Abstract
Arsenic (As) is a classic neurotoxicant; its pathogenesis is associated with oxidative stress and oxidative stress-mediated cholinergic deficits. This study explored antioxidant activity of virgin coconut oil (VCO) against sodium arsenite-induced oxidative stress-mediated cerebral neurotoxicity in rats. Eighteen rats were divided into 3 groups- Normal control, As control and VCO + As. The VCO (5 mL/kg) was given once daily by oral gavage from day 1 to day 21, while As (10 mg/kg) was given once daily by oral gavage from day 15 to day 21. Cerebral superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), malondialdehyde (MDA), adenosine deaminase (ADA) and acetylcholinesterase (AchE) activities were analysed. Nitric oxide (NO), lipid profile, phospholipid (PL), and reduced glutathione (GSH) were also evaluated in cerebral homogenate. The cerebrum was sectioned for histological analysis. Administration of As induced significant depressions in antioxidant enzymes, GSH, PL, and HDL-c compared to normal control. Levels of MDA, NO, total cholesterol and activities of ADA, AchE in the cerebrum were markedly increased by As compared to normal rats. Lipid profile indices and PL were prominently altered by As. Histopathological study supported the biochemical findings through extensive cerebral damage. In contrast, oral supplementation of VCO prior to and along with As treatment significantly attenuated the As-induced biochemical alterations and restored near-normal histology. VCO attenuates cerebral neurotoxicity by strengthening endogenous antioxidant defence and cholinergic function via counteracting free-radical-mediated arsenic toxicity.
Arsenic (As) is a ubiquitous environmental toxicant and a source of health concern worldwide. 1 Inorganic arsenic in drinking water is a global threat to health, and it affects about 200 million people in about 70 countries worldwide. 2,3 It induces pathologies, including neurodegenerative diseases. However, evidence is increasing for health effects of As at lower levels of exposure than previously thought. 4 Arsenic contaminates drinking water via agricultural and industrial use, and water sources without high arsenic levels are scarce. 5,6 On this account, the maximum limit of As in drinking water has been reduced to 0.01 mg/L by the World Health Organisation. 7 Studies indicate that As in drinking water stores in the liver, kidney, lung, spleen, and brain. 5 Arsenic triggers toxicity in the liver, kidney, brain, heart and testes, and induces biological complications including carcinogenesis, mutagenesis, aging, and atherosclerosis. 8 -11
Arsenic is a potent neurotoxicant; it causes subclinical and clinical effects in humans. It can cross the blood–brain barrier and accumulate in brain regions suggesting its role in neurological diseases. 1,12 The increased As levels in the cortex, cerebellum, hypothalamus, striatum, and hippocampus regions of rat brain have been associated with varying degrees of neurotoxicity. 5 In particular, sodium arsenite initiates chromosomal breakage and its toxic effects are amplified by interaction with metals. 8 However, the exact mechanism underlying As neurotoxicity is unclear. Although genetic aberrations and altered signaling transduction have been proposed, 11 systematic investigations have implicated oxidative stress as the overriding mechanism of As neurotoxicity. 13 Arsenic is a sulfhydryl-reactive metalloid. 14 It binds to sulfhydryl-rich proteins to inhibit thioredoxin reductase and glutathione reductase to deplete glutathione (GSH) level. 15 Robust evidence shows that GSH depletion impairs redox balance leading to depression of endogenous antioxidant enzymes and reactive oxygen species (ROS) generation. 1,14,16 The brain is highly vulnerable to oxidative damage due to abundant unsaturated lipids and limited GSH synthesis in neurons. The copious attraction of arsenic to the cerebral cortex aggravates impairment of the cholinergic synapses and communication, motor coordination, and learning ability leading to depressed coordination, memory, and cognitive ability. 1,12 Thus, the presence of As in drinking water and foods necessitate more research into the prevention and mitigation of arsenic neurotoxicity by dietary supplementation.
