Abstract
Objective
Natural product-derived biomaterials are highly desired as possible ingredients in cosmetics, as they are often safe and effective. Salvianic acid A (SAA) and salvianolic acid B (SAB) are the primary water-soluble ingredients of Salvia miltiorrhiza Bunge (Lamiaceae). These compounds have extensive applications as both effective and biocompatible bioactive agents for the treatment of various diseases including cardiovascular disorders, liver ailments, and neuroprotection. This study aimed to evaluate the skin-whitening effects of SAA and SAB on co-cultured human melanoma A375 cells and human keratinocytes HaCaT cells, with the intention of investigating their potential as cosmetic ingredients.
Methods
First, we assessed the SAA and SAB's efficacy on cells proliferation using MTT assay, as well as their inhibition on intracellular tyrosinase (TYR) activity. Next, we evaluated their effects on the mRNA expression levels of TYR and its related proteins. Furthermore, the whitening efficacy of SAA and SAB was assessed by observing the decrease in melanin content in treated human amalignant melanoma A375 and human keratinocyte HaCaT cells.
Results
Both SAA and SAB inhibited the activity of TYR and decreased the mRNA expression of TYR-related proteins, thereby inhibiting melanin synthesis.
Conclusion
Our results suggest that both SAA and SAB can be applied as natural and novel cosmetics materials having a whitening function, which could be beneficial in the cosmeceutical industry.
Introduction
The application of traditional herbs in skin whitening had quite a long history in Asia. Studies have confirmed the pigmentation reduction efficacy of the components from traditional herbs, such as arbutin and kojic acid. 1 In addition, Asian consumers have strong need and trust for traditional herb products in skin whitening because the safety and less side-effect they showed. 2 Salvia miltiorrhiza is a traditional Chinese medicine with the effect of promoting blood circulation and removing blood stasis. 3 It contains various active ingredients such as salvianic acid A (SAA), salvianolic acid B (SAB) (Figure 1), and tanshinone, and has significant effects on cardiovascular disease, liver, neuroprotection, and cancer treatment.4-10 Our study verified the whitening effects of the skin using SAA and SAB that has been traditionally used for medicine through a molecular mechanism approach.

Chemical structure of salvianic acid A (SAA) and salvianolic acid B (SAB).
The difference in the color of human skin usually depends on the content and distribution of melanin and carotenoids in the epidermis, and heme in the dermis, among which melanin plays as the key factor. 11 Skin whitening is affected by many factors, but mainly depends on melanin synthesis ability of melanocytes. L-Tyrosine in melanocytes is oxidized to form dopamine quinone (DQ) under the action of an enzyme (Tyrosinase, TYR). With the participation of TYR-related protein 1 (TRP-1) and TYR -related protein 2 (TRP-2), DQ further oxygenates to form 5,6-dihydroxyindole-2-carboxylic acid and 5,6-dihydroxyindole, both undergo a series of catalytic polymerization to finally synthesize melanin. After that, melanin will be transported to nearby keratinocytes and then moves to the stratum corneum causing pigmentation. 12 Studies have shown that melanogenesis inhibitors can be used to treat hyperpigmentation of the skin.10,13 The expression and activation of TYR have the most immediate impact on melanin synthesis, 14 as the other 2 key enzymes in the melanogenesis pathway, the gene expression of TRP-1 and TRP-2 is also critical in promoting melanin synthesis.15-18 Therefore, to inhibit melanin production by inhibiting TYR or intermediates of melanogenesis in cells is the most common mechanism of cosmetics for skin whitening at present.13,19
In this study, we demonstrated the whitening effects of SAA and SAB using a cell co-culture system to assess the impact on melanocyte activity, melanin and melanin-related enzymes, and their gene expression. Our findings contribute to expanding the scope of applications of Salvia miltiorrhiza by validating its skin whitening effects along with the value of traditional use of the herb.
