, albiflorin, paeoniflorin, liquiritin, nodakenin, hesperidin, naringin, calycosin, liquiritigenin, saikosaponin A, isoliquiritigenin, saikosaponin D, astragaloside I, astragaloside II, ginsenoside Rg6, berberine, pachymic acid, and calycosin-7-O-β-D-glucoside were supplied by Chengdu Preferred Biotechnology Co., Ltd. (Chengdu, China). The purity of these compounds was determined to be above 98% by HPLC-DAD methods.
Dimethyl sulfoxide (DMSO) was purchased from Sigma Chemicals Ltd. (St. Louis, MO, USA), and penicillin, streptomycin, Dulbecco’s Modified Eagle Medium (DMEM), lipopolysaccharide and fetal bovine serum (FBS) from Hyclone (Logan, UT, USA).
Preparation of Sample Solutions
To prepare the sample solution for chemical profile establishment, YSHS Granule samples (0.5 g) were accurately weighed and extracted with 50% methanol-water (v/v) solution (45 ml) in an ultrasonic water bath for 20 minutes at room temperature. The extract was centrifuged at 4,000 g for 15 minutes. The supernatant was transferred to a 100 ml volumetric flask and adjusted to 100 ml with 50% methanol-water (v/v) solution. Before analyses, the sample was filtered through a 0.22 µm pore-size nylon membrane filter.
For bioassays, the extraction method was the same as that for chemical profile establishment. After the extract was centrifuged, the supernatant was transferred and concentrated under reduced pressure. Then, the concentrated extract was frozen at −80 °C, and freeze-dried (VirTis Freezemobile, Virtis Co., Gardiner, NY, USA). The freeze-dried extract was dissolved in DMSO, diluted with DMEM to a concentration of 100 µg/mL and filtered through a 0.22 µm pore-size nylon membrane filter before use.
HPLC Conditions
Liquid chromatography was performed on an Agilent 1260 series system coupled with an Agilent ZORBAX C18 column (4.6 × 250 mm, 5 µm). The mobile phase consisted of solvent A (0.1% formic acid in Milli-Q water) and solvent B (0.1% formic acid acetonitrile), and the gradient elution conditions were: 0‐10 min, 90% A; 10‐25 min, 90 to 82% A; 25‐40 min, 82 to 80% A; 40‐50 min, 80 to 75% A; 50‐80 min, 75 to 60% A; 80‐100 min, 60 to 20% A. The flow rate was 1 mL/min. The column temperature was 30 °C, the injection volume 10 ml, and the detection wavelength 203 nm.
Mass Spectrometry Conditions
Mass spectrometric detection was carried out on an Agilent 6540 Q-TOF mass spectrometer (Hewlett Packard, Agilent, USA) with electrospray ionization (ESI) interface. Positive and negative ion modes were used with the mass range setting at m/z 100‐1700. Ultra-high-purity nitrogen (N2) was used as collision gas in product ion scanning experiments. Ultra-pure N2 was used as the nebulizing and sheath gas.
Cell Culture and Treatment
Rat mesangial cell line HBZY-1 was purchased from American Type Culture Collection (Manassas, VA, USA) and maintained in 5.5 mM glucose DMEM containing 10% FBS and 1% penicillin/streptomycin at 37 °C in a humidified atmosphere of 5% CO2. The cells were seeded on a 24-well plate and allowed to adhere overnight.
Glomerular mesangial cell (GMC) proliferation is one of the pathogenic factors of CGN. Lipopolysaccharide (LPS) stimulation can induce GMC proliferation, causing inflammation and oxidative stress that favor CGN development. 13 Thus, LPS was used to induce HBZY-1 cell proliferation. After the cells had been pretreated with serum-free DMEM medium containing 100 µg/mL of LPS for 24 hours, the medium was removed and 1.5 ml of YSHS Granule DMEM solution (100 µg/mL) was added and incubated for 1 hour. These cells were washed with 1.5 ml of PBS (pH 7.4) 6 times. The last PBS eluate was collected as control for analysis. Finally, 1.5 ml of 10% citric acid (pH 4.0) was added to dissociate the binding components. The last PBS eluate and desorption eluate were frozen at −80 °C and evaporated to dryness under vacuum. The residue was dissolved in 1 ml of methanol and centrifuged at 11,000 g for 10 minutes. The resulting supernatant was used for HPLC-Q-TOF/MS analysis.
