Abstract
Emerging evidence indicates that NOD-like receptor protein 3 (NLRP3) inflammasome-induced inflammation plays a critical role in the pathogenesis of Parkinson’s disease (PD). Baicalein has been considered as a possible option for PD treatment based on its anti-neuroinflammatory effects. However, no studies have elucidated the precise mechanisms underlying the anti-neuroinflammatory activity of baicalein, particularly inflammasome-mediated effects. In this present study, rotenone-induced PD mice and BV2 microglia were used to investigate the anti-neuroinflammatory effects of baicalein and explore its underlying mechanism in vivo and in vitro. The results demonstrated that baicalein alleviated motor impairments and attenuated several inflammatory responses in rotenone-induced PD mice. Also, baicalein inhibited the expression of NLRP3 and activated caspase-1 in brain tissues. Correspondingly, baicalein prominently suppressed the inflammatory response in BV2 microglia induced by rotenone. Furthermore, in vitro data showed that baicalein suppressed the expression of NLRP3 and activated caspase-1 by abrogating the upregulation of ROS, as well as by inhibiting the TLR4/NF-κB signaling cascade. Overall, the results of the present study indicated that baicalein exerted anti-neuroinflammatory effects partly by inhibiting activation of the NLRP3 inflammasome, and targeting NLRP3 inflammasome signaling offers a novel therapeutic strategy for PD treatment.
Both genetic and environmental factors have been implicated in Parkinson’s disease (PD) pathogenesis, and compelling experimental evidence has indicated that inflammation, oxidative stress, mitochondrial dysfunction and protein misfolding are crucial factors in the pathogenesis of PD. 1 -3 Accumulating evidence suggests that neuroinflammation is a central mechanism of neurodegeneration in PD. Neuroinflammation is mediated primarily by microglia, which can produce pro-inflammatory cytokines. 4,5
In particular, interleukin-1 beta (IL-1β) has been recognized to be essential for the initiation and progression of PD and has been detected both in the brain and periphery of PD patients. 6,7 Therefore, suppression of microglial overactivation has been suggested as a potential strategy for delaying the progression of PD. In microglia, IL-1β secretion relies on the functions of inflammasomes. 8 Pro-IL-1β is processed by NOD-like receptor protein 3 (NLRP3), and then mature IL-1β is secreted.
The NLRP3 inflammasome is an apotosis speck-like protein containing CARD (ASC) and caspase-1. 9 The molecular mechanisms of the assembly and activation of the NLRP3 inflammasome involve a 2 step process. First, the priming signal stimulates Toll-like receptor 4 (TLR4) and enhances nuclear factor kappa B (NF-κB) driven transcription of NLRP3. 10 The second signal induces NLRP3 to form a complex with ASC and then induces the activation of caspase-1. The NLRP3 inflammasome can be activated by danger-associated molecular patterns and pathogen-associated molecular patterns, such as the generation of ROS, ATP-induced potassium (K+) efflux, and the release of cathepsin B. 11,12
NLRP3, one of the most intensively investigated inflammasomes, has been reported to play a key role in neurodegenerative diseases. 13 The NLRP3 inflammasome is mainly located in microglia and is essential to the process of neuroinflammation. 14 In particular, the NLRP3 inflammasome plays an important role in the pathophysiological mechanism in several PD models. 15,16 Therefore, pharmacological interference with NLRP3 inflammasome activity may provide novel therapeutic strategies for the treatment of microglia-mediated neuroinflammation. Also, some inhibitors of NLRP3 inflammatory pathways can alleviate the progression of PD by inhibiting central inflammation. 17
Several studies have demonstrated that baicalein (Figure 1), the predominant bioactive component isolated from Scutellaria baicalensis Georgi, protects neurons and may be an ideal drug for treating PD. It has been reported that baicalein exerted neuroprotective effects against MPTP-induced damage in C57BL/6 mice and rotenone-induced PD rats. 18 -20 The underlying mechanisms of baicalein may be related to attenuating reactive oxygen species (ROS) generation and inhibiting the NF-κB, JAK/STAT, and MAPK signaling pathways. 21,22

Chemical structure of baicalein.
A recent study suggested that baicalin mitigated cognitive impairment by suppressing NLRP3 inflammasome activation in Alzheimer’s disease (AD) models. 23 In addition, the NLRP3 inflammasome is involved in the pathogenesis of PD and might be a target for PD therapy. 24 Therefore, in this present study, we investigated the anti-neuroinflammatory mechanisms of baicalein in the context of the NLRP3 inflammasome.