Virgin coconut oil (VCO) is emerging as a functional food oil reputed in the literature to possess health-promoting biological properties. 17,18 Moreover, VCO has shown efficacy in animal studies through its anti-inflammatory, antiarthritic, antidiabetic, and antihyperlipidemic activities. 19 -21 Studies indicate that drug-induced toxicities in the liver, kidney, and testes were attenuated by VCO supplementation. 19,22 In the studies, bioactive phytochemicals such as flavonoids and phenolic acids in VCO are suggested to be responsible for its pharmacological properties. There are reports that the wet method of VCO production from fresh and mature kernels without chemical refining, bleaching and deodorizing conserves VCO bioactive compounds. 18 Recently, a study has shown that oral supplementation of VCO enhances memory and cholinergic activity and modulates oxidative stress in Wistar rats. 23 The beneficial effects of VCO are attributed to its natural antioxidants that scavenge free radicals to block lipid peroxidation and enhance the endogenous antioxidant defence mechanism. 24 However, the efficacy of VCO against As neurotoxicity is unknown. Given the level of As in the environment and growing evidence of its health effects at lower exposure levels than currently thought, the prevention and mitigation of As-induced adverse health outcomes requires more vigorous pursuit. Therefore, this study aimed at investigating the protective effect of VCO against As-induced oxidative stress-mediated cerebral neurotoxicity in rats.
Materials and Methods
Chemicals
Arsenic (sodium arsenite, NaAsO2) with purity ≥98.5%, purchased from BDH Chemicals Ltd Poole, England, was dissolved in distilled water. Some of the kits used for biochemical assays were obtained from Randox Laboratory Ltd., UK, thiobarbituric acid (TBA) from Hi Media Laboratories Pvt. Ltd, India, and commercial kits for nitric oxide analysis from R&D Systems, USA. The dose of sodium arsenite was selected based on previous reports. 25 All other chemicals were of analytical grade.
Animals
Adult male Wistar rats (8, 10 weeks old, weighing 140‐170 g) were purchased from a private Animal Breeding House, near the University of Nigeria, Nsukka, Enugu State, Nigeria. They were kept in the Animal House of the Department of Chemistry/Biochemistry, Alex Ekwueme Federal University, Ndufu-Alike Ikwo, Ebonyi State, Nigeria under controlled conditions of temperature (25 ± 2 °C) and normal photoperiod (12 hours light and 12 hours dark). The animals were allowed free access to pelleted commercial growers mash (Vital Feeds Nigeria Ltd, Jos, Nigeria) and tap water ad libitum. They were acclimatized for 2 weeks preceding treatment and were handled in a humane manner according to the approved animal experimental procedures given by the NIH Publication (NIH Publication No. 85‐23, revised 1996) on Guide for the Care and Use of Laboratory Animals. The animals and experimental procedure were approved by the Physiology Department Research and Ethics Committee, Alex Ekwueme Federal University, Ndufu-Alike Ikwo, Ebonyi State, Nigeria. The VCO used in the current study was prepared from mature coconuts according to a published method, 26 modified by Famurewa et al. 17
Experimental Design
Three groups of rats (n = 6) entered the study following 2 weeks of acclimatization. Arsenic as sodium arsenite (NaAsO2) dissolved in distilled water was administered to rats after 1 hour of oral administration of VCO. The design of experimental treatment was as follows:
Group 1 (Normal control): distilled water for 21 days (3 weeks) (5 mL/kg body weight of rat)
Group 2 (Arsenic control): As (10 mg/kg body weight of rat) from day 15 to day 21 (1 week). 25
Group 3 (VCO +Arsenic): VCO (5 mL/kg body weight of rat) from day 1 to day 21 (3 weeks) +As (10 mg/kg body weight of rat) from day 15 to day 21 (1 week). 19,25
At the end of the treatment period (21 consecutive days = 3 weeks), overnight fasted animals were decapitated under mild diethyl ether anesthesia and the cerebrum was gently removed, washed with an ice-cold saline solution and dried with tissue paper and weighed. One g of cerebrum was cut out and homogenized in phosphate buffered saline (1:5 w/v, pH 6.4) and centrifuged at 4000 g for 20 minutes. The homogenate obtained was used to assay for the biochemical indices evaluated. The remaining part of the cerebrum was immediately fixed in 10% buffered formalin for histopathological examination.
