Abstract
Melanogenesis is a complex process that can lead to pigmentation defects. Various chemical skin-lightening products have been developed to treat pigmentation disorders. However, these chemical products can cause harmful adverse effects. Therefore, the development of safer, natural bleaching ingredients is a trend for sustainability. It has been reported that unsaturated fatty acids exhibit significant antimelanogenic effects. Sapindus mukorossi seed oils contain abundant unsaturated fatty acids; however, these have not yet been investigated for beneficial effects on skin tone evenness. In this study, we tested the possibility of using S. mukorossi oil for the treatment of hyperpigmentation in an in vitro model. Free fatty acid compositions and β-sitosterol were determined by gas chromatography-mass spectrometry and high-pressure liquid chromatography, respectively. The effect of S. mukorossi oil on melanoma B16F10 cell viability was detected using the 3-(4,5-dimethylthiazol-2-yl)- 2,5-diphenyl-tetrazolium bromide assay. The inhibitive effects of fatty acids and β-sitosterol in S. mukorossi oil on α-melanocyte-stimulating hormone (MSH)-induced melanogenesis was evaluated by detecting melanin formation and tyrosinase activity. Our results showed that S. mukorossi oil produced no significant cytotoxicity in B16F10 cells at various concentrations compared with the control group. The enhancement of melanin formation induced by α-MSH was reduced by S. mukorossi oil. We also found that the primary fatty acid contributing to the antimelanogenesis effect was eicosenoic acid. These results suggest that S. mukorossi seed oil can effectively inhibit melanogenesis and has the potential for future development as a de-hyperpigmentation product within a waste utilization context.
The soapnut tree is widely distributed in Asia from Japan to tropical India and has long been known for its triterpenoid saponins in the pericarp of its fruit. 1 -3 Saponins are mainly used as natural washing agents for the body, hair, and clothing. 1,2,4 -6 Soapnut trees are considered an essential economic plant that has multiple biological and pharmacological applications. 6 There are more than 40 wild species in the Sapindaceae family. According to research literature, saponins from Sapindus mukorossi exhibit antibacterial activity, 7 inhibit tumor cell growth, 2 and can be used to treat many skin diseases. 3,8
The majority of previous studies on soapnut fruit have focused on the application of saponins rather than seed products, as the oil in the seed kernel of some varieties of Sapindus contains toxic cyanolipids. Therefore, although the soapnut seed makes up about 30% of the fruit’s total weight and has a seed kernel that is rich in oil (23%), 3,5,9 Sapindus seeds have long been treated as waste. 1,10,11 Recently, sustainability researchers have investigated the possibility of using Sapindus seed oil to produce biodiesel fuel. 10,12,13
Possible medical applications of Sapindus (Wu Huan Zi in Mandarin) seed kernel were recorded in China’s traditional pharmaceutical book, Compendium of Materia Medica (Bencao Gangmu in Mandarin) some 5 centuries ago. In 2019, Chen et al analyzed the composition of S. mukorossi oil by high-pressure liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS) and found that the oil contains abundant phytosterols (β-sitosterol) and a substantial amount of unsaturated fatty acids that account for 85.6% of all the fatty acids in the seed oil. Of these unsaturated fatty acids, 76.8% were monounsaturated. 14 In a series of animal experiments, Chen et al suggested that in addition to S. mukorossi oil’s potential to accelerate skin wound healing, it may also have cosmetological uses for daily face and body care. 14
An even skin tone is a vital issue in cosmetology, and for years, chemical skin-lightening products have been developed to treat pigmentation disorders. Melanin production is the primary cause of skin tanning and freckles, with melanogenesis enhancement being related mechanistically to the activation of tyrosinase due to the absorption of ultraviolet light. Chemicals such as hydroquinone, 15,16 phenols, 17,18 and mercurials 19 help even skin tone by inhibiting pigmentation through various mechanisms, but these skin bleaching products have been proved to cause unwanted harmful adverse effects, such as a high risk of carcinogenesis after long-term exposure to hydroquinone, 18,20 ochronosis caused by phenols, and nephropathy caused by mercurials. 21 Therefore, the development of safer and more effective bleaching ingredients from nature is needed, such as those found in several traditional Chinese medicines that have recently been reported to even skin tone. 22 Since these natural products are milder and safer than synthetic chemicals, they show potential for future application as cosmetic skin tone evening products.
