Abstract
Ginsenoside Rg3 (Rg3), a natural product abundantly present in Korean Red Ginseng, is widely known for its anti-tumor activity. In our previous studies, we had further demonstrated that Rg3 has protective effects on the hearts, kidneys, and aortas of animals with hypertension or hypercholesterolemia, and its main mechanisms include down-regulation of angiotensin II (Ang II) levels and activation of peroxisome proliferator-activated receptor gamma (PPARγ) pathway in those tissues. In this study, the protective effects of Rg3 on liver were determined in db/db mice, a most recognized type II diabetes (T2DM) animal model with nonalcoholic fatty liver disease (NAFLD). The results showed that Rg3 did not have obvious effects to the body weight, blood glucose, and lipids of db/db mice. According to the results of histology examination, Rg3 could not improve steatosis in the hepatic tissue, too. But Rg3 did attenuate alanine aminotransferase (ALT)/aspartate aminotransferase (AST) elevation in serum and collagen deposition in hepatic tissue. Immunohistochemistry (IHC) and enzyme-linked immunosorbent assay (ELISA) showed that Rg3 upregulated PPARγ and downregulated Ang II in hepatic tissue in db/db mice, which resulted in reducing activities of transforming growth factor β (TGF-β)/connective tissue growth factor (CTGF) pathway, downregulating the levels of inflammatory cytokines and attenuating collagen accumulation. In conclusion, although it has no obvious effect on steatosis in the hepatic tissue, Rg3 indeed attenuates early hepatic injury from NAFLD via inhibiting PPARγ- and Ang II-related inflammation and fibrosis in T2DM db/db mice. These effects are independent of reducing blood glucose and lipids, and the mechanisms are similar to the protective effects of Rg3 in hypertension and hypercholesterolemia animals in our previous studies.
Ginsenoside Rg3 (Rg3), a natural product abundantly present in Korean Red Ginseng, 1,2 is widely known for its anti-tumor activity. 1 -3 Rg3 has been proven to have obvious anti-cancer activity by inhibiting cancer cell proliferation, migration, stemness and epithelial-mesenchymal transition, inducing cancer cell apoptosis and boosting immune activation. 1 -8 It has also been reported that Rg3 reduces the toxicity 4 or resistance 5 of chemotherapy in vitro. Of which the principal component is Rg3, Shenyi Capsule produced by our company (Jilin Yatai Pharmaceutical Co., Ltd.) has been applied as an adjuvant to conventional cancer therapies in China. 9,10
In addition to its anti-cancer activity, the cardiovascular protective effects of Rg3 have also been shown by our group and others in a variety of animal disease models, including myocardial infarction, 11 hypertension, 12 and atherosclerosis. 13 Along this line, we found that Rg3 improves hypertensive nephropathy in rats and mice with hypertension. 14 We further demonstrated that Rg3 has satisfactory anti-inflammatory effects on the hearts, 12 kidneys, 14 and aortas 13 of animals with hypertension or hyperlipemia, and its main mechanisms include down-regulation of angiotensin II (Ang II) levels and activation of peroxisome proliferator-activated receptor gamma (PPARγ) pathway in those tissues.
Hypertension and hyperlipemia are the most important risk factors of cardiovascular and metabolic diseases, while the other is hyperglycemia. Patients with type II diabetes (T2DM) usually suffer from both hyperglycemia and hyperlipemia, which causes defects in several important organs, such as liver, 15,16 heart, 17,18 kidneys, 16,19 and vessels. 16,18 Among all these complications of T2DM, Ang II- and PPARγ-related inflammation and fibrosis is one of the most important progressive factors, and thus angiotensin-converting enzyme inhibitor (ACEI), Ang II type I receptor blocker (ARB) and Thiazolidinediones (TZDs) are widely used for improving all these complications in clinic. 15 -18
However, the mechanisms of the protective effects of Rg3 on multi-organ defects in T2DM remains elusive. We had demonstrated that orally Rg3 administration (30 mg·kg-1·d-1 in mice and 20 mg·kg-1·d-1 in rats) has protective effects on kidneys, 14 heart, 12 and vessels 13 in several animal models with hypertension or hyperlipemia via activation of PPARγ and down-regulation of renin-angiotensin system (RAS) activity. Therefore, we hypothesized that Rg3 at the same dose could also have hepatoprotective effects, and its mechanisms might be related to the regulation of Ang II and PPARγ. In this study, the protective effects of Rg3 on liver were determined in db/db mice, a most recognized T2DM animal model.
