Abstract
Nonalcoholic fatty liver disease (NAFLD) is becoming one of the major health issues globally. NAFLD is usually associated with obesity and other metabolic syndromes, and there is no specific cure to address this issue so far. Featured by lipid accumulation in hepatocytes, and later progressing to fibrosis, inflammatory responses are involved in the various levels of the pathological changes. In the present study, we added a natural compound, tyrosol, which in our previous study had demonstrated anti-inflammatory properties, to a high-fat diet-induced NAFLD mouse model and investigated whether tyrosol could mitigate the liver damage by attenuating the inflammation response. The treatment with tyrosol significantly improved the liver function and decreased the fasting glucose level in NAFLD mice. Morphologically, our results showed that tyrosol could reduce the fat deposition and lipid droplets accumulation in liver tissue. The key regulating factors, JAK1 and STAT3, were increased in NAFLD mice, but tyrosol treatment could effectively prevent the upregulation. The expression levels of pro-inflammatory cytokine genes in liver tissue of the NAFLD mice were upregulated, which could be effectively prevented by the treatment with tyrosol. Overall, in the high-fat diet-induced NAFLD mouse model, tyrosol could improve the liver function and, more importantly, ameliorate the inflammatory response triggered by the high-fat diet. Although our data here are consistent with the previous report that tyrosol could exert beneficial effects on the NAFLD animal model, we also provide solid evidence that tyrosol is able to conquer the inflammatory response in liver, which is related to the high-fat-diet feeding. Tyrosol could be a promising candidate for the treatment of NAFLD in the future.
Introduction
Nonalcoholic fatty liver disease (NAFLD) is becoming one of the most prevalent medical conditions globally and affecting around one-third of the general population. 1 Pathologically, NAFLD is featured by steatosis, and nonalcoholic steatohepatitis (NASH), which could deteriorate to fibrosis or cirrhosis or even hepatocellular carcinoma. NAFLD is considered as a hepatic manifestation of metabolic abnormality associated with insulin resistance, obesity, and cholesterol dysregulation.
The etiology of the disease consists of multifactorial components. The onset and deterioration of the disease are greatly influenced by factors such as diet, weight, and genetic background. 2 Among these risk factors, high-fat diet (HFD) is regarded as a key factor that leads to the development of NAFLD. 3 Clinically, HFD is associated with obesity in patients, which is usually concomitant with NAFLD. 4 HFD could increase the exogenous dietary fat accumulation in liver tissue and trigger enhanced endogenous lipid synthesis that promotes the progress of NAFLD. 3 Study has suggested that NAFLD is an inflammation disease and inflammation, not the steatosis is the key determinant for the long-term outcome of NAFLD patients. 4 Pro-inflammatory cytokines and chemokines, collectively known as adipokines, which are generated from adipose tissue, could accelerate the progress of NAFLD to more severe stages. 5 Although advances have been made in the understanding of the pathophysiology of NAFLD, there are no reliable clinical biomarkers of early diagnosis or progression of NAFLD. Consequently, NAFLD patients are not often recognized before reaching a stage where either curative treatment or even disease stabilization has become largely impossible. 6 As a result, researchers have been screening promising functional compounds that could ameliorate or slow the disease progression.
Tyrosol is a natural phenolic compound found in various natural sources, such as olive oil, argan oil, and wine, possessing multiple biological activities such as anti-oxidation, anti-apoptosis, stress-protective, anticancer, cardioprotection, neuroprotection, and anti-inflammation.7–9 Tyrosol was able to attenuate acute and chronic inflammation in mouse models. 10 Especially, one study revealed that tyrosol could attenuate the oxidative stress in the liver of HFD-induced NAFLD mice. 11 Tyrosol could improve ovalbumin caused asthma in animal models through inhibiting the airway inflammation. 12 Our previous study also demonstrated that tyrosol could reduce the synthesis of cytokines from astrocytes, central nervous system specialized immune cells, and protect neurons against oxygen deprivation injury. 7 The Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling cascade is one of the central pathways that regulate various biological processes aggravating NAFLD. Also, JAK/STAT is a major mediator of cytokine cascade and is closely involved in the deterioration in liver disease.
In the present study, we examined whether tyrosol could ameliorate the inflammatory response and promote liver function in an HFD-induced NAFLD mouse model. We also investigated whether the possible beneficial effects are achieved by regulating the JAK/STAT signaling pathway and the triggered cytokine cascade.
