Abstract
Objective/background
The potential for natural products to work synergistically with chemotherapy to reduce side effects and enhance its effectiveness has garnered significant attention. This study aims to evaluate the potential synergistic effect of baicalein, a flavone, in enhancing the chemosensitivity of cisplatin in the MDA-MB-231 triple-negative breast cancer cell line.
Methods
The impact of baicalein-cisplatin combination on cytotoxicity, migration, colony formation, and apoptosis was investigated.
Results
Baicalein exhibited a cytotoxic effect against the MDA-MB-231 cell line, with an IC50 of 44 µM, whereas cisplatin demonstrated an IC50 of 3.5 µM. Cells treated with cisplatin-baicalein combination at 1:4 ratio demonstrated increased sensitivity to cisplatin, showing a twofold reduction in the IC50. Notably, reductions of 7.23, 12.99, 23.37, and 34.84-fold in the ED50, ED75, ED90, ED95, respectively of cisplatin against MDA-MB-231 cells were observed in the baicalein-cisplatin combinations. The combination of 1.5 µM of cisplatin with 6 µM of baicalein resulted in a significant reduction in the number of colonies compared to 1.5 µM of cisplatin alone (p < .05). Baicalein failed to enhance the anti-migratory effect of cisplatin at all studied combinations after 72 h in comparison with cisplatin alone. Baicalein (6 and 3 µM) and cisplatin (3 µM) increased CASP3 while baicalein (6 and 3 µM) and cisplatin (3, 1.5 µM) decreased BCL2 gene expression in MDA-MB-231 cells. Their combinations were not significantly different from the corresponding concentration of cisplatin alone.
Conclusion
This study demonstrated for the first time that baicalein enhances the cytotoxicity of cisplatin in MDA-MB-231 cells by increasing apoptosis, lowering cisplatin's IC50 and inhibiting colony formation. This synergy may reduce the required doses and side effects, supporting baicalein's potential as a chemotherapy adjuvant.
Introduction
Breast cancer is a leading cause of mortality in women.1–4 Triple-negative breast cancer (TNBC) is an aggressive type of cancer that accounts for about 15% of breast cancer cases. Compared to other types, it is associated with a worse prognosis and a higher rate of recurrence. 5 TNBC lacks hormone receptors and HER2 expression, which precludes the use of hormonal and targeted HER2 therapies.6–8 Various chemotherapeutic agents and regimens have been utilized to treat TNBC, aiming to improve survival rates of patients and reduce recurrence. For example, platinum agents (Carboplatin, cisplatin) promote DNA cross-linking and are effective in BRCA1/2-mutated TNBC. 9 However, these agents have severe side effects, including multiple organ toxicity and drug resistance. 10 Similarly, other chemotherapeutic agents used in the treatment of TNBC also cause adverse effects. These include anthracyclines (such as doxorubicin and epirubicin), taxanes (including paclitaxel and docetaxel), and capecitabine, a prodrug that is metabolized into 5-fluorouracil. 11
Combining two or more drug therapies has proven effective in breast cancer treatment by enhancing therapeutic outcomes and overcoming drug resistance through synergistic interactions.12–15 Examples of combinations include FEC regimens, which comprise 5-Fluorouracil, Epirubicin, and Cyclophosphamide, and AC-T regimens, which incorporates Doxorubicin, Cyclophosphamide, and Paclitaxel. 16 Despite being effective, combination therapies also come with increased toxicity. 17
The potential use of natural products in combination with chemotherapeutic medications for TNBC treatment has garnered significant attention with the goal of achieving synergistic effects, and minimizing the side effects associated with chemotherapy. 18 Several clinical studies have been conducted to validate the efficacy of such combinations. In a randomized, double-blind, placebo-controlled study, combining curcumin with paclitaxel demonstrated greater effectiveness in treating patients with metastatic and advanced breast cancer compared to paclitaxel alone. 19 An open-label, phase I dose-escalation trial was conducted to evaluate the feasibility and tolerability of curcumin-docetaxel combination in 14 patients diagnosed with metastatic or locoregionally recurrent advanced breast cancer. Improvements in biological and clinical responses were observed in most patients, with no disease progression noted in any case. Additionally, the combination of curcumin and docetaxel significantly reduced vascular endothelial growth factor levels after three treatment cycles. 20 Clinical studies have demonstrated that treatment with ginsenoside Rg3 can enhance the anti-tumor efficacy of chemotherapeutic agents—such as capecitabine, docetaxel, and cisplatin—in patients with cancer. 21
Several combinations of natural products and chemotherapy were tested in vitro, yielding promising results.
