Abstract
Keywords
Introduction
Parkinson's disease (PD), characterized by dopaminergic neuronal loss in the striatum (ST) and substantia nigra (SN) pars compacta (SNPC), is one of the most common chronic neurodegenerative disorders worldwide. 1 Patients with dopamine deficiency in the basal ganglia exhibit movement disorders such as tremors, rigidity, akinesia, bradykinesia, and abnormal postural adjustments. 2
PD is commonly treated with drugs that enhance intracerebral dopamine concentrations or stimulate dopamine receptors, the most commonly used drug being levodopa (L-DOPA).3,4 However, currently available therapies for PD offer only symptomatic relief.3,4 In addition, deep brain stimulation (DBS) can be used to treat PD; however, both L-DOPA and DBS have little to no impact on slowing or stopping the underlying neurodegenerative processes. 5 As such, there is an urgent need to develop therapeutic medicines that have fewer side effects and that impede disease progression.
PD is often accompanied by chronic neuroinflammation, mitochondrial dysfunction, and the presence of intracytoplasmic α-synuclein-rich inclusions known as Lewy bodies.1,6 Persistent inflammatory responses contribute to the progressive loss of dopaminergic neurons in PD. 7 Further, it is well known that neuroinflammation is predominantly mediated by activated glial cells and is accompanied by the production of inflammatory cytokines. 8 Astrocytes and microglia are cellular mediators of brain inflammation, and the production of pro-inflammatory cytokines by activated glial cells can result in neuroinflammation and neuronal dysfunction.8,9 Inflammasome-related neuroinflammation plays a crucial role in the progression of PD. Inflammasomes are multiprotein complexes, of which the NLR family pyrin domain-containing 3 (NLRP3) inflammasome is the most studied protein complex involved in inflammatory processes.10,11 The NLRP3 inflammasome is composed of an NLRP3 sensor, a signaling adapter, apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), and caspase-1 protease. 6
The activation of NLRP3 inflammasome cleaves pro-caspase-1 into its bioactive form, which subsequently induces the caspase-1-mediated release of interleukin 1β (IL-1β). 6 The NLRP3 inflammasome has attracted attention as a pivotal player in the pathophysiology of PD. In the brain of patients with PD, the NLRP3 inflammasome pathway is activated by oxidative stress. 6 Recently, it has been demonstrated that the NLRP3 inflammasome is a potential treatment target for PD, 12 with research demonstrating an elevated NLRP3 expression in the mesencephalic neurons of patients with PD, 13 and an upregulation in microglia during PD. 14
Traditional medicines comprising natural products are promising sources of new ingredients for the development of pharmaceutical interventions. We previously reported the use of medicinal herbs and their active ingredients in the treatment of stroke 15 and cancer. 16 In the present study, we evaluated the neuroprotective effects of the major active ingredients of Saposhnikovia divaricata (Turcz.) Schischkin (SD) on a mouse model of PD, identifying cimifugin as a potential neuroprotective compound in mouse models of PD. This represents the first report of the effects of cimifugin on PD. As such, we investigated whether cimifugin mitigates Parkinson-related motor deficiencies and the underlying mechanisms. Overall, we propose cimifugin as a potential therapeutic agent for the treatment of PD.
Results
Recovery Effects of Cimifugin, POG and SOG on the Motor Dysfunction in MPTP-Induced PD Model
We evaluated the effects of three compounds of Saposhnikovia divaricata Schischkin (SD) extract on PD symptoms in an 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced mouse model (Figure 1). Because we found that SD improved functional motor outcomes in the PD model (Supplementary Figure S1). Dose-dependent effects of SD were observed using three behavioral tests (traction, pole, and rotarod tests). Furthermore, mice treated with SD were found to exhibit significant increases in tyrosine hydroxylase (TH) expression in both the SNPC and ST of the MPTP-induced group (Supplementary Figure S1). Therefore, we tested the effects of three major components of SD extract 17 : cimifugin, prim-o-glucosylcimifugin (POG), and sec-O-glucosylhamaudol (SOG) (Figure 1). Mice were pretreated with cimifugin (1 mg/kg), POG (1 mg/kg), and SOG (1 mg/kg), and functional motor outcomes following MPTP injection were analyzed (Figure 1a). In the traction test to measure muscle strength, the hanging time score decreased in the MPTP group (Veh) and significantly increased in both the cimifugin-treated (Cimi) and POG groups (*P < 0.05 vs control group. #P < 0.05, and ##P < 0.01 vs the MPTP-induced group; Figure 1b). The positive control group treated with ropinirole (1 mg/kg; Ropi) also showed recovery effects (###P < 0.001 vs the MPTP-induced group). Bradykinesia was measured using the pole test. In the pole test, the time to go down was significantly increased in the MPTP group (Veh) compared to the control group (*P < 0.05 vs control group), which was significantly decreased by all of the three tested compounds, cimifugin (Cimi), POG, and SOG, compared to the MPTP group (#P < 0.05, and ##P < 0.01 vs the MPTP-induced group). In the rotarod test, the latency time was significantly shorter in the MPTP group than in the control group (*P < 0.05 vs control group). Moreover, cimifugin (Cimi) treatment significantly increased the latency time on the rod compared to the MPTP group (#P < 0.05, vs the MPTP-induced group). Based on our findings, we further examined the effects of cimifugin, which demonstrated significant effects similar to the positive control, ropinirole (Ropi) in all three analyses.