Results
Tyrosol Treatment Reduced Fat Deposition and Accumulation of Lipid Droplets in the Liver Tissues of NFALD Mice
Twelve weeks of HFD treatment caused notable pathological changes in the liver tissues of mice (Figures 1 and 2). As shown in Figure 1B, some pathological features of NFALD were observed in the HFD liver section with H&E staining, including macrovesicular steatosis, ballooning, and scattered lobular inflammation. Our results showed that tyrosol treatment could ameliorate these above changes. In Oil Red O staining, there was an obvious accumulation of lipid droplets across the liver sections (Figure 2). However, the severity of accumulation was seemingly attenuated in the liver sections from the mice cotreated with tyrosol. These above beneficial effects of tyrosol on the histological appearance validated the treatment effects of tyrosol on the liver damage in these NFALD mice and laid the foundation for the further investigation.

(A) The structural formula of tyrosol. (B) H&E staining showed that tyrosol treatment reduced the pathological changes in the liver tissues of NFALD mice.

Oil Red O staining showed that tyrosol treatment reduced the fat deposition and accumulation of lipid droplets in liver tissues of NFALD mice.
Tyrosol Treatment Improved the Impaired Liver Function and Glucose Abnormality of the NFALD Mice
Next, we found that 12 weeks of HFD treatment caused a significantly higher fasting glucose level in the serum of NFALD mice, but the increased level of glucose could be effectively reversed by tyrosol treatment (Figure 3A). As for liver function, HFD treatment induced higher levels of both ALT and AST in the serum of NFALD mice (Figure 3B and C), which indicated liver damage in these mice. As we expected, tyrosol treatment was able to attenuate the liver tissue damage by decreasing both ALT and AST levels in the serum of these NFALD mice. These above findings indicated that tyrosol could protect the liver against HFD-induced metabolic abnormalities that are characteristic features of NFALD.

