Abstract
Kukoamine A (Kuk A) is a naturally occurring bioactive spermine alkaloid found in the root bark of Lycium chinense, and it exerts various therapeutic effects including antioxidant, neuroprotective, anti-inflammatory, and antidiabetic effects. This study evaluated the anti-inflammatory properties of Kuk A against lipopolysaccharide (LPS)-induced inflammation in RAW 264.7 macrophage cells and carrageenan-induced paw edema in rats. Pretreatment of cells with Kuk A significantly inhibited the production of reactive oxygen species, nitric oxide, prostaglandin E2, cyclooxygenase-2 activity, tumor necrosis factor-α, interleukin-1β (IL-1β), and IL-6 in LPS-treated cells. In addition, pretreatment of rats with Kuk A significantly decreased inflammatory response to carrageenan-induced paw edema by alleviating proinflammatory cytokines in the serum, malondialdehyde levels in the liver and increasing the activities of liver antioxidant enzymes (superoxide dismutase, catalase, and glutathione peroxidase) of carrageenan-treated rats. These results suggest the protective role of Kuk A in acute inflammatory reaction and could be useful in the treatment of inflammatory-related disorders.
Inflammation is the body’s physiological response that occurs in tissues because of damages or injury caused by internal or external stimuli. These responses are associated with alteration in the flow of blood, recruitment, activation, and migration of leucocytes, resulting in the production of reactive oxygen species (ROS) and proinflammatory mediators like prostaglandins (PGs), nitric oxide (NO), tumor necrosis factor (TNF-α), interleukins, leukotrienes, and cyclooxygenases (COXs). 1 Inflammation plays a pivotal role in the development and progression of several ailments including cancer, cardiovascular diseases, rheumatoid arthritis, and type 2 diabetes. 2,3 ROS which is mainly produced in the mitochondria has the ability to act as a signal-transducing molecule, and it can initiate the upregulation of inflammatory cytokine through several molecular pathways. 4 There are several drugs (corticosteroids and nonsteroidal anti-inflammatory drugs) currently available for the treatment of inflammatory-related disorders; however, the use of these drugs for an extended period of time has been associated with some unpleasant side effects such as gastric mucosal lesions, immunosuppression, and susceptibility to infections. 5
Kukoamine A (Kuk A; Figure 1) is a naturally occurring bioactive spermine alkaloid found in the root back of Lycium chinense, and previous studies have indicated the therapeutic potentials of Kuk A, highlighting its antihypertensive, anti-inflammatory, antisepsis, antioxidant, anticancer and neuroprotective properties. 6 -8 Although Kuk A has many pharmacological effects associated with it, there are no evidences in the literature on its effects on acute inflammation. Thus, this study investigated the in vitro and in vivo anti-inflammatory activities of KuK A on lipopolysaccharide (LPS)-stimulated inflammation in RAW 264.7 cells and carrageenan-induced inflammation in rats.

Chemical structure of kukoamine A.
In order to verify the safety of Kuk A on RAW 264.7 cells, we accessed its cytotoxic effect on the cells using MTT assay. As indicated in Figure 2(a), at the highest concentration (40 µM), Kuk A had no negative effect on the viability of treated cells (Figure 2(a)). Thus, the in vitro anti-inflammatory assay was evaluated at concentrations of 5-40 μM.