Data Analysis
Compound identification was based on the accurate mass of quasi-molecular ions and MS/MS fragmentation patterns. A compound library was established by retrieving and summarizing previous publications on compounds from all the 16 herbs of YSHS Granule. The library recorded compound names, molecular formulas, accurate molecular weights, characteristic fragment ions, chemical structures and relevant references. Data were collected with LC-MS-Q-TOF MassHunter Data Acquisition Software Ver. A.01.00 (Agilent Technologies) and analyzed with Agilent Mass Hunter Qualitative Analysis Software B.06.00.
Results and Discussion
Identification of Chemical Compounds
Reference standards, structure cleavage characterizations, and literature, as well as Q-TOF-MS and MS/MS data analyses were employed for structural characterization of constituents in YSHS Granule. Under the optimized conditions, among 105 detected peaks, 99 compounds, structurally belonging to terpenoids, flavonoids, coumarins, alkaloids, phenols and other types of compounds, were either positively identified or tentatively characterized (Figure 1).

Chemical structures of compounds identified in YSHS Granule.
The total ion chromatograms of the YSHS Granule sample and chemical standards in positive and negative ion modes are shown in Figure 2. Fifteen compounds were unambiguously identified by comparing retention times, precursor ions, UV spectra and MS/MS fragment ions with reference standards (Supplemental Figure S1), while others were tentatively identified based on accurate mass, MS/MS spectra and comparison with literature data. The mass accuracy for molecular ions of all identified compounds was within ± 10 ppm. Information about the precursor and product ions, error of mass accuracy retention time and structural class of the compounds is shown in Table 1.

ESI total ion chromatograms of the YSHS Granule sample in positive (
Identification of Chemical Constituents of YSHS Granule by HPLC-MS/MS in Positive and Negative Modes.

Abbreviations: AKD, Alkaloid; CMN, Coumarin; DHIF, Dihydroisoflavone; FVN, Flavonoid; Hex, Hexose; MTP, Monoterpenoid; ND, not detected; OT, other type; PNL, Phenol; TPN, Triterpenoid.
Peaks identified with reference standards are marked with *.
Identification of terpenoids
Thirty-seven terpenoids were either tentatively or unambiguously identified in YSHS Granule. Of these, 19 ginsenosides were from GRR, 4 monoterpenoids from PRA and 14 triterpenoids from GRP, ASR, ALR, BUR, and JUF. As for the ginsenosides, simultaneous or successive losses of sugar units were commonly observed in the negative ion mode. 14 Ginsenoside Re was used as an example to illustrate the fragmentation pathway of ginsenosides in general (Figure 3(A)). Peak 55 displayed [M-H]- ions at m/z 945.5504. The main fragment ions, including those at m/z 799 [M-H-Rha]-, 637 [M-H-Rha-Glu]-, and 475 [M-H-Rha-2Glu]-, were observed in MS/MS spectra. By comparing the MS and MS/MS information with the literature data, 14,15 we tentatively identified this compound as ginsenoside Re.
