Abstract
Introduction
Nelumbo nucifera Gaertn. (N. nucifera) is a perennial, rhizomatous, and aquatic plant, which was distributed throughout Asia and Australia. Among the different organs of N. nucifera, the leaves and the flowers are commonly used in traditional medicine, while the roots and the seeds provide important nutritional foods.
1
Currently, N. nucifera has acquired enormous significance because of its high content of bioactive secondary metabolites, such as flavonoids and anthocyanins in the flower
2
; alkaloids, flavonoids, steroids, and polysaccharides in the plumule
3
; polyphenols, fatty acids, procyanidins, and polysaccharides in the rhizome and seeds4,5; and phenolic acids, flavonoids, alkaloids, polysaccharides, and terpenoids in the leaves.
6
In ancient China, the leaves of N. nucifera were known for diuretic and astringent properties and were used to treat fever, sweating, and strangury and as a styptic.
7
As early as 1991, the leaves of N. nucifera were included in the second list of “food and drugs” by the Ministry of Health of China. Modern medical studies have revealed the extensive pharmacological activities of the leaves, including antioxidant, antimicrobial, anti-obesity, and anti-cardiovascular disease activities. As a part of our study on the hydrophilic bioactive constituents of Chinese medicines,8,9 we systematically studied the water-soluble constituents of N. nucifera. The water-soluble fraction from 60% aqueous acetone extract of the leaves of N. nucifera was subjected to column chromatography on MCI gel CHP 20P, YMC-Gel ODS-AQ-HG, and TSK gel Toyopearl HW-40F to afford one novel flavonoid (

Chemical structures of compounds
Results and Discussion
Compound
In the 1H NMR spectrum (Table 1), the signals at δ 6.76 (1H, br d, J = 8.2 Hz, H-6′) and δ 6.90 (1H, d, J = 8.2 Hz, H-5′) and δ 6.88 (1H, d, J = 1.6 Hz, H-2′) defined a 13,4-trisubstituted aromatic ring, which was further confirmed in the 13C NMR spectrum by three quaternary aromatic carbons at δ 130.83 (C-1′), 144.12 (C-3′, C-4′) and three methine peaks at δ 114.09 (C-2′), 116.24 (C-5′) and 118.80 (C-6′). The signals at δ 6.15 (1H, s) in the low field of the 1H NMR spectrum and five quaternary aromatic carbons and one methine peak in the 13C NMR spectrum indicated the existence of another aromatic ring. In the high field of 1H NMR spectrum, one methylene group δ 2.64 (1H, dd, J = 16.4, 4.6 Hz, H-4a), 2.58 (1H, dd, J = 16.4, 6.1 Hz, H-4b), and two methine groups δ 5.05 (1H, d, J = 5.7 Hz, H-2) and 4.21 (1H, m, H-3) indicated the following CH(2)-CH(3)-CH2(4) fragment by the analysis of 1H-1H correlation spectroscopy (1H-1H COSY) spectrum and heteronuclear multiple bond coherence (HMBC) spectrum (Figure 2). Further analysis of 1H-1H COSY, heteronuclear single quantum coherence and HMBC spectra established the skeleton of catechin, in which HMBC long-range correlations were observed not only between H-2 (δ 5.05), H-3 (δ 4.21), and C-1′ (δ 130.83), but also between H-4a (δ 2.64), H-4b (δ 2.58), and C-5 (δ 158.95), C-9 (δ 152.43), and C-10 (δ 102.37).