Biochemical Analyses
Superoxide dismutase (SOD) was assayed by the method of Marklund and Marklund, 27 catalase (CAT) activity according to the method of Aebi, 28 glutathione peroxidase (GPx) according to the method of Flohe and Gunzler, 29 and reduced glutathione (GSH) content by the Beutler method. 30 Lipid peroxidation in the cerebrum was estimated by measuring thiobarbituric acid reactive substances (TBARS) expressed in terms of malondialdehyde (MDA) content using the method of Ohkawa et al. 31 Cerebral nitric oxide (NO) was measured by the procedure of Green et al, 32 acetylcholinesterase (AchE) activity by the method of Ellman et al., 33 adenosine deaminase (ADA) activity by the method of Giusti, 34 and lipid profile indices according to the instructions indicated in the Randox kits.
Histopathological Examination
Cerebral tissue samples were fixed in 10% formalin for 48 hours and dehydrated in ethanol and then embedded in paraffin blocks. The blocks were cut into 5 µm sections using a microtome, fixed on slides followed by hematoxylin and eosin (H&E) staining. The prepared slides were observed under a light microscope.
Statistical Analysis
Data are expressed as mean ± SEM. Statistical analysis was done using analysis of variance (ANOVA), followed by Tukey’s post hoc test using GraphPad Prism version 5.00. The level of statistical significance was considered as P < 0.05.
Results
Effect of VCO on Body Weight and Cerebrum Weight in Arsenic-Treated Rats
Table 1 depicts the effect of VCO on body weight, cerebrum weight, and relative cerebrum weight of arsenic-treated rats. Arsenic administration for 1 week (day 15 to day 21) exerted no significant changes (P > 0.05) on body weight compared to the controls. However, arsenic induced significant reduction in the cerebrum weight and relative cerebrum weight of rats in comparison to normal control rats (P < 0.05).
Effect of VCO and Arsenic on Body Weight, Cerebrum Weight, and Rel Cerebrum Weight in Rats.
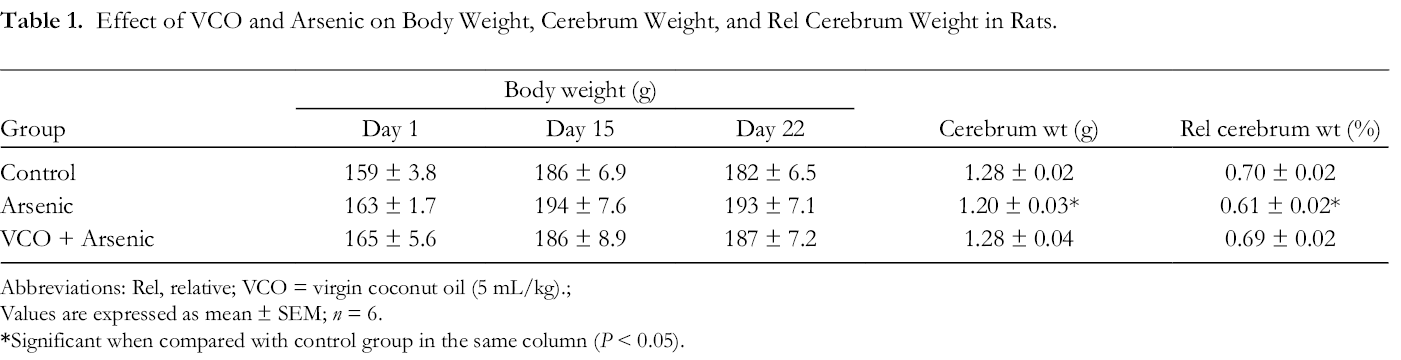
Abbreviations: Rel, relative; VCO = virgin coconut oil (5 mL/kg).;
Values are expressed as mean ± SEM; n = 6.
Effect of VCO on Oxidative Stress Markers in Arsenic-Treated Rats
Arsenic administration to rats resulted in significant decreases in cerebral activities of SOD, CAT, GPx and GSH levels, whereas the cerebral MDA level prominently increased (P < 0.05) compared to normal control rats (Table 2). Conversely, oral VCO supplementation prior and along with arsenic administration (VCO + arsenic) in rats resulted in attenuation of arsenic-elicited cerebral alterations in oxidative stress markers (P < 0.05).