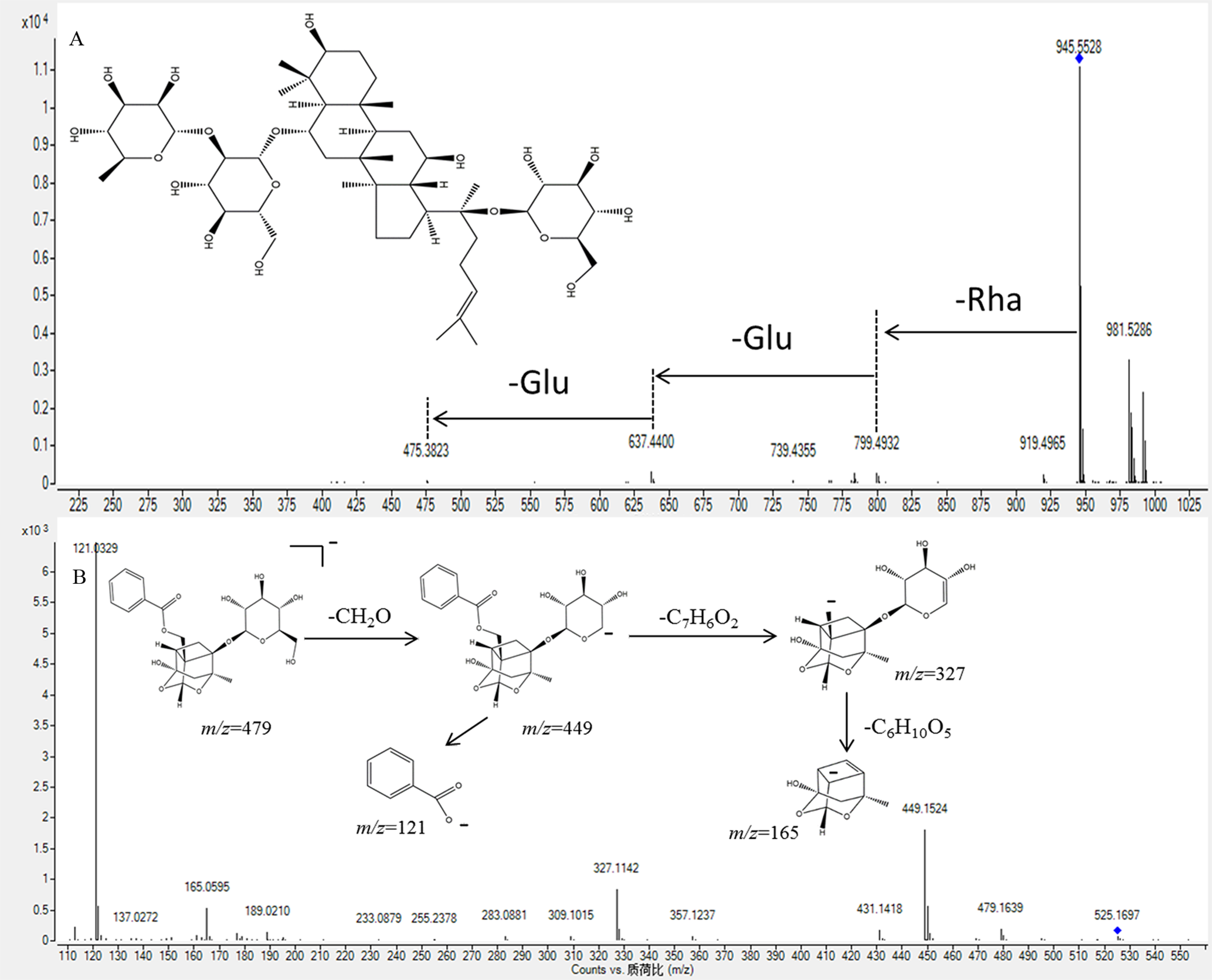
MS/MS spectra and possible fragment pathways of ginsenoside Re (
Four monoterpenoids derived from PRA were identified. They display molecular ions [M + H]+ and [M + Na]+ in positive ion mode, and [M-H]- and [M + HCOO]- in negative ion mode. Albiflorin was unambiguously identified by comparing its retention time, and MS and MS/MS spectral data with a reference sample. The tandem mass spectra and possible fragmentation pathways of paeoniflorin are illustrated in Figure 3B. Peak 22 gave [M + HCOO]- and [M-H]- ions at m/z 525.1697 and 479.1639, respectively. The fragment ions at m/z 449, 327 and 165 were attributed to the loss of a formaldehyde unit, a benzoic acid and a glucosyl, respectively. So, it was tentatively identified as paeoniflorin. 16 Peak 42 possessed a [M + H]+ ion at m/z 633.1797, which is 152 Da greater than that of paeoniflorin. Its fragment ion at m/z 313 is presumed to have been generated by a galloyl glucose residue, so it is tentatively identified as galloylpaeoniflorin.
