Abstract
Laminarin is a component of brown seaweed, especially isolated from
Seaweed has been found to comprise certain bioactive compounds with antibiotic
1,2
and antitumor
3
effects and is used in a variety of functional agents. Particularly, seaweed polysaccharides, including bioactive compounds, contain cellulose and viscous polysaccharides. Seaweed is also composed of about 30% to 60% soluble polysaccharides, including alginates, laminarin, and fucoidan.
4,5
Laminarin from
The AMP-activated protein kinase (AMPK) plays a key role to maintain ATP homeostasis. 8 -10 The activity of AMPK is correlated with an increase in the AMP:ATP ratio in the cells. 11,12 AMPK switches on/off of ATP-generating and ATP-consuming pathways to maintain ATP homeostasis, respectively. AMPK is a key regulator in metabolism, especially in diabetes. 13 -16 The activation of AMPK requires phosphorylation of the catalytic α subunit on Thr-172. 10,17,18 As an AMPK upstream, LKB1 and calcium/calmodulin-dependent protein kinase (CaMKK) are well known as upstream regulators of AMPK. 19 -23 In addition, AMPK regulates glucose uptake in skeletal muscles by physiological and pharmacological stimuli. 24,25
The p38 mitogen-activated protein kinase (MAPK) is known to be associated with AMPK. 26,27 p38 Mitogen-activated protein kinase involves in glucose uptake-related signals in response to insulin 28,29 and skeletal muscle contraction. 30,31 Also, it has been demonstrated that p38 MAPK has a role as one of the downstream of AMPK. 28,32 In this study, we made to verify the metabolic function of laminarin by investigating the effect of laminarin on AMPK activity in muscle.
To determine whether laminarin stimulates metabolic functions in muscles, we examined that laminarin effects on glucose uptake. Glucose uptake was stimulated by laminarin treatment in L6 cells (Figure 1(a)). Insulin was utilized as a positive control. To investigate the molecular mechanisms of laminarin, we evaluated that laminarin effects on AMPK activation, a key molecule in glucose uptake. Laminarin administration induced a time- and dose-dependent increase in AMPK and ACC activity in muscles (Figure 1(b) and (c)). The concentration of laminarin (100 µg/mL) at 10 to 30 minutes increased the activity of AMPK and ACC to the maximum. ACC is known as a direct downstream of AMPK. In addition, to clearly investigate glucose uptake through laminarin-mediated AMPK signaling, we confirmed glucose uptake by laminarin treatment after the inhibition of AMPK activity using compound C, an AMPK inhibitor. The laminarin-induced glucose uptake was decreased by blocking AMPK activity with compound C (Figure 1(d)). These results indicate that laminarin may play some roles in glucose metabolism in L6 myotubes via upregulating an AMPK activity.