A previous study has shown that plant oils, such as sunflower and safflower oils, inhibit melanogenesis in melanoma cells. 23 Unsaturated fatty acids are reported to play a crucial role in inhibiting the transcription of tyrosinase with a mechanism similar to that of vitamin A acid. 24,25 Another unsaturated fatty acid that exhibits skin pigmentation reduction is stearidonic acid, which binds to peroxisome proliferator-activated receptor-gamma (PPAR-γ) to inhibit tyrosine messenger ribonucleic acid (mRNA) and protein expression. 26 In contrast to unsaturated fatty acids, saturated fatty acids such as palmitic acid and stearic acid have the opposite effects on tyrosinase activity and increase melanin production. 27
Since S. mukorossi seed oil contains abundant unsaturated fatty acids, it could provide a skin tone evening effect and act as a natural skin-lightening agent. However, whether or not skin tone evening can be enhanced by S. mukorossi seed oil remains unknown. The purpose of this study was to evaluate the melanogenesis inhibition property of S. mukorossi seed oil in an in vitro cellular model. The inhibitive effects of fatty acids in S. mukorossi oil on melanogenesis were evaluated by detecting the melanin formation and tyrosinase activity, and the compositions of free fatty acids and β-sitosterol were determined by GC-MS and HPLC, respectively.
Materials and Methods
Chemicals and Reagents
The free fatty acid standards, β-sitosterol, and melanin were purchased from Sigma-Aldrich (St. Louis, MO, USA); Triton X-100, 3,4-dihydroxy-
Plant Material
Sapindus mukorossi Gaertn. (family Sapindaceae) used in this study was purchased from He He Co. Ltd. (Taipei, Taiwan). Oil extraction was performed according to the method described in a previous study. 14 Briefly, after the pericarp and fruit pulp were removed, the remaining seed parts were cleaned with distilled water and dried in an oven at 40 °C for 72 hours. A grinder was used to crush the seeds, and the kernels were separated from their shells and collected. Cold-press extraction was performed to remove the oil from the kernels. The extracted oil was filtered (0.45 µm pore size) and stored at −20 °C before the experiments.
GC-MS Analysis
For the GC-MS analysis, the extracted S. mukorossi oil was transesterified to produce fatty acid methyl esters (FAMEs). Briefly, the extracted oil was first mixed with 1 N sodium hydroxide and stirred at room temperature for 30 seconds. The resulting mixture was saponified for 15 minutes. After saponification, 1 mL of boron trifluoride (in a methanol solution) was added to the sample, which was then kept at 110 °C in a dry bath for 15 minutes. To extract FAMEs, 1 mL of n-hexane was added to the sample. The FAMEs were then injected into the GC-MS system (GCMS-QP2010, Shimadzu, Tokyo, Japan) equipped with a BPX70 capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness). Helium was used as the carrier gas at a pressure of 75 kPa to separate the component oils. The injection port was set at 250 °C and the oven temperature at 120 °C and held for 0.5 minutes for the solvent delay, then increased at 10 °C/min to 180 °C. Then the temperature was increased at a rate of 3 °C/min to 220 °C, followed by an increase of 30 °C/min to 260 °C, and then held for 5 minutes. For mass analysis, the temperature of the ionization chamber was set at 200 °C. Molecules separated by GC were identified by MS in the electron impact (EI) mode at 70 eV. The NIST/EPA/NIH Mass Spectral Library was searched for matching spectra.
HPLC Analysis
To determine the amount of β-sitosterol in the S. mukorossi seed oil, HPLC was performed. The HPLC system included a low-pressure mixing pump (L2130, Hitachi, Tokyo, Japan), a CBM-20A interface module (Shimadzu Technology, Kyoto, Japan), and an ultraviolet detector (Waters 486, Waters Corporation, Milford, MA, USA). Separation was performed using a Mightysil RP-18 GP column (250 mm × 4.6 mm i.d., Mightysil RP 18 GP Cica, Tokyo, Japan) at 30 °C. The mobile phase was a mixture of 96% methanol, 3% tetrahydrofuran, and 1% deionized water. The flow rate was set at 0.5 mL/min, and separated samples were monitored at 280 nm. During detection, diluted S. mukorossi seed oil (1%) was used to inject into the column. β-Sitosterol was identified by comparing the detected retention time to its standard. Quantification was achieved using linear regression analysis obtained from various concentrations of the β-sitosterol standard.