Results
Effects of SAA and SAB on Cell Viability
We evaluated the effects of co-cultured cells of human melanoma cells A375 and human keratinocytes HaCaT under SAA and SAB at 4 different concentrations through MTT assay. The results showed that SAB exhibited no toxicity to the cells up to 1.25 μg/mL, but decreased cell viability at 2.5 and 5.0 μg/mL. SAA did not block cell viability at 0.625 μg/mL, but slightly decrease viability at 1.25 μg/mL, the cell viability was the lowest at the highest concentration of 5 µg/mL, and all cell viability was near or above 80% (see Figure 2).

Effects of SAA (A) and SAB (B) on the viability of A375 and HaCaT co-cultured cells. Compared with the blank control group, *P < .05, **P < .01.
SAA and SAB Inhibit Melanin Synthesis in A375 and HaCaT Co-cultured Cells
To identify the inhibitory effect of SAA and SAB on melanin synthesis, A375 and HaCaT cells were treated at concentrations of 0.625, 1.25, 2.5, and 5.0 μg/mL to compare melanin inhibition rates. Arbutin was regarded as a whitening ingredient 20 and has been used as a positive control in this study. The results showed a concentration-dependent inhibition on melanin production by SAA and SAB (see Figure 3). SAB showed the highest inhibitory effect at the concentration of 5.0 μg/mL. With the increase of concentration, the inhibitory effect of SAA on melanin synthesis was on the rise, there was a significant difference (P < .01).