22
Dietary isothiocyanate Erucin (4-methylthiobutyl isothiocyanate), found in cruciferous vegetables, increased Paclitaxel's antiproliferative and pro-apoptotic actions in human breast cancer cells.
23
Another example is the co-administration of Rosmarinic acid and Thymoquinone with Docetaxel, which has shown promise as an adjuvant therapy in MDA-MB-231 TNBC cells.
24
Various naturally occurring flavonoids have demonstrated synergistic effects with chemotherapy in TNBC, offering potential to enhance treatment efficacy and reduce drug resistance.
25
Quercetin, found in onions, enhanced the antitumor efficacy of docetaxel by inducing apoptosis and modulating the PI3K/AKT, MAPK/ERK, and JAK/STAT3 signaling pathways in the MDA-MB-231 cells.
26
Isorhamnetin, found in sea buckthorn (Hippophae rhamnoides), induced cell cycle arrest and apoptosis through promoting DNA damage and modulating the AMPK/mTOR/p70S6K signaling pathway in doxorubicin-resistant breast cancer.
27
(-)-Epigallocatechin gallate, present in green tea, sensitized breast cancer cells to paclitaxel in a murine model of breast carcinoma.
28
Combining the citrus flavonoid naringenin and metformin with doxorubicin enhanced its anticancer activity against TNBC in vitro and in vivo.
29
Additionally, Rhoifolin, found in citrus limon
Baicalein (5,6,7-trihydroxyflavone) (Figure 1A) is a naturally occurring flavonoid found in the roots of the plant Scutellaria baicalensis Georgi (Lamiaceae) and Oroxylum indicum (L.) Kurz (Bignoniaceae). 31 Baicalein demonstrated significant antioxidant, anti-inflammatory, and anticancer activities against different cancer cell lines including MDA-MB-231 cells, a TNBC cell line. 32 This flavonoid has multifaceted anticancer properties. It inhibits proliferative signaling pathways like PI3K/Akt and MAPK, induces apoptosis, 33 halts the cell cycle, reduces angiogenesis 34 and inhibits metastasis by targeting matrix metalloproteinases (MMPs) 33 and epithelial mesenchymal transition. 35 Moreover, baicalein's anti-inflammatory effects suppress cancer progression. Importantly, it overcomes radioresistance and chemoresistance, enhancing the efficacy of existing treatments. 35

Structure of Baicalein (A) and Cisplatin (B). The Images Were Obtained via the PubChem Web Platform (https://pubchem.ncbi.nlm.nih.gov).
Cisplatin (Figure 1B) is a small inorganic molecule with a central platinum (Pt2+) atom coordinated in a square planar geometry. It has two ammonia ligands and two chloride ions arranged in a cis configuration, which is essential for its anticancer activity. 36 Cisplatin and baicalein may interact through coordination between the platinum center of cisplatin and the hydroxyl groups of baicalein, potentially forming Pt–O bonds. Additionally, baicalein's hydroxyls could form hydrogen bonds with cisplatin's ammine ligands. These interactions may influence drug stability, solubility, or cellular uptake, enhancing cisplatin's cytotoxicity.
Synergistic effects were demonstrated using baicalein in combination with chemotherapeutic agents in different cancer types. For example, baicalein enhances the sensitivity of A549 lung adenocarcinoma cells to cisplatin. 37 In a hepatocellular carcinoma cell line baicalein enhanced the cytotoxicity of 5-Fluorouracil. 38 In TNBC MDA-MB-231 cells, it enhanced the cytotoxic effect of Doxorubicin. 32 In the present investigation, the presence of a synergistic effect of the cisplatin-baicalein combination on TNBC was investigated in vitro. The effect of the baicalein–cisplatin combination was investigated in MDA-MB-231 cells, which represent a triple-negative, mesenchymal-like, and highly aggressive subtype of breast cancer. This subtype is typically more resistant to conventional therapies and associated with a poorer prognosis compared to hormone receptor-positive lines like MCF7.
This study aims to investigate whether the combination of baicalein and cisplatin produces a synergistic effect in treating TNBC, as suggested by preclinical evidence from other cancer types. The MDA-MB-231 cell line, representative of aggressive TNBC, serves as a suitable model to explore this potential therapy. If effective, the combination could enhance treatment efficacy and reduce the required cisplatin dose, thereby minimizing its dose-limiting toxicities—such as nephrotoxicity, neurotoxicity, and gastrointestinal side effects—while improving patient tolerance and quality of life. Baicalein's anti-inflammatory, antioxidant, and anti-metastatic properties may further support systemic tolerance and reduce long-term complications, including treatment-induced secondary malignancies and chronic organ damage. Furthermore, this strategy may help overcome cisplatin resistance, expanding therapeutic options for patients with recurrent or chemoresistant TNBC. In neoadjuvant and adjuvant settings, the combination could improve tumor response, surgical outcomes, and recurrence prevention. Personalized treatment approaches, guided by tumor profiling, may further enhance its clinical utility. Further in vivo and clinical studies are needed to confirm safety, determine optimal dosing, and assess long-term benefits before clinical application.