Cell Viability Assay
In this study, murine melanoma B16F10 cells (BCRC 60031) were used as an in vitro model to test the antimelanogenesis effect of S. mukorossi seed oil. Cells were purchased from the Bioresource Collection and Research Center, Food Industry Research and Development Institute (Hsinchu, Taiwan), and the first 6 passages were used in this study. The cells were cultured in Dulbecco’s modified Eagle medium, 10% fetal bovine serum, and 1% penicillin-streptomycin. Cells were maintained in 6-well plates at a concentration of 1 × 105 cells/mL and incubated in 5% carbon dioxide at 37 °C and 95% humidity. Sapindus mukorossi oil was saponified by mixing the oil with polyoxyethylene sorbitan mono-oleate (Tween 80) 14 at an oil/emulsifier ratio of 5:1 (v/v) to dissolve the salt in the medium. After saponification, the pH of the solution was adjusted to 7.4.
For the assessment of possible toxicity of S. mukorossi oil to B16F10 cells, the cells were seeded in 24-well culture plates at a density of 1 × 105 cells/mL. After the cells were attached to the plate, saponified S. mukorossi oil at various concentrations (0, 100, 250, and 500 µg/mL) was added to the culture medium, and the cell viability was recorded at 48 hours. The cells were seeded at a density of 1 × 104 cells/mL and cultured with the S. mukorossi oil at a concentration of 250 µg/mL to evaluate the influence of S. mukorossi oil on the growth curves of the B16F10 cells, as recorded every 24 hours for 3 days. Sham-exposed cells were treated as control. Cell viability was detected using the MTT method. According to the manufacturer’s protocol, after the cells were incubated with tetrazolium salt for 4 hours, 500 µL of dimethyl sulfoxide (DMSO) was added to solubilize the formazan crystals overnight. Optical density of tested cells was measured at 570 nm with a microplate reader (EZ Read 400, Biochrom, Holliston, MA, USA).
Melanin Content
To test the antimelanogenesis effect of S. mukorossi oil on B16F10 cells, the melanin activity of the cells was determined according to a previous report. 28 For this assay, the blank group was treated with medium only. For the test group, α-MSH was used as a stimulator. After being treated with 200 ng/mL α-MSH for 24 hours, the B16F10 test cells were treated with various concentrations of either 20 μL pure Tween-80 (20 and 100 µg/mL) or 20 µL Tween-80 saponified S. mukorossi oil (100, 200, 300, and 500 µg/mL). After being co-cultured for an additional 24 hours, test cells were washed twice with cold phosphate-buffered saline, followed by a wash using 10 mM Tris buffer at 4 °C for 5 minutes. Washed cells were centrifuged for 20 minutes at 1000×g. After removing the supernatant (collected for analysis of tyrosinase activity), samples were lysed with 1 N sodium hydroxide at 60 °C for 120 minutes. Absorbance of the extracted melanin was measured at 405 nm using a microplate reader (EZ Read 400, Biochrom).
To test for total sample protein, cells treated using the same procedure above were aspirated and washed twice with phosphate-buffered saline. Then 300 µL 0.05% (V/V) Triton X-100 was added per dish. After cell disruption by 3 freeze/thaw cycles, 50 µL of cell lysate from each dish were transferred to a 96-well microtiter plate. To determine the total amount of protein contained in cells, bicinchoninic acid (BCA Protein Assay Kit; Pierce, Rockford, IL, USA) was added to the cell lysate and incubated for 10 minutes. Absorbance was read spectrophotometrically at a wavelength of 590 nm. Specific melanin content was calculated using absorbance measured at 405 and 590 nm.
Effect of Composition of Oil on Anti-Melanogenesis
The effect of β-sitosterol on melanin production of α-MSH-treated B16F10 cells was tested by co-culturing the cells with 0.1 and 0.5 µg/mL β-sitosterol using the previously mentioned culture method. The free fatty acid content greater than 1% in S. mukorossi oil was also tested. The concentrations of these saponified free fatty acids added to the α-MSH-treated B16F10 cells were set according to the content ratio detected in the GC-MS analysis (Table 1). Cells were treated with the following fatty acids: palmitic acid (1.0 µg/mL), oleic acid (11 µg/mL), linoleic acid (0.5 µg/mL), linolenic acid (1.3 µg/mL), arachidic acid (1.1 µg/mL), and eicosenoic acid (5.1 µg/mL). To assess the effect of the free fatty acids of S. mukorossi oil on tyrosinase activity, buffer-lysed cells from the above procedure were used as samples; 2.5 mM L-DOPA was added to the supernatant samples in a ratio of 1:1. After incubation at 37 °C for 1 hour, the solution absorbance was measured spectrophotometrically at 475 nm.