Five triterpenoids were detected and identified from GRP. The main fragmentation pattern for these triterpenoids was the cleavage at the glycosidic linkage due to the loss of glucuronic acid (GluA, 176 Da). 17,18 Peak 70 showed a [M-H]- ion at m/z 821.4023 in negative ion mode, and its MS/MS spectral ions were at m/z 759 [M-H-CO-H2O]-, 645 [M-H-GluA]-, and 351 [2GluA-H]-. Peak 69 was tentatively identified as glycyrrhizic acid by comparison with the data of previous studies. 19 Peaks 49, 63, 64, and 66 displayed [M-H]- ions at m/z 823.4122, 819.3883, 837.3914, and 863.4071 in negative ion mode, respectively. They showed a similar fragmentation pattern to that of peak 69. Peaks 48, 62, 63, and 65 were tentatively identified as licorice saponin J2, licorice saponin E2, licorice saponin G2 and 22β-acetoxy licorice saponin C2 by comparing the characteristic molecular ions and MS/MS fragment information with literature data. 20 Other triterpenoids were tentatively characterized based on MS and MS/MS spectra from available literature data. 19,21
Identification of flavonoids
Twenty-eight flavonoids, including isoflavone, flavanones, flavanonol and chalcones, were either tentatively or unambiguously characterized.
Calycosin was identified by comparing the retention time, molecular ion and fragment ions with a reference standard. Peak 31 showed a [M + H]+ ion at m/z 447.1282, 162 Da more than that of calycosin. It also showed fragment ions at m/z 270 [M + H-Glu-CH3]+ and 213 [M + H-Glu-CH3-CHO-CO]+. All these data indicate that Peak 31 could be calycosin-7-O-β-D-glucoside. 22 Peak 49 provided a [M + H]+ ion at m/z 431.1331, and fragment ions at m/z 269 and 254, corresponding to the loss of glucosyl (-Glu/−162 Da) and a side chain radical (-CH3/−15 Da). The Retro-Diels-Alder (RDA) fragment ions at m/z 137 and 134 could also be found in positive ion mode of Peak 49. So, it is tentatively identified as ononin. 22 Peak 50 displayed a [M + H]+ ion at m/z 269.0814 in positive ion mode. In the MS/MS spectrum, Peak 50 yielded fragment ions at m/z 254, 137 and 134, which are similar to the fragmentation ions of peak 49 (except for the loss of glucosyl). Thus, peak 50 is tentatively identified as formononetin, the aglycone of Peak 49. 22