Materials and Methods
Cell Proliferation Assay
The MDA-MB-231 cell line was purchased from the American Type Culture Collection (ATCC® HTB-26™), USA. The cell line (passage number 22) was grown in a humidified 5% CO2 incubator at 37 °C and in DMEM-high glucose medium (Euroclone, Italy) containing 10% fetal bovine serum, 10 g/L penicillin-streptomycin and 10 g/L L-glutamine. Baicalein (Sigma-Aldrich, USA) was dissolved in dimethyl sulfoxide (DMSO; ChemCruz, USA) to prepare the stock solution and ensuring that the final DMSO concentration remained below 0.1%. Cisplatin was obtained from Biosynth Carbosynth (UK).
Cytotoxicity of cisplatin, baicalein and their combinations was assessed using the MTT assay (3-[4,5-dimethyl-2-thiazolyl]-2,5-diphenyl tetrazolium bromide) with an MTT assay kit (Invitrogen, USA). MTT is a colorimetric method based on the reduction of MTT by mitochondrial dehydrogenases in viable cells. This assay was selected for its sensitivity, reproducibility, and compatibility with high-throughput screening in multi-well plates, allowing semi-automated processing and rapid result acquisition using a microplate reader. In this test, active mitochondria in viable cells reduce the yellow tetrazolium salt MTT to form an insoluble purple formazan crystal. The intensity of the blue color corresponds to the number of viable cells, with a darker blue color indicating a higher cell count.
Cells seeded into 96-well plates at a density of 8 × 103 cells per well in DMEM were incubated for 24 h and then treated with either vehicle, baicalein, cisplatin, or a combination of both drugs for 72 h. Synergistic effects were assessed using CompuSyn software (version 1.0), which is freely available online at https://compusyn.software.informer.com/1.0. Combination treatments of cisplatin and baicalein were applied at fixed ratios of 1:1, 1:2, and 1:4. The combination index (CI) values were then calculated, where a CI less than one indicates synergism, CI that equals one indicates an additive effect, and CI more than one indicates antagonism. 30
Colony Formation Assay
The colony-forming or clonogenic assay is a quantitative in vitro test used to evaluate the ability of a single cell to proliferate and form a colony through clonal expansion. Six hundred cells were seeded in DMEM medium supplemented with 10% fetal bovine serum using 6 well plates. After 24 h, the cells were treated with cisplatin, baicalein or their combination at the indicated concentrations and incubated at 37 °C in 5% CO2 for 72 h. Then, the medium was replaced with fresh one and cells were allowed to grow for a duration of 15 days. Cells were then washed with phosphate buffered saline (PBS), fixed with 4% paraformaldehyde for 1 h, rinsed again with PBS and stained with a 0.1% crystal violet solution. 30 Subsequently, plates were photographed, and colonies containing ≥50 cells were counted manually within each well.
Scratch (Wound Healing) Assay
The scratch (wound healing) assay is a simple and widely used in vitro technique for evaluating cell migration and proliferation. It involves creating a linear gap in a confluent monolayer of cells, after which the cells at the edges of the wound begin to migrate and proliferate toward the center until the gap is closed. In this assay, MDA-MB-231 cells were seeded in 12-well plates at a density of 9 × 104 cells/ml. Once confluence was achieved, a uniform scratch was made using a sterile 200 μL pipette tip. The wells were then gently rinsed with PBS to remove cellular debris. Cells were subsequently treated with cisplatin, baicalein, or their combination. All treatments were performed in triplicate. Plates were imaged after 24, 48, and 72 h of treatment using a Nikon microscope camera (Japan), and wound areas were quantified using Motic software version 2 (Motic China Group Co., Ltd) as previously described. 30 The wound area percentage was calculated using the formula: Wound area percent = wound area at specific time in hours/wound at zero time in hours *100.