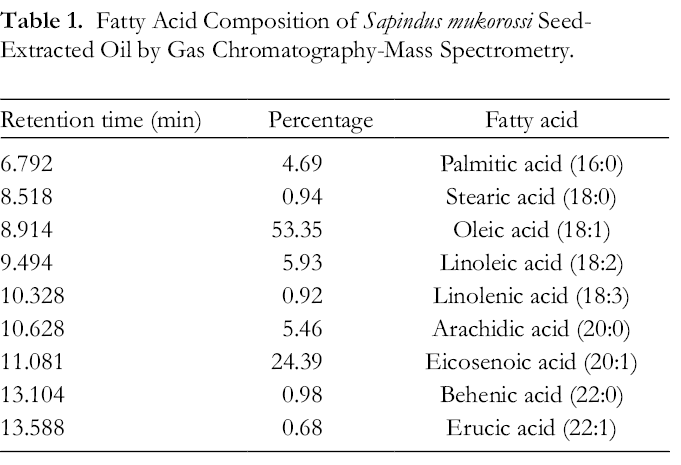
Fatty Acid Composition of Sapindus mukorossi Seed-Extracted Oil by Gas Chromatography-Mass Spectrometry.
Statistical Analysis
Results are presented as mean ± SD for the 4 samples. Intragroup differences were tested using a one-way analysis of variance, and a Scheffe post hoc test was performed to demonstrate the difference. A P value less than 0.05 was considered statistically significant.
Results and Discussion
Melanogenesis is a mechanism for preventing skin damage upon exposure to ultraviolet light. The primary molecule involved in melanogenesis is melanin. Both overexposure to ultraviolet rays and skin injuries may result in the overproduction of melanin and hyperpigmentation disorders. 29 Our GC-MS and HPLC analysis showed that S. mukorossi seeds contain abundant unsaturated fatty acids (85.9%) (Table 1) and β-sitosterol (14.2 mg/mL). Among these unsaturated fatty acids, 90% are monounsaturated. This result is similar to the finding reported by Chen et al. 14 Since both monounsaturated fatty acids and β-sitosterol may cover the skin barrier, act as permeability enhancers, and have a positive effect on wound healing, 30 our results suggest that S. mukorossi seed oil is suitable as a cosmetic material for the daily care of the face and body.
To test the effect of plant oils on melanogenesis in the murine B16F10 melanoma cell model, saponification is an essential process before the oils can be tested. 23 Our results showed that neither the viability nor the growth curve of treated B16F10 cells was affected by the saponified S. mukorossi oil. To test the effect of the emulsifier on the melanin formation, we co-cultured Tween-80 with the α-MSH-induced B16F10 cells. Figure 1(A) shows that even when cells were cultured with Tween-80 at a concentration of 100 µg/mL, no significant reduction in melanin production was found. This result indicates that saponified S. mukorossi oil itself and not the emulsifier significantly reduced α-MSH-enhanced melanin expression (Figure 1(B)) and contributed to the reduction of melanogenesis.

(A) Melanin formation analysis showed that Tween-80 used in this study does not affect antimelanogenesis. (B) The addition of S. mukorossi oil significantly reduced melanin production in a dose-dependent manner. Data are presented as the mean ± SD. Values not sharing a letter are significantly different (P < 0.05).
Figure 1(B) shows that the effect of S. mukorossi oil on melanin inhibition is statistically significant. When the added S. mukorossi oil was lower than 300 µg/mL, the melanin induced by α-MSH was reduced by about 21%. This value increased to 50% when the concentration of the added oil increased to 500 µg/mL. This dose is almost 10-fold higher than that of sunflower (50 µg/mL) and safflower (25 µg/mL) oils reported by Koo et al. 23 The reason for the different melanogenesis inhibition abilities among these plant oils is the various free fatty acid compositions of the plant oils. It has been reported that unsaturated fatty acids, including oleic acid, linoleic acid, and linolenic acid, decrease melanin synthesis, whereas saturated fatty acids such as palmitic acid and stearic acid increase melanin expression. 25,27,31 Among the unsaturated fatty acids, linolenic acid appears to play an essential role in the inhibition of melanogenesis. Since the linoleic acid content in sunflower (66.2%) and safflower (76.6%-79%) oils is much higher than in the tested S. mukorossi oil (6%) (Table 1), it is not surprising that the antimelanogenic effect of sunflower and safflower oils is higher than that of S. mukorossi oil.