Effect of Oral VCO Supplementation on Cerebral Oxidative Stress Markers-SOD, CAT, and GPx (U/mg Protein), GSH (Mg/g Protein) and MDA (Nmol/mg Protein) of Arsenic-Treated Rats.

Abbreviation: VCO, Virgin coconut oil.
Values are mean ± SEM (6 rats/group). *P < 0.05: significant when compared to control group in the same column;
Effect of VCO on Cerebral Lipid Profile of Arsenic-Treated Rats
Data in Table 3 show the effect of VCO on lipid profile of rat cerebrum treated with arsenic. Arsenic exposure significantly increased total cholesterol and LDL-C, while total phospholipid and HDL-C markedly decreased (P < 0.05). Interestingly, VCO supplementation significantly reversed the lipid alterations in comparison to the arsenic group rats (P < 0.05).
Effect of VCO on Cerebral Levels of Total Cholesterol (TC), Total Phospholipid (TPL), HDL-C and LDL-C (Mg/g Protein) of Arsenic-Treated Rats.

Abbreviation: VCO, Virgin coconut oil.
Values are mean ± SEM (6 rats/group). *P < 0.05: significant when compared to control group in the same column;
Effect of VCO on Cerebral Activities of Acetylcholinesterase (AchE), Adenosine Deaminase (ADA), and Nitric Oxide (NO) Level in Arsenic-Treated Rats
Figure 1 shows the effect of VCO supplementation on cerebral AchE, ADA and NO in arsenic-treated rats. Arsenic administration induced significant increases (P < 0.05) in AchE and ADA activities, and NO level in comparison to normal control. However, supplementation with VCO in VCO + arsenic group decreased cerebral AchE and ADA activities (P < 0.05), as well as NO compared to the arsenic group.

Effect of VCO supplementation on activities of cerebral acetylcholinesterase (AchE), adenine deaminase (ADA) and nitric oxide (NO) in arsenic-intoxicated rats. VCO: Virgin coconut oil; values are mean + SEM (6 rats/group). *P < 0.05: significant when compared to normal control group; #P < 0.05: significant when compared to arsenic group.
Effect of VCO on Arsenic-Induced Cerebral Histopathological Alteration in Rats
Effects of VCO and arsenic on histological architecture of rat cerebrum are shown in Figure 2. Arsenic-treated rats showed vacuolar and inflammatory changes as well as neuronal necrosis (arsenic group). The supplementation with VCO inhibited the arsenic-induced histopathological changes in the cerebrum comparable to the normal control (VCO + arsenic group).