Effects of cimifugin, POG, and SOG on motor dysfunction in MPTP-induced PD model mice. (a) Experimental protocol. Mice were injected with a single compound (cimifugin, prim-o-glucosylcimifugin, or sec-O-glucosylhamaudol, 1 mg/kg dissolved in 10% methanol in distilled water, ip, n = 4 per each group) for 7 days. MPTP was intraperitoneally injected into mice for 5 consecutive days starting on the third day (n = 4). The vehicle group (MPTP only, no drug) was intraperitoneally administrated an equal volume of 10% methanol in distilled water. Ropinirole was used as a positive control (n = 4). Behavioral tests were conducted on the fifteenth and sixteenth day. (b) The improvement effect on functional motor outcome was assessed through the traction test (hanging time score), pole test (time to go down), and rotarod test (latency time). Ropinirole was used as a positive control. Cimi, Cimifugin; POG, Prim-o-glucosylcimifugin; SOG, Sec-o-glucosylhamaudol; Ropi, Ropinirole. *P < 0.05 versus control group. #P < 0.05, ##P < 0.01, and ###P < 0.001 versus the MPTP-induced group. PD, Parkinson's disease ; MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine.
Dose-Dependent Effects of Cimifugin on Motor Dysfunction and Dopaminergic Neuronal Loss in MPTP-Induced Mouse PD Model
To examine the dose-dependent effects of cimifugin on motor function in the PD model, mice were intraperitoneally injected with different concentrations of cimifugin (0.3, 1, and 3 mg/kg) and subjected to three tests (traction, pole, and rotarod) (Figure 2). In the traction test, the hanging time score significantly increased with doses of 1 and 3 mg/kg compared to the MPTP-induced group (#P < 0.05, and ##P < 0.01 vs the MPTP group; Figure 2a). In the pole test, the 1 and 3 mg/kg cimifugin groups showed a shorter time to fall (#P < 0.05, and ##P < 0.01 vs the MPTP group; Figure 2b). In the rotarod test, the latency time was significantly increased in the cimifugin-treated groups at doses of 1 and 3 mg/kg compared to that in the MPTP-induced group (#P < 0.05 vs the MPTP group; Figure 2c).

Dose-dependent effect of cimifugin on motor function recovery. (a) Traction test (hanging time score), (b) pole test (time to go down), and (c) rotarod test (latency time) were performed. Cimi; cimifugin. Ropi; ropinirole. Control, n = 8; MPTP, n = 8; Cimi, n = 8/each concentration; Ropi, n = 5. *P < 0.05, **P < 0.01, and ***P < 0.001 versus control group. #P < 0.05 and ##P < 0.01 versus the MPTP group. MPTP, 1-methyl-4-phenyl-1,2,3,6- tetrahydropyridine.
Subsequently, we investigated whether dopaminergic neurons in the SNPC and ST were affected by cimifugin (Figure 3a-c). Mouse brain sections were immunostained for TH, revealing low TH levels in the SNPC and ST of the MPTP group. In contrast, mice treated with cimifugin exhibited a significant increase in TH expression in both the SNPC and ST regions compared to MPTP group (Veh) (##P < 0. 01 and ###P < 0.001 vs the MPTP group; Figure 3). Treatment with the positive control ropinirole triggered an increase in TH immunostaining in both SNPC and ST regions as compared to Veh. These findings suggest that cimifugin administration mitigates MPTP-induced dopaminergic neuronal loss.
