Abstract
In this study, we investigated the effect of a mixture of Platycladus orientalis (L.) Franco leaf extract and α-terpineol, a natural monoterpene alcohol (PEaT), on hair growth and its mechanisms. C57/BL6 mice (total n = 14) in the telogen phase of hair growth were used. Either distilled water as vehicle or PEaT was topically applied to the dorsal skin for 17 days. Chronological hair growth change was examined by hair growth-promoting scores. In addition, to find out mechanisms of PEaT on hair growth, insulin-like growth factor-1 (IGF-1), vascular endothelial growth factor (VEGF), Ki-67, wnt3, and β-catenin expressions were investigated by using immunohistochemistry. We found that PEaT remarkably promoted hair growth by inducing early anagen transition compared with the control group. In addition, treatment with PEaT significantly increased numbers of Ki-67-positive cells and expressions of IGF-1, VEGF, wnt3, and β-catenin in the outer root sheath. These results indicate that PEaT used in this study might be a good hair growth promoter, showing that PEaT treatment increased growth factors and cell proliferation through upregulation of wnt3 and β-catenin expressions.
Hair consists of the follicle and hair shaft, and the lowermost portion of the hair follicle includes dermal papilla and hair matrix, which contain active cells (hair germ). 1 Adult hair follicle cells have a high turnover rate and go through a continuous cycle of anagen, catagen, and telogen to produce new hairs throughout the life of the hair follicle. 2 The anagen phase is one of active growth; the cells in the hair bulb divide rapidly to produce an entire hair shaft growth. The catagen phase is a very short period of hair regression; hair stops growing and forms a club hair, including apoptosis of epithelial cells in the bulb. The final telogen stage is a resting period; club hairs shed finally while follicles reset and prepare their stem cells (germ) to start the next hair cycle. 1 -3
Loss of hair is considered a common aging phenomenon, 4,5 which is caused by genetic factors. 6 Hair loss is increased by stress 7 and nutrient deficiency. 8 Recently, the population suffering from hair loss has been increasing. 9 Hair loss (alopecia) is not limited to men, but also occurs in women, but they have a different pattern. 10 Namely, bitemporal recession and vertex baldness is shown in men, and diffuse alopecia over the mid-frontal scalp occurs in women. Hair loss is distressing and decreases the individual’s quality of life. 10
To date, plenty of drugs, such as minoxidil, finasteride, fluridil, and cyproterone acetate, have been studied to either prevent or treat hair loss. 11 However, redness, irritant and allergic contact dermatitis, pruritus, scaling of the scalp (limited to the scalp), and chest pain are known to be common side effects of them. 12,13 Hair loss treatment takes a long time, and, therefore, it is necessary to develop medication with fewer side effects and using better formulation methods. 11
Platycladus orientalis (L.) Franco is an ever-green tree and an extract of its leaves has been used for hair growth promotion and hair loss inhibition.
14,15
The leaves have been reported to contain abietane, isopimarane, diterpenes, dihydrobenzofuran, neolignane, sesquiterpene glycosides, phenolic glycosides,
16
monoterpenes, such as thujone, cedrol, and isopimaric acid, diterpenes, including pinusolide, lambertianinc acid, hinokiol, and sandaracopimaric acid, glycosides (4-E-propenyl-phenol-1-O-β-
The monoterpene cedrol, isolated from P. orientalis leaves, showed hair growth-promoting activity with safe status. 19 In addition, it has been reported that hair growth promoted by Thuja orientalis extract is prompted when it is treated with monoolein cubosomal suspensions that enhance skin permeation of the herbal extracts. 15 α-Terpineol, a major monoterpene alcohol, is a volatile compound found in many plant aromatic species. 20 It is known to be a skin penetration enhancer at concentrations of 1%, 21 2.5%, 22 and 5% 23 by either increasing solubility within the stratum corneum (the outermost layer of the skin) or lipid fluidity of the intracellular lipid bilayers, which can retain substances in the skin. 20,24 However, there are few studies so far on hair growth promotion effects of a mixture of P. orientalis leaves extract and α-terpineol (PEaT) and its subsequent mechanisms. Therefore, in this study, we investigated the effect of PEaT on hair growth and its mechanisms in C57BL/6 mice, which are good animals for hair cycle study because they show truncal pigmentation only during the anagen phase. 25,26
Materials and Methods
Experimental Animals
Male C57BL/6 mice (6-week-old in the telogen stage, 20-25 g of body weight) were purchased from the Central Lab. Animal Inc. (Seoul, Republic of Korea). The animals were housed under conventional temperature (about 23°C), humidity (about 60%), and given free access to food and water under 12 hours of light and dark cycle conditions. All experimental processes for the handling and care of animals were conducted in compliance with the current international laws and policies of the NIH Guide for the Care and Use of Laboratory Animals (The National Academies Press, 8th ed., 2011). All operations used in this study were performed to minimize the number of animals used and their suffering. Finally, the protocol of this research was approved by the Animal Care and Use Committee of Kangwon National University of Korea (approval no. KW-190128-1).