Tyrosol treatment improved the impaired liver function and glucose abnormality of the NFALD mice. (A) Tyrosol treatment reduced the fasting glucose level in the serum of NFALD mice. (B) Tyrosol treatment reduced the AST level in the serum of NFALD mice. (C) Tyrosol treatment reduced the ALT level in the serum of NFALD mice. Values are expressed as means ± SEM n = 6-7 in A; n = 11-14 in B and C. *P < .05 versus Cont; #P < .05 versus HFD.
Tyrosol Treatment Prevented JAK1/STAT3 Activation in the Liver Tissues of NFALD Mice
Next, we investigated whether the inflammation-related signaling pathway in liver tissues was regulated by tyrosol treatment. We focused on the expression levels of JAK1 and STAT3 that are important proteins of the inflammatory response in liver tissues under NFALD. Western blot assays showed that HFD caused a significantly increased expression level of JAK1 protein in liver tissues of NFALD mice, which could be brought down by tyrosol treatment (Figure 4A). Meanwhile, the protein level of STAT3 was boosted by the HFD treatment, while the tyrosol treatment could prevent the increase of STAT3 protein (Figure 4B).
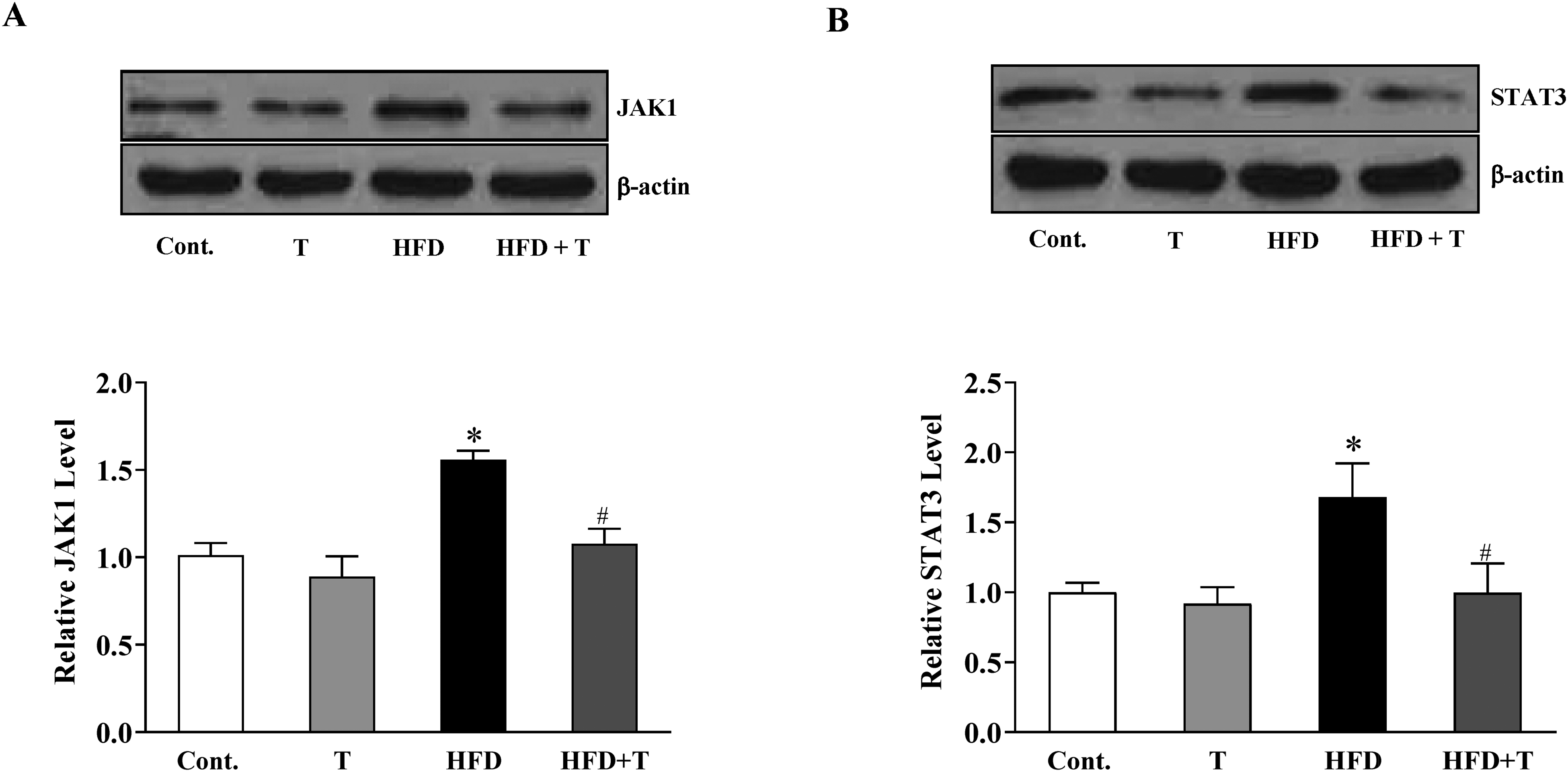
Tyrosol treatment prevented the JAK1/STAT3 activation in the liver tissues of NFALD mice. (A) Tyrosol treatment reduced the JAK1 protein level in the liver tissues of NFALD mice. (B) Tyrosol treatment reduced the STAT3 protein level in the liver tissues of NFALD mice. Values are expressed as means ± SEM n = 5. *P < .05 versus Cont; #P < .05 versus HFD.
Tyrosol Treatment Inhibited the Expression Levels of IL-6, TNF-α, and IL-10 in the Liver Tissues of NFALD Mice
Last, we postulated that inhibition of the JAK1/STAT3 signaling pathway could consequently exert regulating effects on the expression levels of pro-inflammatory cytokines. Therefore, we explored whether the gene expression levels of IL-6, TNF-α, and IL-10 could be affected by tyrosol treatment in the liver tissues of NFALD mice. As shown in Figure 5, HFD caused a significantly increased expression of IL-6, TNF-α, and IL-10. As we expected, these increased expression levels of cytokines were prevented by treatment with tyrosol. These results verified our hypothesis that tyrosol might rescue the liver damage from HFD-induced NFALD by attenuating the inflammatory response in the liver tissues.
