Abstract
Introduction
Hair loss is a prevalent concern prompting both male and female patients to seek treatment. 1 Substantial advancements in stem cell research and tissue engineering have opened avenues for employing stem cell techniques in both hair regeneration and the prevention of hair loss. 2 In particular, extensive research has highlighted the potential of hair stem cells, especially dermal papilla (DP) cells, in promoting hair regeneration and induction. 3 DP cells are known for their ability to communicate with neighboring cells by secreting various growth factors and cytokines, thus influencing hair growth and the maintenance of the hair cycle. 4
Therefore, the targeting for activation of Wnt/β-catenin signaling is crucial for hair follicle regeneration. Numerous plant-derived compounds have been shown to enhance hair growth by stimulating Wnt/β-catenin signaling in both vivo and vitro studies. 4 Activated Akt can phosphorylate GSK3β at Ser9, leading to its inactivation and subsequently regulating the cell cycle. 5 The stabilize β-catenin into the nucleus can activate the stem cells markers that leading to cell survival and proliferation. 6 The stem cell properties, or stemness, of DP cells are essential for their role in inducing hair growth and secreting growth factors. 7 Therefore, a treatment approach aimed at enhancing the stemness of these cells could promote hair regrowth and reduce hair loss. 8 This is where the importance of research on plant-derived compounds and extracts comes into play.
Aloe vera (AV) extracts are widely used in cosmetics, pharmaceuticals, and food industries for their medicinal properties, such as treating burns, healing wounds, and reducing skin inflammation. The plant is rich in antioxidants, enzymes, minerals, sugars, anthraquinones, fatty acids, and hormones.9,10 Research indicates that Aloe vera extracts and gel promote wound healing by reducing inflammation, increasing epidermal thickness, and enhancing collagen deposition when applied topically. 11 Centella asiatica (L.) (CA) is a highly valued medicinal herb recognized for its antioxidant, anti-inflammatory, neuroprotective, cardioprotective, and wound healing properties. 12 Its primary active constituents are pentacyclic triterpenoid saponins, including asiaticoside, madecassoside, asiatic acid, and madecassic acid. 13 CA treatment boosted the expression of hair growth-related genes and reduced phosphorylated STAT proteins in 3D sphered DP cells, thereby enhancing the hair inductive potential of these cells. 14 Green tea (Camellia sinensis) (GT) leaves exhibit various pharmacological properties, including antioxidant, anticancer, cardiovascular disease prevention, cholesterol-lowering effects, hypoglycemic, antibacterial, antiviral, and neuroprotective effects. Key components include polyphenolic compounds like catechins, especially (-)-epigallocatechin-3-gallate, as well as caffeine and amino acids.15,16 Pueraria mirifica (PM), part of the Leguminosae family, has potent phytoestrogenic properties, which means it contains non-steroidal plant compounds that imitate estrogenic activity. Its extracts include isoflavonoids, coumestrans, and chromenes.17,18
The effects for combination of extracts on the stemness properties of DP cells via stabilize β-catenin are not well understood. The insights gained from studying stem cell-stimulating properties of these extracts via stabilized β-catenin could lead to new therapeutic options for hair rejuvenation.
Materials and Methods
Cells and Reagents
Human DP cells were obtained from Applied Biological Materials Inc. (Richmond, BC, Canada) and cultured in Dulbecco's Modified Eagle's Medium (DMEM) with 10% fetal bovine serum and 2 mM L-glutamine at 37 °C with 5% CO2. Alexa Fluor™ 594 goat anti-rabbit IgG secondary antibody was from Invitrogen (Carlsbad, CA, USA), and primers for OCT4, NANOG, SOX2, and GAPDH were sourced from Eurofins Genomics (Plantside Drive Louisville, KY, USA).