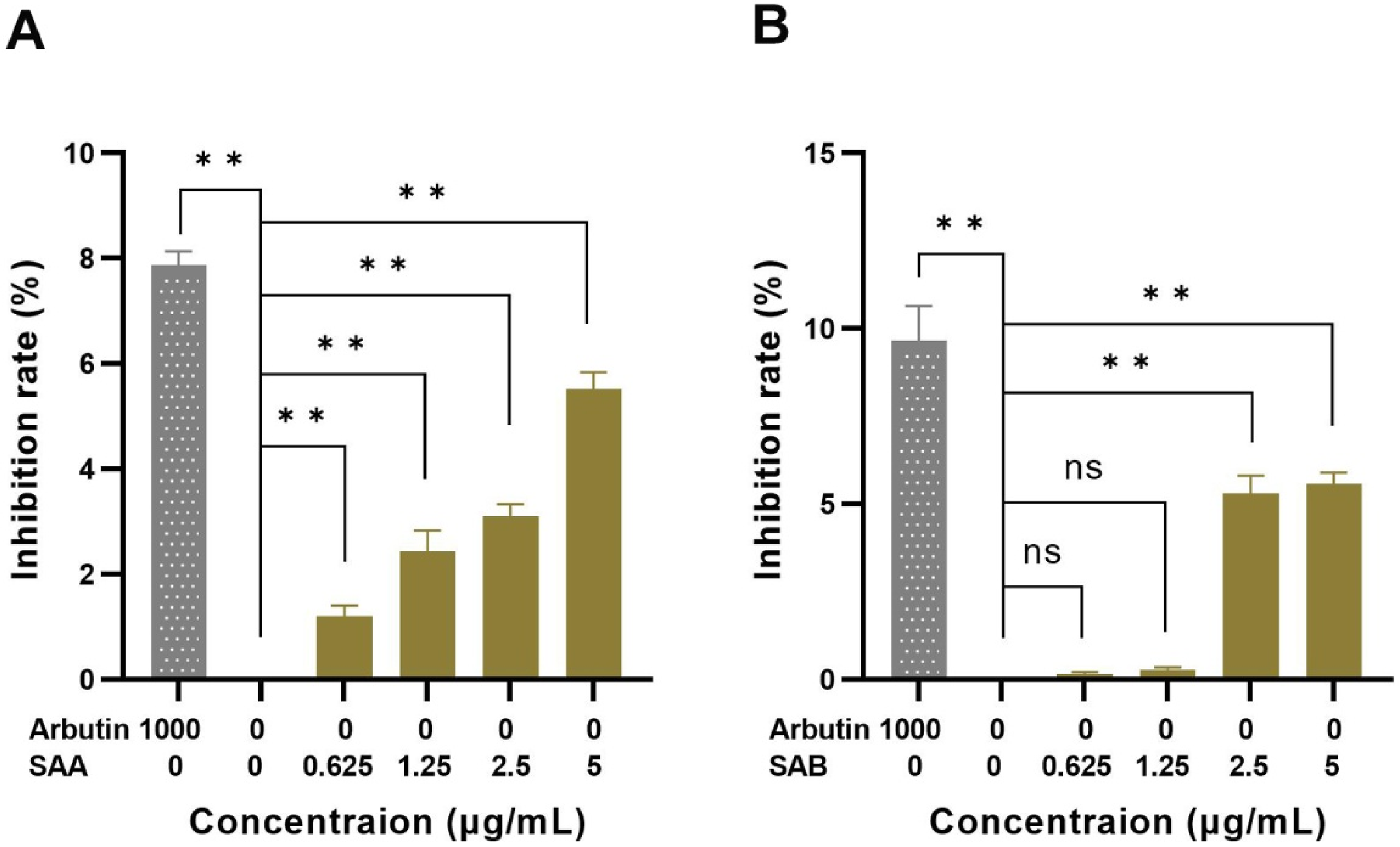
Effect of SAA (A) and SAB (B) on melanin synthesis. Compared with the negative control group, *P < .05, **P < .01.
SAA and SAB Inhibit TYR Activity
TYR activity assay was performed to verify that the melanogenesis inhibitory effects of SAA and SAB caused by the regulation of TYR. The results showed that SAA and SAB directly inhibited TYR activity at 3 different concentrations (see Table 1).
Inhibition Rates of SAA and SAB on TYR Activity.

SAA and SAB Inhibit TYR Activity in A375 and HaCaT Co-cultured Cells
To further analyze the effects of SAA and SAB on melanin synthesis in co-cultured cells, we investigated the inhibitory effects of SAA and SAB on TYR in co-cultured cells (see Figure 4). It was found that SAA and SAB showed different inhibitory effects on TYR activity at different concentrations, and the results of experimental groups were significantly different from that of the negative control groups (P < .01). SAB had the greatest inhibitory effect on TYR at the concentration of 1.25 μg/mL, and then showed a decreasing trend. SAA had the greatest inhibitory effect on TYR at the concentration of 0.625 μg/mL.
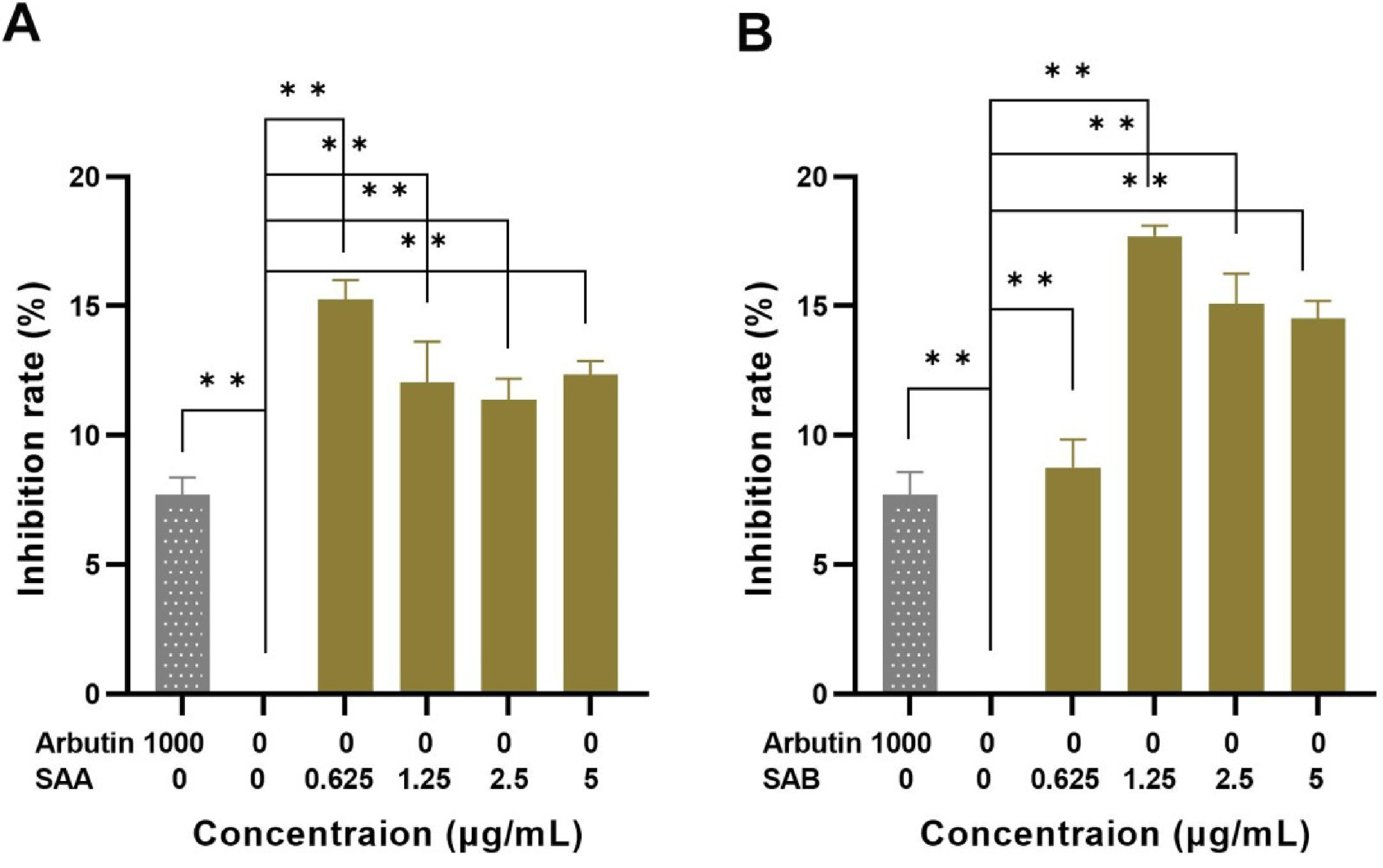
Effect of SAA (A) and SAB (B) on TYR activity. Compared with the negative control group, *P < .05, **P < .01.
SAA and SAB Inhibit the Expression of Melanosome Biogenesis-related Factors
