Abstract
Coumarin derivatives, such as esculetin, have various physiological functions, including antioxidant, anti-inflammatory, antibacterial, antiviral, and anti-cancer. 6,7-Dihydroxy-4-methylcoumarin (6,7-DH-4MC) is a derivative of esculetin, and its anti-inflammatory effect and mechanism in macrophages have not been studied. In this study, the anti-inflammatory activity of 6,7-DH-4MC was evaluated by measuring the expression of inflammatory factors (NO and PGE2) and pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) in LPS-stimulated RAW 264.7 macrophages. The results revealed that 6,7-DH-4MC significantly reduced NO levels and PGE2 expression without inducing cytotoxicity; it was confirmed that the inhibition of NO and PGE2 expression was related to iNOS and COX-2 downregulation in response to 6,7-DH-4MC treatment. Moreover, 6,7-DH-4MC decreased the levels of pro-inflammatory cytokines, such as IL-1β and IL-6, in a dose-dependent manner. Mechanistic studies revealed reduced phosphorylation of ERK and p38-MAPK upon 6,7-DH-4MC treatment. Furthermore, the degradation of IκB-α and phosphorylation of NF-κB in cells treated with LPS were interrupted by 6,7-DH-4MC treatment. These results suggest that 6,7-DH-4MC is a potential therapeutic agent for inflammatory diseases. To the best of our knowledge, this is the first report demonstrating the anti-inflammatory effects of 6,7-DH-4MC in RAW 264.7 cells via MAPK and NF-κB signaling pathways.
Inflammation, the primary immune response to a physical or chemical stimulus, such as a wound or bacterial infection, regenerates damaged tissue and protects the body from infection. However, these inflammatory reactions cause elevated blood flow, increased body temperature, flushing, swelling, and pain. Furthermore, persistent inflammatory reactions have been shown to cause chronic inflammatory diseases and tumors. 1 -4
Macrophages are immune cells that are well-known for their roles in inflammatory responses. They are activated by cytokines secreted by immune cells as well as various other stimuli, resulting in the production of pro-inflammatory cytokines (interleukin-1β, IL-1β; interleukin-6, IL-6; tumor necrosis factor-α, TNF-α) and inflammatory factors (nitric oxide, NO; prostaglandin E2, PGE2). 5 -7 Nitric oxide synthase (NOS) exists in 3 similar forms: neuronal NOS (nNOS), endothelial NOS (eNOS), and inducible NOS (iNOS). 8 Among these, iNOS has been reported to promote NO production by being expressed upon stimulation with interferon-γ, lipopolysaccharide (LPS), and pro-inflammatory cytokines. 9,10 NO plays an important role in the physiological transmission of the nervous signals, inflammatory response, cell differentiation, and intracellular signal transmission. Furthermore, it is reported to be critically involved in various physiological functions, such as vascular relaxation, inhibition of platelet aggregation, and immunity. 11 However, NO is a free radical and toxic in vivo. In particular, the overproduction of NO owing to persistent inflammation is associated with rheumatoid arthritis and cancer. 12,13