Laminarin stimulates glucose uptake through AMPK pathway in L6 myotube cells. (a) Differentiated L6 myotube cells were incubated in 60 mm dishes for 30 minutes with laminarin (100 µg/mL) or insulin (100 nM) alone and then assayed for 2-DG uptake, as described in the Materials and Methods section. *
The previous studies demonstrated that
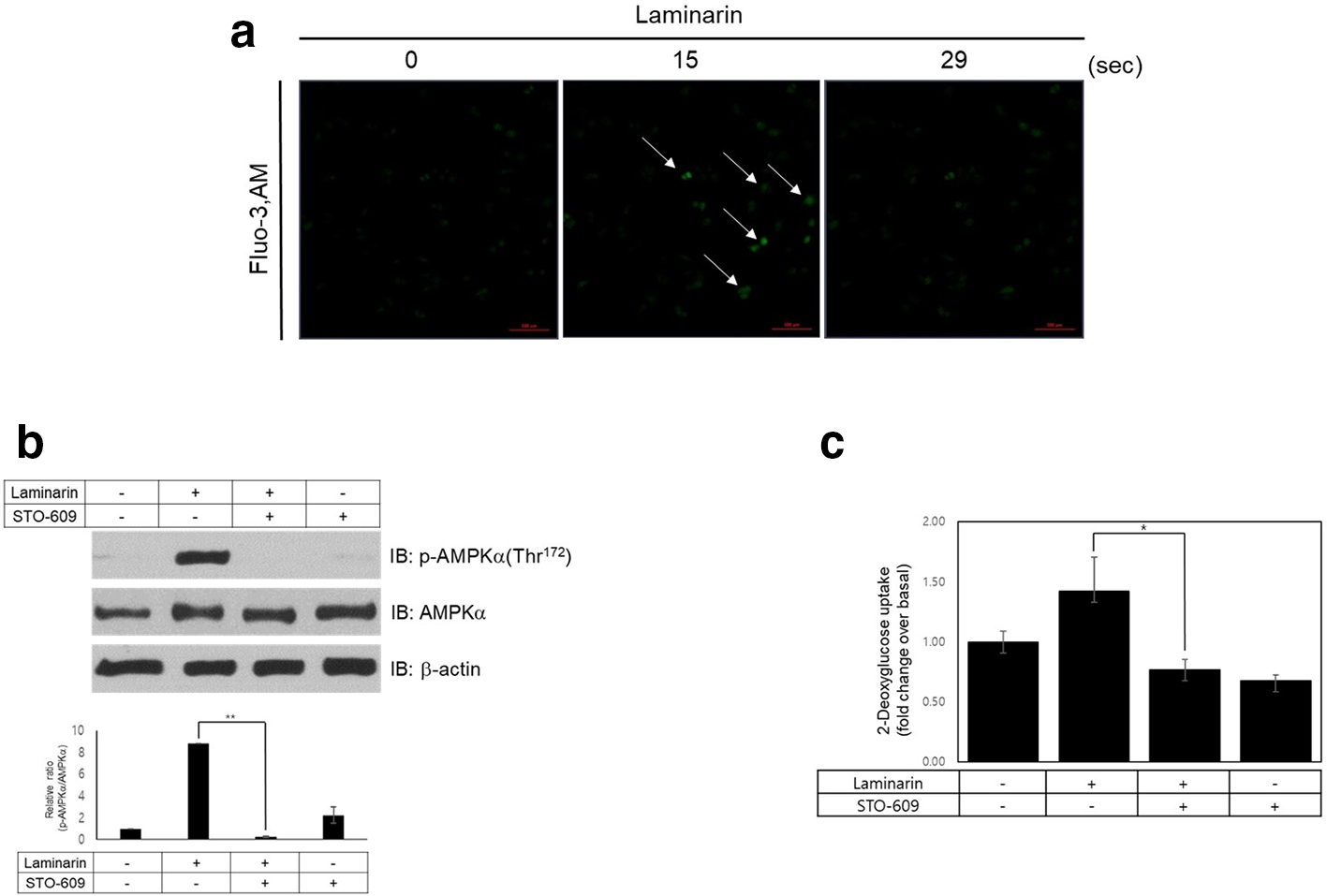
Laminarin increases intracellular calcium release in rat L6 myotubes. (a) L6 cells were pretreated with 10 µM fluo-3 for 30 minutes and treated with laminarin (100 µg/mL). The intracellular calcium (green) in images was obtained by confocal microscopy (×20). (b) Rat L6 cells were treated with laminarin for 30 minutes after pre-incubating with the CaMKK inhibitor, STO-609 (10 µM). Western blot was performed using an antibody against phospho-AMPK, while AMPK and β-actin antibodies were used as a control for protein loading. **
To explain the signal pathways involved in laminarin-mediated glucose uptake, we examined the effects of laminarin on p38 MAPK, a key agent for glucose uptake. 28,30,31,45 We figured out that laminarin (100 µg/mL) activated p38 MAPK in a time- and dose-dependent manner in the L6 myotubes (Figure 3(a) and (b)). The phosphorylation level of p38 MAPK reached a highest level at 30 minutes. These results have been demonstrated as crucial activation steps in correlation in AMPK-mediated glucose uptake. 26,27,29 To determine whether p38 MAPK is downstream of AMPK, we investigated p38 MAPK activation following the treatment of an AMPK inhibitor, compound C. Laminarin-mediated activation of p38 MAPK was decreased in the compound C pretreated L6 cells (Figure 3(c)). To validate the roles of p38 MAPK in the laminarin-mediated signaling pathway, we evaluated the pretreatment effects of inhibitors of the p38 MAPK on glucose uptake. The effect of laminarin on glucose uptake was attenuated in L6 cells in the presence of SB203580, p38 MAPK inhibitor (Figure 3(d)). Together, these results imply that p38 MAPK plays as a downstream of AMPK in the laminarin-mediated signaling pathway.