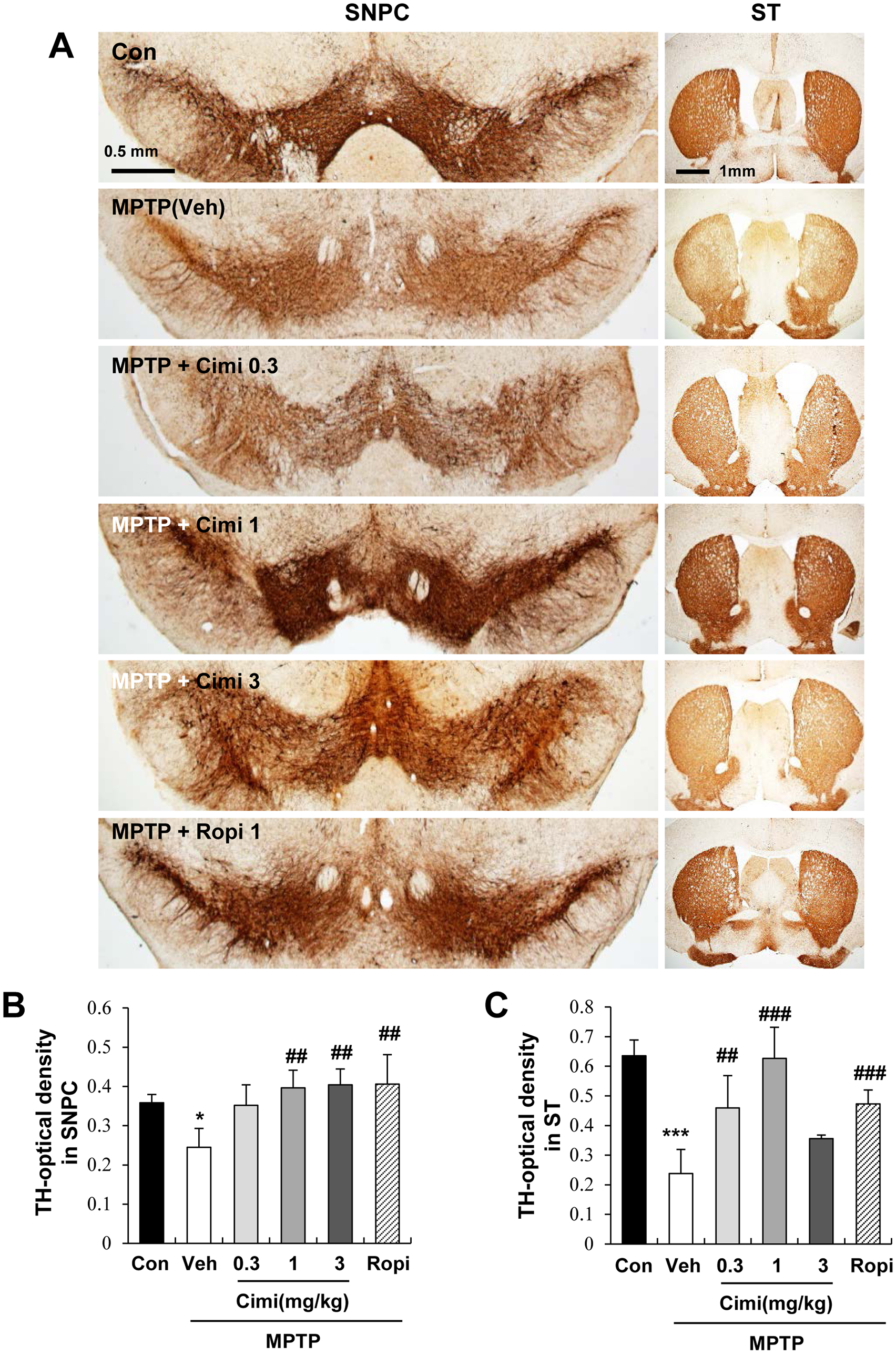
Dose-dependent effect of cimifugin on dopaminergic neuron rescue. (a) Brain sections were stained with tyrosine hydroxylase (TH). Mice treated with cimifugin (0.3, 1, 3 mg/kg) showed increased expression of the dopaminergic neuron marker, TH. Ropinirole was used as a positive control (Ropi). (b, c) Quantification graphs of TH immunostaining in SNPC (b) and ST (c) (n = 5 each). ∗P < 0.05 and ***P < 0.001 versus control group. ##P < 0. 01 and ###P < 0.001 versus the MPTP group. MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; ST, striatum; SNPC, substantia nigra pars compacta.