Materials and Methods
Chemicals and Reagents
Rg3 (95% purity) provided by Jilin Yatai Pharmaceutical Co., Ltd. (Changchun, China) was dissolved in 0.5% sodium carboxymethyl cellulose solution (0.5% CMC-Na) for use. The remaining chemicals were of analytical grade. 14
Animals
Sixteen db/db mice (BKS-
Experimental Protocols
Sixteen WT mice were randomly divided into 2 groups, (i) Group WT and (ii) Group WT+Rg3. Sixteen db/db (abbreviated as DB in group-names) mice were randomly divided into 2 groups, (iii) Group DB, and (iv) Group DB+Rg3. Mice in Group WT+Rg3 and Group DB+Rg3 were orally administered 30 mg·kg-1·d-1 Rg3, while mice in Group WT and Group DB were orally administered an equal volume of 0.5% CMC-Na. Rg3 or placebo administration was performed daily for 8 weeks. Body weight and blood glucose measurement was performed weekly during 8 weeks of treatment.
After those treatments, all mice were sacrificed. Then hepatic tissue samples and blood samples (prepared into serum by centrifugation) were collected. The serum or tissue specimens were snap-frozen with liquid nitrogen and kept at −80 °C for histopathology (Oil Red O staining, ORO), biochemical assays, enzyme-linked immunosorbent assay (ELISA), and reverse transcription-quantitative polymerase chain reaction (RT-qPCR). Some hepatic tissue specimens were fixed in 4% formaldehyde and embedded in paraffin, and 4-micrometer-thick sections were used for histopathology and immunohistochemistry (IHC).
Fasting Blood Glucose Measurement
Throughout the 8 weeks of the treatment process, the fasting blood glucose of lateral tail vein was monitored weekly using Blood Glucose Test Meter and Strips (GlucoLab, Infopia Co., Ltd., Kyunggi, Korea) according to the manufacturer’s protocol.
Serum Biochemical Assays
Blood specimens were subjected to centrifugation (1500 g, 4 °C for 15 minutes), and the serum was kept at −80 °C for further biochemical assays. Biochemical assay kits were purchased form Nanjing Jiancheng Bioengineering Institute (Jiangsu, China): alanine aminotransferase (ALT, C009-2-1), aspartate aminotransferase (AST, C010-2-1), triglyceride (TG, A110-1-1), total cholesterol (TC, A111-1-1), high-density lipoprotein cholesterol (HDL, A112-1-1), and low-density lipoprotein cholesterol (LDL, A113-1-1). The activities of ALT, AST, and levels of TG, TC, HDL, LDL in serum were assayed in accordance with the manufacturer’s protocols.
Histopathological Assessment
Hepatic tissue specimens fixed in 4% formalin were embedded in paraffin, cut into 4-micrometer-thick sections, and then stained with hematoxylin and eosin (H&E) and Masson trichrome stain (Masson). ORO staining of lipid droplets was performed on 6-micrometer-thick cryostat liver sections, using an ORO kit (BSBA-4081, ZSGB-BIO, Beijing, China) according to the manufacturer’s protocol. Sections stained with H&E, Masson, and ORO were examined and photomicrograph images were captured using a Nikon E100 light microscope (Nikon Corporation, Tokyo, Japan).