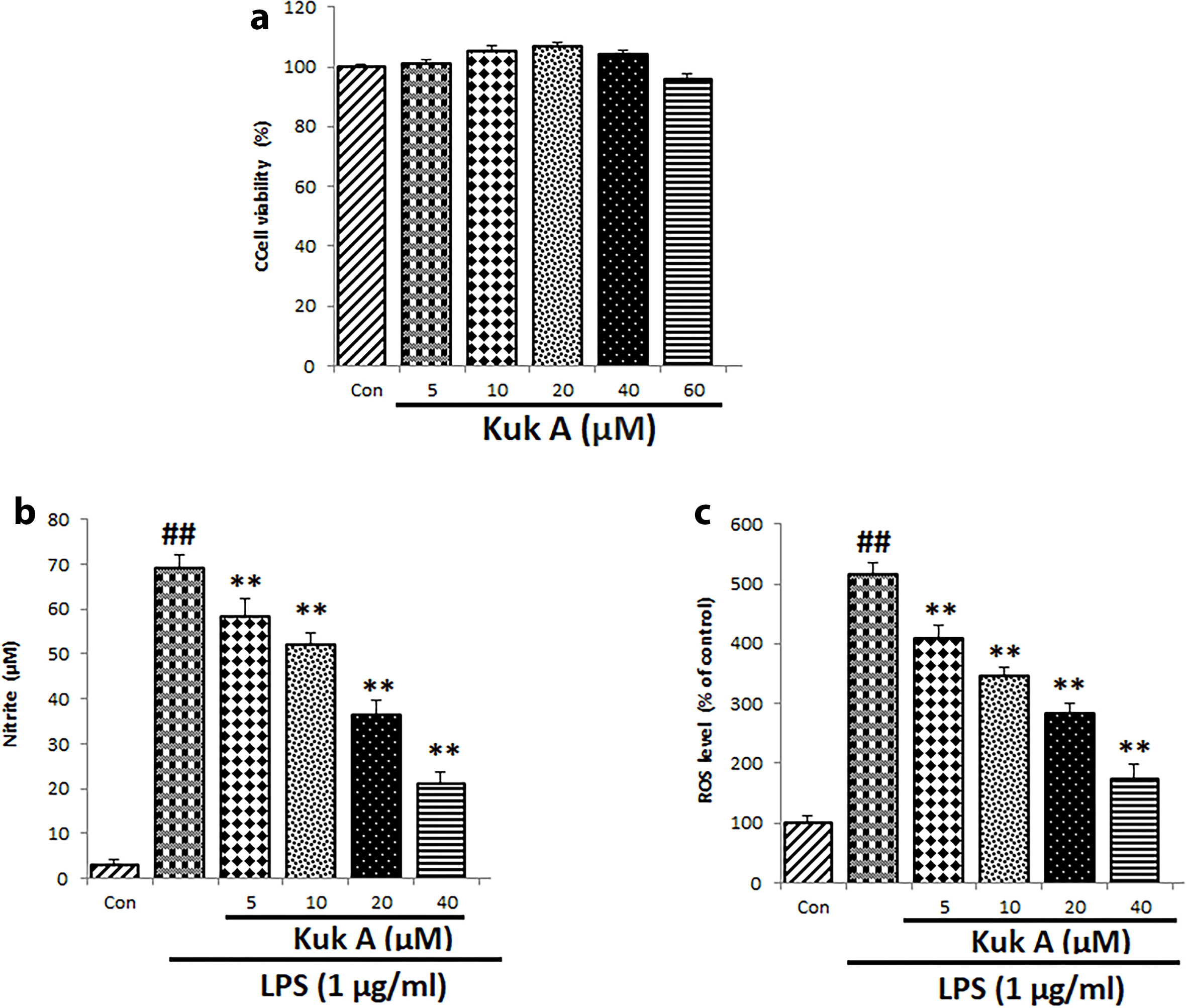
(a) Effect of Kuk A on RAW 264.7 macrophage cell viability. RAW 264.7 cells were treated with various concentrations of Kuk A for 24 hours. (b) Effects of Kuk A on LPS-induced nitric oxide and (c) ROS level in RAW 264.7 macrophage cells. The results are expressed as mean ± standard deviation (n = 5). ## P < .05 compared with the control group; **P < .05 compared with LPS control group. Kuk, kukoamine; LPS, lipopolysaccharide; ROS, reactive oxygen species.