Cimifugin Protects Neuroinflammation in MPTP-Induced Mouse PD Model
As cimifugin was found to reduce dopaminergic neuronal loss, we further evaluated its effects on neuroinflammation (Figure 4). We observed immunoreactivity against GFAP (an astrocyte marker) and Iba-1 (a microglial marker). Significantly higher levels of GFAP and Iba-1 immunoreactivity were observed in the SNPC of the MPTP group than in those of the control group (Figure 4a-c). In contrast, cimifugin treatment (1 mg/kg) significantly decreased the GFAP and Iba-1 levels in the SNPC region. We also observed immunoreactivity against GFAP in the ST region. Significantly higher level of GFAP immunoreactivity was observed in the ST of the MPTP group than in that of the control group (Supplementary Figure S2a-S2b). In contrast, cimifugin treatment also significantly decreased the GFAP level in the ST region. These data indicate that cimifugin protects against the neuroinflammation and neuronal dysfunction caused by astrogliosis and microgliosis.
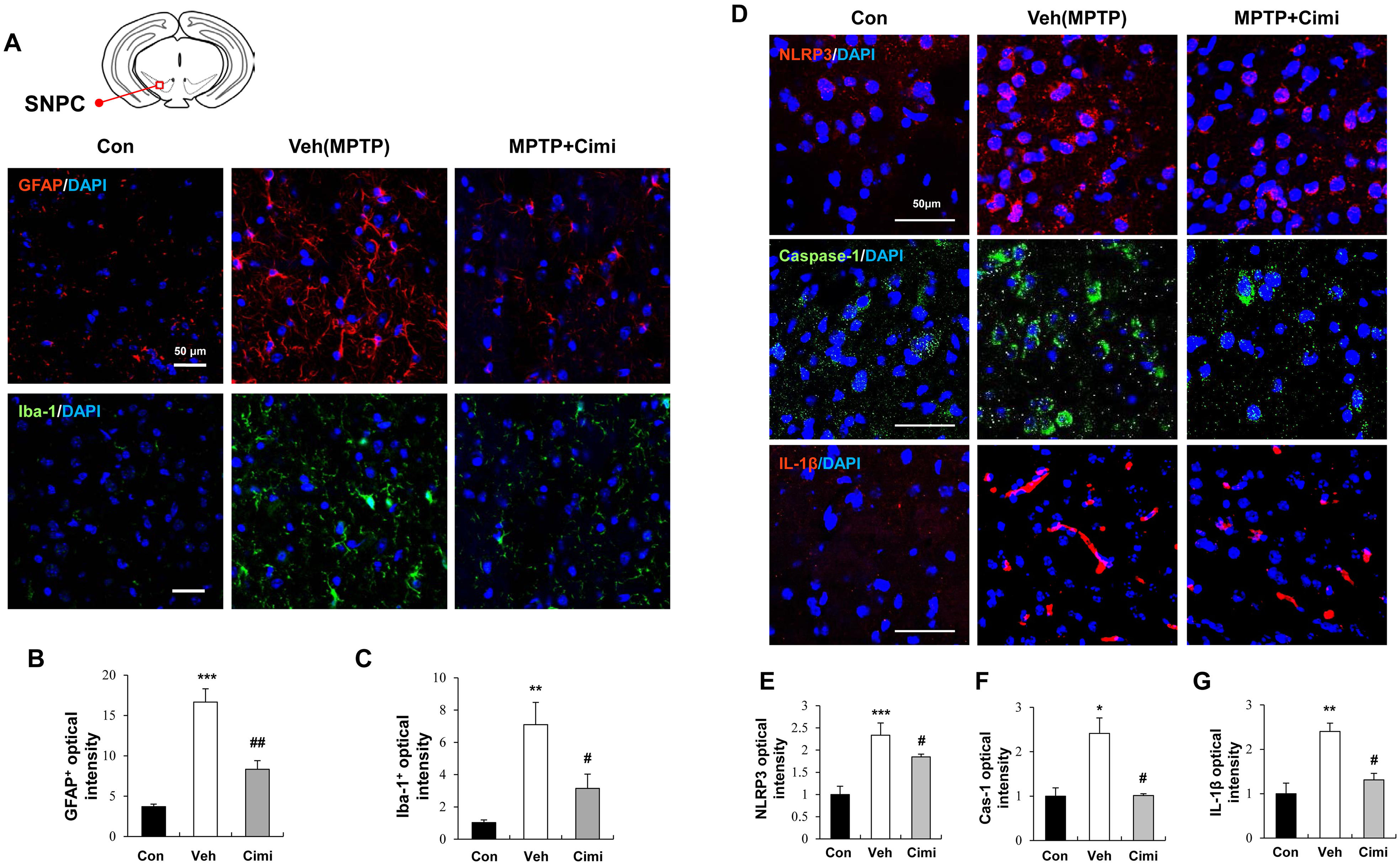
The effect of cimifugin on neuroinflammation signals in the SNPC of PD model mice. (a) Immunofluorescence staining for GFAP (red) and Iba-1 (green). Nuclear DNA was counterstained with DAPI (blue). Magnification:×200. Scale bar = 50 μm. The schematic diagram shows the regions of the SNPC of the brain. (b, c) Quantification of GFAP and Iba-1 fluorescence intensity. Data are expressed as the mean ± SEM (n = 5). Veh (MPTP group); Cimi (MPTP + cimifugin 1 mg/kg). **P < 0.01 and ***P < 0.001 versus control group. #P < 0.05 and ##P < 0.01 versus veh group. (d) Immunofluorescence staining for NLRP3 (red), caspase-1 (green), and IL-1β (red) in the SNPC. Nuclear DNA was stained with DAPI (blue). Magnification:×400. Scale bar = 50 μm. (e-g) Quantification of NLRP3, caspase-1 (Cas-1), and IL-1β fluorescence integrated optical density in SNPC. Data are expressed as the mean ± SEM (n = 4). *P < 0.05, **P < 0.01, and ***P < 0.001 versus control group. #P < 0.05 versus the vehicle group. PD, Parkinson's disease. MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; IL-1β, interleukin 1β; SNPC, substantia nigra pars compacta; SEM, standard errors of the mean.