Another representative inflammatory response factor, PGE2, is the product of arachidonic acid metabolism as a result of the catalytic action of cyclooxygenase-2 (COX-2). PGE2 has been reported to be involved in tumorigenesis by inhibiting tumor apoptosis and inducing angiogenesis. Macrophages stimulated by LPS and cytokines, such as interleukin-1, overexpress COX-2 and consequently upregulate PGE2 production, thereby maintaining inflammation. 14,15
The mitogen-activated protein kinase (MAPK) and nuclear factor-κB (NF-κB) signaling pathways regulate the expression of inflammatory factors after LPS stimulation in macrophages, such as RAW 264.7 cells. MAPK family members, such as extracellular signal regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38, act as regulators of signals induced by cytokines, growth factors, and environmental stress and are involved in various cellular functions. In addition, it has been reported that the increased phosphorylation of MAPKs in macrophages increases the inflammatory response by controlling the synthesis of inflammatory mediators at the transcriptional and translational levels. 16 -20
Nuclear factor kappa B (NF-κB), generally bound to inhibitory factor kappa B alpha (IκB-α), is present in the cytoplasm in an inactive state. Upon LPS stimulation, the complex dissociates owing to phosphorylation and decomposition of IκB-α, and NF-κB is released into the cytoplasm. The released NF-κB is phosphorylated and migrates into the nucleus, where it acts as a transcription factor that promotes the synthesis of iNOS, COX-2, and inflammatory cytokines. Therefore, the inhibition of NF-κB and MAPK signaling pathways can serve as a potential strategy for the inhibition of iNOS and COX-2, which induce the expression of NO and PGE2. This approach can, thus, inhibit the secretion of pro-inflammatory cytokines owing to NO and PGE2 expression and trigger anti-inflammatory effects. 21 -26
Coumarin is synthesized from shikimic acid pathways in bacteria, fungi, and plants. Its chemical structure is derived through the hydroxylation, saccharification, and cyclization of cinnamic acid; multiple variants of coumarin derivatives have also been reported. These include umbelliferone, esculetin, herniarin, psoralen, and imperatorin. Furthermore, studies have reported various physiological functions of these coumarin derivatives, including antioxidant, anti-inflammatory, antibacterial, antiviral, and anti-cancer effects. 27 -32
6,7-Dihydroxy-4-methylcoumarin (6,7-DH-4MC; Figure 1) is a derivative of esculetin, which has been well-documented for its antioxidant and anti-inflammatory effects in the intestine. 33 However, few reports are available on the anti-inflammatory effects and mechanisms of action of 6,7-DH-4MC using the RAW 264.7 macrophage cell model. To the best of our knowledge, this study is the first to investigate the role of MAPK and NF-κB signal transduction pathways in mediating the anti-inflammatory effects of 6,7-DH-4MC.