Immunohistochemistry
Primary antibodies against PPAR-γ (bs-4590R), transforming growth factor β1 (TGF-β1, bs-0103R), and connective tissue growth factor (CTGF, bs-0743R) were purchased from Bioss Inc. (Beijing, China). DAB kit (ZLI-9017) and 2 step rabbit IHC kit (PV-9001) were purchased from ZSGB-BIO (Beijing, China). IHC was performed in accordance with the manufacturer’s protocols of the IHC kit and DAB kit. Photomicrograph images were then captured with light microscope as mentioned above and were further analyzed using Image Pro Plus 6.0 (Media Cybernetics, Inc., Rockville, MD, USA).
Assessment of RAS Activity in Hepatic Tissue
Angiotensin converting enzyme (ACE) ELISA kits (CSB-E04490r) and Ang II ELISA kits (CSB-E04494r) was purchased from Cusabio Biotech Co., Ltd. (Hubei, China). A total of 100 mg of hepatic tissue was homogenized in 900 µL of ice-cold normal saline and subjected to centrifugation (1000 g, 4 °C for 15 minutes), and the supernatant was used to measure the level of Ang II in accordance with the manufacturer’s protocol.
RNA Purification and RT-qPCR
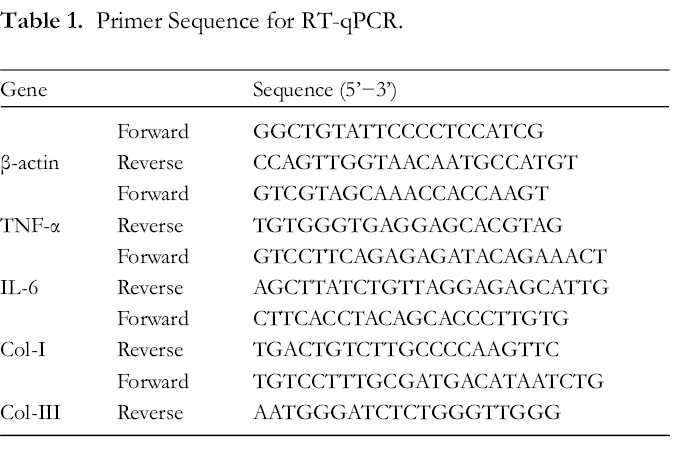
Isolation of total RNA was carried out using TRIzol reagent (Thermo Fisher Scientific, Inc., MA, USA) according to the manufacturer’s protocol. Reverse transcription and qPCR were performed with TransScript Green Two step qRT-PCR SuperMix (TransGen Biotech, Beijing, China) on a Stratagene Mx3000P Real-Time PCR System (Agilent Technologies, Inc., CA, USA). The 2−ΔΔCt method 21 was employed for analysis of the expression of genes of interest, and β-actin was used as a housekeeping gene. All primers are listed in Table 1.
Primer Sequence for RT-qPCR.
Statistical Analysis
SPSS 16.0 (IBM Corporation, NY, USA) was employed for all statistical analyses. Data are mean ± standard deviation (SD). One-way analysis of variance (ANOVA) with Tukey’s post hoc test was employed for group comparisons, with
Results
Effects of Rg3 on Body Weight, Blood Glucose and Lipids
The body weight between the 2 WT groups and 2 db/db groups were significantly different during 8 weeks of treatment. Similar result was observed regarding the blood glucose. The blood glucose of mice in 2 db/db groups continuously increased during the treatment, while the body weight did not. Rg3 administration did not obviously affect the body weight and blood glucose in both WT and db/db mice (Figure 1).