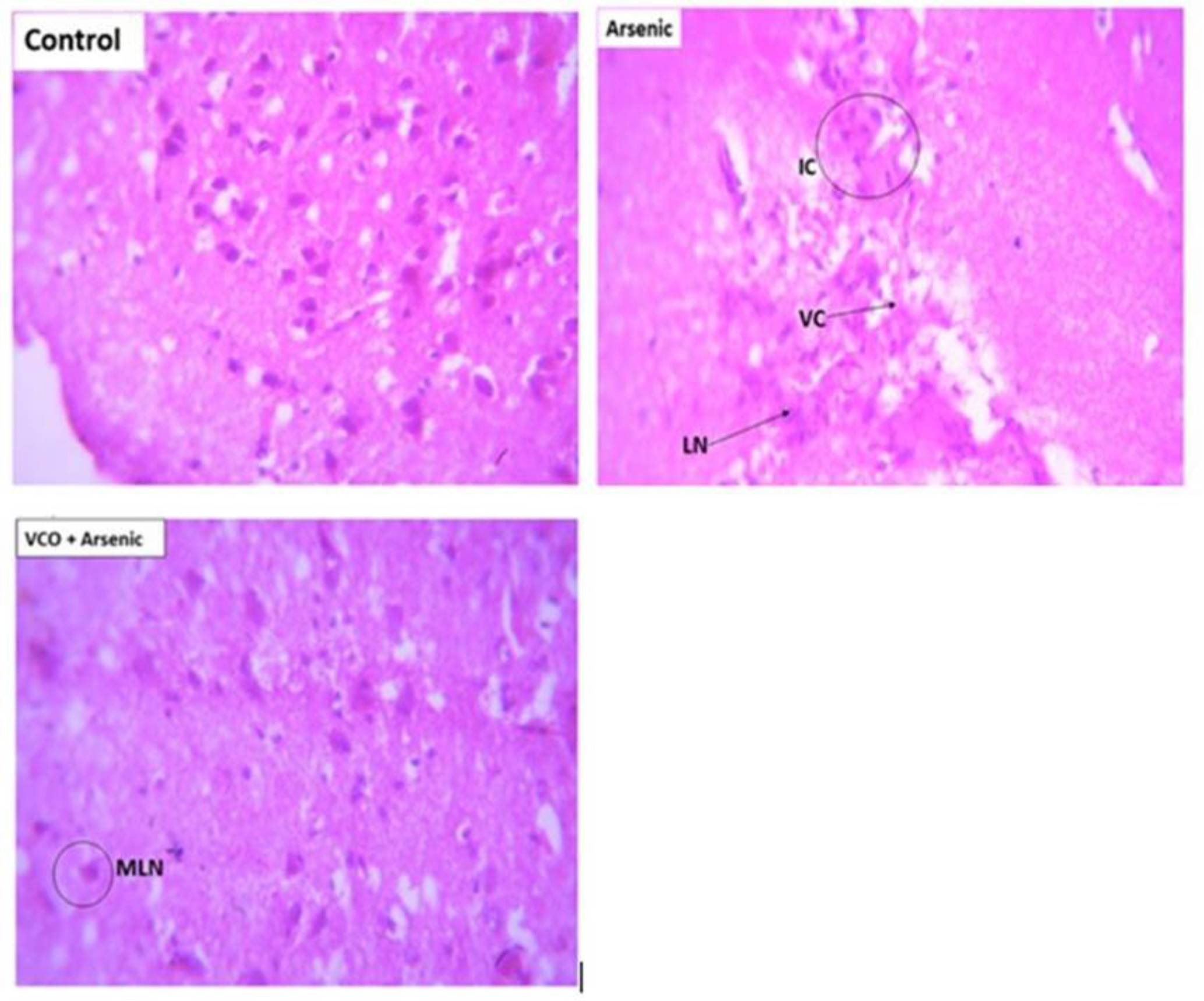
Effect of VCO on cerebral histopathology in arsenic-treated rats. Representative photomicrographs of cerebrum sections stained with haematoxylin–eosin (400 x). Control: show normal architecture. Arsenic: shows severe vacuolar changes (VC), inflammatory cells (IC) and liquefactive necrosis (LN). VCO +Arsenic: shows mild liquefactive necrosis (MLN).
Discussion
Arsenic (As) is a ubiquitous metalloid toxicant that has gained considerable attention due to its neurotoxicity strongly associated with oxidative stress mechanism via ROS generation. Some compounds, including chelating agents, have been evaluated to modulate As toxicity. 1,5 In fact, a recent study shows an ameliorative effect of combinatorial therapy of chelators with metal ions against As-induced mitochondrial oxidative stress and neurotoxicity in rat brain. 35 However, these regimens remain to be readily amenable to daily diet to prevent or reverse As toxicity. Further, the intriguing report that some synthetic antioxidants play a role in carcinogenesis has led to increasing exploration of natural products to mitigate disease development. 36 Recent studies show that natural dietary oils such as flaxseed oil, Moringa oleifera oil and Nigella sativa oil mitigate toxicities induced by arsenic, mercury and cadmium in rat models, respectively. 37 -39 Although evidence is accumulating on the pharmacological activity of VCO, its efficacy to prevent As toxicity is lacking in the literature.
The results of the present study indicate that As administration caused significant decreases in the cerebral activities of SOD, CAT, GPx, and GSH, whereas ADA, MDA, AchE, and NO increased considerably in rats. The cerebrum is a target for As toxicity associated with impaired memory, coordination and cognitive ability. 1,12 Moreover, histopathological findings supported the biochemical alterations induced by the oral As intoxication. Interestingly, VCO ingestion has a protective mechanism by restoring the altered biochemical indices near to the control levels.