Chemical structure of 6,7-dihydroxy-4-methylcoumarin.
Results and Discussion
Measurement of Cell Viability
To assess the non-toxicity of 6,7-DH-4MC at various doses, LPS-stimulated RAW 264.7 macrophages were treated with various concentrations of 6,7-DH-4MC, and a 3-(4,5-dimethylthiazol-2-yl)−2,5-diphenyltetrazolium bromide (MTT) assay was performed. 6,7-DH-4MC treatment had no cytotoxic effect on the macrophage cells at concentrations ranging from 5 to 40 µM compared with the untreated control cells (Figure 2(A)). Consequently, these 6,7-DH-4MC doses were used in the subsequent experiments.
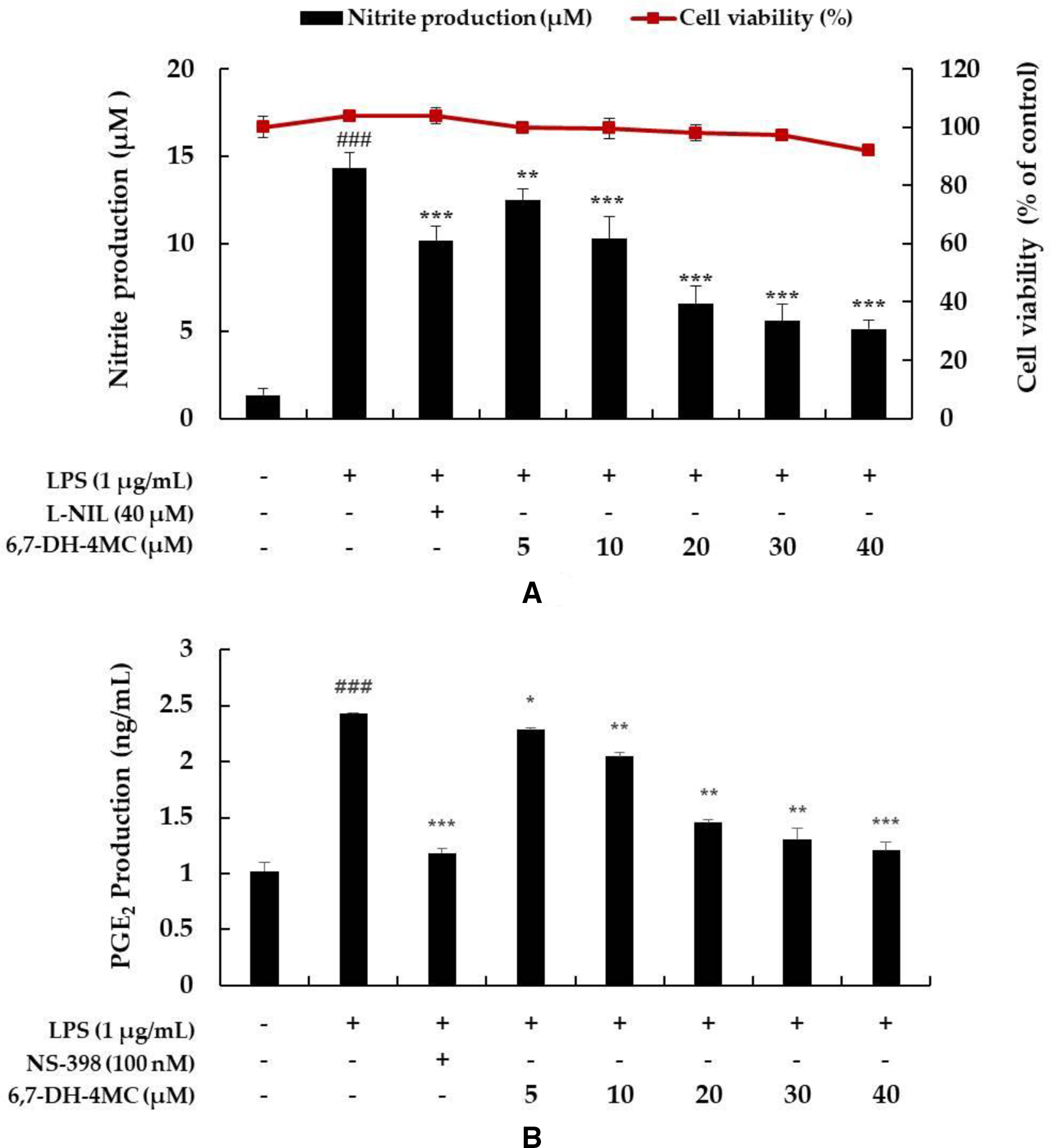
Effect of 6,7-dihydroxy-4-methylcoumarin (6,7-DH-4MC) on the viability and NO and PGE2 production in LPS-induced RAW 264.7 cells. Cells were pretreated with 6,7-DH-4MC for 1 hour followed by LPS stimulation for 24 hours. (A) Viability of LPS-induced RAW 264.7 cells subjected to 6,7-DH-4MC treatment was evaluated using an MTT assay. The amount of (A) NO and (B) PGE2 in the medium was measured using Griess reagent and an ELISA kit, respectively. L-N6-(1-Iminoethyl)lysine dihydrochloride (L-NIL) and N-[2-cyclohexyloxy-4-nitrophenyl] methane sulfonamide (NS-398) were used as a positive control for NO and PGE2 production, respectively. The results are presented as the mean ± SD from 3 independent experiments. ###P < 0.001 vs untreated control group. *P < 0.05, **P < 0.01, ***P < 0.001 vs group treated with LPS alone.
Effect of 6,7-DH-4MC on NO and PGE2 Production
A recent study reported that inflammation induces the expression of iNOS, an NO synthesis enzyme that causes NO free radicals to react with oxygen to produce peroxynitrite, thereby increasing NO production. 34 Therefore, to determine the effect of 6,7-DH-4MC on NO production, LPS-stimulated RAW 264.7 cells were treated with different concentrations of 6,7-DH-4MC (5, 10, 20, 30, and 40 µM) and NO production was determined. NO production was 8.5-fold higher in cells stimulated with LPS alone than in untreated control cells. However, N-[2-(cyclohexyloxy)−4-nitrophenyl] methanesulfonamide (NS-398; 100 nM) and 6,7-DH-4MC (5, 10, 20, 30, and 40 µM) reduced NO production by 30.3%, 15.2%, 27.6%, 53.1%, 59.3%, and 64.8%, respectively, compared with LPS-treated cells (Figure 2(A)). The results of this study suggest that 6,7-DH-4MC treatment considerably reduces the inflammatory response, cytotoxicity, and harmful factors affecting macrophages, as NO activity was significantly inhibited in the 6,7-DH-4MC treatment groups compared with the group treated with LPS alone. Similarly, the inhibition of PGE2 production in macrophage cells was confirmed using the PGE2 ELISA kit upon 6,7-DH-4MC treatment. The results revealed that 6,7-DH-4MC suppressed the production of PGE2 by 6.25%, 12.5%, 39.6%, 47.9%, and 50.8% at 5, 10, 20, 30, and 40 µM, respectively, compared with LPS alone (Figure 2(B)).