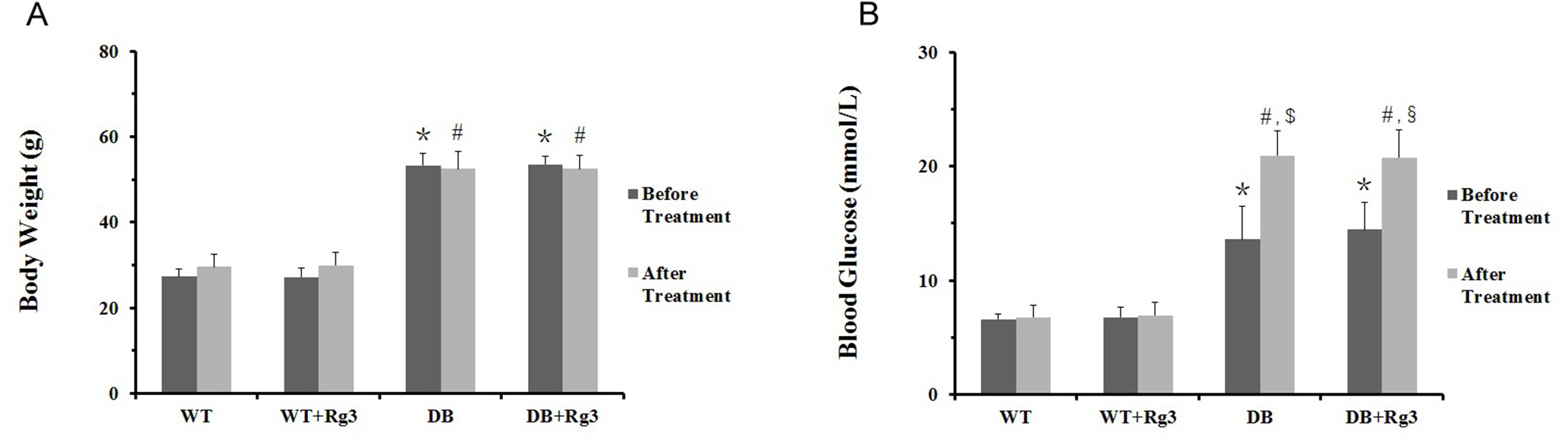
Body weight and blood glucose in mice. A, body weight of mice prior to and following 8 weeks of treatment; B, blood glucose of mice prior to and following 8 weeks of treatment. Data are mean ± SD, and n = 8. One-way ANOVA with Tukey’s post hoc test was employed for group comparisons, with
The blood lipids, including TG, TC, HDL, and LDL, in 2 db/db groups were significantly higher than those in 2 WT groups. Rg3 administration did not obviously affect the blood lipids in both WT and db/db mice (Figure 2).

Blood lipids in mice. (A) TG levels in serum in mice; (B) TC levels in serum in mice; (C) HDL levels in serum in mice; (D) LDL levels in serum in mice. Data are mean ± SD, and
Effects of Rg3 on Activities of ALT and AST in Serum
The activities of ALT and AST in DB Group were significantly higher than those in WT Group, suggested that hepatic injury occurred in animals in DB Group. Rg3 administration improved the hepatic injury in db/db mice, as the activities of ALT and AST in DB+Rg3 Group were reduced markedly comparing to DB Group. Between WT Group and WT+Rg3 Group, there were no statistic difference regarding the activities of ALT and AST (Figure 3).

Activities of ALT and AST in serum in mice. A, activities of ALT in serum; B, activities of AST in serum. Data are mean ± SD, and
Effects of Rg3 on Hepatic Histology
According to the results of histology examination, including H&E, Masson and ORO staining, there were obvious steatosis in hepatocytes and mild disorder of hepatic lobule in the 2 db/db groups other than the 2 WT group. Rg3 administration did not improve the steatosis and mild disorder, but it did inhibit hepatic fibrosis in animals in DB+Rg3 Group. There was mild collagen deposition in the livers of mice in DB Group, and this phenomenon was attenuated by Rg3 (Figure 4).