The synthesis of human melanin was synergistically affected by TYR, TRP-1, and TRP-2. To further investigate the effects of SAA and SAB on TYR content, mRNA expression levels of TYR-related proteins in A375 and HaCaT co-cultured cells were analyzed using RT-PCR. After the PCR was done, the cDNA was synthesized from RNA, separated on 1% agarose gel, and evaluated. In comparison, beta-actin was used as internal reference. The results of the band density of gel electrophoresis were shown in Figure 5. Transcription levels of TYR, TRP-1, and TPR-2 were significantly decreased by SAA (0.625 μg/mL) compared with the negative control group (Figure 5A-D and Table 2). Treatment of SAB did not significantly alter the ratio of TYR (Figure 5A-C, 5E and Table 2) while it had significantly depressed the levels of TRP-1 and TRP-2 in the cells. These results suggest that the inhibitory effects of SAA were much greater than that of SAB, thus effectively inhibiting the expression of melanogenesis-related factors.

(A-C) The expression levels of TYR, TRP-1, and TRP-2 treated with SAA (0.625 μg/mL) and SAB (1.25 μg/mL) were measured, respectively, untreated genes was used as negative control. (D) and (E) the ratio of band density was evaluated, *P < .05 versus untreated control group, **P < .01 versus untreated control group.
Ratio of Band Density in RT-PCR Experiments (

*P < .05 compared with negative control. **P < .01 compared with negative control.