Measurement of iNOS and COX-2 Protein Expression
Previous studies have reported that inflammatory factor (NO and PGE2) production is related to the expression of iNOS and COX-2 at the protein level. 9,10,14,15 Therefore, a Western blot assay was performed to confirm whether the inhibition of NO and PGE2 by 6,7-DH-4MC was associated with the expression of iNOS and COX-2. iNOS was 29.1-fold higher in the cells stimulated with LPS alone than in the untreated control cells; however, in the presence of 6,7-DH-4MC (5, 10, and 20 µM), iNOS production was reduced by 6.9%, 35.6%, and 51.7%, respectively, compared with LPS treatment alone (Figure 3(A)). In addition, COX-2 expression was higher in cells stimulated with LPS than in untreated control cells. However, COX-2 expression decreased with 6,7-DH-4MC (5, 10, and 20 µM) treatment by 31.1%, 44.9%, and 63.8%, respectively, compared with LPS treatment alone (Figure 3(B)). These results indicate that 6,7-DH-4MC inhibits NO and PGE2 by downregulating iNOS and COX-2 protein expression.

Effect of 6,7-DH-4MC on the protein expression of (
Effect of 6,7-DH-4MC on the Production of Pro-Inflammatory Cytokines
Recent studies have shown that the increased expression of pro-inflammatory cytokines, such as TNF-α, IL-6, and IL-1β, mediates inflammation in LPS-stimulated macrophages 5 -7 ; therefore, we investigated the effect of 6,7-DH-4MC on the expression of pro-inflammatory cytokines in LPS-stimulated RAW 264.7 cells using ELISA. To confirm the inhibition of pro-inflammatory cytokine expression, LPS-stimulated cells were treated with various concentrations (5, 10, 20, 30, and 40 µM) of 6,7-DH-4MC. The production of each cytokine was significantly higher in cells stimulated with LPS alone than in untreated control cells. In addition, IL-1β and IL-6 levels in LPS-stimulated cells pretreated with various concentrations of 6,7-DH-4MC significantly decreased in a dose-dependent manner. However, TNF-α production was slightly increased at concentrations of 5, 30, and 40 µM 6,7-DH-4MC compared with LPS treatment alone (Figure 4). This result shows that, unlike that of other cytokines (IL-6 and IL-1β), TNF-α production was not different between 6,7-DH-4MC- and LPS-treated cells.

Effect of 6,7-DH-4MC on the production of pro-inflammatory cytokines in LPS-induced RAW 264.7 cells. Cells were pretreated with 6,7-DH-4MC for 1 hour and subsequently stimulated for 24 hours with LPS. (
Effect of 6,7-DH-4MC on the Phosphorylation of MAPKs
It has been reported that the phosphorylation of MAPKs (ERK, JNK, and p38) is closely associated with the expression of inflammatory cytokines. ERK is phosphorylated by various stimulating factors, whereas p38 and JNK constitute a stress response pathway and are phosphorylated by cellular stress induced by factors, such as inflammatory cytokines. JNK has been reported to regulate the maturation and activity of T cells and synthesis of pro-inflammatory cytokines, especially TNF-α. 16 -20 Therefore, to determine whether the inhibitory effect of 6,7-DH-4MC on inflammatory cytokine production is related to the downregulation of ERK, JNK, and p38 phosphorylation, Western blot analysis was performed on LPS-stimulated cells treated with various concentrations of 6,7-DH-4MC. There was significantly more phosphorylation of ERK and p38 in cells stimulated with LPS alone than in untreated control cells. It was also confirmed that the phosphorylation of ERK and p38 in LPS-stimulated cells pretreated with various concentrations of 6,7-DH-4MC was significantly reduced in a concentration-dependent manner (Figure 5). However, 6,7-DH-4MC did not affect JNK phosphorylation (data not shown). Therefore, these results indicate that 6,7-DH-4MC suppresses the expression of inflammatory factors and pro-inflammatory cytokines via the inhibition of ERK and p38 phosphorylation in the MAPK signaling pathway.