Preparation of PEaT
P. orientalis leaves were purchased by the Leefarm Co., Ltd. (Hongcheon, Republic of Korea). The dried powdered leaves were extracted with 10 volumes (v/w) of 70% ethanol and refluxed 3 times. The extract was filtered through Whatman filter paper (No. 2), concentrated using a vacuum evaporator, and freeze-dried. α-Terpineol was purchased from Sigma-Aldrich Co. (St Louis, MO, USA). The concentrated leaves extract was combined with α-terpineol; the final concentrations of the extract and α-terpineol were 3% 15 and 1%, 21 respectively.
Experimental Groups and PEaT Application
Mice were randomly divided into 2 groups (n = 7 per group): (1) control group, to which 250 µL of distilled water (DW) was applied to the dorsal skin, and (2) the PEaT group, to which 250 µL of PEaT was applied to the dorsal skin.
Hairs on the dorsal skin were shaved from a 2 × 4 cm2 area by using animal clippers 1 day before application of either DW or PEaT. Experiments started when the animals were 7 weeks old and in the telogen phase 27 ; all treatments were performed twice daily for 2 minutes for 17 days by using a syringe. Hair growth was recorded by taking pictures at 1, 4, 7, 10, 14, and 17 days after all the treatments.
Tissue Processing for Histology
To obtain the dorsal skin, the mice (n = 7 in each group) were anesthetized with pentobarbital sodium (60 mg/kg, intraperitoneal injection; JW Pharm. Co., Ltd, Republic of Korea) 17 days after the treatments. The anesthetized mice were transcardially perfused with 0.1 M phosphate-buffered saline (PBS, pH 7.4), followed by 4% paraformaldehyde in 0.1 M phosphate buffer (PB, pH 7.4). The dorsal skin tissues of the mice were removed and post-fixed in the same fixative for 1 day and embedded in paraffin. Finally, the embedded tissues were sectioned into 8 µm samples (Leica, Wetzlar, Germany).
Immunohistochemistry
Immunohistochemistry was carried out to investigate (1) growth factors by using rabbit anti-IGF-1 (diluted 1:200, Santa Cruz, CA, USA) and mouse anti-VEGF (diluted 1:300, Abcam, Cambridge, MA, USA), (2) proliferation by using rabbit anti-Ki-67 (diluted 1:300, Abcam, Cambridge, MA, USA), and (3) the induction of the anagen phase by using goat anti-wnt3 (diluted 1:500, Santa Cruz, CA, USA) and rabbit anti-β-catenin (diluted 1:2000, Abcam, Cambridge, MA, USA). The dorsal skin tissue sections were immunohistochemically stained according to our published method. 28 In short, the sections were treated with 0.3% hydrogen peroxide (in 0.1 M PBS, pH 7.4) and immersed in 10% normal goat, horse, or rabbit serum (Vector Laboratories, Inc., Burlingame, CA, USA) in 0.05 M PBS for 30 minutes at room temperature. Next, the sectioned dorsal skin tissues were reacted in a solution of each primary antibody at 4 ℃ for 24 hours. Thereafter, these sections were subjected to biotinylated horse anti-mouse IgG (diluted 1:250, Vector, CA, USA), goat anti-rabbit IgG (diluted 1:250, Vector, CA, USA), or rabbit anti-goat IgG (diluted 1:250, Vector, CA, USA) as secondary antibody. These sections were then reacted in a solution of avidin–biotin complex (diluted 1:300, Vector, CA, USA), and, finally, visualized by treatment with a solution of 3,3′-diaminobenzidine tetrahydrochloride (Sigma, MO, USA) (in 0.1 M PBS, pH 7.4).