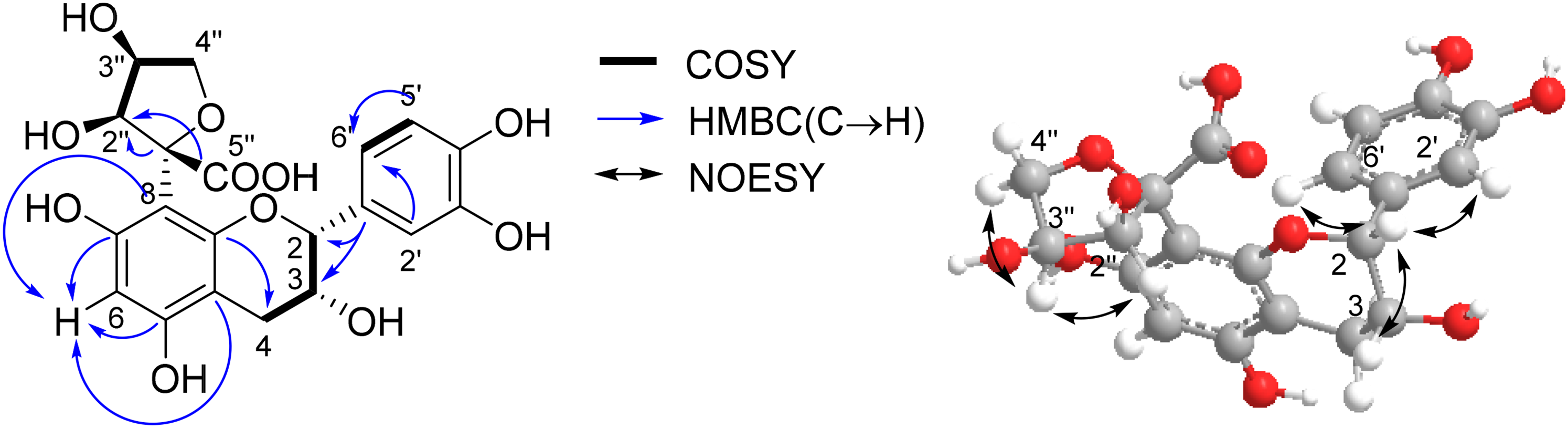
Key COSY, HMBC, and NOESY correlations for compound
NMR Data for Compound

600 MHz, D2O; chemical shifts in ppm; coupling constant (J) in Hz.
150 MHz, D2O; multiplicity was established from the DEPT data.
HMBC, heteronuclear multiple bond coherence; NMR, nuclear magnetic resonance.
In addition, the low field of the 13C NMR spectrum still showed the presence of one carbonyl carbon, one quarternary carbon, two methines, and one methylene group. The analysis of 1H-1H COSY showed another remaining -CH(O)-CH(O)-CH2-O- fragment. The HMBC long-range correlations between H-2″ (δ 4.51) and C-1″ (δ 110.80), C-5″ (δ 174.20) confirmed the presence of CH(2″)-C(1″)-COOH(5″) fragment. The methylene group δ 3.86 (1H, dd, J = 11.8, 4.3 Hz, H-4″a), 3.75 (1H, dd, J = 11.8, 6.6 Hz, H-4″b) in 1H NMR and C-4″ (δ 62.11) in 13C NMR suggested the formation of ether ring between C-4″ and C-1″, instead of forming lactone between C-4″ and C-5″. The HMBC long-range correlations between H-6 (δ 6.15) and C-5 (δ 158.95), C-7 (δ 156.3), C-8 (δ 100.77), C-10 (δ 102.37) confirmed the site of C-8 is a quaternary carbon. Therefore, the five-membered heterocyclic ring should be directly connected to the C8.
The relative configuration of epicatechin was deduced from the results of a nuclear Overhauser effect spectroscopy (NOESY) spectrum (Figure S7), in which NOESY signals were observed between H-2 (δ 5.05) and H-3 (δ 4.21), H-2′ (δ 6.88), H-6′ (δ 6.76). Similarly, NOESY signals were observed between H-3″and H-2″ and H-4″a/b, indicating that H-3″ and H-2″ are on the same side of the five-membered heterocyclic ring. The chemical shift of H-2″ is located in a lower field relative to H-3″, which may be caused by intramolecular hydrogen bonding of -COOH and -OH are on the same side. The absolute configuration is determined as (−)-2R, 3R, 1″R, 2″S, 3″S by comparing the calculated electronic circular dichroism (ECD) spectroscopy with the measured circular dichroism (CD) spectroscopy results (Figure S8). Consequently, the chemical structure of
The eleven known compounds were identified as Quercetin 3-O-β-D-Glucuronide (
The C. elegans. model was employed to explore the potential of compounds

Caenorhabditis elegans survival curves of selected compounds (100 μg/ml).
The Mean Lifespan After Treatment of Selected Compounds.