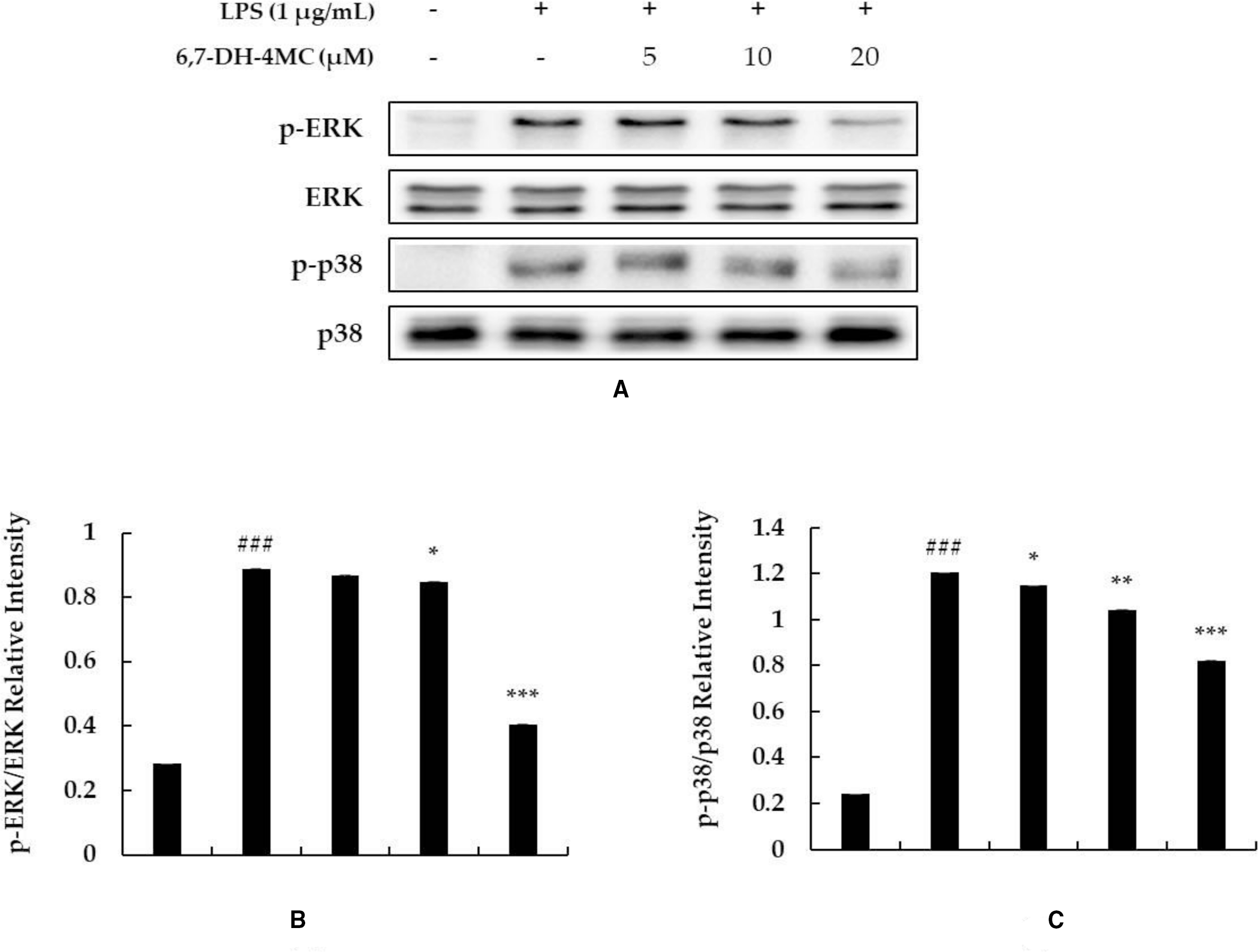
Effect of 6,7-DH-4MC on the phosphorylation of MAPKs in LPS-induced RAW 264.7 cells. Lysates were prepared from cells pretreated with 6,7-DH-4MC for 1 hour followed by LPS treatment for 30 minutes. β-Actin was used as a loading control. The cellular proteins were separated using SDS-PAGE, transferred to PVDF membranes, and detected using specific antibodies against p-ERK, ERK, p-p38, p38, and β-actin. (
Effect of 6,7-DH-4MC on the Expression Levels of IκB-Α and P-NF-κB
A previous study reported that in LPS-stimulated macrophages, IκB-α is phosphorylated by IκB-α kinase and subsequently ubiquitinated. Consequently, IκB-α is released from the NF-κB complex and undergoes degradation. In addition, phosphorylated NF-κB (p50 and p65) is translocated to the nucleus, increasing the production of inflammatory mediators. 35 To determine whether 6,7-DH-4MC treatment prevents IκB-α degradation, Western blot analysis was performed to detect the IκB-α protein levels. The expression level of IκB-α was lower in macrophages stimulated with LPS alone than in untreated control cells. However, the expression level of IκB-α in LPS-stimulated cells pretreated with various concentrations of 6,7-DH-4MC was significantly higher than that in LPS-stimulated cells without pretreatment (Figure 6(A)). In addition, to determine whether 6,7-DH-4MC inhibits the phosphorylation of NF-κB by preventing the decomposition of IκB-α, we confirmed the phosphorylation of p65, an NF-κB subunit. There was more p65 phosphorylation in macrophages stimulated with LPS alone than in untreated control cells. However, the phosphorylation level of p65 in LPS-stimulated cells pretreated with various concentrations of 6,7-DH-4MC decreased in a concentration-dependent manner (Figure 6(B)). These results suggest that 6,7-DH-4MC prevents IκB-α degradation and NF-κB phosphorylation, thereby suppressing the nuclear translocation of NF-κB and impeding inflammation.