Next, we investigated whether cimifugin affects the NLRP3 inflammasome (Figure 4d-g, Supplementary Figure S2a and S2c). Once activated, the NLRP3 inflammasome cleaves pro-caspase-1 into the bioactive form caspase-1, which subsequently induces the release of the pro-inflammatory cytokine IL-1β. 6 We found that NLRP3, caspase-1, and IL-1β immunoreactivity were high in the MPTP group, but were significantly decreased by cimifugin treatment (1 mg/kg). These results indicate that cimifugin may protect neuronal cell death by suppressing the NLRP3/caspase-1/IL-1β pathway.
To confirm this, we investigated whether cimifugin functionally improved the NLRP3-mediated aggravation of motor function. As shown in Figure 5, treatment with monosodium urate (MSU) crystals (an NLRP3 agonist) combined with cimifugin reversed the protective effect of cimifugin on motor function in the PD model (†P < 0.05 vs the cimifugin group; Figure 5). These findings indicate that pretreatment with cimifugin decreases the aggravation of motor function, possibly by suppressing the NLRP3 inflammasome.

The protective effect of cimifugin on motor dysfunction is mediated by NLRP3 reduction. (a) Experimental protocol. Mice were injected with cimifugin (1 mg/kg, ip, once a day for 7 days). Mice were further intraperitoneally injected with MPTP for 5 consecutive days starting on the third day. Mice were injected with an NLRP3 agonist, MSU crystals, or PBS (10 mg/kg, intraperitoneally) once a day for 4 days before the behavioral test. Behavioral tests were conducted on the fifteenth and sixteenth day. (b) Quantification graphs of traction test (hanging time score), pole test (time to go down), and rotarod test (latency time) were shown to evaluate functional outcomes. Con (n = 3); Veh (n = 4), MPTP only, Cimi (n = 5), cimifugin; Cimi + M (n = 5), cimifugin + MSU crystal. *P < 0.05 versus control group. #P < 0.05 versus the vehicle group. †P < 0.05 versus the cimifugin group. MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MSU, monosodium urate; PBS, phosphate-buffered saline.