Materials and Methods
Materials
The mouse microglial cell line BV2 was purchased from the American Type Culture Collection (Rockville, MD, USA), baicalein (molecular formula: C15H10O5, molecular weight: 270.24 Da, purity ≥98%) from Macklin (Shanghai, China), rotenone (C23H22O6) from Aladdin (Beijing, China), 1-(4,5-dimethylthiazol-2-yl)−3,5-diphenylformazan (MTT) from Sigma (St. Louis, MO, USA), and Dulbecco’s modified Eagle’s medium (DMEM) and fetal bovine serum from Gibco (Gibco-BRL, Gaithersburg, MD, USA). The enzyme-linked immunosorbent assay (ELISA) kits for mouse TNF-α and IL-1β were purchased from eBioscience (San Diego, CA, USA), the reactive oxygen species kit and caspase-1 activity kit from Beyotime (Beijing, China), antibodies against caspase-1 from Abcam (Cambridge, UK), and antibodies against NLRP3, TLR4, phospho-NF-κB p65 (p-p65), NF-κB p65, phospho-κBα, IκBα and GAPDH from Cell Signaling Technology (Beverly, MA, USA). Other reagents were commercially available and of analytical grade.
Cell Culture and Treatment
C57BL/6 mice (male, 18‐22 g) were purchased from Jinan Pengyue Experimental Animal Breeding Co., Ltd. (license: SCXK (LU) 20140007). All mice were housed under standard laboratory conditions. The mice were randomly assigned to 5 groups according to their body weight (control group, model group, and 3 baicalein-treated groups). As previously described, 25,26 the model group was treated with rotenone alone (1 mg/kg/day i.p. in sunflower oil), and the baicalein groups were treated with rotenone and different doses of baicalein (10 mg/kg, 30 mg/kg, and 100 mg/kg). Baicalein was prepared in physiological saline containing 1% DMSO and was intraperitoneally injected once a day 1 hours before rotenone for 3 weeks. On the 21st day, the behavior patterns of all animals were evaluated. On day 22, all mice were sacrificed, and the brains were excised and washed in ice-cold phosphate-buffed saline (0.1 M, pH 7.4) for further investigation of biochemical parameters. All data were collected and analyzed by 2 independent observers who were blinded to the group assignments of the animals.
BV2 microglial cells were cultured in DMEM containing 10% fetal bovine serum, streptomycin (100 U/mL) and penicillin (100 U/mL). The cells were maintained at 37 °C in a 5% CO2 incubator. BV2 cells were seeded in 96-well culture plates and incubated for 24 hours. Then, the cells were treated with different concentrations of either rotenone or baicalein. After 24 hours of incubation, MTT solution was added to each well and incubated for 4 hours at 37 °C. The medium was removed, the blue formazan crystals that had formed in intact cells were dissolved in DMSO, and the absorbance at 570 nm was measured using a microplate reader (Synergy H2, BioTek, USA). To measure the anti-inflammatory effects of baicalein, BV2 cells were seeded in either 96-well culture plates or 60 mm culture dishes and pretreated for 1 hours with baicalein, followed by the addition of rotenone. Cells were then incubated for different times until the respective measurements were taken.
Animals and Treatment
Rotarod Test
The rotarod test was used to assess balance and motor status in a rotarod apparatus (ZB-200, Techman Soft) in which animals walked on a rotating rod (3 cm in diameter). 27,28 For 3 days before starting the experiment, all mice were pretrained to maintain balance on the rotating rod for 30 s; during this time, any animal that fell was placed back onto the rod. In the formal experiment, the animal was placed on the rod and timed to assess their locomotor skill at a speed of 18 rpm, with a predetermined cut-off time of 120 s; the animals were assessed 3 times with an interval of 30 minutes between each round. The average retention time on the rod was calculated.
Hang Test
For the hang test, a wire (50 cm long and 0.14 cm diameter) was placed horizontally 40 cm above a foam pad. The fore paws of the mice were placed onto the wire, and the prehensile duration on the wire was recorded. The maximum descent latency time was fixed at 3 minutes. 19
Cytokine Analyses
For measuring the pro-inflammatory cytokines in mice with PD, brains were harvested quickly, and protein extracts were isolated in lysis buffer containing a proteinase inhibitor cocktail. For in vitro studies, following rotenone treatment, the cell culture supernatants were harvested, centrifuged, and stored at −80 °C until analysis. TNF-α and IL-1β levels in either the brains or cell culture supernatants were measured using specific ELISA kits (eBioscience). The procedures were performed according to the instructions with the related kits.