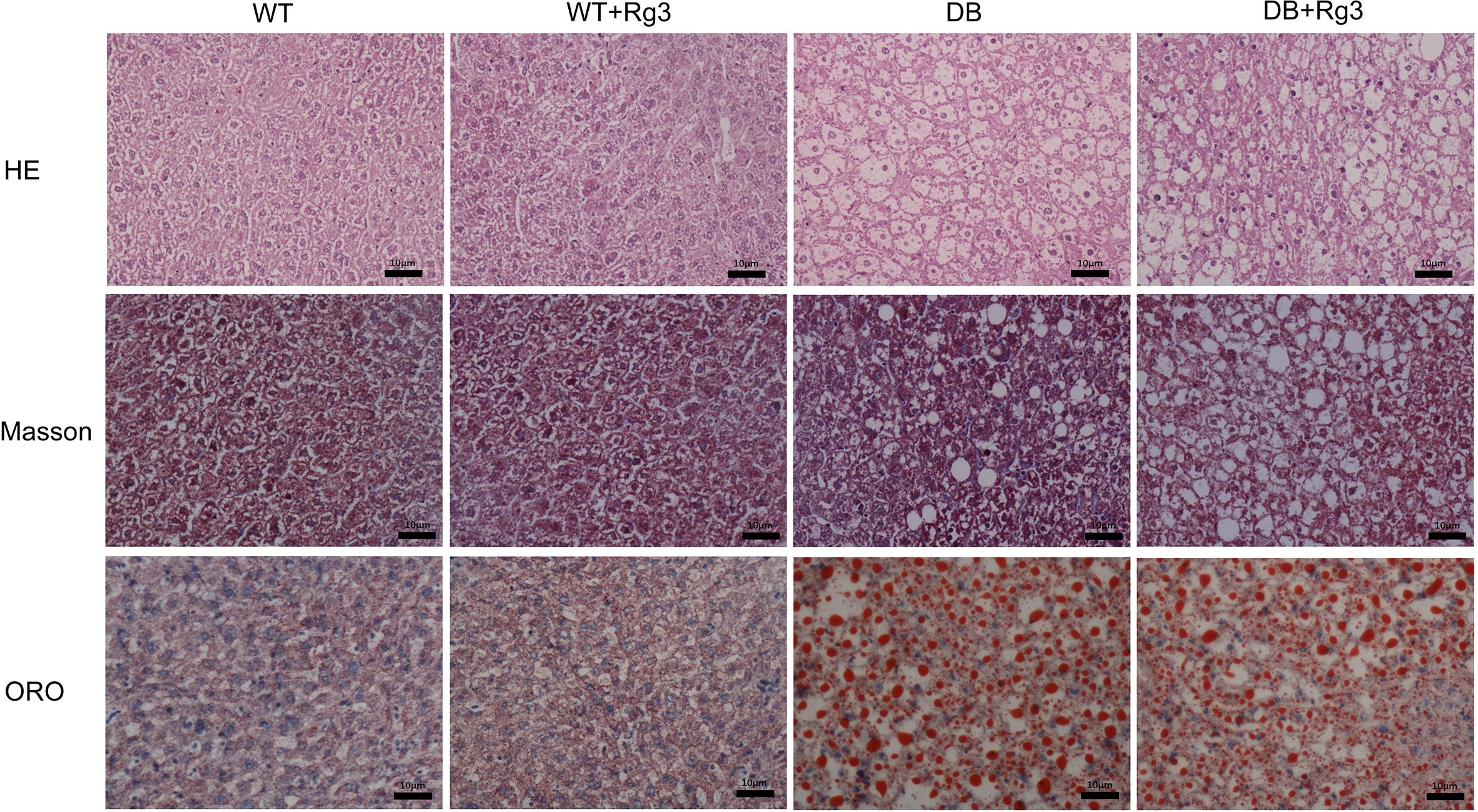
Representative H&E, Masson, and ORO staining histology photomicrographs of the hepatic tissue in mice. All scale bars represent 10 µm.
Effects of Rg3 on Levels of PPARγ, TGF-Β1, and CTGF in Hepatic Tissue
As it was shown in the IHC, the expression of PPARγ in hepatic tissue in DB Group was compensated up-regulated comparing to that in WT Group. Rg3 administration further upregulated the expression of PPARγ in hepatic tissue in db/db mice. TGF-β1 and CTGF are 2 important factors of inflammation and fibrosis, whose levels were significantly upregulated in DB Group comparing to those in WT Group. The levels of these 2 factors were significantly reduced in DB+Rg3 Group by Rg3 administration, suggested that inflammation and fibrosis might be improved by Rg3. Between WT Group and WT+Rg3 Group, there were no statistic difference regarding the expression of PPARγ, TGF-β1, or CTGF levels in hepatic tissue (Figure 5).