Gene Expression of Caspase-3, BCL2 and BAX
RNA extraction and q-PCR were conducted as follows: The MDA-MB-231 cell line (106 cells) was seeded in DMEM medium. On the following day, the culture medium was aspirated and replaced with fresh DMEM. Subsequently, the cells were treated with baicalein (12, 6 or 3 μM), cisplatin (3, 1.5 or 0.75 μM), or a combination of both drugs at the indicated concentrations. Gene expression analysis was performed 72 h post-treatment with the drugs. Cells were then washed with PBS and collected by scraping for further analysis. Total RNA was extracted using a Quick- RNA Miniprep Kit (Zymo, USA Cat. # R1055) according to the manufacturer's instructionsTotal RNA (1 µg), extracted from cells, was used for cDNA synthesis using a 2720 Thermal Cycler (Applied Biosystems, USA) and the ProtoScript First Strand cDNA Synthesis Kit (New England Biolabs, USA; Cat. #E6300S), in accordance with the manufacturer's instructions. The GAPDH gene served as the internal control. Real-time q-PCR was conducted using TB green master mix (Takara, Japan, Cat. # RR82LR) and the QuantStudio5 (Applied Biosystems by Thermo Fisher) under the following conditions: 95 °C for 30 s one cycle. Then, 95 °C for 5 s and 62 °C (GAPDH and Caspase 3), 65.5 °C (BCL2 and BAX) for 60 s, 72 °C for 60 s for 40 cycles. A subsequent melting curve step at 60–95 °C. Primers were procured from Macrogen (South Korea), and the sequences of sense and antisense primers for CASP3, BCL2, BAX, and GAPDH genes are listed in Table 1. All gene expression data were normalized by dividing the expression level of each target gene by that of GAPDH, which was used as an internal control for each sample.
Sequences of Primers.

Statistical Analysis
The data were subjected to analysis using one-way analysis of variance (ANOVA), followed by post-hoc Tukey's test, utilizing GraphPad Prism version 8 (San Diego, USA). Statistical significance was defined as p value less tahn .05. Values were presented as mean ± standard deviation (SD).
Results
Baicalein Synergistically Enhances the Cytotoxic Effect of Cisplatin on MDA-MB-231 Cell Line
As shown in Figure 2, both baicalein and cisplatin decreased the viability of MDA-MB-231 cells after 72 h, with IC50 values of 44.0 ± 2.0 µM for baicalein and 3.50 ± 0.70 µM for cisplatin (Figure 2, Table 1 Supplementary)

Cell Viability of MDA-MB-231 Cell Line Tested by the MTT Method After 72 h of Incubation with (A) Baicalein (B) Cisplatin. Values are the Average of Triplicate and Presented as Mean ± SD. Cis: Cisplatin.
Cells treated with cisplatin-baicalein at 1:4 ratio demonstrated increased sensitivity to cisplatin with 2 folds reduction in the IC50 while the other ratios showed reduction in the cisplatin sensitivity (Figure 3B). The data for the proliferation inhibitory rates of the 1:4 ratios were substituted into Compusyn software for further analysis.