A negative control test was used to establish the specificity of each immunostain with pre-immune serum instead of each primary antibody. No immunoreactivity was observed in any of the sections (data not shown).
Data Analyses
Hair growth activity was evaluated by hair growth-promoting scores based on different hair growth stages according to a previously published study. 29 The scoring criteria are as follows: 0 points: the skin of the shaved area is pink; 1 point: the skin of the shaved area is gray; 2 points: the skin of the shaved area is black; 3 points: the hair in the shaved area is starting to grow; 4 points: short-hair growth is complete.
The numbers of Ki-67-positive cells were quantitatively analyzed as follows. Five sections were selected on each mouse. As previously described, 28 digital images of Ki-67-stained dorsal skin tissues were captured using a light microscope (Olympus, Japan) equipped with a digital camera (DP7) connected to a PC monitor. Ki-67-positive cells were counted in a 300 × 300 µm square on the dorsal skin. The count was evaluated by averaging total numbers by using an image analyzing system (software: Optimas 6.5) (CyberMetrics, Scottsdale, AZ, USA).
Immunoreactivity of insulin-like growth factor-1 (IGF-1), vascular endothelial growth factor (VEGF), wnt3, and β-catenin, respectively, was quantitatively analyzed according to our published protocol. 28 Briefly, we captured a digital image of each immunoreactive structure in the dorsal skin by using a light microscope (BX53, Olympus, Tokyo, Japan). Each structure was evaluated as relative immunoreactivity (RI) as follows. First, on the basis of the optical density (OD) of the image, OD was obtained after the transformation of the mean gray level of each immunoreactive image using the formula: OD = log (256/mean gray level). Next, the background was taken from the areas adjacent to the measured areas. Finally, the RI of IGF-1, VEGF, wnt3, and β-catenin in the dorsal skin was calibrated as percentage after background was subtracted and compared with the control group designated as 100% by using Adobe Photoshop (version 8.0) (San Jose, CA, USA) and NIH Image software (version 1.59) (NIH, Bethesda, MD, USA).
Statistical Analysis
The data shown in this study represent the means ± standard error of the mean. Differences of the means among the groups were statistically analyzed by two-way analysis of variance with a post hoc Bonferroni’s multiple comparison test to determine differences among groups. Statistical significance was considered at P <.05.
Results
Effects of PEaT on Hair Growth
To investigate the effects of PEaT on hair growth promotion, we compared skin color, and hair growth speed and amount by observing the shaved dorsal skin at 1, 4, 7, 10, 14, and 17 days after the topical application of either DW or PEaT. The hair growth-promoting scores in each group between 7 and 14 days after shaving are presented in Table 1.
Hair Growth-Promoting Scores in the Control and PEaT Groups.

Abbreviation: PEaT, mixture of Platycladus orientalis leaves extract and α-terpineol.
The hair growth-promoting scoring criteria are as follows: 0 point: the skin of the shaved area is pink; 1 point: the skin of the shaved area is gray; 2 points: the skin of the shaved area is black; 3 points: the hair in the shaved area starts to grow; 4 points: the short-hair growth is complete. Data are expressed as the mean ± standard error of the mean (n = 7 per group; *P < .05 vs control group).
In the control group, the color of the dorsal skin was mostly pink until 10 days after shaving (Table 1, Figure 1). Thereafter, dark skin was shown at 14 days, and very short dorsal skin hairs were found at 17 days (Figure 1).