Discussion
Due to the potential cytotoxicity and skin irritation of artificially synthesized chemical components, screening and developing of natural herb ingredients as raw materials for skin whitening has become important in the cosmetics industry. Because of the richness in pharmacological effects, Salvia miltiorrhiza has been used as a traditional medicine for a long time. However, the potential use of Salvia miltiorrhiza in the cosmetic industries has yet to be explored. Therefore, we performed studies to evaluate the skin whitening activity of SAA and SAB. It was found that SAA and SAB, as the main active ingredients in Salvia miltiorrhiza, can inhibit the melanogenesis in melanocytes, which makes Salvia miltiorrhiza has potential skin whitening value in the cosmetic field.
Previously, the pharmacological effects of whitening ingredients were predominantly tested using melanoma cells.21-23 In human epidermis, each melanocyte forms an “epidermal melanin unit” with neighboring keratinocytes. 24 Under normal conditions, keratinocytes regulate melanocyte morphology, melanin synthesis, and transport through the secretion of various cytokines including basic fibroblast growth factor, stem cell factor, extracellular matrix,25,26 endothelin, and leukotriene. These cytokines directly stimulate melanocytes, promoting cell proliferation and melanin synthesis. 27 Therefore, it is crucial to consider the interaction between melanocytes and keratinocytes especially in the drug screening and validation of skin-whitening. In this experiment, a co-cultured model of human melanoma A375 cell line and human keratinocyte HaCaT cell line was utilized to stimulate the interactions between skin-resident cells. This model allowed for a comprehensive evaluation of the effects of Salvia miltiorrhiza on melanogenesis, thereby augmenting its potential value in clinical applications. Importantly, the inhibitory effect of arbutin on melanogenesis in A375 cells cultured alone has been confirmed. To validate the results of this experiment, we conducted tests using co-cultured A375 and HaCaT cells, with arbutin serving as a control.
The mechanism of action for cosmetics with whitening effects can be categorized based on the factors influencing skin color and the biogenesis of melanosome. These mechanisms include inhibiting melanin synthesis, inhibiting melanocyte proliferation, preventing melanin transfer to keratinocytes, and reducing expression levels of proteins involved in melanin synthesis. The assay results for melanin content demonstrated that both SAA and SAB exhibited a concentration-dependent inhibition of melanin synthesis.
TYR, a key enzyme in melanin synthesis, plays a decisive role in the entire enzymatic catalytic process. 28 Inhibiting the activity of TYR can effectively inhibit melanin synthesis, thereby achieving skin whitening effects. Therefore, we further investigated the effect of SAA and SAB on TYR in cells. The results showed that SAB significantly inhibited TYR activity at all treatment concentrations, its inhibitory effect on TYR was even one time higher than that of arbutin at the concentration of 1.25 μg/mL, indicating that SAB is effective in whitening through inhibiting TYR activity. SAA was also found to significantly inhibit the synthesis of melanin and TYR activity, with the strongest inhibitory effect on TYR occurring at a concentration of 0.625 µg/mL. There are several factors that contribute to the effects of compounds on intracellular TYR activity. Some compounds directly inhibit enzyme activity s by competitively binding to the enzyme's binding site. Others indirectly influence cellular activity and enzyme expression, which ultimately impacts the activity of the targeted enzyme. Although arbutin demonstrated a noticeable inhibitory function on TYR activity during the in-vitro test, SAA and SAB exhibited significant inhibitory effects in the cellular assay, surpassing arbutin. This difference may be attributed to the complex interaction mechanisms between the compounds and enzymes in the cells, contrasting the more straightforward situation in the in-vitro test. In the intracellular assay, the lowest TYR activity was observed at a concentration of 1.25 μg/mL when the co-cultured cells were treated with SAB. As the concentration increased, the TYR activity also increased. Similarly, the activity of TYR increased at higher concentrations of SAA. This can be attributed to the inhibitory effect of the compounds on cellular activity at low concentrations, resulting in a decrease in TYR expression and subsequent activity. At higher concentrations, the compounds directly inhibited TYR activity, although the inhibitory effect was not significant. Consequently, the compounds did not display concentration-dependent effects on intracellular TYR activity in the experiment.