Cell Viability Assay
Cells were seeded in a 96-well plate and cultured for 24 h at 37 °C. Following this, the culture medium was aspirated, and varying concentrations (0-4000 μg/ml) of different extracts were added for another 24 h. After removing the extract-containing medium, MTT reagent at a final concentration of 0.4 mg/(ml) was added and allowed to incubate for 3 h at 37 °C. The formed formazan crystals were dissolved in 100 μl of DMSO, and the absorbance was measured at 570 nm using a microplate reader.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR)
After treatment, RNA was isolated from the cells using GENEzol reagent and converted to cDNA with SuperScript III reverse transcriptase. For RT-qPCR, 100 ng of cDNA was used with Luna Universal qPCR Master Mix in a 20 µl reaction volume, run on the CFX 96 real-time PCR system (Bio-Rad, Hercules, CA). The protocol included an initial denaturation at 95 °C for 1 min, followed by 45 cycles of denaturation at 95 °C for 10 s, and annealing/extension at 60 °C for 30 s. Melting curve analysis verified primer specificity. Gene expression was normalized to GAPDH, and relative mRNA levels were calculated from comparative Cq values.
Immunofluorescence
The cells were fixed with 4% paraformaldehyde for 15 min. and permeabilized with 0.5% Triton-X for 5 min., Then, non-specific proteins were blocked with 10% FBS in 0.1% Triton-X PBS for 1 h at room temperature. The cells were then incubated with primary antibodies (β-catenin, pAkt, OCT4, NANOG, and SOX2) and left overnight at 4 °C. A secondary antibody solution containing Alexa Fluor 594-conjugated goat anti-rabbit IgG (H + L) was applied. Nuclei were stained with Hoechst 33342 for 1 h at RT. Fluorescence images were captured using a fluorescence microscope (Olympus IX 51 with DP70; Olympus America Inc., Center Valley, PA, USA), and the fluorescence intensity was quantified using Image J software.
Proliferation Assay
DP cells were seeded 2 × 103 cells per well in a 96-well plate. The cells were treated with concentration of 1000 and 2000 μg/ml of AV, CA, GT, PM, and ACGP with different time points such as 0 h, 24 h, 48 h, and 72 h. After that, the cell viability was measured by using MTT at 0 h, 24 h, 48 h, and 72 h.
Western Blot Analysis
DP cells were seeded cell density for 3 × 105 cells per well in a 6-well plate and treated with varying concentrations (0, 1000 and 2000 μg/ml) of AV, CA, GT, PM, and ACGP for 24 h. After treatment, the cells were collected, lysed and measured protein concentration. The proteins were separated via SDS-PAGE, then transferred onto nitrocellulose membranes. These membranes were blocked with nonfat dry milk and incubated overnight with primary antibodies targeting specific proteins (Akt, pAkt, and β-catenin), with β-actin serving as a loading control. After that, the membranes were incubated with HRP-conjugated secondary antibodies and visualized using a chemiluminescent substrate. The protein band intensities were quantified using ImageJ software.
Statistical Analysis
The results are expressed as mean ± standard deviation (SD) from a minimum of three separate biological experiments. Multiple comparisons were conducted using one-way ANOVA analysis followed by a post hoc test in GraphPad Prism software version 9.0 (GraphPad Software, La Jolla, CA, USA). Statistical significance was defined as a p-value < 0.05.
Results
Cell Viability of Dermal papilla Cells in Response to Natural Product Extracts
We initially assessed the effect of the extracts on DP cell viability using the MTT assay, as detailed in the Materials and Methods section. Various natural product extracts, including AV, CA, GT, PM, and a combination of extracts (ACGP), were applied to cultured human DP cells at concentrations ranging from 0 to 4000 μg/ml for 24 h. Figure 1 illustrates that none of the extracts affected cell viability, even at concentrations up to 4000 μg/ml. These findings suggest that the extracts have low cytotoxicity on DP stem cells.

Cytotoxicity of natural products on human DP cells. (A-E) The effect of extracts from AV, CA, GT, PM, and ACGP on DP cell viability at 24 h, was evaluated using MTT assays. Data are presented as mean ± SD (n = 3). Not significant compared to the untreated control group.
The Natural Product Extracts Induced Stabilize β-Catenin Expression via Induction of Akt Level in DP Cells
The cell proliferation rate was assessed using an MTT assay over a period of three consecutive days. The results revealed that the proliferation rate in DP cells increased significantly, by 1.5-fold, when treated with a combination of ACGP compared to single treatment (Figure 2A).