Progress of hair growth in the dorsal skin in the control and PEaT groups (n = 7 per group) for 17 days. Color of the dorsal skin is pink until 10 days after shaving in the control group. However, color of the dorsal skin begins to change from light to dark at 7 days in the PEaT group. Note that hair growth area is wider and hair length is longer at 17 days in the PEaT group compared with that in the control group. PEaT, mixture of Platycladus orientalis leaves extract and α-terpineol.
In the PEaT group, light dark skin was shown at 7 days after shaving. This phenomenon was significantly earlier than that of the control groups (Table 1, Figure 1). At 10 days after shaving, much darker blue skin was observed compared with that in the control group, and the hair in the shaved area had started to grow (Table 1, Figure 1). At 14 days after shaving, hair growth was shown in about 70% of the area, and the hair growth-promoting scores were significantly higher than those that in the control group (Table 1, Figure 1). Compact and longer hairs were shown on the entire dorsal skin at 17 days after shaving (Figure 1).
Effects of PEaT on Growth Factors Immunoreactivity
In all groups, IGF-1 immunoreactivity was easily detected in the outer epithelial root sheath of the hair follicle in the dorsal skin at 17 days after shaving (Figure 2(A) and (B)). In the control group, IGF-1 immunoreactivity was weak (Figure 2). However, the IGF-1 immunoreactivity was significantly increased by about 329% in the PEaT group compared with that of the control group (Figure 2(B) and (C)).

Immunohistochemistry of IGF-1 (A, B) and VEGF (D, E) in the dorsal skin of the control (A, D), and PEaT (B, E) groups at 17 days after shaving. IGF-1 and VEGF immunoreactivity is detected in the outer root sheath in all the groups. IGF-1 and VEGF immunoreactivity is significantly increased in the PEaT group compared with that in the control group. Scale bar = 100 µm. (C, F) RI of IGF-1 (C) and VEGF (F) was calibrated as percentage, with the control group designated as 100%. The bars indicate the means ± standard error of the mean (n = 7 per group; *P < .05 vs control group). IGF-1, insulin-like growth factor-1; PEaT, mixture of Platycladus orientalis leaves extract and α-terpineol; RI, relative immunoreactivity; VEGF, vascular endothelial growth factor.
VEGF immunoreactivity was found in the outer epithelial root sheath of the hair follicle in the dorsal skin of all the groups at 17 days after shaving (Figure 2(D) and (E)). In the PEaT group, VEGF immunoreactivity was significantly increased by about 200% compared with that of the control group (Figure 2(E) and (F)).
Effects of PEaT on Cell Proliferation
In the control group, a few Ki-67 immunoreactive cells were observed in the hair follicles at 17 days after shaving (Figure 3(A)). However, in the PEaT group, the number of these cells in the hair follicles was markedly increased by about 298% compared with the control group (Figure 3(B) and (C)).