LPS is a complex molecule made up of polysaccharides, lipid A core, and carbohydrate chains. 9 LPS has the ability to stimulate the immune response in macrophages and it has been extensively used as a model for in vitro inflammatory and immunomodulatory studies in murine macrophage RAW 264.7 cells through the induction of systemic inflammation. 10,11 Excessive production of ROS and NO has been linked to several inflammatory-related disorders such as arteriosclerosis, cancer, diabetes, ischemic reperfusion, hypertension, and septic shock. 12 Furthermore, ROS has been implicated in the expression of inflammatory mediators through the activation of the nuclear factor kappa-light-chain-enhancer of activated B cells signaling pathway. Previous reports have confirmed that macrophages stimulated by LPS displayed increased levels of ROS and NO. 13 As indicated in Figure 2(b and c), the levels of NO and ROS were significantly increased in LPS stimulated cells when compared with the control cells. However, in Kuk A-pretreated cells, the levels of ROS and NO were significantly reduced in a concentration-dependent manner when compared with LPS-treated group.
Proinflammatory cytokines are small proteins that are secreted by activated macrophages and they are vital modulatory agents of inflammatory reactions. Several body of evidence has suggested the active roles that certain proinflammatory cytokines like TNF-α, IL-1β, and IL-6 play in the onset and progression of acute and chronic inflammation. 14 The secretion of LPS-activated proinflammatory cytokines, such as TNF-α and IL-1β, can enhance the production of secondary proinflammatory cytokines such as IL-6 and IL-8. 15 TNF-α in the LPS-stimulated cells was markedly increased as compared with the control cells, whereas, treatment with different concentrations of Kuk A significantly attenuated the level of TNF-α (Figure 3(a)). Furthermore, as illustrated in Figure 3(b and c), exposure of cells to LPS triggered a significant increase in IL-1β and IL-6 levels in comparison with the control cells. Kuk A significantly lowered IL-1β and IL-6 levels when compared with the LPS-stimulated cells.

Effects of Kuk A on LPS-induced TNF-α, IL-1β and IL-6 production in RAW 264.7 macrophage cells. The results are expressed as mean ± standard deviation (n = 5). ## P < .05 compared with the control group; **P < .05 compared with LPS control group. IL, interleukin; Kuk, kukoamine; TNF-α, tumor necrosis factor-alpha.
As shown in Figure 4(a), the treatment of RAW 264.7 cells with LPS led to a marked increase in PGE2 levels, but Kuk A treatment significantly reduced the upregulated levels of PGE2. Likewise, in the LPS-treated cells, the levels of cyclooxygenase-2 (COX-2) was 5.5-fold higher than the control cells. However, Kuk A markedly inhibited the COX-2 level in the treated cells (Figure 4(b)). The activity of COX-2 has been shown to be upregulated in inflammatory conditions and this can be associated with the induction of proinflammatory cytokines. PGE2, a product formed from the enzymatic catalytic action of COX upregulates proinflammatory cytokines. The activation of the COX-2 pathway plays a pivotal role in promoting the production of ROS. 16,17

Effects of Kuk A on LPS-induced PGE2 level and COX-2 activity in RAW 264.7 macrophage cells. The results are expressed as mean ± standard deviation (n = 5). ## P < .05 compared with the control group; **P < 0.05 compared with LPS control group. COX-2, cyclooxygenase-2; Kuk, kukoamine; LPS, lipopolysaccharide; PGE2, prostaglandin E2.
Carrageenan-induced rat paw edema is a widely used model of acute inflammation, and it is employed for the screening of anti-inflammatory agents. Carrageenan is a group of sulfated polysaccharides obtained from seaweed and algae, and it has been extensively used for inducing in vivo models of acute inflammatory reaction. It acts by instigating the release of histamine, bradykinin, serotonin, and PGs leading to systemic or local inflammation. 18 As illustrated in Figure 5(a), the evidence of inflammatory reaction in the hind paw of the rats was observed after the subplantar injection of carrageenan. There was a marked increase in the paw volume with a peak value of 1.08 mL after 4 hours of carrageenan injection. Kuk A (25 and 50 mg/kg) significantly reduced the paw edema volume in a concentration-dependent fashion.