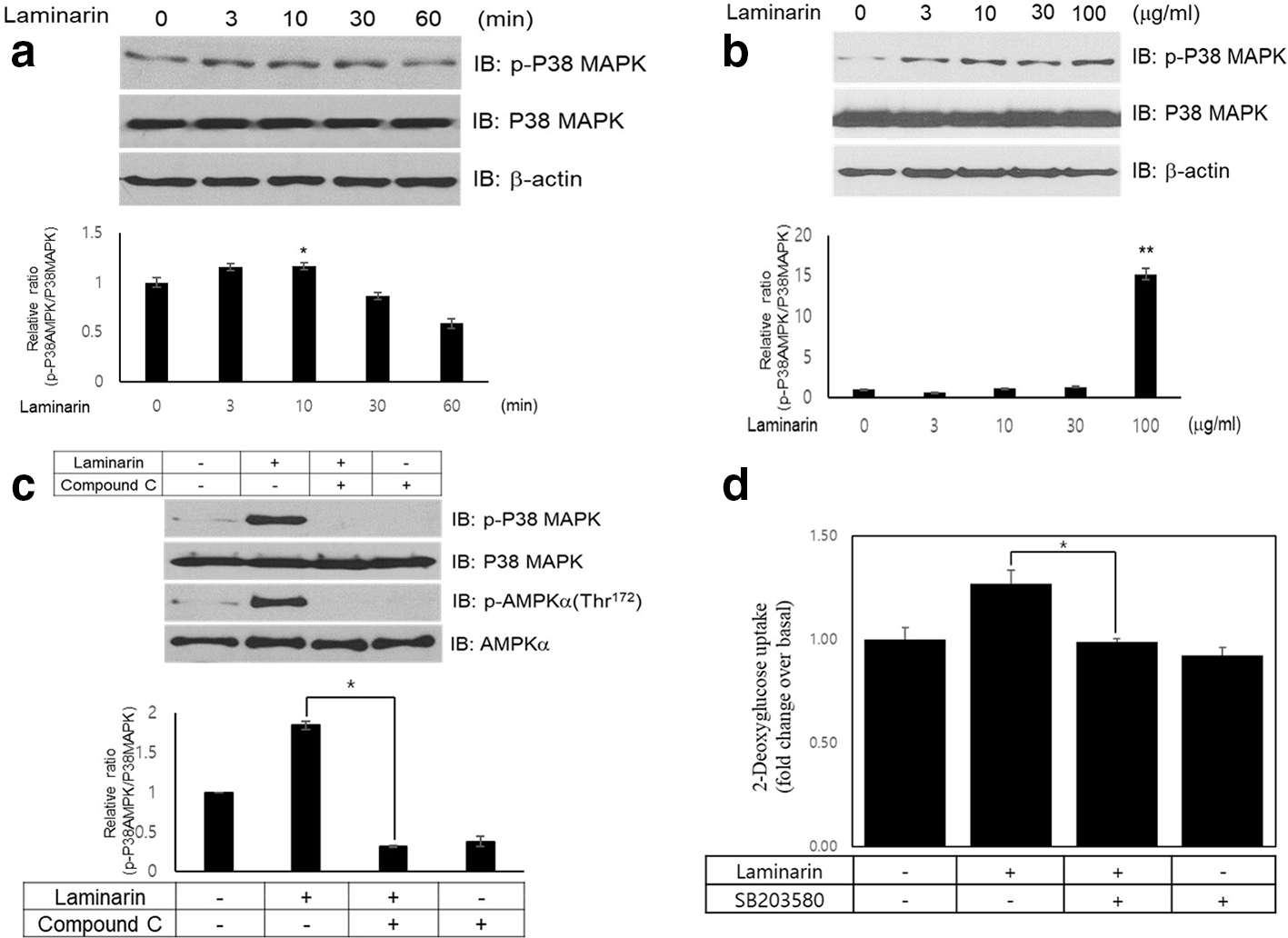
Laminarin activates p38 mitogen-activated protein kinase signal pathway via AMPK signaling. (a) L6 myotube cells were activated for various times with laminarin (100 µg/mL). Western blot analysis was performed using p38 mitogen-activated protein kinase phosphorylation antibody. Blotting was conducted with anti-p38 mitogen-activated protein kinase, and β-actin antibodies were as a control for protein loading. The results are representatives of 4 independent experiments. *
Finally, to establish physiological relevance, we corroborated Glut4 translocation to the plasma membrane using myc-Glut4-tagged L6 cells. Laminarin stimulated Glut4 translocation to the plasma membrane from the cytosol in L6 cells (Figure 4(a)). Insulin was employed as a positive control for Glut4 translocation. Furthermore, we measured a Glut4 translocation from cytosol to the plasma membrane using myc-tagged-Glut4 expressed L6 cells. Using the p38 MAPK inhibitors, SB203580, we confirmed that laminarin-mediated translocation of Glut4 was inhibited when the p38 MAPK signal was blocked (Figure 4(b)). Taken together, our findings suggest that laminarin stimulates glucose uptake via Glut4 translocation to the plasma membrane in skeletal muscle L6 myotube cells.

Laminarin induces Glut4 translocation in rat L6 muscle cells. (a) L6-myc-Glut4 cells were stimulated with laminarin (100 µg/mL) or insulin (100 nM) for 30 minutes. Cells were processed for cell surface Glut4-myc detection with an anti-myc antibody followed by a cy3-conjugated secondary antibody. Cell surface myc-Glut4 density with laminarin treatment increased. Cytological images of Glut4 are shown in the control. The results shown are representatives of at least 4 experiments. The cells were then incubated with 100 nM insulin for 20 minutes. (b) The differentiated myc-Glut4 expressing L6 cells were pretreated with 10 µM SB203580, p38 mitogen-activated protein kinase inhibitor, for 30 minutes. Cell were then treated with 100 µg/mL laminarin for 30 minutes. Cell surface expression of myc-Glut4 was detected by performing an antibody-coupled colorimetric absorbance assay. Cell surface myc-Glut4 levels on intact cells were determined as described in the Materials and Methods section. The results are mean ± SEM from 3 experiments. *
Experimental
Reagents
Laminarin, glucose, insulin, and STO-609 (CaMKK inhibitor) were purchased from the Sigma Chemical Company (St. Louis, MO, United States).