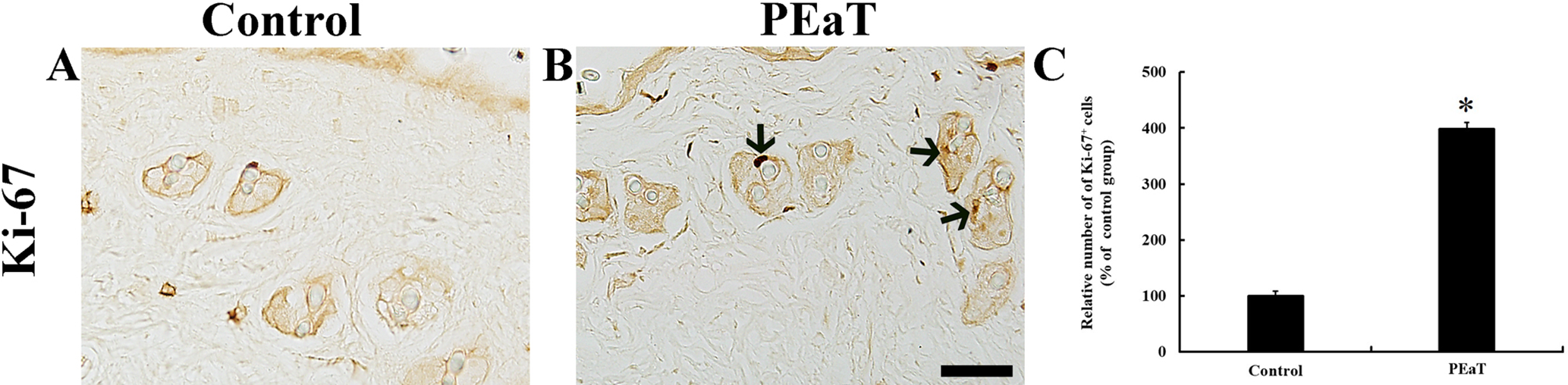
Immunohistochemistry of Ki-67 (A, B) in the dorsal skin of the control (A), and PEaT (B) groups at 17 days after shaving. Ki-67-positive cells are rarely found in hair follicles in the control group. However, the numbers of Ki-67-positive cells (arrows) are increased in the PEaT group. Scale bar = 100 µm. (C) Relative number of Ki-67-positive cells was calibrated as a percentage, with the control group designated as 100%. The bars indicate the means ± standard error of the mean (n = 7 per group; *P < .05 vs control group). PEaT, mixture of Platycladus orientalis leaves extract and α-terpineol.
Effects of PEaT on wnt3 and β-Catenin Immunoreactivity
Wnt3 immunoreactivity was detected in the outer epithelial root sheath of the hair follicles in the dorsal skin of all the groups at 17 days after shaving (Figure 4(A) and (B)). Wnt3 immunoreactivity in the PEaT group was significantly increased (about 22% of the control group) compared with that of the control group (Figure 4(B) and (C)).

Immunohistochemistry of wnt3 (A, B) and β-catenin (D, E) in the dorsal skin of the control (A, D), and PEaT (B, E) groups at 17 days after shaving. In the control group, weak wnt3 immunoreactivity is observed in the outer root sheath. In the EO group, wnt3 immunoreactivity is significantly increased compared with that in the control group. Strong β-catenin immunoreactivity is shown in the outer root sheath in the control group. β-Catenin immunoreactivity in the PEaT group is significantly increased compared with that in the control group. Scale bar = 100 µm. (C, F) RI of wnt3 (C) and β-catenin (F) was calibrated as a percentage, with the control group designated as 100%. The bars indicate the means ± standard error of the mean (n = 7 per group; *P < .05 vs control group). PEaT, mixture of Platycladus orientalis leaves extract and α-terpineol; RI, relative immunoreactivity.
β-Catenin immunoreactivity was well observed in the outer epithelial root sheath of the hair follicles in the dorsal skin in all the groups at 17 days after shaving (Figure 4(D) and (E)). In the PEaT group, this was increased by about 109% compared with that of the control group (Figure 4(E) and (F)).
Discussion
Hair is important not only in maintaining body temperature and protecting the skin, but also in determining the first impression, so patients with hair loss show psychosocial symptoms, such as loss of self-respect. 30 In this regard, healthy hair maintenance is very important in life. Some herbs have been used to either prevent or treat hair loss. 11,31 In the present study, we developed a natural hair tonic, which was a mixture of P. orientalis leaf extract and α-terpineol, and investigated the effects of PEaT on hair growth in mouse dorsal skin. C57/BL6 mice showed that the growth speed of hair was fastest in the PEaT group compared with that of the control group. In addition, the density of hair was most compact in the PEaT group compared with that of the control group.