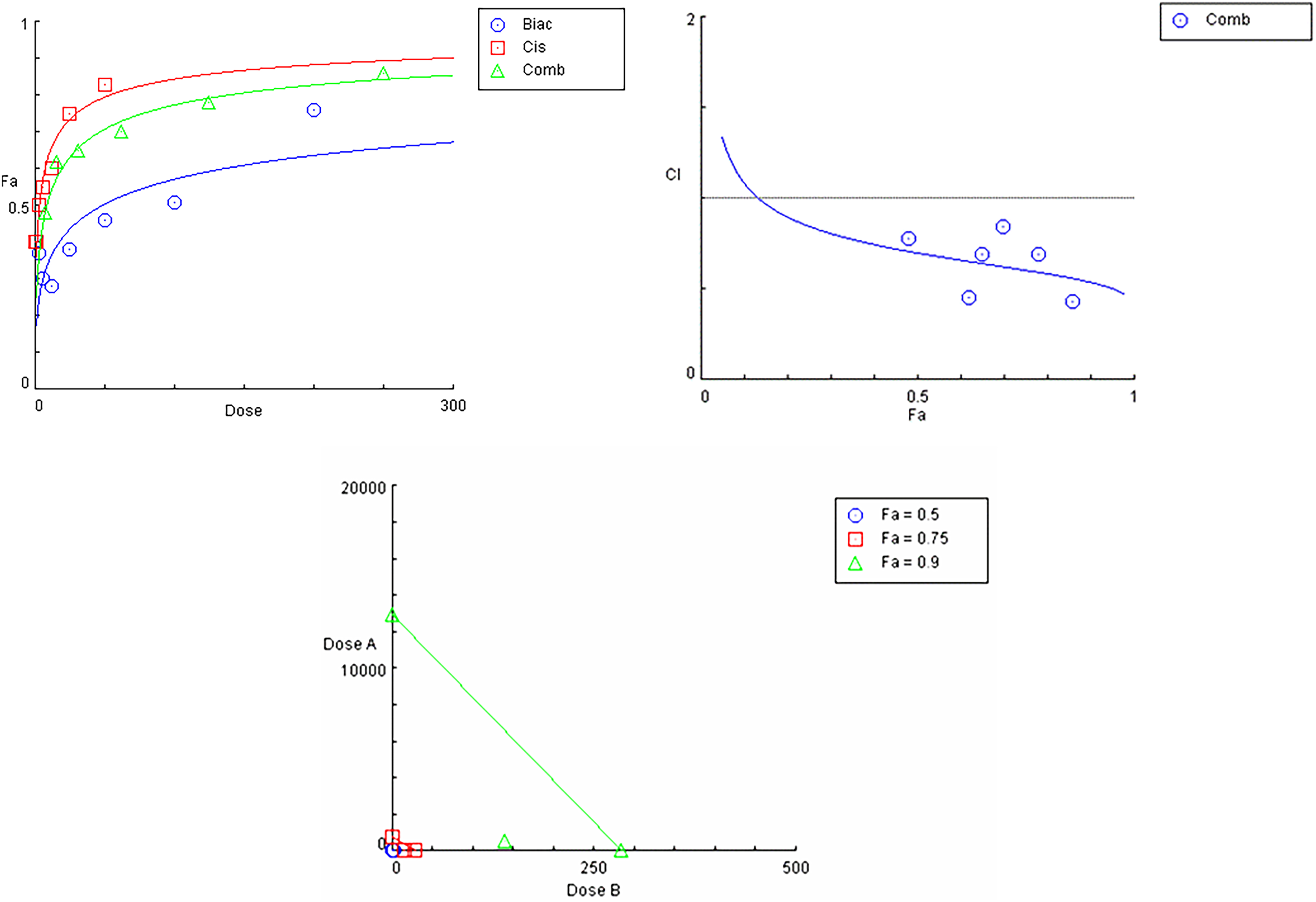
Effect Curves were Generated from the CompuSyn Calculation. A, Dose Response Curve for (1:4 Cisplatin; Baicalein), B. Combination Index Plot Among 6 Combinations. C. Isobologram for Combination of the Data Indicates a Synergistic (CI < 1).
The lowest combination index was found at the combination ratio of 1:4 (cisplatin: baicalein) indicating the presence of a synergistic effect. Therefore, this ratio was employed for further experiments. A flat sigmoidal (m < 1) curve was observed for cisplatin and baicalein with a linear correlation coefficient (r) of approximately 0.98 (Figure 3A). The CompuSyn software calculated combination index (CI) values for the experimental data, revealing synergistic interactions, as evidenced by CI values ranging from 0.428 to 0.798. These results indicate that most tested ratios exhibited a synergistic effect against MDA-MB-231 cells. As illustrated in Figure 3B, the CI–Fa plots further confirmed that CI values at various effect levels were consistently below 1. However, at the 1:1 and 1:2 ratios, the CI values ranged from 3.293 to 0.854 and from 3.078 to 0.562, respectively, suggesting less synergy or even antagonism at lower effect levels for these specific ratios.
Isobologram analysis at ED90 (Figure 3C) demonstrated that the data points for the drug combinations fell below the line of additivity, indicating a synergistic interaction against MDA-MB-231 cells. To identify the most synergistic combination, CI values for the cisplatin: baicalein (1:4) ratio were examined and found to be less than 1 at ED50, ED75, ED90, and ED95, confirming a consistent synergistic effect across multiple effect levels (Table 2). Also, analysis of dose-reduction index (DRI) data was performed. Results showed that a 7.23, 12.99, 23.37 and 34.84- fold reduction in the ED50, ED75, ED90, and ED95, respectively of cisplatin against MDA-MB-231 cells in the baicalein combinations were found.
Summary of CompuSyn Simulated CI and DRI Values for Cisplatin and Baicalein Combination in MDA-MB-231 Cell Line at 50%, 75%, 90% and 95% Growth Inhibition.

Colony Formation Assay
Treatment with cisplatin alone (3, 1.5, 0.75 µM) or baicalein alone (12, 6, 3 µM) resulted in a significant decrease in colony formation compared to the control (Figure 4, Figure 1 Supplementary). Interestingly, combining 3 µM of cisplatin with 12 µM of baicalein or 1.5 µM of cisplatin with 6 µM of baicalein resulted in a significant reduction in the number of colonies compared to cisplatin alone (p < .05). However, the combination of low doses of cisplatin (0.75 µM) and baicalein (3 µM) produced no significant reduction in colony formation compared to each drug alone.