The combination of natural products ACGP targeted for stabilizing β-catenin signaling pathway. (A) The proliferative effect of DP cells treated with a combination of ACGP was assessed using an MTT assay at 0 h, 24 h, 48 h, and 72 h, with results analyzed relative to the control group. (B) DP cells were exposed to AV, CA, GT, PM, and a combination of extracts (ACGP) (0, 1000, 2000 µg/ml) for 24 h. The levels of the Akt/β-catenin signaling pathway were assessed using Western blot analysis. The membrane was subsequently probed with β-actin to ensure equal protein loading. (C) The protein expression levels of pAkt, β-catenin were determined using immunofluorescence. The fluorescence intensity was determined using Image J software. Data are presented as mean ± SD (n = 3). Significant compared to the control group, *p < 0.05, **p < 0.01, ***p < 0.001 versus untreated control cells.
Numerous studies have shown that the activation of the Wnt/β-catenin pathway is a key regulator in hair rejuvenation development. 6 The Akt signaling pathway is activated by the phosphorylation of Akt at the specific site Ser473. This Akt phosphorylation is crucial for regulating kinase activity and its subsequent signaling pathways. Akt is involved in various cellular mechanisms, including cell proliferation and survival. 4 The expression of pAkt was 1.5-fold in DP cells treated with a combination of the natural product extract (ACGP). Furthermore, β-catenin protein levels were also significantly increased by 1.5-fold in ACGP-treated DP cells compared to control DP cells (Figure 2B).
Immunofluorescence analysis confirmed the expression levels of pAkt and β-catenin proteins in DP cells treated with ACGP. Our findings indicated that DP cells treated with a combination of ACGP exhibited a 3-fold higher fluorescence intensity for pAkt compared to control DP cells. Additionally, the fluorescence intensity of β-catenin protein was 1.5-fold higher in ACGP-treated DP cells (Figure 2C).
The Natural Product Extracts Induced High Expression of Stemness Transcription Factors mRNA Expression Level in DP Cells by RT-qPCR Method
To investigate the effect of extracts on stemness, DP cells were treated with extracts at 1000, 2000 µg/(ml) for 24 h. Subsequently, mRNA levels of stem cell transcription factors were assessed by RT-qPCR analysis.
The results showed that treatment of the cells with AV, CA, GT, and PM at the dose of 2000 µg/ml could induce approximately 10-fold increase in OCT4 mRNA level. Surprisingly, the combination of extracts (ACGP) at the same dose could induce dramatically high mRNA of OCT4 (approximately 40 folds), comparing to those of control non-treated cells, implying the synergistic effect of such extracts on stem cell induction (Figure 3A).

The effect of natural products on the expression of stemness transcription factors in DP cells. (A-C) DP cells were treated with extracts (0, 1000, 2000 µg/ml) for 24 h, and the mRNA levels of stemness transcription factors, OCT4, NANOG, and SOX2 were determined. The mRNA level was normalized using the housekeeping mRNA GAPDH. The relative mRNA expression was calculated by using comparative Ct cycles. Data are presented as mean ± SD (n = 3). Significant compared to the control group, *p < 0.05, **p < 0.01, ***p < 0.001 versus untreated control cells.
The expression of NANOG mRNA increased 40-fold in DP cells treated with AV extract and 20-fold in DP cells treated with CA, GT, and PM extracts at a concentration of 2000 µg/ml. Moreover, the combination of extracts (ACGP) resulted in a significantly higher expression level (110-fold) compared to treatment with individual extracts on DP cells. (Figure 3B).
The SOX2 mRNA was substantially raised around 10-fold, in DP cells when treated with extracts of AV, CA, and PM at a dosage of 2000 µg/ml. For the mixture (ACGP), SOX2 mRNA was approximately 30-fold significantly higher compared to treatment with non-treated control (Figure 3C).
Overall, these findings indicate that the named extracts have inductive effects on stem cell factors. Importantly, the combination of these four extracts significantly enhances the stemness of DP cells.
The natural product extracts induced high expression of stemness transcription factors
Immunofluorescence analysis was employed to evaluate the protein levels of stemness transcription factors in DP cells treated with natural product extracts.