Analysis of NO
NO production was measured as previously described. 29 The isolated supernatants were mixed with an equal volume of Griess reagent (1% sulfanilamide and 0.1% naphthylenediamine in 5% phosphoric acid) and incubated at room temperature for 10 minutes. The optical density was measured at 540 nm using a microplate reader (Synergy H2, BioTek, USA). The concentration of NO was determined using a standard curve generated with NaNO2.
Measurement of Caspase-1 Activity
The activity of caspase-1 was determined using a caspase-1 activity kit based on the ability of caspase-1 to change acetyl-Tyr-Val-Ala-Asp p-nitroanilide (Ac-YVAD-pNA) into the yellow formazan product p-nitroaniline (pNA). The procedure was performed according to the protocol. Cellular extracts were incubated with 20 nmol Ac-YVAD-pNA overnight at 37 °C in 96-well microplates. The absorbance values of pNA were measured at 405 nm using a microplate reader (Synergy H2, BioTek, USA).
Detection of ROS Generation
ROS level was determined by using dichlorodihydrofluorescein (DCF-DA). BV2 cells were cultured in 96-well plates and pretreated with baicalein for 1 hours prior to incubation with rotenone for 24 hours. DCF-DA was added for 30 minutes, and then, the live cells were imaged with an Operetta high-content imaging system (PerkinElmer, Fremont, USA).
Immunofluorescence Staining
BV2 cells were cultured in 96-well plates and treated with baicalein (1, 3, and 10 µg/mL) for 1 hours prior to incubation with rotenone (1 µM) for either 30 minutes or 24 hours. The cells were then washed and fixed with 4% paraformaldehyde at 37 °C for 30 minutes, permeabilized with 0.3% Triton X-100 for 10 minutes and blocked with 3% BSA for 30 minutes at room temperature. Then, the cells were incubated with either anti-NF-κB p65 or anti-NLRP3 (1:200) overnight at 4 ℃ and washed 3 times with phosphate buffer saline (PBS). Next, the fluorescent secondary antibody (1:200) was added and incubated for 30 minutes at room temperature. After being washed with PBS, cells were incubated in 4',6-diamidino-2-phenylindole (DAPI) for 10 minutes in the dark. Then, the cells were washed and imaged with an Operetta high-content imaging system (PerkinElmer, Fremont, USA).
Western Blot Analysis
After the indicated treatments, either BV2 cells or brain tissues were homogenized with protease and phosphatase inhibitors. The protein concentration was determined with a BCA protein assay kit (Beyotime, Shanghai, China). Lysates (25 µg/lane) were separated by 12% SDS-PAGE and transferred onto nitrocellulose membranes. The membranes were blocked with blocking buffer (5% BSA in TBS-T solution) for 2 hours at room temperature. Then, the membranes were subsequently probed with anti-NLRP3, anti-caspase-1, anti-phospho-IκBα, anti-phospho-NF-κB p65, anti-IκBα, anti-NF-κB p65 and anti-GAPDH overnight at 4 °C. These blots were washed with TBS-T 3 times (10 minutes each) and incubated with peroxidase-conjugated anti-mouse/rabbit IgG for 2 hours at room temperature. Following a further 4 washings in TBS-T (15 minutes each), the expression of each protein was detected by the ECL Western blotting detection system (ChemiDoc TMXRS, Bio-Rad). The Western blotting results were quantified using Quantity One software (Bio-Rad), and all the band intensities were expressed relative to the control.
Statistical Analysis
All data are expressed as the means ± S.D. Statistical significance was evaluated by one-way ANOVA and post hoc tests with the LSD test for comparisons of 2 groups. A value of P < 0.05 or P < 0.01 was considered statistically significant.
Results
Effects of Baicalein on Behavioral Impairments in Rotenone-Induced PD Mice
Behavioral assessments were performed to determine the extent of rotenone-induced injury. The rotarod test has been widely used to assess coordinated motor skills, and the hang test has been used to study the neuromuscular strength of PD mice. As shown in Figure 2, rotenone injection for 21 consecutive days significantly decreased the retention time and average hanging time compared with those of the control mice (P < 0.01). However, administration of baicalein significantly improved the retention time (df = 31, F = 3.75, P < 0.05) and hanging time (df = 31, F = 9.468, P < 0.01) compared to the model group.