Effect of Kuk A on (a) carrageenan-induced rat paw edema, (b) TNF-α, (c) IL-1β, and (d) IL-6 in the serum of rats 4 hours after carrageenan injection. The results are expressed as mean ± standard deviation (n = 6). ##
P
As shown in Figure 5(a-c), the levels of TNF-α, IL-1β, and IL-6 in the serum were obviously upregulated in carrageenan
Quite a number of inflammatory-related reactions including carrageenan-induced acute inflammation has been linked to an increase in the generation of reactive radical species and lipid peroxidation as well as the impediment of antioxidant defense activities. 21 Malondialdehyde (MDA), a marker of cellular injury caused by ROS and oxidative stress, is also considered as a mediator of inflammatory processes. Cellular antioxidant enzymes such as superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and catalase (CAT) are mainly responsible for annihilating the damaging effects of oxidative stress and ROS. 22 In the liver of carrageenan-treated rats, the activities of SOD, CAT, and GSH-Px were evidently decreased, while MDA content in the liver was significantly increased in comparison with the control group (Figure 6(a-d)). However, in groups pretreated with Kuk A marked enhancement in the activities of SOD, CAT, and GSH-Px, as well as a reduction in the MDA levels, were observed when compared with the Carr group.

Effect of Kuk A on (a) SOD, (b) CAT, (c) GSH-Px, and (d) MDA levels in the liver tissue homogenates 4 hours after carrageenan injection. The results are presented as mean ± standard deviation (n = 6). ##
P
In conclusion, the protective effect of Kuk A on LPS-stimulated inflammation in RAW 264.7 cells and carrageenan-induced acute inflammation in rats was investigated. Kuk A displayed anti-inflammatory effects in vitro through the inhibition of ROS and NO, TNF-α, IL-1β, IL-6, COX-2 and PGE2 levels. In addition, Kuk A successfully inhibited carrageenan-induced inflammation in rats by suppressing the release of proinflammatory mediators and oxidative stress. These findings suggest that Kuk A may have potential use as an anti-inflammatory agent.
Experimental
Chemicals and Reagents
Kuk A was procured from Chengdu Biopurify Phytochemicals Ltd. (Chengdu, China). LPS (Escherichia coli; O55:B5), sodium nitrite Griess reagent, and carrageenan were purchased from Sigma (St. Louis, MO, USA). PGE2, TNF-α, IL-1β, and IL-6 enzyme-linked immunosorbent assay (ELISA) assay kits were obtained from R&D Systems (Minneapolis, MN, USA). COX-2 assay kit was procured from Cayman Chemical Co. (Ann Arbor, MI, USA). NO and ROS assay kits were procured from Beyotime (Nanjing, China). Dulbecco modified eagle’s medium (DMEM), fetal bovine serum (FBS) and penicillin–streptomycin were purchased from Gibco Industries Inc. (Green Island, NY, USA). All other chemicals and reagents used were of analytical grade.
Cell Culture
RAW264.7 cells were cultured in DMEM supplemented with 10% FBS, 100 U/mL of penicillin, and 100 µg/mL of streptomycin. The cells were maintained at 37°C in a humidified condition of 95% air and 5% carbon dioxide.
Determination of Cell Viability
The effect of Kuk A on RAW 264.7 cell viability was accessed by the MTT method. Cells were seeded in a 96-well plate at a density of 1.5 × 106 cells/well for 24 hours and exposed to different concentrations of Kuk A for an additional 24 hours. Thereafter, the culture medium was removed and 10 µL of MTT solution was added to each well and incubated for 4 hours at 37°C. The purple formazan crystals were dissolved in 150 µL of dimethyl sulfoxide, and the absorbance was read at 570 nm. Cell viability was expressed as a percentage of control.