Numerous studies have associated As neurotoxicity with depressed antioxidant mechanisms in brain regions. 5,40,41 SOD, CAT and GPx act in synergy to reduce ROS to maintain redox homeostasis, as the first line of defence against oxygen free radicals or ROS. However, As is a thiol (SH) group-binding toxicant; it inactivates glutathione reductase by binding to its thiol moiety which in turn culminates in a reduced level of GSH, as demonstrated in the current study. 40 The depletion of GSH by As impairs the antioxidant defence to exacerbate ROS generation and overwhelms the activities of the antioxidant enzymes shown in the findings, leading to oxidative stress and lipid peroxidation, as further indicated by increased levels of MDA. The observed MDA increase in the cerebrum is associated with oxidative stress-induced lipid peroxidation. MDA is a product of ROS attack on unsaturated fatty acid constituents of the cell membrane. The oxidative stress-mediated ROS attack and the consequent lipid peroxidation concomitantly exerted inflammatory damage on cells of the rat cerebrum in this study. This was demonstrated in our histopathological observations with inflammatory changes. Robust evidence is available on association of inflammation and increase production of NO, 42 as found in this study. The increased cerebral NO could be explained by inflammatory upregulated inducible NO synthase. 43 NO can react with superoxide radicals to produce the more potent radical peroxynitrite that causes DNA damage and cell death. However, this may suggest the markedly increased activity of ADA found in this study. It is known that DNA damage and cell death increase levels of adenosine and deoxyadenosine. 44 Extreme levels of adenosine, which is a substrate for ADA may amplify the activity of ADA in the damaged neural cells. 45 These findings are in harmony with previous studies in the liver, and brain of rats exposed to sodium arsenite. 46,47 This shows that increased levels of NO and ADA activity are involved in the pathogenesis of As-mediated oxidative damage and cerebral neurotoxicity. Our findings in this study are consistent with previous studies suggesting As-induced oxidative stress in the brain regions. 1,5,41
On the contrary, however, oral supplementation of VCO prior to and along with As ingestion inhibited the alterations observed in As-induced oxidative stress and neurotoxicity in this study. Literature suggests antioxidant supplementation to combat deleterious effects of oxidative stress in biological processes. 48 Antioxidant mechanisms target free radicals to reverse signaling cascades that favor toxicity and pathologies. Our findings here indicate that VCO possesses antioxidant activity capable of suppressing the oxidative neurotoxic effect of As in the rat cerebrum. VCO supplementation significantly increased SOD, CAT, and GPx activities comparable to the control. As a result of the improved antioxidant defence, the cerebral levels of MDA and NO, and ADA levels decreased considerably. Although As is considered a potent neurotoxicant, natural antiradical phenolic compounds, including ferulic, p-coumaric, protocatechuic, vanillic, and caffeic acids, 2 in VCO resist its mechanism of neurotoxicity and histological changes. The natural phenolics such as those found in VCO prevent ROS production, as well as neutralizing/scavenging free radicals that might inhibit the activities of antioxidant enzymes. 17,18 It is thus possible that the VCO phenolics preserve the SOD, CAT, and GPx activities by either inhibition or neutralization of ROS generation. VCO is increasingly attracting significant research attention due to its emerging pharmacological properties. 19,20,23,49 To our knowledge, this is the first study to report beneficial effects of VCO against toxicity of metals, including As. Our findings are consistent with recent evidence suggesting the role of VCO against toxicity induced by non-metals in animal models. 20,22,50