Effects of baicalein on the behavioral deficits in rotenone-induced PD mice. Rotarod test (
Effects of Baicalein on the Neuroinflammatory Response in Rotenone-Induced PD Mice
The inflammatory cytokines IL-1β and TNF-α play important roles in the pathogenesis of PD. Therefore, the influence of baicalein on the secretion of IL-1β and TNF-α were examined in vivo. IL-1β and TNF-α were markedly increased in the brain tissues of rotenone-induced PD mice (P < 0.01, Figure 3(A-B). Administration of baicalein greatly reduced the levels of IL-1β (df = 31, F = 4.696, P < 0.01) and TNF-α (df = 31, F = 10.192, P < 0.01) compared with those of the model group. Additionally, rotenone treatment markedly increased the protein expression of iNOS, COX-2, Iba-1 and GFAP (P < 0.01, Figure 3(C-F)). Pretreatment with baicalein significantly decreased the protein expression levels of iNOS (df = 19, F = 15.291, P < 0.01), COX-2 (df = 19, F = 20.389, P < 0.01), Iba-1 (df = 19, F = 11.375, P < 0.01) and GFAP (df = 19, F = 8.15, P < 0.01) compared with those of the model group, in a dose-dependent manner. These results confirm the anti-inflammatory activities of baicalein.

Effects of baicalein on the neuroinflammatory responses in rotenone-induced PD mice. (
Effects of Baicalein on the Rotenone-Induced BV2 Microglial Inflammation
First, the effects of baicalein on the viability of rotenone-stimulated BV2 microglia were measured. As shown in Figure 4(A),1 1 μM and 2 µM rotenone significantly reduced the viability of BV2 cells (df = 14, F = 14.701, P < 0.01). The viability of rotenone-induced cells that were pretreated with baicalein (2 µg/mL or 10 µg/mL) was significantly higher than that of cells induced with rotenone alone (P < 0.05) (Figure 4(B)). The concentrations of baicalein used alone had no effect on the viability of BV2 cells (Figure 4(C)). Moreover, rotenone significantly increased the production of NO, IL-1β and TNF-α compared to those of control BV2 cells, and pretreatment with baicalein greatly reduced the secretion of NO, IL-1β and TNF-α in a concentration-dependent manner (P < 0.05 or P < 0.01) (Figure 4(D)). Furthermore, rotenone strikingly increased the protein expression of iNOS and COX-2, which was ameliorated by pretreatment with baicalein (P < 0.05 or P < 0.01) (Figure 4(G) and (H)).

Effects of baicalein on the inflammation in BV2 microglia induced by rotenone. (
Effects of Baicalein on the Activation of the NLRP3 Inflammasome in the Brains of PD Model Mice and BV2 Microglia
To understand better the underlying anti-inflammatory mechanism of baicalein, the effects of baicalein on NLRP3 inflammasome expression were investigated in vitro and in vivo. In the in vivo study, compared with those of the model group, significant decreases in the expression levels of NLRP3 (df = 19, F = 9.357, P < 0.01, Figure 5(A)) and caspase-1 p20 (df = 19, F = 3.629, P < 0.05, Figure 5(B)) were observed in the brain tissues of baicalein-treated mice. Moreover, incubation of BV2 cells with rotenone resulted in increases in the protein expression of NLRP3 and caspase-1 p20 (P < 0.01) (Figure 5(C) and (D)). Following pretreatment with baicalein, the expression of NLRP3 and caspase-1 p20 were significantly decreased in a dose-dependent manner (Figure 5(C) and (D)).