In the cosmetic industry, β-sitosterol is valued for its anti-inflammatory and antioxidant properties 32,33 and is reported as a safe chemical without undesirable adverse effects. 34,35 The most famous plant oil enriched with β-sitosterol is shea butter, 30,36 which has an unsaponifiable matter ranging from 1.2% to 10%, 37 -39 with approximately 10% phytosterols found in the unsaponifiable matter. With β-sitosterol accounting for 68% of these phytosterols, it makes up about 0.082%-0.68% of shea butter. 39 Our results indicate that S. mukorossi oil contains about 1.42%, which is 2.1-17 times higher than that reported for shea butter. 37 -39 However, our results indicated that β-sitosterol had no antimelanogenic effect even when 1 µg/mL was co-cultured with α-MSH-induced B16F10 cells. This result confirms a previous study that also found β-sitosterol to have no effect on melanin inhibition. 40
It is well known that several melanogenic enzymes regulate melanin synthesis. Among these enzymes, tyrosinase plays a pivotal role in regulating melanin production, especially in fatty acid-induced regulation of melanogenesis, 41 and is the major melanogenesis-inducing enzyme. The tyrosinase-dependent mechanism in melanogenesis occurs by catalyzing the oxidation of tyrosine to dopaquinone, which leads to the production of eumelanin and pheomelanin. 17 Thus, the inhibition in tyrosinase activity is an indicator of the reduction of melanogenesis. Figure 2 shows that both palmitic acid and arachidic acid exhibited no regulatory function on tyrosinase activity, but the unsaturated fatty acids, such as oleic acid, linoleic acid, and eicosenoic significantly decreased tyrosinase activity. These results confirm previous studies that found while unsaturated fatty acids play a significant role in inhibiting tyrosinase activity, saturated fatty acids have the opposite effect, increasing tyrosinase activity and thus increasing melanin production. 24,25,41

Eicosenoic acid significantly reduced tyrosinase activity in alpha-melanocyte-stimulating hormone (α-MSH)-induced B16 F10 cells. Data are presented as the mean ± SD. Values not sharing a letter are significantly different (P < 0.05).
As shown in Figure 3, palmitic acid displayed the lowest antimelanogenic effect. This result is consistent with previous findings that palmitic acid has the opposite effect on melanin production. 25,41 Ando et al compared the difference in antimelanogenic results between saturated and unsaturated fatty acids and found that unsaturated fatty acids, especially linoleic acid, significantly decreased melanin production. 25 However, such an antimelanogenic effect is not observed in Figure 3 because the quantity of linoleic acid in S. mukorossi oil, at only 5.93%, is low. In addition, this phenomenon also confirmed a previous report that the alteration of melanogenesis by palmitic acid and linoleic acids is independent of the change in mRNA levels for melanogenic enzymes. 41