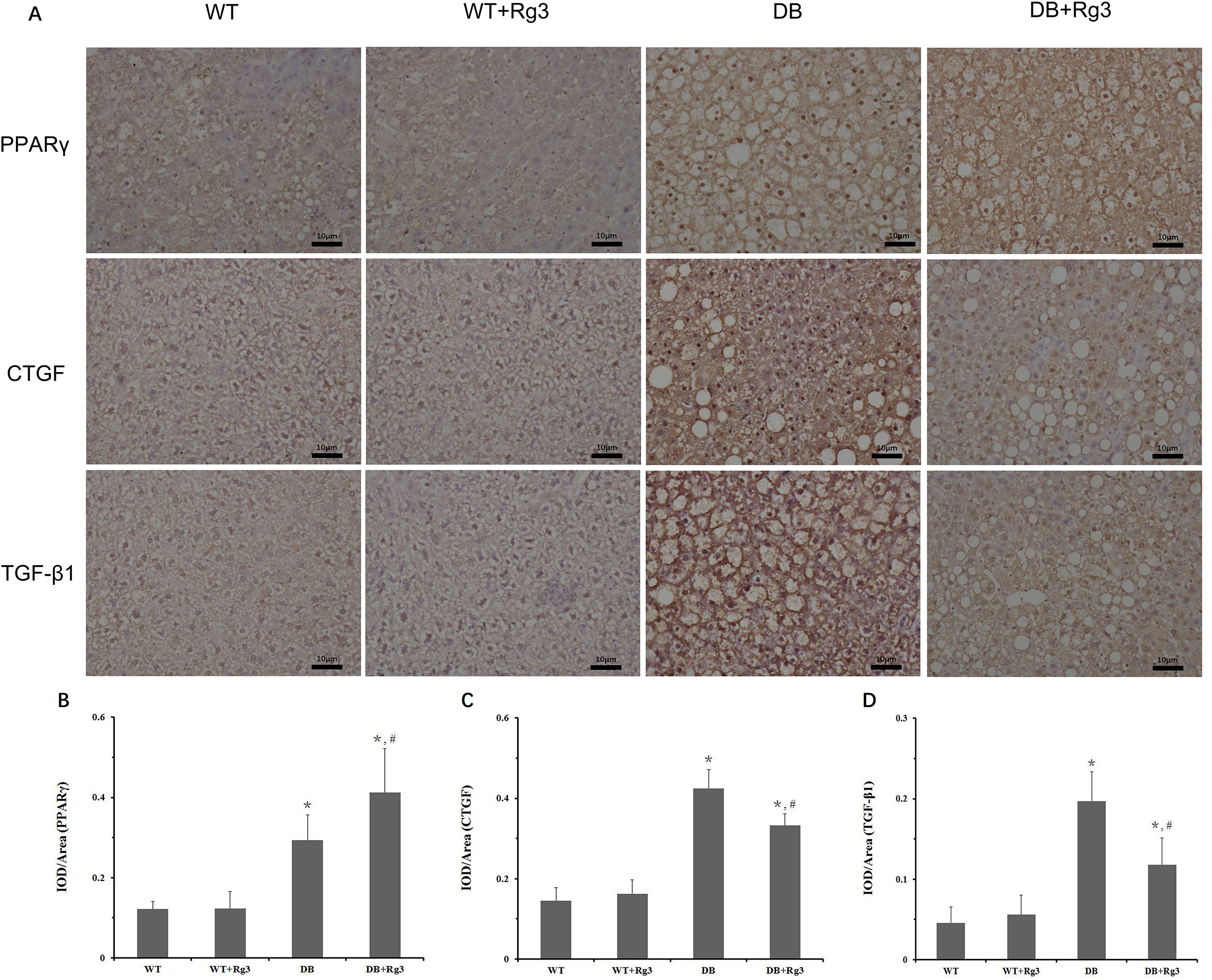
Levels of the PPARγ, TGF-β1, and CTGF in the hepatic tissue in mice. A, representative IHC staining photomicrographs of the hepatic tissue in mice; quantitative results of IHC staining, which were presented as IOD/area and were proportional to the levels of PPARγ (
Effects of Rg3 on RAS Activities in Hepatic Tissue
The elevated RAS activities would induce hepatic injury in db/db mice. The results of ELISA showed that the levels of ACE and Ang II in hepatic tissue were significantly higher in DB Group than those in WT Group. Rg3 administration downregulated both of them significantly in DB+Rg3 Group, while Rg3 has no obviously effects on the activities of RAS in hepatic tissue in WT+Rg3 Group (Figure 6).