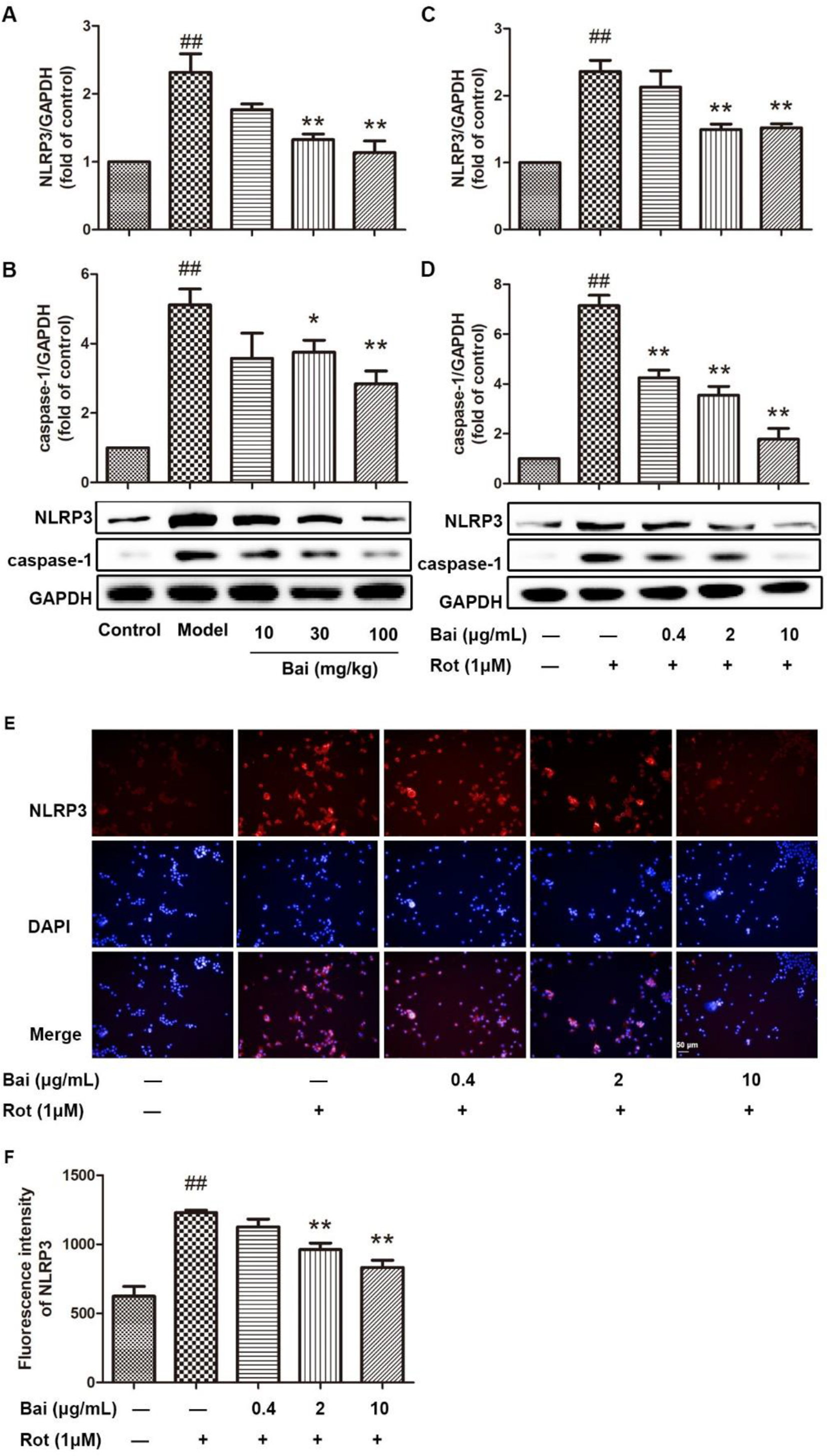
Effects of baicalein on the activation of the NLRP3 inflammasome in the brains of PD model mice and BV2 microglia. (
In addition, the effects of baicalein on NLRP3 expression were assayed using immunofluorescence staining of BV2 cells. As shown in Figure 5(E), rotenone stimulation increased the protein expression of NLRP3 in BV2 cells. Pretreatment with baicalein also significantly inhibited NLRP3 protein expression (Figure 5(E) and (F)).
Effects of Baicalein on the Activation of the NF-κB Pathway in BV2 Microglia
It is well known that the TLR4/NF-κB pathway plays an essential role in regulating the protein expression of NLRP3. The effects of baicalein on the TLR4/NF-κB pathway were investigated in microglia. Rotenone induced the phosphorylation of IκBα and p65 at 30 minutes (Figure 6(A) and (B)) and increased the protein expression of TLR4 in BV2 cells at 24 hours (Figure 6(C)). As shown in Figure 6, pretreatment with baicalein markedly decreased the protein expression of TLR4 and inhibited the phosphorylation of IκBα and p65 induced by rotenone.