Discussion
Overall, this study investigated the neuroprotective effects of cimifugin, one of the main components of the dried roots of Saposhnikovia divaricate. Cimifugin was found to protect against motor deficits and dopaminergic neuronal death. Furthermore, cimifugin reduced neuroinflammation by inhibiting NLRP3/caspase-1/IL-1β signaling in an MPTP-induced mouse PD model.
The dried roots of Saposhnikovia divaricate, traditionally called “Fang-Feng,” are used for the treatment of pyrexia, rheumatism, headaches, vertigo, generalized aches and pains, and arthralgia in traditional Korean medicine. 18 Prior studies have reported that the major active ingredients of SD are POG, SOG, and cimifugin. 17 SOG and POG were found to suppress LPS-induced inflammation in RAW264.7 macrophages.19,20 Cimifugin was shown to ameliorate lipotoxicity-induced hepatocyte damage by inhibiting the TLR4/p38 MAPK pathway 21 and suppresses allergic inflammation.22,23 Lipopolysaccharide-induced inflammation was also suppressed by cimifugin through inhibition of the signaling MAPKs and NF-κB pathways. 24 Among these three ingredients, the cimifugin-treated group showed significantly improved Parkinson-related motor deficiency, similar to the positive control ropinirole-treated group in behavioral tests (Figure 1).
PD is accompanied by chronic neuroinflammation, which contributes to the progressive loss of dopaminergic neurons in PD. 7 Cimifugin represses inflammation;22,23 however, whether cimifugin exerts neuroprotective effects against PD currently remains unclear. In this study, we observed that cimifugin protected against dopaminergic neuronal cell death in the MPTP-induced mouse model in a dose-dependent manner, as measured by TH-positive staining (Figure 3).
Neuroinflammation is mediated by activated glial cells and is accompanied by the production of inflammatory cytokines. 8 The NLRP3 inflammasome has been reported to be a potential target for PD. 12 The NLRP3 inhibitor MCC950 was further shown to suppress inflammasome activation and mitigate motor deficits in multiple rodent models of PD. In addition, NLRP3 deficiency significantly reduced motor dysfunction and dopaminergic neurodegeneration in a mouse model of PD. 25 Caspase-1 deficiency alleviated dopaminergic neuronal death in an MPTP-induced mouse model of PD. 26 In the present study, we found that cimifugin inhibits NLRP3 inflammasome activation and reduces active caspase-1 and IL-1β (Figure 4). Moreover, MSU crystal, an NLRP3 agonist, reversed the protective effects of cimifugin on motor function in the PD model (Figure 5), suggesting the possible involvement of the NLRP3 inflammasome in cimifugin-mediated neuroprotection.
Our study has several limitations. While cimifugin was found to mitigate motor deficits and suppress the NLRP3 inflammasome, and NLRP3 agonist treatment eliminated the neuroprotective effect exhibited by cimifugin, our results have not established the precise mechanisms by which sustained NLRP3 activation drives dopaminergic neuron survival. Another limitation of our study is that the mechanism of cimifugin-mediated NLRP3 reduction was not explored.
In conclusion, cimifugin, which exerts a treatment benefit on Parkinson-related motor deficiency, showed neuroinflammation suppression and inhibited NLRP3/caspase-1/IL-1β signaling in a mouse PD model. These findings suggest that cimifugin is a potential therapeutic agent targeting the NLRP3 inflammasome to suppress neuroinflammation in patients with PD.
Materials and Methods
Animal Experiments and MPTP Mouse Model of PD
Male C57BL/6 mice were purchased from Hana Biotech (Ansan, Korea). The mice were housed under a 12-h light/dark cycle and allowed ad libitum access to food and water. The animal protocol used in this study was reviewed and approved by the Pusan National University-Institutional Animal Care and Use Committee (PNU-IACUC) as per their ethical procedures and scientific care, and it has been approved (PNU-2019-2224). To generate the MPTP-induced PD model, mice were intraperitoneally injected with MPTP hydrochloride (dissolved in 0.9% saline, 30 mg/kg, ip; M0896, Sigma-Aldrich, St. Louis, MO, USA) for 5 consecutive days (vehicle group), while the control group was intraperitoneally administrated an equal volume of normal saline. 27
Drug Administration
Cimifugin, POG, SOG, and ropinirole (Sigma-Aldrich, St. Louis, MO, USA) were dissolved in 10% methanol in distilled water at a concentration of 1 mg/kg. Cimifugin (0.3 mg/kg, 1 mg/kg, and 3 mg/kg, n = 4-8 each), POG (1 mg/kg, n = 4), SOG (1 mg/kg, n = 4), and ropinirole (1 mg/kg, n = 4-5) (Sigma-Aldrich, St. Louis, MO, USA) were intraperitoneally administrated to the mice once daily for 7 days before and during MPTP injection. The vehicle group (MPTP only, no drug, n = 4-8) was administrated an equal volume of 10% methanol in the distilled water intraperitoneally. The NLRP3 agonist, MSU crystals, were injected into mice (dissolved in 0.9% saline, 10 mg/kg, intraperitoneally, n = 5; Tocris, Bristol, UK) once a day for 4 days before the behavioral test.