Studies suggest an association between As exposure and neurodegenerative diseases. 2 Arsenic induces alterations in brain levels of dopamine, norepinephrine, serotonin and AchE activity. 5,51 In our study, we found that As considerably increased the AchE activity in the brain. However, the use of AchE inhibitors in the treatment of Alzheimer’s disease suggests the pivotal role of this enzyme in the brain. 51 A cholinergic deficit in Alzheimer’s disease is characterized by a significant decrease in acetylcholine, which may be associated with increased activity of AchE. 52 By implication, increased AchE activity observed in our study may decrease the acetylcholine level to promote cognitive dysfunction. Our finding is consistent with previous studies that metal toxicants could activate AchE activity. 53 However, some studies suggest that As decreases AchE activity. 13 In these studies, duration, brain region and route of exposure vary in the experimental designs. 5,13 These factors are well known to influence the toxicity or effect of toxicants on biochemical molecules. It is interesting that oral VCO supplementation modulated cerebral AchE activity in the current study. It is known that bioactive agents in natural products can modulate AchE activity. 52
In the current study, As exposure impaired lipid levels in the rat cerebrum. Arsenic markedly increased TC and LDL-C levels, whereas HDL-C and TPL levels decreased significantly. Exposure to aluminum has been reported to increase TC, TG, and LDL-C in brain. 54 Literature shows that As disrupts lipid metabolism in major organs of the body. 55 Due to the blood brain barrier, circulating lipoproteins cannot reach the brain except for small HDL particles. 56 However, the lipoproteins in the brain are synthesized and transferred by the astrocytes within the brain. 57 Arsenic has been implicated to pervert astrocytic process. 58 Thus, an arsenic-induced oxidative stress damage to the blood brain barrier and astrocytes might be responsible for the lipid alterations observed in the cerebrum of the arsenic-exposed animals. Interestingly, VCO, reported to possess hypolipidemic activity, modulated the impaired levels of the lipids in this study. Studies repeatedly show reduction in body and brain weight of animals exposed to As. 58 However, As prominently reduced cerebrum weight, but not body weight of rats in this study. The effect of As in this regard corroborates the earlier report of Nagaraja and Desiraju. 59 The mechanism underlying reduction in brain weight is currently unclear, although previous studies indicate As inhibiting neurite growth via signaling pathways that trigger oxidative stress. 60 Supplementation of VCO inhibited As-induced reduction of cerebrum weight. The VCO inhibitory activity on oxidative stress may equally contribute to the beneficial effect associated with cerebrum weight.
Conclusion
Altogether, findings from this study converge to indicate that arsenic exposure induced cerebral neurotoxicity through deficits in acetylcholinesterase and adenosine deaminase activities, lipid imbalance and oxidative stress mechanism. It is interesting that VCO supplementation resisted and reversed the arsenic-induced alterations via improvement in antioxidant defence systems, and balance in adenosine deaminase and acetylcholinesterase activities in the rats’ brain.
Supplemental Material
Online supplementary file 1 - Supplemental material for Virgin Coconut Oil Resists Arsenic-Induced Cerebral Neurotoxicity and Cholesterol Imbalance via Suppression of Oxidative Stress, Adenosine Deaminase and Acetylcholinesterase Activities in Rats
Supplemental material, Online supplementary file 1, for Virgin Coconut Oil Resists Arsenic-Induced Cerebral Neurotoxicity and Cholesterol Imbalance via Suppression of Oxidative Stress, Adenosine Deaminase and Acetylcholinesterase Activities in Rats by Sharon O. Azubuike-Osu, Ademola C. Famurewa, Japheth C. David, Innocent Abi, Patience N. Ogbu, Chiedozie K. Oparaji, Konyefom G. Nwaeze and Godson G. Akunna in Natural Product Communications
Supplemental Material
Online supplementary file 2 - Supplemental material for Virgin Coconut Oil Resists Arsenic-Induced Cerebral Neurotoxicity and Cholesterol Imbalance via Suppression of Oxidative Stress, Adenosine Deaminase and Acetylcholinesterase Activities in Rats
Supplemental material, Online supplementary file 2, for Virgin Coconut Oil Resists Arsenic-Induced Cerebral Neurotoxicity and Cholesterol Imbalance via Suppression of Oxidative Stress, Adenosine Deaminase and Acetylcholinesterase Activities in Rats by Sharon O. Azubuike-Osu, Ademola C. Famurewa, Japheth C. David, Innocent Abi, Patience N. Ogbu, Chiedozie K. Oparaji, Konyefom G. Nwaeze and Godson G. Akunna in Natural Product Communications
Footnotes
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted according to the approved animal experimental procedures given by the NIH Publication (NIH Publication No. 85–23, revised 1996) on Guide for the Care and Use of Laboratory Animals, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