Effects of baicalein on the activation of the NF-κ B pathway in BV2 microglia. (
To investigate further the effects of baicalein on the NF-κB signaling pathway, the translocation of cytosolic p65 into the nucleus was evaluated by immunofluorescence analysis. As shown in Figure 6(E), in untreated cells, the majority of the p65 subunit was distributed in the cytoplasm, as indicated by green fluorescence, and the nucleus displayed blue fluorescence due to DAPI. The addition of rotenone stimulated nuclear translocation of the majority of p65 in BV2 cells. However, pretreatment with baicalein obviously inhibited rotenone-induced p65 nuclear translocation induced by rotenone in a concentration-dependent manner (Figure 6(D) and (E)).
Effects of Baicalein on ROS Production in BV2 Microglia
The ROS has been recognized as the second signal that promotes the activation of the NLRP3 inflammasome. The effect of baicalein on ROS generation was investigated based on the fluorescence of DCF-DA. The results showed that rotenone increased the level of ROS in BV2 cells (Figure 7). As expected, baicalein significantly inhibited ROS production in a dose-dependent manner (Figure 7(A) and (B)).

Effects of baicalein on the ROS production in BV2 microglia. BV2 cells were pretreated with baicalein for 1 h prior to incubation with rotenone for 24 h. Live cell images of ROS generation based on the fluorescence of DCF-DA (green) (scale bar = 50 µm). All data are presented as mean ± SD of triplicate tests. ## P < 0.01 compared with the control group, * P < 0.05, ** P < 0.01, compared with the rotenone alone group.
Effects of Baicalein on the Activation of Caspase-1 in BV2 Microglia
NLRP3 inflammasome activation can induce caspase-1 activation. Caspase-1 activation in BV2 cells was measured by the cleavage of the caspase-1 substrate Ac-YVAD-pNA. As shown in Figure 8, rotenone increased the level of activated caspase-1 compared with that of control cells. When pretreated with baicalein, caspase-1 activation was significantly reduced.

Effects of baicalein on the activation of caspase-1 in BV2 microglia. BV2 cells were pretreated with baicalein for 1 h prior to incubation with rotenone for 24 h. Analysis of caspase-1 activity using Ac-YVAD-pNA; the activity of caspase-1 was reflected by the production of pNA. All data are presented as mean ± SD of triplicate tests. ## P < 0.01 compared with the control group, * P < 0.05, ** P < 0.01, compared with the rotenone alone group.
Discussion
In the present study, baicalein exerted anti-neuroinflammatory effects in rotenone-induced PD mice and BV2 microglia. The results demonstrated that baicalein improved behavioral impairments in PD mice. We further found that baicalein suppressed the secretion of inflammatory mediators and the expression of inflammation-related proteins both in vitro and in vivo. Moreover, baicalein inhibited NLRP3 inflammasome activation by suppressing the TLR4/NF-κB pathway and ROS production.
Previous studies suggested that baicalein has the potential to slow the progression of neurodegenerative diseases in MPTP-induced PD mice, 6-OHDA-lesioned rats, and a rotenone-induced PD model, possibly through blocking NF-κB and MAPK signals, restoring autophagy, and protecting mitochondrial function 19,20,30,31 In this study, the effectiveness of baicalein was evaluated in rotenone-induced PD mice, which offers many more advantages than other PD models. 32,33 In the present study, injection of rotenone (1 mg/kg) for 21 days induced Parkinsonian-like symptoms in mice, consistent with another report. 26 Additionally, baicalein showed dose-dependent trends in improving rotenone-induced motor impairments based on the rotarod and hang tests (Figure 2).
The inflammatory response is increasingly recognized as an important component of PD etiopathogenesis. The accumulation of pro-inflammatory and cytotoxic factors is able to destroy pathogens, but continued production can also induce toxicity in neurons, which occurs in PD. 34,35 The results indicated considerable alterations in the production of IL-1β and TNF-α in the brain tissue from mice injected with rotenone (Figure 3). The pro-inflammatory cytokines IL-1β and TNF-α can mediate the release of a large number of downstream inflammatory factors and act as potent enhancers of neuroinflammatory reactions. 36 As expected, baicalein suppressed the secretion of IL-1β and TNF-α. Moreover, baicalein inhibited the protein expression of iNOS, COX-2, Iba-1 and GFAP (Figure 3), which was consistent with previous reports. 19 Various studies have indicated that increased concentrations of iNOS and COX-2 have neurotoxic effects and are associated with several neurodegenerative diseases 37,38 The upregulation of IBA1 and GFAP expression in the PD model is an indicator of microglial and astrocyte activation. 39,40 The present data indicated that the protective effects of baicalein in rotenone-induced PD mice may be related to inhibiting neuroinflammation by acting on microglia and astrocytes to suppress pro-inflammatory cytokines.