TYR-related proteins (TRP-1, TRP-2) also played a key role in melanosome biogenesis,29,30 therefore, we further studied whether SAA and SAB could decrease the expression of TYR related genes in melanocytes. The optimal concentration for subsequent experiments was selected based on the inhibition rate of SAA and SAB on TYR. The results demonstrated that a concentration of 0.625 µg/mL of SAA inhibits the expression of TYR and TYR-related proteins (TRP-1, TRP-2), thereby decreasing the content of TYR, TRP-1, and TRP-2 and subsequently inhibiting melanin synthesis. SAA exhibits better whitening effects compared to SAB as it is more effective in down-regulating the expression of TYR and related genes. Our findings demonstrated that SAA and SAB reduce the levels and synergistically inhibit the activities of TYR and related genes, resulting in the inhibition of melanin synthesis and the achievement of the skin whitening effect. In conclusion, our study has shown that SAA and SAB exhibit a whitening effect through multiple mechanisms, including the inhibition of melanin synthesis and TYR and its related genes. Therefore, SAA and SAB could be used as new whitening materials for the development of effective whitening products. Due to time limitation, the inhibitory effect of SAA and SAB on TYR is either competitive or non-competitive requires further investigation. In addition, this study primarily focuses on exploring the inhibitory effects of SAA and SAB on melanogenesis. It is important to note that exposure to ultraviolet (UV) radiation is a crucial factor in pigment accumulation 31 as it stimulates melanin biosynthesis in epidermal melanocytes. 32 Therefore, future research should aim to investigate the anti-UV radiation effects of SAA and SAB on cells to provide a more comprehensive understanding of their potential for skin whitening.
Materials and Methods
Materials and Reagents
SAA and SAB were purchased from Nanjing Zelang Pharmaceutical Co., Ltd (Nanjing, China); L-DOPA was purchased from Shanghai Sculler Biotechnology Co., Ltd (Shanghai, China); TYR was obtained from Roche (Basel, Switzerland); Arbutin was purchased from Sigma Aldrich (Saint Louis, MO, USA). Other chemicals (Beijing Analytic Co., ltd, Beijing, China) were of reagent grade.
Cell Lines and Culture Conditions
Human amalignant melanoma A375 cells used in this experiment were purchased from Cell Center of Chinese Academy of Sciences (Beijing, China), human keratinocyte HaCaT cells were obtained from China Center for Type Culture Collection (Wuhan, China). The cells were cultured in MEM containing 10% fetal bovine serum and 1% penicillin–streptomycin at 37 °C and under the condition of 5% CO2.
MTT Assay
A375 and HaCaT cells in logarithmic growth phase were seeded in 96-well plate with a ratio of 1:2 at a density of 1.5 × 104 cells/mL and incubated for 24 h. SAA and SAB were diluted in MEM producing 0.625, 1.25, 2.5, 5.0 μg/mL concentrations and added to each well with 200 μL. After incubation for 48 h, 20 μL of 5 mg/mL MTT was treated to each well, 6 replicate wells were set in each group. Subsequently, the supernatants were removed from each well and replaced with 150 μL DMSO. Absorbance was measured at 490 nm using a microplate reader (Thermo Fisher Scientific Inc., Waltham, MA, USA).
Melanin Content Assay
A375 cells and HaCaT cells were seeded at the density of 4.5 × 104 cell/mL in a 96-well plate and incubated at 37 °C for 24 h. They were then treated by various concentrations of SAA and SAB (0.625, 1.25, 2.5, 5.0 μg/mL) according to the results of MTT assay. Arbutin was dissolved in MEM to a final concentration of 1000 μg/mL. After incubation for 48 h, the culture medium was removed and the cells were washed twice with PBS. After digestion with 0.25% trypsin 1 mL per hole, the cell suspension was collected and centrifuged. Then, the cells were dissolved with 100 μL of 1 mol/L NaOH and incubated at 37 °C for 1 h. Six replicate wells were set in each group, the OD value of each well was measured at a wavelength of 490 nm. The inhibition rate was calculated as: [(OD value/cell number of the experimental group) / (OD value/cell number of negative control group)] × 100%.