The levels of ACE and Ang II in hepatic tissue in mice. (A) levels of ACE in hepatic tissue; (B) levels of Ang II in hepatic tissue. Data are mean ± SD, and
Effects of Rg3 on Levels of Inflammatory Cytokines and Procollagen in Hepatic Tissue
Relative levels of mRNA of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), procollagen I (Col-I), and procollagen III (Col-III) in hepatic tissue were measured (Figure 7). All these levels were significantly higher in DB Group than those in WT Group. The upregulation of inflammatory cytokines and procollagen levels represent the inflammation and fibrosis existing in livers of mice in DB Group. The levels of all these markers were reduced in DB+Rg3 Group by Rg3 administration, demonstrating the anti-inflammation and anti-fibrosis effects of Rg3. Among WT Group, WT+Rg3 Group and DB+Rg3 Group, there were no statistic difference regarding the levels of inflammatory cytokines and procollagen.

Levels of mRNA associated with inflammation and fibrosis in the hepatic tissue in mice. Relative mRNA levels of TNF-α (
Discussion
Nonalcoholic fatty liver disease (NAFLD) is one of the most common cause of abnormal liver function among adults and one of the most common and important complications of T2DM. Studies have shown that NAFLD may be present in up to 70% of patients with diabetes. 22 -24 Studies have also demonstrated that NAFLD and nonalcoholic steatohepatitis (NASH) are risk factors for higher prevalence of cardiovascular disease (CVD) independent of classical risk factors, glycemic control, medications and metabolic syndrome (MS) features. 16,24 The spectrum of NAFLD ranges from simple steatosis to NASH, which can progress to end-stage liver disease. 24 In NAFLD treatment, it is important to improve early hepatic injury and prevent simple steatosis from developing into NASH.
Simple steatosis in liver is inevitable in most patients with obesity, MS or T2DM, which are all exist in db/db mice. Insulin resistance, together with hyperglycemia and hyperinsulinemia, are all the most important reasons for the development from simple steatosis to NASH and hepatic fibrosis, in both T2DM patients and db/db mice. According to the “multi-hit hypothesis” of NAFLD, 25 -28 insulin resistance within the liver is implicated in the pathogenesis of NAFLD. Activation of PPARγ pathway could attenuate this progress of NAFLD via improving the insulin resistance as well as the PPARγ-related inflammation and fibrosis in hepatic tissue, in clinic or in vivo animal research. 22,29,30
In our previous research, 13 Rg3 improved atherosclerosis in ApoE-knockout mice, a well-recognized animal model of hypercholesterolemia, by upregulating PPARγ and suppressing inflammation in aortic endothelial tissue. In this research, we also observed that Rg3 administration increased the expression of PPARγ in hepatic tissue of db/db mice (Figure 5). Although histopathological examination indicated that mice in these 2 db/db mice groups all had NAFLD, there was mild hepatocytes injury and collagen deposition in the DB Group, while there was only simple steatosis in the DB+Rg3 Group (Figures 3, 4 and 7). As mentioned above, activation of the PPARγ pathway could attenuate inflammation and fibrosis, which would improve the hepatic injury in NAFLD patients or animals and prevent the progress from simple steatosis to NASH and hepatic fibrosis. 15,16 In our study, it seemed that the hepatoprotective effects were also associated with inhibition of PPARγ-related inflammation and fibrosis.