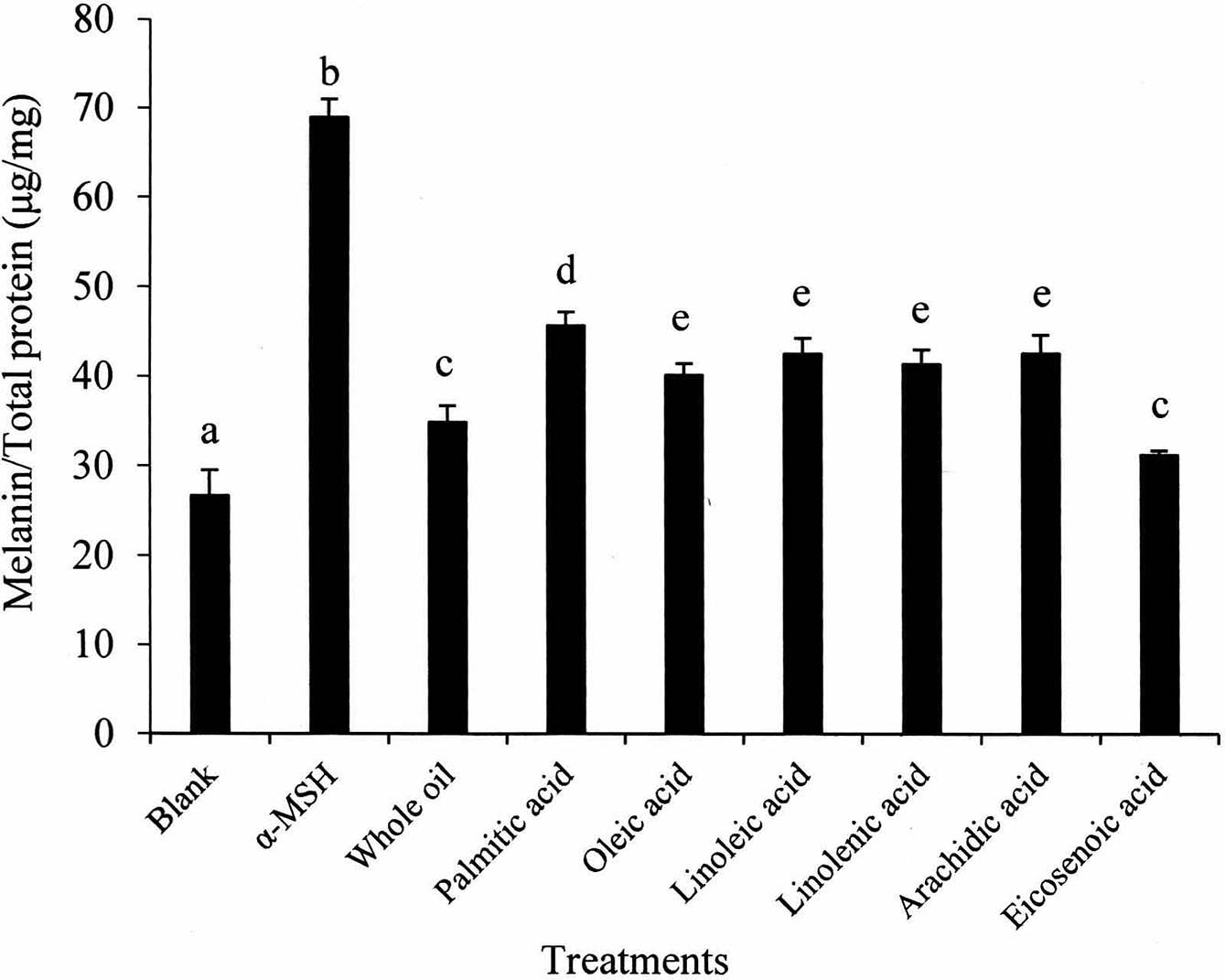
The inhibitive effect of free fatty acids in S. mukorossi oil on alpha-melanocyte-stimulating hormone (α-MSH) induced melanogenesis shows that the major constituent contributing to the antimelanogenic effect is eicosenoic acid. Data are presented as the mean ± SD. Values not sharing a letter are significantly different (P < 0.05).
Sapindus mukorossi oil contains a characteristically large amount of eicosenoic acid which plays a direct role in enhancing skin cell proliferation. 30,42 The eicosenoic acid content detected by GC-MS is about 24.4% (Table 1). However, the antimelanogenic effect of eicosenoic acid has not previously been studied systemically. We found that the melanin expression of cells treated with eicosenoic acid is almost the same as that of pure S. mukorossi oil (Figure 3). This finding suggests that eicosenoic acid plays a pivotal antimelanogenic role in S. mukorossi oil. In addition, since the reduction in tyrosinase activity (Figure 2) and melanin production is consistent, it is reasonable to suggest that the alteration of melanogenesis by eicosenoic acid is related to the change in tyrosinase activity. However, the tyrosinase-dependent mechanism underlying how unsaturated fatty acids reduce melanin deposition at the biomolecular level remains unclear 24,25,41 and will become an important subject of future investigation.
As an inedible oil that contains abundant free fatty acids, S. mukorossi kernel oil has been considered as an alternative, sustainable, and environmentally friendly source of biodiesel, an alternative fuel source that itself reflects reduced pollution and environmental sustainability. 43,44 However, the fabrication of biodiesel from S. mukorossi oils is complicated by a complex transesterification process that results in large amounts of organic solvent waste that in turn requires further treatment. Since our data demonstrates that the cold-pressed oil exhibits a significant antimelanogenic effect, it provides another economic value of being used to replace the existing chemical bleaching agent. Although more investigation is needed, the cultivation and harvesting of these alternative sources for the cosmetics industry could have even wider-ranging positive effects on improving agricultural and rural economies. 45 -48
Conclusions
Although various chemicals are used to treat uneven skin tone, scientific evidence for the antimelanogenic benefits of S. mukorossi seed oil is incomplete. To the best of our knowledge, our results are the first to indicate that S. mukorossi seed oil has a remarkable antimelanogenic effect. In addition, the principal constituent in the extract that contributes to the inhibition of melanin formation is eicosenoic acid. We suggest that S. mukorossi seed oil has the potential for development as a skin tone evening agent in the cosmetic industry.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