In summary, compound
Conclusion
One novel compound
Experimental
General Experimental Procedures
Optical rotation data were obtained on a Perkin–Elmer 241 automatic digital polarimeter. 1H, 13C NMR, 1H-1H COSY, HSQC and HMBC spectra were recorded on a Bruker DRX-600 spectrometer (1H 600 MHz and 13C 150 MHz). The carbon multiplicities were obtained by the distortionless enhancement by polarization transfer experiment. ESI-MS data was measured on an ESI-MS HP5989A mass spectrometer. TSK gel Toyopearl HW-40F (30-60 μm, Toso Co., Ltd), MCI gel CHP 20P (75-150 μm, Mitsubishi Chemical Industries Co., Ltd) and YMC-Gel ODS-AQ-HG (42-105 μm, YMC Group) were used for column chromatography. Thin layer chromatography was performed using a Kiesel gel 60 F254 pre-coated plate. (0.2 mm, Merck). HR-ESI-MS data were measured on AB Qtrap5500.
Plant Material
The 5 kg fresh leaves of N. nucifera were collected in Chongming Island, Shanghai, People's Republic of China in autumn 2021, and certified by Professor T. Guo. A voucher specimen has been deposited in the School of Pharmacy, Fudan University.
Extraction and Isolation
Fresh leaves of N. nucifera (5 kg) were subjected to two extractions using 60% aqueous acetone at room temperature (10 L). The solvent was evaporated under reduced pressure to 1 L and subsequently filtered through cotton and celite. The resulting filtrate was concentrated in vacuo, yielding a black oil, of approximately 100 mL. Water was then supplemented to 300 mL and subjected to chromatography using MCI gel CHP 20P (8 × 60 cm) eluted with methanol (MeOH)/H2O [300 mL H2O → 10% MeOH → 30% MeOH → 50% MeOH → 70% MeOH → 100% MeOH] and acetone/H2O [300 mL 60% acetone → 100% acetone] to obtain fraction 1 and fraction 2. Fraction 1 was further chromatographed on Toyopearl HW-40F (6 × 60 cm), resulting in four sub-fractions 1A-1D [400 mL H2O → 300 mL 50% MeOH → 300 mL 100% MeOH → 400 mL 60% acetone]. Fraction 1C was then purified using YMC-Gel ODS-AQ-HG (4 × 30 cm) with elution of H2O → 70% MeOH and middle-pressure liquid chromatography (C18-MPLC) with elution of [H2O → 50% MeOH], yielding compound
Nuciferanoid (1 )
White amorphous powder; [α]25 D −34.9 (c 0.10, EtOH); ESI-MS m/z 435 [M - H]−; HR-ESI-MS m/z 436.1005 [M]+ (calcd for C20H20O11, 436.1011); 1H and 13C NMR, see Table 1.
ECD Calculation
The random conformational searches were performed by Spartan’14 using Molecular Merck force field. The conformers with a Boltzmann population of over 5% were subsequently optimized by using Gaussion09 software at the B3LYP/6-31G (d) level in MeOH using the IEFPCM model. The optimized stable conformers were selected for further ECD calculations at the B3LYP/6-311G (d, p) level in MeOH. The overall ECD data were weighted by Boltzmann distribution, and the ECD curves and enantiomeric ECD curves were produced by SpecDis 1.70.1 software with a half-bandwidth of 0.3 eV, according to the Boltzmann-calculated contribution of each conformer after UV correction.
Anti-Aging Tests
Compounds
Statistical Analysis
Survival curves were plotted and analyzed using GraphPad Prism 9.5.1.
The formula for calculating the mean lifespan of C. elegans is:
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241271623 - Supplemental material for Research on Water-Soluble Constituents and Anti-Aging Effect of the Leaves of Nelumbo nucifera Gaertn
Supplemental material, sj-docx-1-npx-10.1177_1934578X241271623 for Research on Water-Soluble Constituents and Anti-Aging Effect of the Leaves of Nelumbo nucifera Gaertn by Rong-Qian Cheng, Heyanhao Zhang and Jun Chang in Natural Product Communications
Footnotes
Acknowledgements
This project was supported financially by Science and Technology Commission of Shanghai Municipality (21S11907400).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Commission of Shanghai Municipality, (grant number 21S11907400).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