Effect of 6,7-DH-4MC on the levels of (A) IκB-α and (B) p-NF-κB (
These mechanistic studies suggest that the inhibition of ERK and p38 phosphorylation and prevention of IκB-α degradation are associated with the inhibitory effect of 6,7-DH-4MC on LPS-stimulated inflammatory factors and cytokines. Furthermore, these effects were mediated via the MAPK and NF-κB signaling pathways in RAW 264.7 macrophages.
Conclusions
In conclusion, this study is the first to evaluate the inhibitory effect of 6,7-DH-4MC on inflammation in RAW 264.7 macrophage cells. Moreover, 6,7-DH-4MC was shown to exert anti-inflammatory effects in cells by inhibiting the production of inflammatory mediators and pro-inflammatory cytokines via the inhibition of ERK and p38 phosphorylation in the MAPK signaling pathway. In addition, the degradation of IκB-α in cells treated with LPS was prevented by 6,7-DH-4MC treatment (Figure 7). These findings suggest that 6,7-DH-4MC is a potential therapeutic agent for inflammatory diseases, such as dermatitis, arthritis, cardiovascular disease, and cancer.
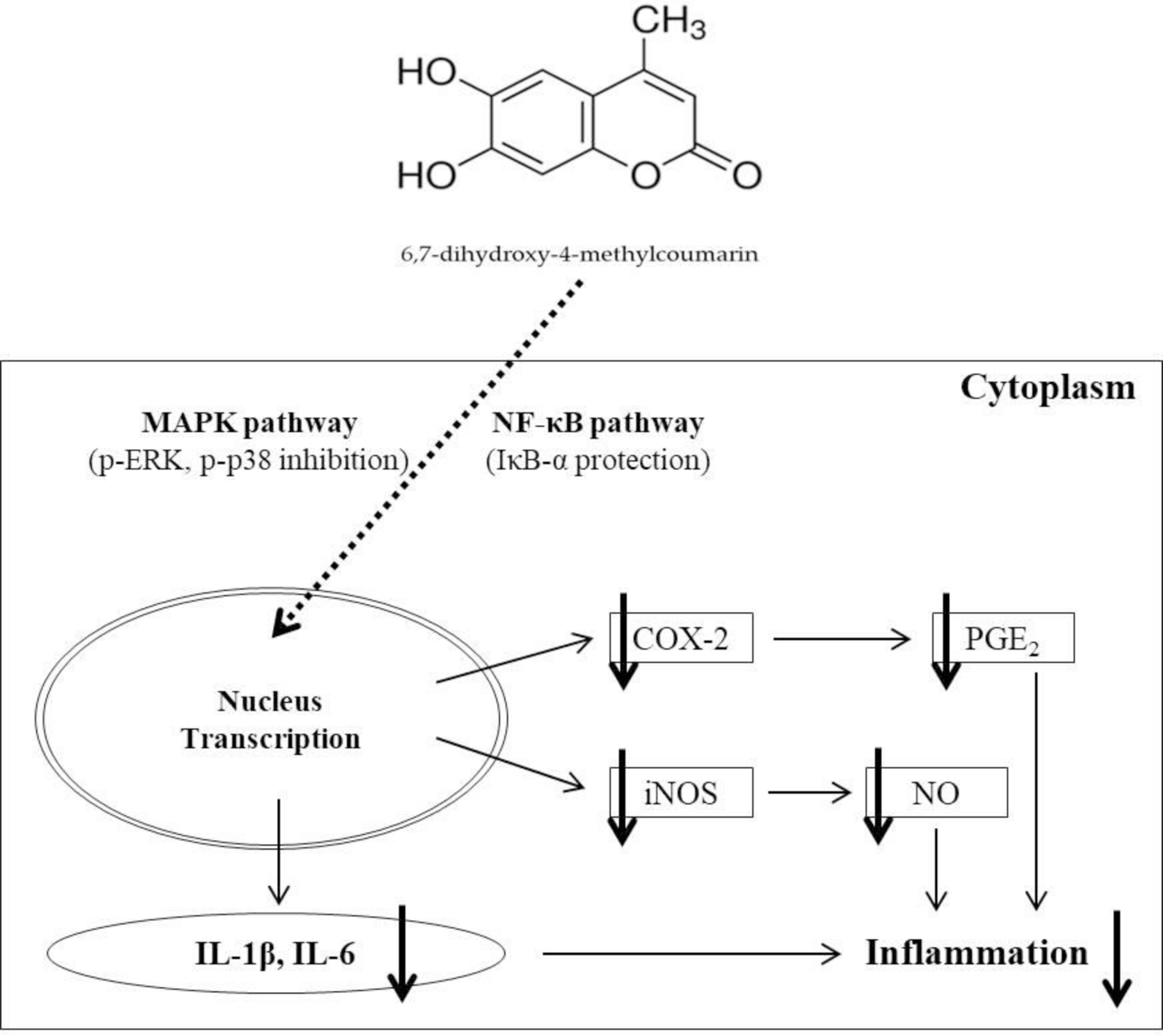
6,7-DH-4MC induces anti-inflammatory effects by inhibiting the expression of iNOS, COX-2, and inflammatory cytokines, such as PGE2, IL-1β, and IL-6, and production of NO in LPS-stimulated macrophage cells via the MAPK and NF-κB pathways.
Experimental
Chemicals and Reagents