Effect of Baicalein, Cisplatin and Their Combination on Colony Formation Capability by MDA-MB-231. (A) Colony Number is Shown After 12 µM of Baicalein and 3 μμ of Cisplatin Treatment Versus Control. (B) Colony Number is Shown After 6 µM of Baicalein and 1.5 μμ of Cisplatin Treatment, Their Combination and the Control (C) Colony Number is Shown After 3 µM of Baicalein and 0.75 μμ of Cisplatin Treatment, Their Combination and the Control After 15 Days. Results are Presented as Mean ± SD of Three Repeats. *p < .05, **p < .01, ***p < .0001, ****p < .0001.
Baicalein Enhanced the Anti-Migratory Effect of Cisplatin on MDA-MB-231 Cell Line
The closure percent of MDA-MB-231 was reduced following baicalein and cisplatin (Figure 5). Baicalein (12 µM) had a wound area percentage of 70% ± 7.1, compared to 9.3 ± 3.27of the control at 72 h. The wound area percentage was significantly higher than the control in all treatment groups—baicalein (6 µm: 84.5% ± 0.5), cisplatin (1.5 µM: 64.3% ± 2.6), and the combination therapy (75.0% ± 4.0)—indicating impaired wound closure. The low dose of baicalein (3 µM) showed the same trend following 72 h (63% ± 5.7) compared to the control (17% ± 1.2). Also, cisplatin had a significant anti-migratory effect against MDA-MB-231 at the three tested doses. The effect of baicalein-cisplatin combination on migration was also evaluated. Baicalein failed to enhance the anti-migratory effect of cisplatin at all studied combinations after 72 h in comparison with cisplatin alone (Figure 5).

Representative Images of the Scratch Assay in MDA-MB-231 Following Treatment with Baicalein, Cisplatin, or their Combination for 72-h. Results are Presented as mean ± SD of Three Repeats. *p < .05, **p < .01, ***p < .0001, ****p < .0001.
Effect of Baicalein, Cisplatin and Their Combination on the Expression of Genes Related to Apoptosis in MDA-MB-231 Cell Line
A significant increase in the expression of CASP3 was observed after baicalein (12, 6 and 3 µM), cisplatin (3 µM) and two of their combinations (Figure 6). Also, cisplatin (3, 1.5 µM) and baicalein (12, 6 and 3 µM) decreased BCL2 and two of their combination (Figure 6). Similarly, baicalein, cisplatin, and their combination produced a significant decrease in BAX expression in the MDA-MB-231 cell line.

Effect of Baicalein, Cisplatin, and Their Combination on Caspase 3 (A-C), BAX (D-F) and BCL2 (G-I) Gene Expression After 72 h. MDA-MB-231 Cells Were Treated with 0.75 μM of Cisplatin, 3 μM of Baicalein, or Their Combination, or 6 µM of Baicalein, 1.5 μμ of Cisplatin or Their Combination or 12 µM of Baicalein, 3 μμ of Cisplatin or Their Combination. Results are Presented as Mean ± SD of Three Repeats. *p < .05, **p < .01, ***p < .0001, ****p < .0001.
Discussion
In the current study, baicalein reduced MDA-MB-231 cell viability and enhanced cisplatin's cytotoxicity synergistically at a 1:4 ratio, significantly lowering cisplatin's IC50 and enabling dose reduction. This combination inhibited colony formation and apoptosis-related gene expression by increasing CASP3 and decreasing BCL2. Baicalein chemosensitized TNBC cells to doxorubicin by restoring autophagy markers, suppressing CDK1 upregulation, and reducing Drp1 expression and phosphorylation. These effects were reversed by the autophagy inhibitor 3-methyladenine, which also blocked baicalein's ability to enhance apoptosis and reduce cell viability in doxorubicin-treated MDA-MB-231 cells. 32 Likewise, baicalein (5-20 µM) sensitized A549/CDDP cells to ciprofloxacin rather than being cytotoxic. 37 Baicalin, baicalein, and wogonin are key active flavonoids in Scutellaria baicalensis Georgi (Chinese skullcap), a traditional herb widely used in Chinese medicine. This plant has been shown to enhance the anticancer effects of cisplatin by inhibiting tumor growth in vitro and in vivo, while also alleviating cisplatin-induced cachexia and acute kidney injury. 39
MTT primarily assesses mitochondrial activity; thus, baicalein's direct effects on mitochondria may result in outcomes that reflect either metabolic disruption or actual cytotoxicity. To accurately distinguish between these possibilities, complementary assays—such as neutral red uptake, resazurin reduction, sulforhodamine B (SRB), or Annexin V/Propidium iodide staining—should be utilized to assess diverse cellular functions. Incorporating assay validation and methodological diversity is essential for reliable interpretation of cytotoxic effects.