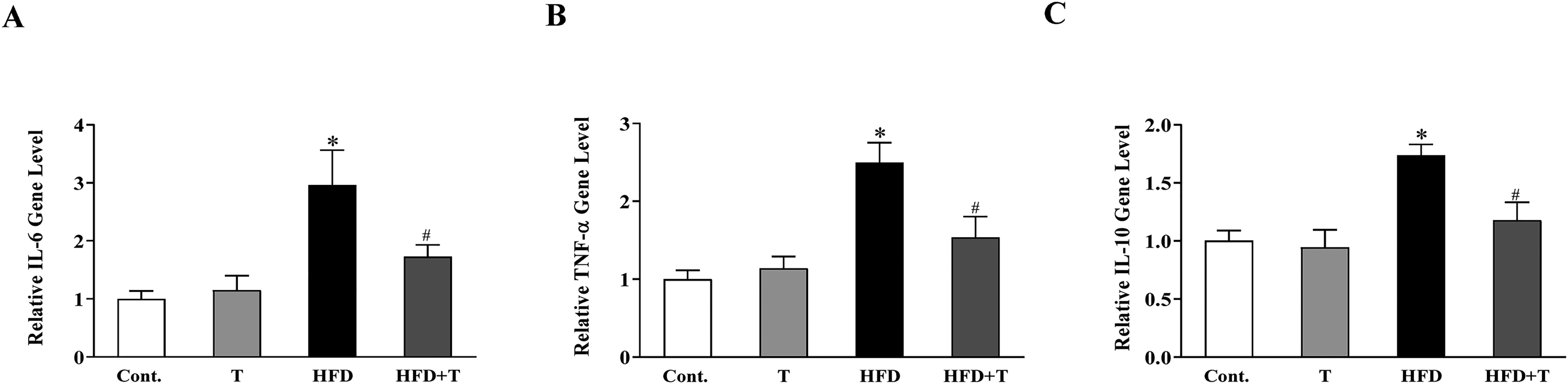
Tyrosol treatment inhibited the expression levels of IL-6, TNF-α, and IL-10 in the liver tissues of NFALD mice. (A) Tyrosol treatment reduced the IL-6 protein level in the liver tissues of NFALD mice. (B) Tyrosol treatment reduced the TNF-α protein level in the liver tissues of NFALD mice. (C) Tyrosol treatment reduced the IL-10 protein level in the liver tissues of NFALD mice. Values are expressed as means ± SEM n = 5. *P < .05 versus Cont; #P < .05 versus HFD.
Discussion
Nonalcoholic fatty liver disease is becoming a major health concern globally. It is a key link between several medical conditions, such as obesity, diabetes, and cardiovascular and kidney disease. 13 However, no effective treatment is available so far. More and more effort has been made in this area by using in vivo animal models, such as the high-fat diet (HFD) induced NAFLD mouse model. 3 Chronic inflammation is one of the characteristic features of NAFLD. 14 The roles of inflammation in NAFLD have attracted substantial attention, and a study has suggested that inflammation is an essential part of the development of metabolic syndrome and insulin resistance. Both of these 2 conditions contribute to the development of NAFLD. The pro-inflammatory cytokines could directly facilitate fat synthesis and result in the accumulation of triglycerides and cholesterol in liver cells. 15
Interestingly, in the early stage of NAFLD, inflammation was not obvious in animal model studies. 16 Therefore, in the present study, we established an HFD-induced NAFLD mouse model by feeding the mice for 12 weeks instead of 5 weeks. After the 12 weeks of treatment, we found fat deposition, accumulation of fat droplets, obvious cytokines increase, and JAK1/STAT3 signaling pathway activation in the livers of these HFD mice. The JAK1/STAT3 signaling pathway was reported to play an important role in the development of NAFLD and could be one of the promising therapeutic targets to conquer the disease. 15 These above results validated the establishment of a NAFLD model in mice for our study. More importantly, consistent with our major hypothesis here as to whether the natural compound, tyrosol, could rescue the mice from the HFD-induced NAFLD, our results showed that tyrosol could improve the glucose metabolism and repair damaged liver function. These results were also in line with the previous report. 3 The primary goal of this study was to evaluate the anti-inflammation role of tyrosol in a mouse model of NAFLD. With support from the encouraging histological results that showed that tyrosol ameliorated the fat accumulation and deposition in the liver tissue, we expanded our investigation to the effects of cytokines. The pro-inflammatory cytokines, TNF-α and IL-6, are extensively involved in the pathophysiology of NAFLD. 17 In the present study, we found a notable increase in these 2 cytokines, but tyrosol could effectively bring them down. Interestingly, one of the anti-inflammatory cytokines, IL-10, was also upregulated with the HFD treatment of NAFLD mice. One of the explanations is that IL-10 might exert an anti-inflammatory role in the early stage of the NAFLD mouse model and then turn into being pro-inflammatory in the later stage since 12 instead of 5 weeks of treatment of HFD was used in our study. A short-term treatment protocol of HFD is warranted for future study to verify further the changes in the IL-10 expression level in the NAFLD model.