Determination of PGE2 and ProInflammatory Cytokines
Cells were pretreated with Kuk A (10, 20, and 40) for 12 hours and stimulated with LPS (1 µg/mL) for 12 hours. The supernatants from the culture medium were collected for the determination of PGE2, IL-1β, IL-6, and TNF-α by using ELISA kits (R&D Systems; Minneapolis, MN, USA) based on the guidelines of the manufacturer.
Determination of Nitrite Levels
The levels of NO were measured using Griess reagent. 23,24 After cell treatment, 100 µL of the supernatant from the cell culture was mixed with 100 µL of Griess reagent and incubated in the dark for 10 minutes at room temperature. The absorbance of the mixture was measured at 550 nm using a microplate reader. The concentration of nitrite was determined based on a sodium nitrite standard curve for the quantification of NO production.
Determination of Intracellular ROS Levels
The levels of ROS were determined using dichloro-dihydro-fluorescein diacetate (DCFH-DA) protocol. 24 Briefly, after cell treatment, cells were washed with phosphate-buffered saline (PBS) and incubated with 20 µM of DCFH-DA at 37°C for 30 minutes in the dark. The fluorescence intensity was measured at an excitation wavelength of 485 nm and an emission wavelength of 535 nm.
Determination of COX-2 Activity
COX-2 enzyme activity was determined based on the ELISA kit manufacturers protocol (Cayman Chemical Co., Michigan, USA). After treatments, the cells were homogenized in a cold Tris–hydrochloric acid buffer (0.1 M, pH: 7.8), centrifuged for 15 minutes at 4°C. The supernatant was obtained and used for the determination of COX-2 activity based on the manufacturer’s protocols.
Experimental Animals
Specific pathogen-free male Wistar rats (180-220 g) were accommodated at a facility with the following standard conditions of temperature 22 ± 2°C, relative humidity 50 ± 10, and 12-hour light–dark cycle. The rats were allowed 7 days of adjustment to the laboratory conditions, and they were given unlimited access to food and water during this period. All the procedures used during the animal treatments were in compliance with the National Institutes of Health Guide of the Care and Use of Laboratory Animals. The study protocols also received approval from the Committee in charge of Animal Ethics of Wannan Medical College (YJS 2019-2-012).
The anti-inflammatory effect of Kuk A was evaluated against carrageenan-induced paw edema according to the previous method. 25 The rats were randomly divided into 5 groups (6 rats in each group). The groupings were assigned as follows: group 1—normal control group; group 2—carr control group; group 3—indomethacin group (10 mg/kg); groups 4 and 5—Kuk A (25 and 50 mg/kg, respectively). Kuk A dosage selection was based on the previous report. 26,27 Kuk A and indomethacin were dissolved in normal saline prior to administration. The rats in groups 1 and 2 were orally administered with normal saline, while the rats in groups 3-5 were treated with indomethacin and Kuk B by oral gavage for 5 days prior to carrageenan injection. 21,28 Thirty minutes after the last treatment, edema was induced by administering 50 µL of 1% carrageenan by subplantar injection at the right hind paw. The rats designated as normal control were administered with an equal volume of saline through the intraperitoneal route and by subcutaneous injection in the paw. 19 Changes in paw volume were recorded hourly for 4 hours using a plethysmometer. The rats were then sacrificed by cervical dislocation and blood samples were obtained by cardiac puncture for the estimation of proinflammatory cytokine. The liver of the rats was immediately excised, washed with PBS, homogenized, and the homogenate supernatant was used for estimating SOD, CAT, GSH-Px, and MDA levels.
Statistical Analysis
All data were analyzed by Graph Pad Prism software and the results are presented as mean ± standard deviation. One-way analysis of variance followed by Tukey’s post hoc test was used for statistical analysis. P < .05 was considered statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are sincerely grateful to the Key Project of Natural Science Foundation of Anhui Province for College Scholars (KJ2019A0416) for the financial assistance.