PPARγ pathway is crucial in regulating glucose and lipid metabolism. Though its activation promotes fat accumulation, it is generally beneficial to patients with T2DM based on the improvement of hyperglycemia, hyperinsulinemia, insulin resistance, inflammation and fibrosis. 31 In our study, the expression of PPARγ was upregulated in hepatic tissue in db/db mice relative to that in WT mice. Of note, the expression of PPARγ in hepatic tissue of db/db mice with Rg3 treatment was further increased compared to that in db/db mice treated with vehicle (Figure 5), suggesting that Rg3 could not improve the steatosis in hepatic tissue of db/db mice although it did attenuate early hepatic injury by inhibiting inflammation and fibrosis.
The role of RAS in NAFLD progress has gained more attention in the last decade, and ACEI/ARB have been used for treatment of NAFLD in some situation. 32 -34 Ang II is associate with inflammation and fibrosis in most cardiovascular and metabolic diseases such as T2DM and NAFLD. In our previous research, 12,14 we have demonstrated that Rg3 improved Ang II-mediated cardio and renal injury in early stage in rats and mice. In current study, we also observed that Rg3 downregulated hyperglycemia-induced elevation in the activity of RAS in hepatic tissue of db/db mice (Figure 6). As a result, the downregulation of Ang II inhibited Ang II-related inflammation and fibrosis, which is another mechanism of the hepatoprotective effects of Rg3.
Taken together, the 2 mechanisms of cardio protective effects that Rg3 represented in our previous researches in hypertension or hyperlipidemia animals are also represented in this study in hyperglycemia animals. As it has been reported in our previous researches, 12 -14 the regulation of PPARγ pathway and RAS reduced activities of TGF-β/CTGF pathway (Figures 5 and 6), attenuated collagen accumulation, and downregulated the levels of inflammatory cytokines. These effects would inhibit inflammation and fibrosis in hepatic tissue, which might be the reasons of Rg3 in improving early hepatic injury in db/db mice. Moreover, the situations were similar that Rg3 had not improved the hypertension or hyperlipidemia in animals in our previous researches, 12 -14 and Rg3 did not has obvious effects on the blood glucose and lipids in db/db mice (Figures 1 and 2). Transgenic animals or small molecular inhibitors/activators are needed to elucidate the detailed mechanisms in the further.
Rg3 and other ginsenosides have intricate biological activities according to decades of global research. Besides the regulation of PPARγ pathway and RAS, some researchers believe that suppression of oxidative stress might be an important mechanism for the protective effects of Rg3 on the heart, nerves, kidneys or liver. 35 -37 Interestingly, other researchers believe that activation of oxidative stress might be an important mechanism of Rg3’s anti-tumor effects. 38 -40 It is hard to tell the causal relationship between Rg3’s effects on oxidative stress and its effects on cancer cells or normal tissues. Since oxidative stress is a double-edged sword, the paradoxical effects of oxidative stress on killing cancer cells and protecting normal tissues worth further elucidation.
Conclusion
In conclusion, although it has no obvious effect on steatosis in the hepatic tissue, Rg3 indeed attenuates early hepatic injury, including mild ALT/AST elevation and collagen deposition, via inhibiting PPARγ- and Ang II-related inflammation and fibrosis in T2DM db/db mice. These effects are independent of reducing blood glucose and lipids, and the mechanisms are similar to the protective effects of Rg3 in hypertension and hypercholesterolemia animals in our previous studies.
Footnotes
Statement of Human and Animal Rights
All the experimental procedures involving animals were conducted in accordance with the institutional Animal Care guidelines of Jilin University, China and approved by the Laboratory Animal Ethics Committee of Jilin University, Jilin Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (Grant 81473378), Science and Technology Development Projects of Jilin Province (Grant 20170101002JC), and Outstanding Doctoral Cultivation Program of Norman Bethune Health Science Center of Jilin University (2014).