Cell Culture
Rat L6 myoblast cell lines were maintained in Dulbecco’s modified Eagle’s medium (DMEM; GIBCO, Auckland, NZ; containing 0.584 g/L of
Western Blot Analysis
L6 myoblast cells were grown in 6-well plates. The cells were subjected to 6 hours of serum starvation prior to all experiments at 37°C. Next, the cells were washed twice in ice-cold phosphate-buffered saline (PBS) and lysed in 100 µL of lysis buffer (0.5% deoxycholate, 0.1% SDS, 1% Nonidet P-40, 150 mM NaCl, and 50 mM Tris-HCl, pH 8) containing proteinase inhibitors (0.5 µM aprotinin, 1 µM phenyl-methyl-sulfonyl-fluoride, and 1 µM leupeptin) (Sigma). The supernatants were sonicated briefly, heated for 5 minutes at 95°C, centrifuged for 5 minutes, separated on SDS-PAGE (8% to 16%) gels, and finally transferred to polyvinylidene difluoride membranes. The blots were then incubated overnight at 4°C with primary antibodies and washed 3 times in Tris-buffered saline/0.1% Tween 20 prior to 1 hour of probing with horseradish peroxidase (HRP)-conjugated secondary antibodies at room temperature. The blots were then visualized with ECL (Amersham Biosciences, Buckinghamshire, United Kingdom).
Intracellular Ca2+ Measurement Using Fluo-3, AM
Ca2+ concentration was determined using confocal microscopy (Zeiss LSM 510 Meta; Zeiss, Oberkochen, Germany). The cells were loaded with 5 mM Fluo-3, AM for 30 minutes. The culture plates were then placed on a microscope stage and observed with 200× microscope magnification. The excitation and emission wavelengths for signal detection were 488 and 515 nm, respectively.
Immunocytochemistry
Briefly, myc-Glut4-expressing L6 myoblasts were seeded onto collagen-coated glass cover slips. After 24 hours, the cells were serum-starved for 3 hours and stimulated with either 100 nM insulin or 100 µg/mL laminarin for 30 minutes at 37°C. The cells were fixed with 4% paraformaldehyde in PBS for 20 minutes at 4°C. The cells were then incubated for 3 hours with primary c-myc antibody at a dilution of 1:200 in buffer (3% BSA in PBS) at 4°C, followed by incubation with Cy3-conjugated secondary antibody at a dilution of 1:1000 for 1 hour at room temperature. The cells were observed using a Zeiss LSM 700 confocal fluorescence microscope with 400× microscope magnification.
2-Deoxyglucose (DG) Uptake Assay
Myotube L6 cells were grown in DMEM containing 2 % v/v FBS until fully differentiated. The uptake of 2-deoxyglucose (DG) was examined. 100 nM insulin was used as a positive control. In brief, the cells were washed twice in PBS containing 2.5 mM MgCl2, 1 mM CaCl2, and 20 mM HEPES at pH 7.4, followed by incubation in test compounds in the same buffer at 37°C. The transport assay was then initiated via addition of 2-[3H]-deoxy-
Immunodetection of Myc-Glut4
Myotube L6 overexpressing myc tagged Glut4 cells were grown in DMEM containing 2% v/v FBS for 6 to 7 days. The cell surface expression of myc-Glut4 was quantified using an antibody-coupled colorimetric absorbance assay. After stimulation with laminarin and the p38 MAPK inhibitor, L6 myoblasts stably expressing myc-Glut4 were incubated with anti-myc antibody (1:1000) for 1 hour, and then incubated with HRP-conjugated anti-mouse IgG (1:1000) for 1 hour. Cells were washed with PBS and incubated in 200 µL of OPD reagent (0.4 mg/mL) (Sigma-Aldrich, St. Louis, MO, United States) for 30 minutes at room temperature in the dark. The absorbance of the supernatant was measured at 450 nm.
Data Analysis
We used one-way ANOVA and Holm-Sidak comparisons followed by the post hoc Fisher’s test and analyzed band intensity. The mean values were considered statistically significant when the probability of the event was determined to be below 5% (*
Footnotes
Acknowledgment
This research was a part of the project titled “The base study to discover and to commercialize for the resources of sea healing to activate marine industry.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Ministry of Oceans and Fisheries, Korea (KIMST, 2019R1707173).