Pole Test
The pole test was performed to measure bradykinesia in the MPTP-induced mouse model. Experiments were conducted on a wooden pole with a diameter of 1 cm and a height of 50 cm. The mice were held head-downward at the top of the pole, and the time required for them to come down to the floor and place their feet was recorded. Each mouse underwent three trials, from which the times were averaged.
Rotarod Test
Balance and motor coordination were assessed using a rotarod test. The mice were placed on a rod (Panlab S.L.U., Barcelona, Spain), and the average latency until falling from the spinning rod under continuous acceleration (4-40 rpm) was measured. Mice underwent pre-training for 3 days prior to MPTP injection, after which they were placed on a rotating rod at a speed of 8 rpm on the first day, 15 rpm on the second day, and 18 rpm on the third day. Each mouse was subjected to five trials.
Traction Test
The traction test was performed to measure the muscle strength of mice in the MPTP-induced PD model. Each mouse was suspended on a horizontal metallic wire, and forced to hang with both forepaws. The hanging time was scored as follows: 0 = 0-4 s; 1 = 5-9 s; 2 = 10-14 s; 3 = 15-19 s; 4 = 20-24 s; 5 = 25-29 s; and 6 = ≥ 30 s
Immunohistochemistry
After the behavioral tests, mouse brains were harvested, perfused with cold phosphate-buffered saline (PBS), and fixed for 24 h in 4% paraformaldehyde at 4 °C, after which they were cryoprotected in 30% sucrose for 72 h at 4 °C. All brains were then individually frozen in optical cutting temperature compound (Sakura Finetek, Torrance, CA) and stored at −80 °C. Frozen brains were cut into 40 µm slices using a CM 3050 cryostat (Leica Microsystems, Wetzlar, Germany) and stored in storage buffer (30% ethylene glycol and 0.01% polyvinylpyrrolidone phosphate, pH 7.4) at −20 °C until analysis. The serial coronal sections (thickness, 40 µm) in the ST (bregma:0.97 to 0.13) and SN (bregma: −2.79 to −3.39) were permeabilized with PBS containing 0.3% Triton X-100 for 30 min and then incubated with 3% hydrogen peroxide (H1009, Sigma-Aldrich, St. Louis, MO) for 10 min. The brain sections were blocked with 10% normal goat serum (S-1000; Vector Labs, Burlingame, CA, USA) or 10% normal horse serum (S-2000; Vector Labs) for 1 h. Immunohistochemistry was performed using the Vectastain Elite ABC reagent kit (PK-6100, Vector Labs, Burlingame, CA, USA). For dopaminergic neuron staining, the sections were immunostained with anti-TH (MAB318, Millipore, Burlington, MA) overnight at 4 °C, followed by biotinylated anti-mouse IgG (BA-9200, Vector Labs) at room temperature for 1 h. The 3,3′-Diaminobenzidine substrate kit (SK-4100, Vector, Stuttgart, Germany) was used as a chromogenic agent to visualize the staining. Images were obtained using an AxioScope.A1 microscope (Carl Zeiss, Jena, Germany) and analyzed using ImageJ software (NIH, Bethesda, MDUSA).