It has been reported that the leaf extract of P. orientalis shows hair growth-promoting activity. 15,16 In addition, it has been demonstrated that the extract from a herbal complex containing P. orientalis, Cnidium officinale Makino, and Angelica gigas Nakai shows a hair growth-stimulating effect. 30,32,33 In our current study, we mixed P. orientalis leaf extract with α-terpineol. It has been reported that α-terpineol has antibacterial activity, 34,35 which can be helpful in removing noxious microbial cells in the scalp of alopecia patients. 36,37 In addition, 3% lavender oil, including α-terpineol (0.6%), displayed a remarkable hair growth-promoting effect in C57BL/6 mice by reducing the number of perifollicular mast cells, which may induce hair loss. 38 Furthermore, rosemary oil containing α-terpineol (6.8%-8.2%) 39 shows a hair growth property by stimulating the scalp. 40 It has been reported that alcoholic terpenes, such as α‐terpineol, linalool, and carvacrol in essential oils, have relatively low toxicity and skin irritancy compared with synthetic penetration enhancers. 23,41 Taken together, P. orientalis and/or α-terpineol could be used as functional hair growth promoters, and we, in this study, found that PEaT was a potent hair growth promoter.
A hair follicle, as part of the skin, grows a hair by packing old cells together. In C57BL/6 mice, numbers and size of hair follicles increase when the hair cycle transits from the telogen phase to the anagen phase, 26 and follicular melanocytes produce dark pigment during the anagen phase, thus leading to truncal pigmentation. 26,42,43 During the hair cycle, a bright pink skin color indicates the telogen phase, and gray to black skin indicates the beginning of the anagen phase. 27 In the present study, spontaneous transition from the telogen to the anagen phase began at 14 days after shaving in the control mice, but the transition in the PEaT group began 7 days after the shaving, which was earlier than that in the control group. In addition, the topical application of P. orientalis water extract apparently enhanced hair growth, promoted the progression of the hair cycle from the telogen to the anagen phase, and prolonged the mature anagen phase compared with those in the control group. 14
It is known that VEGF mRNA expression is upregulated by follicular keratinocytes and that follicle-derived VEGF increases perifollicular angiogenesis during the anagen phase, resulting in increases in hair regrowth rate, follicle number and size, and hair size. 44 In addition, dermal papilla cells play a key role in inducing transition from the telogen phase to the anagen phase and maintaining hair growth. 44 In the phases, growth factors and development-related molecules, such as IGF-1 and VEGF, modulate follicular growth and hair cycle; namely, dermal papilla cells regulate proliferation of the follicular epithelium. 45,46 Many studies have investigated various mechanisms of action of natural extracts that enhance hair growth and either treat or prevent hair loss. In our current study, PEaT induced significant increases of IGF-1 and VEGF expressions and numbers of Ki-67 positive cells, along with hair growth promotion compared with the control group. Therefore, based on the previous research results and our present study, it is likely that PEaT used in this study has a follicular growth property, which supports hair growth through increases in cell proliferation and expressions of growth factors.
In the present study, wnt3 and β-catenin expressions were significantly increased in the outer root sheath in the PEaT group. It has been reported that wnt/β-catenin signaling, considered as a dominant pathway regulating hair regeneration, activates hair follicle stem cells to promote development of new hair follicles, and that the stimulation of β-catenin activity induces follicular proliferation and differentiation, which leads to transition of the hair growth cycle from the telogen phase to the anagen phase and prolongs the anagen phase in the dermal papilla. 47 -49 Therefore, it is likely that the hair growth promotion by PEaT may be closely related to upregulation of wnt3 and β-catenin expressions in the outer root sheath.
In summary, PEaT (a mixture of 3% P. orientalis leaf extract and 1% of α-terpineol) significantly enhanced hair growth rate in mouse dorsal skin, showing that IGF-1 and VEGF immunoreactivities, numbers of Ki-67-positive cells, and wnt3 and β-catenin immunoreactivities were significantly increased in the dorsal skin compared with the control group. These results indicate that PEaT can be a hair growth promoter. In the future, we have to identify other components of PEaT and their mechanisms in either preventing hair loss or promoting hair growth.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by 2019 Industry Research Grant from Leefarm Co., Ltd., (C1014940-01-01) through Kangwon National University.