TYR Activity Test
Various concentrations (0.01, 0.1, 1.0 mg/mL) of SAA, SAB, and arbutin, 0.1 mol/L PB (pH 6.8), and 0.5% L-DOPA were added to 96-well plate to mix well, 6 replicate wells were designed in each group (see Table 3), they were then treated with 100 U/mL TYR and incubated at 37 °C for 10 min, the absorbance of the mixtures was measured with a spectrophotometer at 490 nm. The inhibition rate is calculated as: [(OD value of Group A−OD value of Group B)−(OD value of Group C−OD value of Group D)/(OD value of Group A−OD value of Group B)] × 100%.
Composition of the Test Solution.

Intracellular TYR Activity Assay
A375 cells and HaCaT cells in the logarithmic growth phase were digested with 0.25% trypsin and seeded in a 96-well at a density of 1.5 × 104/mL, and incubated at 37 °C for 24 h. According to the results of MTT assay, they were then treated with various concentrations of SAB and DSU, 6 replicate wells were designed in each group. After incubation for 48 h, each well was treated with 200 mL of lysis buffer (1% Triton-X) to collect the lysate. Next, each group was incubated with 50 μL of 0.5% L-DOPA for 30 h at 37 °C. The OD value of each well was measured at a wavelength of 490 nm. The inhibition rate is calculated as: [(OD value of experimental group − OD value of blank group) / (OD value of negative control − OD value of blank group)] × 100%.
Gene Expression Analysis (RT-PCR)
To determine the gene expression of TYR, A375 cells and HaCaT cells in logarithmic growth phase were seeded in 96-well plate at a density of 1.5 × 104 cells/well and incubated for 16 h, and further treated with SAA and SAB and then subjected to mRNA extraction. A negative control was treated with MEM medium. The total RNA was extracted from the cells using RNA extraction kit (Sangon Biotech, Shanghai, China) and reverse transcribed into cDNA, and cDNA synthesis was performed using RT-PCR premix (Sangon Biotech, Shanghai, China). Then, RT-PCR was performed to evaluate the expression of TYR, TRP-1, and TRP-2. The primers (Shengong Bioengineering, Shanghai, China) used in this experiment are listed in Table 4.
Primer Sequences.

The cDNA samples of each group were mixed with 5 μL of 10 × reaction buffer, 5 μL of dNTP, 0.2 μL of Taq enzyme (5 U/μL), and 3 μL of MgCl2, following the addition of primers of TYR, TRP-1, TPR-2, and internal reference (β-actin). Finally, the PCR tube was put into the PCR amplifier (Biometra, Göttingen, Germany) according to the steps listed in Table 5. The DNA samples of each group were obtained after 35 consecutive cycles from step No. 2 to No. 5. Subsequently, mRNA expression patterns were identified by agarose gel electrophoresis.
PCR Amplification Procedure.

Statistical Analysis
All the data were processed by SPSS16.0 statistical software package and tested by ANOVA.
Footnotes
Acknowledgements
The authors thank the editor and reviewers for their comments and efforts toward improving our manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China. Youth Science Foundation(No. 82204562); the National Key Research and Development Program of the 13th Five-year Plan in China-R & D and Demonstration of Ecological Forest Exploitation and Utilization Technology of Forest Drugs in Northeast Forest Region (Study on the Key Technologies in the Production of Bulbus Fritillaria ussuriensis, No. 2016YFC0500303-03); Heilongjiang Traditional Chinese Medicine Young and Middle-aged Science and Technology Tackling Project (No. 2QG-038).
Supporting Information
The preparation details of solutions used in the assays are available on request to the corresponding author.