6,7-DH-4MC was obtained from Alfa Aesar (Ward Hill, MA, USA), LPS from Escherichia coli, MTT, protease inhibitor cocktail, and L-N6-(1-iminoethyl)lysine dihydrochloride from Sigma-Aldrich (St. Louis, MO, USA), sodium nitrite and anti-iNOS antibodies from Merck Millipore (Burlington, MA, USA), Dulbecco’s modified Eagle’s medium (DMEM), penicillin/streptomycin, Nonidet P-40, and BCA protein assay kits from Thermo Fisher Scientific (Waltham, MA, USA), fetal bovine serum (FBS) from Merck KGaA (Darmstadt, Germany), DMSO, radioimmunoprecipitation assay (RIPA) buffer, phosphate-buffered saline (PBS), enhanced chemiluminescence (ECL) kit, and Tris-buffered saline (TBS) from Biosesang, Inc. (Seongnam, Korea) Griess reagent from Enzo Life Sciences (Farmingdale, NY, USA), 2X Laemmli sample buffer and Tween 20 from Bio-Rad (Hercules, CA, USA), NS-398 from Cayman Chemical Company (Ann Arbor, MI, USA), PGE2 and IL-1β ELISA kits from R&D System Inc. (Minneapolis, MN, USA), IL-6 and TNF-α ELISA kits and anti-COX-2 antibodies from BD Biosciences (Franklin Lakes, NJ, USA), and antibodies against ERK, p-ERK, JNK, p-JNK, p38, p-p38, IκB-α, and β-actin and horseradish peroxidase-linked secondary antibodies (anti-rabbit and anti-mouse) from Cell Signaling Technology (Danvers, MA, USA). All reagents used in this study were of analytical grade.
Cell Culture
RAW264.7 mouse macrophage cells were obtained from the Korean Cell Line Bank (Seoul, Korea). The cells were sub-cultured every 2 days in DMEM containing 10% FBS and 1% penicillin/streptomycin at 37 °C in a humidified incubator with 5% CO2.