The capacity of baicalein to hinder colony formation suggests that it might exert its effect by inhibiting the cell cycle. In the present investigation, baicalein demonstrated a synergistic impact when combined with cisplatin in clonogenic assay. This agrees with previous studies that have documented the cell cycle inhibitory effect of cisplatin. 40 Furthermore, it was reported that baicalein induced G1 phase arrest in MDA-MB-231 cells. This was accompanied by a reduction in cyclin D1 and cyclin-dependent kinase 4 expression, along with an increase in CDK inhibitors such as p21 and p27. These alterations disrupt cell cycle progression and inhibit cellular proliferation. 34
In the current investigation, baicalein had anti-migratory effect against human TNBC cell line. Similar findings were obtained by other researchers who reported that baicalein suppresses the metastatic potential of MDA-MB-231 cells by inhibiting MMPs, particularly MMP-2 and MMP-9, which play a crucial role in extracellular matrix degradation and cancer cell invasion. 41 Similarly, cisplatin had pronounced anti-migratory activity as reported earlier. 42 However, when combined together, baicalein failed to enhance the anti-migratory activity of cisplatin.
Natural polyphenolic compounds like ellagic acid derivatives exhibit antioxidant and antiproliferative effects in breast cancer cells, supporting their therapeutic potential. 43 This aligns with our findings that baicalein enhances cisplatin's cytotoxicity and modulates apoptosis-related genes in MDA-MB-231 cells, suggesting that such compounds can improve chemotherapy efficacy through complementary mechanisms. The findings that baicalein increased CASP3 and reduced BCL2 in the current investigation agrees with previous studies which reported that baicalein induced apoptosis in MDA-MB-231 cells via both intrinsic and extrinsic pathways. Moreover, baicalein upregulated pro-apoptotic protein expression such as BAX and CASP3, while it downregulated anti-apoptotic proteins like BCL2 in a time-dependent manner. 44 A significant increase in CASP3 expression observed after treatment with baicalein (3, 6, and 12 µM), cisplatin (1.5 and 3 µM), and two of their combinations suggests a potential activation of apoptotic pathways. This upregulation, coupled with the concurrent decrease in the anti-apoptotic protein BCL2 under the same treatment conditions, further supports the induction of apoptosis. However, to determine whether these increases in CASP3 expression are biologically meaningful, it is essential to correlate them with functional apoptosis assays such as Annexin V staining, which detects early apoptotic events. Without such correlation, elevated CASP3 expression alone does not confirm caspase-3 activation or effective apoptosis, since caspase-3 must be cleaved to its active form to execute cell death. Therefore, integrating CASP3 expression data with Annexin V results or other apoptosis markers is necessary to validate that the observed molecular changes translate into increased apoptotic cell death. Future studies have to give a detailed investigation of the signaling pathways of apoptosis.
The observed upregulation of CASP3 and downregulation of BCL2 in MDA-MB-231 cells treated with baicalein and cisplatin suggests the activation of apoptotic pathways, indicating a pro-apoptotic shift in cellular signaling. Although the combination treatments did not show a statistically significant difference compared to cisplatin alone, these findings still offer valuable mechanistic insight into how baicalein may influence apoptosis-related gene expression in the context of chemotherapeutic treatment. Apoptosis is regulated by a complex network of signaling pathways, with CASP3 serving as a key executioner protease that orchestrates the dismantling of the cell during the terminal stages of apoptosis. The downregulation of BCL2 further promotes apoptotic signaling by disrupting mitochondrial membrane integrity and facilitating cytochrome c release. This mitochondrial (intrinsic) pathway is particularly relevant in cancer cells, where overexpression of BCL2 contributes to resistance against chemotherapy-induced apoptosis. 33 Baicalein has been shown in other cancer models to modulate several upstream regulators of apoptosis, including the PI3 K/Akt, MAPK (ERK/JNK/p38), and NF-κB signaling pathways.33,37 For example, inhibition of the PI3K/Akt pathway can lead to reduced phosphorylation of Bcl-2-associated death promoter, thereby promoting its pro-apoptotic activity and enhancing caspase-3 activation. Similarly, baicalein-mediated suppression of the NF-κB pathway may reduce the transcription of anti-apoptotic genes such as BCL2 and survivin. 37 In parallel, activation of JNK and p38 MAPK pathways may promote mitochondrial-mediated apoptosis by increasing the expression or activity of pro-apoptotic factors such as Bax and CASP3. 45 Cisplatin is known to exert its cytotoxic effect by forming DNA adducts that trigger DNA damage responses and activate p53-dependent and p53-independent apoptotic pathways. In future studies, investigating the impact of baicalein–cisplatin treatment on key nodes within these pathways—such as Akt phosphorylation, NF-κB nuclear translocation and mitochondrial membrane potential would significantly enhance the mechanistic depth of the research. Elucidating these molecular interactions may help identify predictive biomarkers of response and inform rational design of combination therapies aimed at overcoming drug resistance in TNBC.