Peak 34 displayed a [M + H]+ ion at m/z 307.1185 in the positive ion mode. The main fragment ions of peak 34 were observed at m/z 289 [M + H-H2O]+, 259 [M + H-H2O-2CH3]+, 235 [M + H-C4H8O]+, 221 [M + H-H2O-2CH3-C3H2]+, and 205 [M + H-C4H8O-CH2O]+. Comparing with the literature, 23 peak 34 was tentatively characterized as cimifugin. Figure 4A shows the possible fragmentation pathway of cimifugin in YSHS Granule. Peaks 24, 41 and 58 gave [M + H]+ ions at m/z 469.171 453.1765 and 291.1233, respectively, and they demonstrate MS/MS spectra similar to that of peak 34. Furthermore, peak 24 and 41 showed the loss of glucosyl (-Glc/−162 Da). Therefore, peaks 24, 41 and 58 were characterized as prim-O-glucosylcimifugin, 5-O-methylvisammioside and 5-O-methylvisamminol, respectively. 24
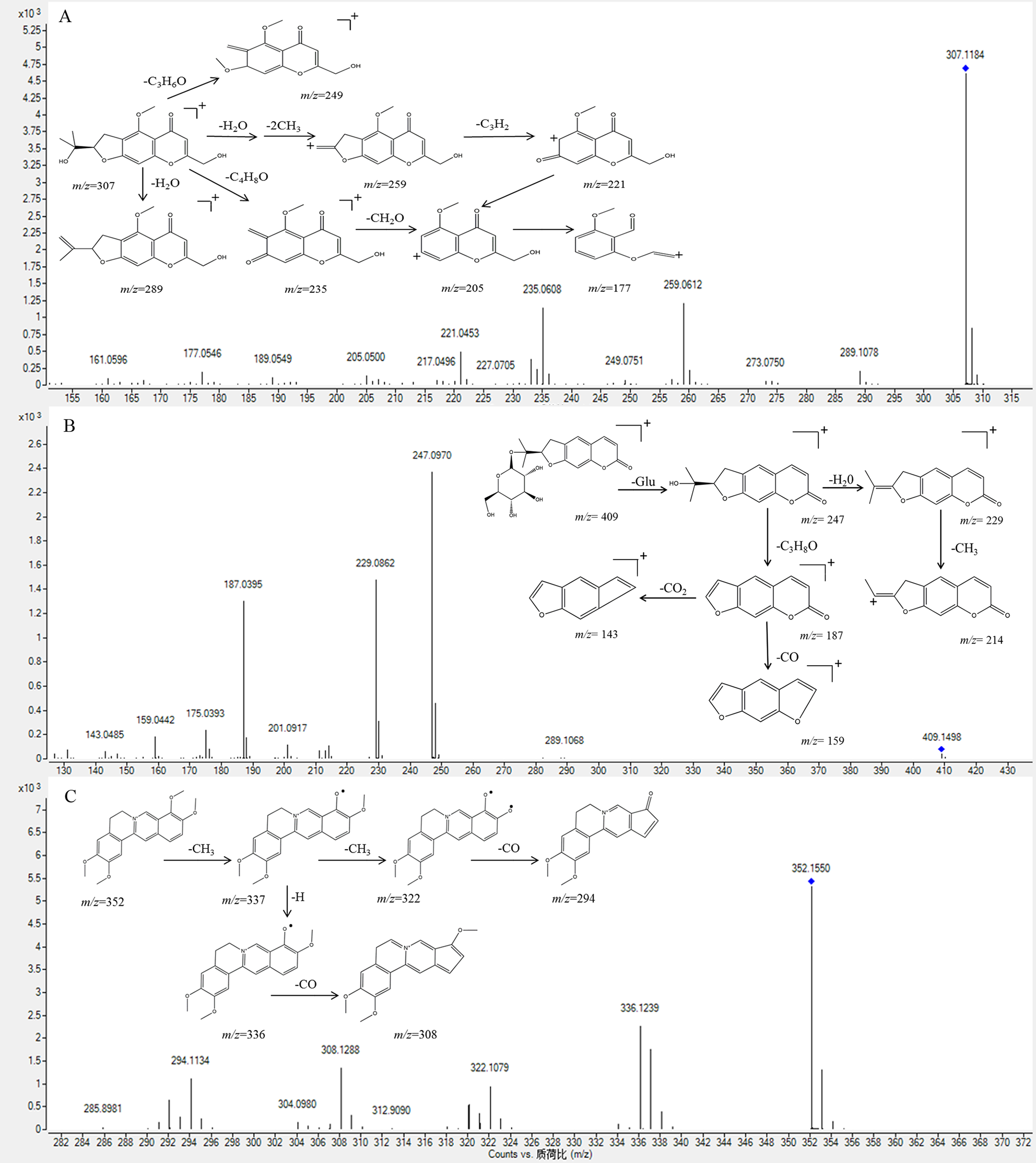
MS/MS spectra and possible fragment pathways of cimifugin (
Peak 38 provided a [M-H]- ion at m/z 417.1175 in the negative ion mode and a [M + Na]+ ion at m/z 441.1159 in the positive ion mode. The MS/MS spectrum showed a fragment ion at m/z 255 [M-H-Glu]-. We also observed that peak 38 underwent RDA fragmentation and displayed fragment ions at m/z 135 [C7H4O3-H]- and 119 [C7H5O3-H]-. According to the published MS data, 20 peak 38 was tentatively characterized as liquiritin. Peak 37 gave [M-H]- and [M + H]+ ions at m/z 549.1682 and 551.1749 in negative and positive ion modes, respectively. MS/MS fragment ions were observed at m/z 417 [M-H-C5H10O4]- and 255 [M-H-C5H10O4-Glu]- in negative ion mode and m/z 419 [M + H-C5H10O4]+ and 257 [M + H-C5H10O4-Glu]+ in positive ion mode. Peak 37 was tentatively identified as liquiritin apioside based on this information and the literature. 19 Peaks 56 and 59 gave [M-H]- ions at m/z 549.1656 and 417.1181, and showed the same MS/MS patterns as liquiritin apioside and liquiritin, so they were tentatively identified as isoliquiritin apioside and isoliquiritin, 19 respectively.