Immunofluorescence Staining
Frozen brains were sectioned (thickness, 40 µm) using a CM 3050 cryostat (Leica Microsystems, Wetzlar, Germany). Brain sections were permeabilized with 0.5% Tween-20 for 30 min, and then immunostained with anti-glial fibrillary acidic protein (GFAP; Z0334, Dako, Glostrup, Denmark), anti-Iba-1 (019-19741, Wako, Osaka, Japan), anti-NLRP3/NALP3 (NBP1-77080, Novus biological, Centennial, CO), anti-caspase-1 (14F468, Novus biological, Colorado, USA), and anti-IL-1β (SC-32294, Santa Cruz, Biotechnology Inc. Dallas, TX) overnight at 4 °C, following which they were incubated with Alexa 488 (A-11001, A-11008; Invitrogen Corporation, Carlsbad, CA) or Alexa 594-conjugated secondary antibodies (A-11005, A-11037; Invitrogen Corporation) for 2 h in total darkness. Nuclei were stained with 4’,6-diamidino-2-phenylindole (DAPI; D1306, Invitrogen Corporation) and mounted with Vectashield mounting medium (H-1400, Vector Labs). Fluorescent images were visualized using a laser scanning confocal microscope (FV1000; Olympus, Tokyo, Japan) and quantified using the ImageJ software (National Institutes for Health, Bethesda, MD, USA).
Statistical Analysis
Data are represented as the means ± standard errors of the mean. All analyses of significance were performed using Sigma plot version 11.2 software (Systat Software, San Jose, CA, USA). Two groups were compared using unpaired Student's t-tests, whereas the analysis of more than two groups was performed using one-way or two-way analysis of variance, followed by Tukey's post-hoc test. Statistical significance was set at P < 0.05.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241265938 - Supplemental material for Cimifugin Improves Motor Function Through Suppression of the NLRP3 Inflammasome in an Animal Model of Parkinson's Disease
Supplemental material, sj-docx-1-npx-10.1177_1934578X241265938 for Cimifugin Improves Motor Function Through Suppression of the NLRP3 Inflammasome in an Animal Model of Parkinson's Disease by Ji Yun Lee, Min Jae Kim, Byung Tae Choi, Young Ju Yun, Seo-Yeon Lee and Hwa Kyoung Shin in Natural Product Communications
Supplemental Material
sj-jpg-2-npx-10.1177_1934578X241265938 - Supplemental material for Cimifugin Improves Motor Function Through Suppression of the NLRP3 Inflammasome in an Animal Model of Parkinson's Disease
Supplemental material, sj-jpg-2-npx-10.1177_1934578X241265938 for Cimifugin Improves Motor Function Through Suppression of the NLRP3 Inflammasome in an Animal Model of Parkinson's Disease by Ji Yun Lee, Min Jae Kim, Byung Tae Choi, Young Ju Yun, Seo-Yeon Lee and Hwa Kyoung Shin in Natural Product Communications
Supplemental Material
sj-jpg-3-npx-10.1177_1934578X241265938 - Supplemental material for Cimifugin Improves Motor Function Through Suppression of the NLRP3 Inflammasome in an Animal Model of Parkinson's Disease
Supplemental material, sj-jpg-3-npx-10.1177_1934578X241265938 for Cimifugin Improves Motor Function Through Suppression of the NLRP3 Inflammasome in an Animal Model of Parkinson's Disease by Ji Yun Lee, Min Jae Kim, Byung Tae Choi, Young Ju Yun, Seo-Yeon Lee and Hwa Kyoung Shin in Natural Product Communications
Footnotes
Author Contributions
Participated in research design: Ji Yun Lee, Seo-Yeon Lee, Byung Tae Choi, and Hwa Kyoung Shin. Conducted experiments: Ji Yun Lee and Min Jae Kim. Contributed new reagents or analytic tools: Byung Tae Choi and Young Ju Yun. Performed data analysis: Ji Yun Lee, Seo-Yeon Lee, and Hwa Kyoung Shin. Wrote or contributed to the writing of the manuscript: Ji Yun Lee, Seo-Yeon Lee, and Hwa Kyoung Shin.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal protocol used in this study was reviewed by the Pusan National University-Institutional Animal Care and Use Committee (PNU-IACUC) as per their ethical procedures and scientific care, and it has been approved (PNU-2019-2224).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (grant number 2019R1A2C1087515, to YJY), National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (grant number 2020R1A2C1012564, to SYL).
Institutional Review Board Statement
Not applicable
Statement of Human and Animal Right
All procedures in this study were conducted in accordance with the Pusan National University-Institutional Animal Care and Use Committee (PNU-IACUC) as per their ethical procedures and scientific care, and it has been approved (PNU-2019-2224).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