Given the critical role of the NLRP3 inflammasome in the process of neuroinflammation, BV2 microglia were used as a model to investigate the effects of baicalein on the NLRP3 inflammasome. 17 First, the inhibitory effect of baicalein on rotenone-induced cytotoxicity in BV2 cells was studied. Treatment with 1 µM rotenone significantly diminished the survival rates of BV2 cells (Figure 4), similar to the results of a previous report. 41 Additionally, the production of NO, IL-1β and TNF-α were highly increased by rotenone (Figure 4). These results revealed that rotenone induced the activation and generation of pro-inflammatory factors in microglia. Additionally, baicalein increased the viability of BV2 cells and inhibited the production of NO, IL-1β and TNF-α while suppressing the expression of iNOS and COX-2 in rotenone-induced BV2 microglia (Figure 4). These results demonstrated that baicalein pretreatment significantly reduced rotenone-induced cytotoxicity and activation in BV2 microglia.
The accumulation of rotenone can activate NLRP3 in microglia or in an environmental pesticide toxicant-driven mouse model of PD. 42 As expected, the expression of NLRP3 and cleaved caspase-1 were significantly increased in rotenone-induced BV2 microglia and PD mice (Figure 5). Previous studies reported that baicalein reduced NLRP3 activation and IL‐1β production in LPS-stimulated macrophages/BV2 cells and AD mice. 23,43 In contrast, it has still not been demonstrated that baicalein can inhibit the NLRP3 inflammasome in a PD model. In our study, we first found that pretreatment with baicalein effectively decreased the protein expression of NLRP3 and cleaved caspase-1 in both PD mice and BV2 cells (Figure 5).
It is well known that NLRP3 inflammasome activation depends on the NF-κB signaling pathway (first signal) and ROS (second signal). 44 -46 Additionally, numerous studies have demonstrated that baicalein suppresses inflammation through the inhibition of the TLR4/NF-κB signaling pathway in different cells or disease models. 47 In this study, Western blotting and immunofluorescence assays were used to study TLR4/NF‐κB signaling. These results showed that TLR4 was activated, as indicated by its upregulation in rotenone‐induced BV2 cells (Figure 6). In contrast, baicalein significantly inhibited the upregulated expression of TLR4 and suppressed the phosphorylation of IκBα and p65 (Figure 6). In particular, baicalein inhibited the subsequent nuclear translocation of NF‐κB p65. All those data suggest that the effects of baicalein on the activation of NLRP3 inflammasome were mediated via downregulation of the TLR4/NF‐κB signaling pathway.
ROS have been shown to be a critical mechanism that triggers NLRP3 inflammasome formation and activation. 6 Moreover, accumulating evidence has suggested that baicalein is a free radical scavenger in various neurological diseases. 48,49 As shown in Figure 7, rotenone-induced ROS production was significantly reduced by baicalein in BV2 cells. Additionally, baicalein inhibited the activation of caspase-1 (Figure 8), which is responsible for the secretion of IL-1β. These findings indicate that baicalein attenuated NLRP3 inflammasome activation by suppressing ROS production.
In addition to ROS production, more recent publications reinforce the idea that mitochondrial impairment in microglia can amplify NLRP3 inflammasome signaling. 42 Furthermore, rotenone, a specific mitochondrial complex I inhibitor, can induce mitochondrial dysfunction. 50 Furthermore, a previous report showed that baicalein partially enhanced mitobiogenesis to restore mitochondrial function, thus exerting therapeutic effects in rotenone-induced PD models. 28 Baicalein may prevent NLRP3 inflammasome activation by restoring mitochondrial function. These hypothesis require further study.
Conclusion
Overall, this study demonstrated that baicalein could improve behavioral impairments, inhibit microglial activation, and alleviate neuroinflammation. Moreover, the mechanisms underlying the anti-inflammatory effects of baicalein may involve the suppression of NLRP3 inflammasomes and blockage of the TLR4/NF‐κB signaling pathway and ROS production. These findings provide a new basis for the beneficial effects of baicalein administration during PD development.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science Foundation of China (NO.81803546), Natural Science Foundation of Shandong Province (No. ZR2018LH024) (ZR2017MH068).