In a nutshell, we employed a natural compound in a mouse model to investigate its potential beneficial effects on HFD-induced NAFLD. The previous study had shown that another component of olive oil, hydroxytyrosol, was mainly responsible for the anti-inflammation effect in liver disease. 18 We focused on the role of inflammation in this mouse model and validated that tyrosol alone could inhibit the cytokine increase in liver tissue by regulating the JAK1/STAT3 signaling pathway. As summarized in Figure 6, tyrosol could attenuate the generation of cytokines at the transcriptional level and then reduce the amount of cytokines that could activate the cytokine receptors on the cell membrane. Tyrosol could be a promising candidate compound to treat NAFLD in the future.

A sketch showing the possible mechanism of tyrosol attenuating the inflammation in the mouse model.
Methods and Materials
Animals
Sixty 6-week-old male C57BL/6 mice were housed in standard cages at 22 °C in a 12:12-h light-dark cycle and randomly grouped into 4 groups (15 mice/group): control (Cont.); tyrosol alone (T); high-fat diet alone (HFD); high-fat diet + tyrosol (HFD + T). Control mice were fed with a standard formula comprised of 10% kcal fat, 20% kcal protein, and 70 kcal carbohydrate. The HFD consisted of 60% kcal fat, 20% kcal protein, and 20% kcal carbohydrate. Mice were allowed ad libitum access to water and food. Mice were sacrificed after 12 weeks of the abovementioned treatments. Liver tissue and serum were collected from the mice that were fasted overnight for further analysis. Tyrosol was purchased from Sigma and its structural formula is presented in Figure 1A. All procedures related to animals were approved by the Animal Care Committee of Henan University, Henan, China.
Western Blot
Total protein was extracted from liver tissue with RIPA buffer and protein concentration was determined by BCA Protein Assays (ThermoFisher). An equal amount of protein from each group was loaded and run on 12% SDS-PAGE gel after being denatured with boiling water. Proteins were then transferred onto a PVDF membrane followed by 2 h incubation with 5% skim milk in TBST for blocking. The membranes were immersed in primary antibodies overnight at 4 °C. The antibodies were JAK1 (1:500, cell signaling), STAT3 (1:1000, cell signaling), and β-actin (1:5000, Santa Cruz). Next day, corresponding secondary antibodies were employed to incubate the membranes for 2 h. Lastly, the membranes were developed and visualized by chemiluminescence in ECL solution (ThermoFisher). Quantitative results are expressed as a ratio of each target protein to their internal control.
Serum and Liver Tissue Analysis
After being anesthetized, blood and liver tissues were collected from the mice. Fasting glucose, serum alanine aminotransferase (ALT), and aspartate aminotransferase (AST) levels in serum were measured by employing commercial kits. Total RNA was extracted using buffer RLT (Qiagen) with 0.12 m β-mercaptoethanol (Sigma). RNA was purified with an RNeasy Protect Mini kit (Qiagen), following the recommendations of the manufacturer. One microgram of total RNA in each sample was used for reverse transcription, and PCR was then performed. The primers for the mouse DNA are listed in Table 1.19–21
Sequences of Primers Used.

Tissue Histological Analysis
Liver tissues were fixed in 4% paraformaldehyde after perfusion and stained with hematoxylin-eosin for pathological analysis. To detect the lipid droplets directly in these frozen sections, Oil Red O was applied. For Oil-Red-O (ORO) staining, tissues were sliced into 10 μm sections. The tissue slides were stained with 0.7% (wt/vol) ORO, followed by washing out the excess stain.
Statistical Analysis
All values are expressed as means ± SEM. Comparison between groups was evaluated using one way ANOVA analysis; P < .05 was considered statistically significant.
Footnotes
Acknowledgments
The authors thank the funding support from Beijing Hospitals Authority Youth Program (No. QML20190505).
Authors’ Note
Yinuo Huang, Donghai Li: conceptualization, experimental design, writing draft; Shuai Li, Zifei Han, Juan Du, Xin Liu: project administration and animal maintaining; Zhiyuan Zhu, Lixia Zheng, Suyan Han, Hongbo Shi, Xiaojuan Wang, Xinmei Liu, Zhihong Jiang, Yuxiang Li, Xiaoli Li, Xin Gu, Dazheng Han: investigation, methodology, editing. All animal experiments were conducted in accordance with protocols approved by the Animal Care Committee of Henan University, Henan, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Hospitals Authority Youth Program (grant number No. QML20190505).