While baicalein demonstrated a cytotoxic effect in vitro with an IC50 of 44 µM and enhanced cisplatin efficacy in combination, the clinical relevance remains uncertain due to limited data on its pharmacokinetics and bioavailability in vivo. Studies have shown that after oral administration, baicalein reaches plasma concentrations of approximately 13.6 nM in rats, 36 which is below the IC50 observed in vitro, potentially limiting its standalone therapeutic efficacy. However, its ability to act synergistically at lower concentrations may still hold clinical promise. Baicalein has not yet entered advanced clinical trials for breast cancer but has shown a favorable safety profile in a phase I trial for influenza. Among 80 healthy subjects, 56 mild adverse events occurred, primarily elevated hs-CRP and triglycerides. No serious effects were reported. After single oral doses (200-800 mg), baicalein reached peak plasma levels of 280–845 ng/mL with a half-life of 7.8–14.9 h. Exposure increased less than proportionally with dose. 46 These findings suggest baicalein is well-tolerated, but further studies are needed to evaluate its safety and efficacy in cancer patients.
Importantly, baicalein inhibits cytochrome P450 enzymes (CYP1A2, CYP2D6, and CYP3A4), 47 raising concerns about possible interactions with drugs. While baicalein enhanced cisplatin's efficacy in TNBC cells, several challenges must be addressed before clinical application. Potential pharmacokinetic interactions may alter cisplatin's metabolism or clearance, affecting its efficacy or toxicity. Baicalein's low bioavailability 36 and possible off-target effects, such as hepatotoxicity or interference with drug-metabolizing enzymes, raise safety concerns. Additionally, TNBC heterogeneity may limit the combination's effectiveness across subtypes. The lack of in vivo data further complicates translation to clinical use. Therefore, comprehensive pharmacokinetic and toxicological studies are necessary to evaluate the safety, optimal dosing, and therapeutic relevance of this combination in treating aggressive breast cancers.
The MDA-MB-231 cell line was selected due to its well-characterized nature and widespread use in TNBC research, particularly for studying invasive and mesenchymal-like features. However, TNBC is a highly heterogeneous disease encompassing multiple molecular subtypes. A limitation of this study is the use of one cell line. As such, findings from MDA-MB-231 may not be generalizable to all TNBC cases. Future studies should incorporate additional TNBC cell lines—such as BT-549 or HCC38 (basal-like A subtype) and SUM149 or HCC1937 (BRCA1-mutated)—to better capture the heterogeneity of TNBC and strengthen the translational relevance of the results. Additionally, the lack of in vivo validation raises questions about the translatability of the observed effects, particularly baicalein's limited bioavailability.
Conclusions
Based on the findings of this study, baicalein reduced the viability, migration, and colony formation of MDA-MB-231 cells. Furthermore, it showcased for the first time, a synergistic enhancement of cisplatin's cytotoxic effect when combined with baicalein. This study provides preliminary evidence supporting baicalein's potential as an adjuvant therapy for the treatment of TNBC. Future studies should incorporate multiple TNBC models, mechanistic analyses, and animal studies to support clinical relevance.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251368753 - Supplemental material for Baicalein Sensitizes Triple Negative Breast Cancer Cells to Cisplatin by Activating Apoptosis
Supplemental material, sj-docx-1-npx-10.1177_1934578X251368753 for Baicalein Sensitizes Triple Negative Breast Cancer Cells to Cisplatin by Activating Apoptosis by Aya Y. Al-Kabariti, Manal A. Abbas, Moath Alqaraleh, Razan Madi and Steven D. Shnyder in Natural Product Communications
Footnotes
List of Abbreviations
Ethical Considerations
Ethical approval is not applicable for this study.
Statement of Human and Animal Rights
This study does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this study and informed consent is not applicable.
Author Contributions/CRediT
A.K. M.A. and M.A-Q. conceived and supervised the project. R.M and A.K. performed the experiments, M.A. wrote the manuscript. S.S. critically analyzed the results. All authors read and approved the finalized article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