Measurement of Cell Viability
To determine the range of non-toxic doses of 6,7-DH-4MC, an MTT assay was performed to measure cell viability. RAW 264.7 cells were seeded in 24-well plates at a density of 1.5 × 105 cells/well and incubated for 24 hours. The cells were pretreated with various concentrations of 6,7-DH-4MC (5, 10, 20, 30, and 40 µM) 1 hour before LPS (1 µg/mL) stimulation. After 24 hours, the cells were incubated with 0.2 mg/mL MTT solution at 37 °C for 4 hours. DMSO (1 ml) was added to dissolve the formazan crystals in each well. After 20 minutes, 100 µL was transferred to 96-well plates and the absorbance was measured at 570 nm. Cell viability was evaluated by comparing the absorbance of the sample group with that of the control group and is expressed as a percentage.
Measurement of NO Production
The amount of NO present in the cell culture medium was measured in the form of nitrite using Griess reagent. RAW 264.7 cells were seeded in 24-well plates at a density of 1.5 × 105 cells/well and incubated for 24 hours. The cells were pretreated with various concentrations of 6,7-DH-4MC (5, 10, 20, 30, and 40 µM) 1 hour before LPS (1 µg/mL) stimulation. After 24 hours, equal volumes of the cell culture supernatant and Griess reagent were mixed for 10 minutes in a 96-well plate. The absorbance was measured at a wavelength of 540 nm. After preparing the standard calibration curve of sodium nitrite, the measured absorbance was compared and quantitatively calculated.
Measurement of Prostaglandin E2 and Pro-Inflammatory Cytokines
To determine whether 6,7-DH-4MC inhibits the production of inflammatory mediators in the cell culture supernatant, the concentrations of PGE2 and pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) were estimated using ELISA kits according to the specified protocols. RAW 264.7 cells were seeded in 24-well plates at a density of 1.5 × 105 cells/well and incubated for 24 hours. The cells were pretreated with various concentrations of 6,7-DH-4MC (5, 10, 20, 30, and 40 µM) 1 hour before LPS (1 µg/mL) stimulation. After 24 hours, the supernatant was collected, centrifuged at 11,000 × g for 5 minutes, and frozen at 20 °C until further use.
Western Blotting Assay
RAW 264.7 cells were seeded in 60 mm cell culture dishes at a density of 6.0 × 105 cells/dish, and each sample was pretreated 1 hour before LPS (1 µg/mL) stimulation at the appropriate time before lysis. After 6,7-DH-4MC treatment, the cells were washed once with cold 1X PBS buffer and lysed for 20 minutes using lysis buffer (1X RIPA buffer, 1% NP-40, and 1% protease inhibitor cocktail). The cell lysates were centrifuged at 11,000 × g for 15 minutes. The protein content of the supernatant was measured using the BCA protein assay kit, quantified, and compared to the standard calibration curve of bovine serum albumin. Equal volumes of quantified protein and 2X Laemmli sample buffer were mixed and heated at 100 °C for 5 minutes. The samples were loaded onto a sodium dodecyl sulfate-polyacrylamide gel and separated by size using electrophoresis. After the gel was transferred to the PVDF membrane, the membrane was blocked with 5% skimmed milk for 1 hour at 25 °C. The membrane was washed 6 times at 10 minutes intervals using TBST containing 1X TBS and 0.1% Tween 20, and the appropriate primary antibody was incubated with the membrane at 4 °C overnight. Thereafter, it was washed 6 times with TBST, and the secondary antibody was incubated with the membrane for 2 hours at 25 °C. The protein bands were visualized and imaged by Chemidoc using an ECL kit and analyzed using ImageJ software.
Statistical Analysis
All data are expressed as mean ± standard deviation (SD: n = 3). Each experiment was repeated at least 3 times. Statistical analyses were performed by one-way analysis of variance using SPSS (v. 22.0, SPSS Inc., Chicago, IL, USA). Values of P < 0.05, (*), P < 0.01 (**), and P < 0.001 (***), as well as P < 0.01 (##) and P < 0.001 (###), were considered statistically significant.
Footnotes
Acknowledgements
This research was supported by the Ministry of Trade, Industry & Energy (MOTIE) and the Korea Institute for Advancement of Technology (KIAT) through the Encouragement Program for the Industries of Economic Cooperation Region (P0006063).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