Identification of coumarins
Thirteen coumarins were identified in YSHS Granule, which are from NRR, APR, and SAR. Coumarins have similar fragmentation pathways of successive or simultaneous loss of H2O (18 Da), CO (28 Da) and CO2 (44 Da). 21,23,25 In this study, coumarins showed [M + H]+ and [M + Na]+ ions in positive mode and [M-H]- ions in negative mode. The [M + H]+ ion of peak 40 at m/z 409.1495 produced a fragment ion at m/z 247 in the MS/MS spectrum, which originated from the loss of a glucosyl (162 Da). In addition, a series of fragment ions at m/z 229, 187, 159, and 143 were identified as product ions [M + H-Glu-H2O]+, [M + H-Glu-C3H8O]+, [M + H-Glu-C3H8O-CO]+, and [M + H-Glu-C3H8O-CO2]+, respectively. This coumarin was tentatively characterized as nodakenin by comparing its molecular mass and MS/MS spectral data with that in the literature. 20,26 The fragmentation pathway of peak 40, representative of coumarins, is shown in Figure 4(B). The other 12 coumarins were tentatively assigned on the basis of characteristic fragment ions and the literature.
Identification of alkaloids
Eleven alkaloids were identified in positive mode.
Peaks 52, 70, 86, 94 and 97 are all from COR. These alkaloids exhibited [M]+ precursor ions, and the MS/MS fragmentation of these alkaloids showed successive losses of CO (28 Da), CH4 (16 Da) and methyl (15 Da). As an example, the precise molecular weight of peak 86 (Figure 4(C)) is 352.1550, and the main fragment ions on MS/MS screening were observed at m/z 337 [M-CH3]+, 336 [M-CH4]+, 322 [M-2CH3]+, 308 [M-CH4-CO]+, and 294 [M-2CH3-CO]+. According to the above precursor ions and fragmentation patterns, peak 86 was tentatively identified as palmatine. By comparing the characteristic accurate mass and fragment ions with data in the literature, peaks 52, 70, 94 and 97 were deduced to be demethyleneberberine, jatrorrhizine, berberine and 13-methylberberine, 27 respectively.
Peak 3 gave a [M + H]+ ion at m/z 138.0551 and a MS/MS fragment ion at m/z 109 [M + H-CH3N]+. By comparison with previously reported data, 28 peak 3 was tentatively characterized as trigonelline.
Identification of phenols and other types of compounds
Peaks 11 and 12 are 2 isomers with [M-H]- ions at m/z 153.0203 and 153.0199, respectively. Their MS/MS fragment ions appeared at m/z 135 [M-H-H2O]-, and 109 [M-H-H2O]-. These fragmentation pathways, together with literature data, 29 indicate that peaks 11 and 12 are protocatechuic acid and its isomer, respectively.
Peaks 2, 6, 7, 13 and 102 are tentatively identified on the basis of their exact masses and MS/MS fragmentation patterns in the literature. 25,30,31
Potential Active Compounds Screening and Identification
The last PBS eluate and the desorption eluate were analyzed using the HPLC-Q-TOF-MS method as described in section 3.1. Theoretically, peaks of the potential active compounds observed in the desorption eluate should not be detected in the chromatogram of the last PBS eluate, but should appear in that of YSHS Granule. As shown in Figure 5(C) and (D), after comparing the total ion chromatograms of the last PBS eluate (Figure 5) and the Granule (Figure 2(A) and (B)), 7 common peaks were found. The compounds corresponding to the 7 peaks are the components of YSHS Granule that are, potentially, active in reversing HBZY-1 cell dysfunction. The 7 peaks were identified as calycosin-7-O-β-D-glucoside (31), 6-gingerol (33), naringin (44), ginsenoside Re (55), poncirin (60), liquiritigenin (67) and isoliquiritigenin (83) (Table 2). All 7 compounds can be found in the chemical profile established in section 3.1.

ESI total ion chromatograms of the last PBS eluate in positive (
Characterization of Potential Bioactive Compounds From YSHS Granule by HPLC-MS/MS in Positive and Negative Ion Modes.

Abbreviation: ND, not detected.
Calycosin-7-O-β-d-glucoside, one of the most abundant flavonoids of ASR (the principal herb of the formula), has many pharmacological activities, including anti-inflammatory, anticancer and cardioprotective properties. 32 It was reported that calycosin-7-O-β-d-glucoside can inhibit advanced glycation end product (AGEs)-induced human glomerular endothelial cell apoptosis and high glucose-induced rat mesangial cell proliferation. 33
6-Gingerol has been reported as an important active constituent of ginger and has various biological effects, including antioxidant, anti-inflammatory and immunomodulatory activities. 34 A study showed that 6-gingerol has protective effects against carbendazim-mediated hematotoxicity and prevents oxidative damage in the kidney of rats. 35
Naringin and poncirin from CRP are flavonoid glycosides. They are also abundant in the peel of oranges and grapefruits. 36 A study showed that naringin significantly reduces collagen deposition and renal interstitial fibrosis in streptozotocin (STZ)-induced diabetic rats through inhibiting the NF-κB signaling pathway, and inhibits high glucose-induced oxidative stress injury in HBZY-1 cells by activating the Nrf2 antioxidant pathway. 37 The role of poncirin in nephropathy has not been reported.
Ginsenoside Re is one of the saponins of ginseng. Wang et al. found that this compound exerts a protective effect on renal function in a mouse model of nephrotoxicity induced by cisplatin. That study also showed that pretreatment with ginsenoside Re significantly reduces cisplatin-induced oxidative stress, inflammation, and apoptosis. 38
Liquiritigenin and isoliquiritigenin are flavonoids. 39 They have various pharmacological properties, such as anti-inflammatory, antioxidant and detoxification effects. Zhu et al. found that liquiritigenin can attenuate high glucose-induced mesangial matrix accumulation, inflammation and oxidative stress in HBZY cells by suppressing the NFκB and NLRP3 inflammasome signaling pathways. 40 Li et al. found that isoliquiritigenin significantly reduces fibrosis in human renal mesangial cells by inhibiting the TGF-β signaling pathway. Another study showed that isoliquiritigenin alleviates chronic kidney injury, renal inflammation and fibrosis in a mouse model. 41
All of these compounds, except poncirin, have been reported to have bioactivities beneficial for treating CGN. Poncirin, whose structure is similar to that of naringin, needs further study to determine its pharmacological effects related to CGN management.
Conclusions
In the chemical profiling, 105 peaks were detected, and 99 compounds were identified based on an established HPLC–Q-TOF-MS/MS method. These compounds include 37 terpenoids, 28 flavonoids, 13 coumarins, 11 alkaloids, 5 phenols and 5 other types of compounds. Of the 99, 15 were unambiguously identified. In the screening for bioactive components, 7 potentially bioactive components were detected; they were identified as: calycosin-7-O-β-D-glucoside, 6-gingerol, naringin, ginsenoside Re, liquiritigenin, isoliquiritigenin and poncirin. The first 6 compounds have been reported to have anti-CGN related pharmacological effects.
Establishment of the chemical profile should help in the identification of bioactive and/or toxic components that can be used as quantitative assay markers. The outcomes of the CMIC provide a chemical basis for clinical use of the Granule in managing CGN, and provide quality control markers for the Granule’s production and use.
Supplemental Material
Online supplementary file 1 - Supplemental material for Chemical Profile Assessment and Potential Bioactive Component Screening of a Chinese Patent Herbal Drug Yi-Shen-Hua-Shi Granule
Supplemental material, Online supplementary file 1, for Chemical Profile Assessment and Potential Bioactive Component Screening of a Chinese Patent Herbal Drug Yi-Shen-Hua-Shi Granule by Chan Yuen-Cheung, Zhao Jing, Hu Qin, Guo Hui, Liu Yu-Xi, Wu Jia-Ying, Zheng Zhao-Guang and Yu Zhi-Ling in Natural Product Communications
Footnotes
Acknowledgments
This research was supported by Consun Pharmaceutical Co., Ltd.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship,and/or publication of this article: This research was supported by Science and Technology Key Project of Covid-19 in Foshan City (grant No.: 2020001000206) and the Innovation and Technology Commission of Hong Kong (grant No.: UIM/290).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