The fluorescence intensity of OCT4 was significantly increased 2-fold in DP cells treated with PM extracts at a concentration of 2000 µg/ml. Moreover, the fluorescence intensity of the combination of extracts (ACGP) was significantly higher compared to treatment with individual extracts on DP cells (Figure 4A).

The effect of natural products on the expression of stemness transcription factors in DP cells. (A-C) DP cells were treated with natural product extracts (0, 1000, 2000 µg/ml) for 24 h, and the protein expression levels of stemness transcription factors, OCT4, NANOG, and SOX2 were determined using immunofluorescence. The stemness transcription factors, OCT4, NANOG, and SOX2 were captured using immunofluorescence and the fluorescence intensity was determined using Image J software. Data are presented as mean ± SD (n = 3). Significant compared to the control group, *p < 0.05, **p < 0.01, ***p < 0.001 versus untreated control cells.
Furthermore, the fluorescence signal of NANOG exhibited a notable increase of 3-fold in DP cells following treatment with these natural product extracts (Figure 4B).
Remarkably, the protein expression level indicated by the SOX2 fluorescence intensity increased by 2-fold in DP cells treated with a combination of extracts (ACGP) (Figure 4C).
Overall, the expression levels of the transcription factors OCT4, NANOG, and SOX2 increased in DP cells treated with natural product extracts, with the combination of AV, CA, GT, and PM (ACGP) showing the most significant enhancement.
Discussion
Skin stem cells are fundamental for the renewal and repair of skin and its associated structures. 19 Various types of stem cells are present in the skin, such as hair follicle stem cells, which are crucial for regulating hair growth, and melanocyte stem cells, which are involved in hair pigmentation. 20 The DP is vital for initiating and maintaining hair follicles, essential for hair growth and regeneration. 21
The β-catenin signaling pathway is essential for activating hair follicle stem cells and enhancing hair regeneration by stabilizing β-catenin and activating genes that promote cell proliferation and survival. 22 Cell proliferation, assessed using an MTT assay over three days, showed a 1.5-fold increase with ACGP combination treatment compared to single treatment (Figure 2A). In DP cells treated with the ACGP combination, the expression of both pAkt and β-catenin proteins was 1.5 times higher than in control cells (Figure 2B).
Moreover, our research indicates that a combination of ACGP extracts effectively enhances stem cell characteristics in DP cells (Figure 3 and 4). Stemness is vital for maintaining the self-renewal and properties of hair follicle stem cells, which are key in wound healing and hair follicle regeneration. These stem cells can reprogram and change their fate. 23 Dermal papilla-induced pluripotent stem cells from hair follicles can be reprogrammed using transcription factors like POU class 5 homeobox 1 (OCT4), Nanog homeobox (NANOG), and SRY-box transcription factor 2 (SOX2). 24
The mRNA expression levels of the stemness markers OCT4, NANOG, and SOX2 increased by 40-fold, 110-fold, and 30-fold, respectively, in DP cells treated with a combination of extracts_ACGP compared to individual treatments in those cells (Figure 3). Immunofluorescence analysis revealed that the protein expression levels of stem cell markers were significantly higher in DP cells treated with a combination of extracts_ACGP (Figure 4).
These markers are known for their vital role in regulating the self-renewal abilities necessary for the formation of hair follicles. OCT4 serves as the primary gene at the beginning of the signaling pathway that governs the self-renewal capacity of stem cells. Additionally, OCT4 serves as an enhancer of the cell cycle, aiding in the progression from the G1 phase to the S phase. This unique cell cycle is essential for maintaining the self-renewal capability of stem cells. 25
The expression of NANOG is recognized as a critical element in preserving the self-renewal capacity of cells, a process directly governed by OCT4 and SOX2 regulation. 26 The accumulation of NANOG and OCT4 within DP cells promotes the prolonged maintenance of self-renewal capability and stemness, thus preventing these cells from differentiation. 27
The current research findings indicate that in a cell-based model, extracts from AV, CA, GT, PM, and their combination (ACGP) promote stemness characteristics by stabilizing β-catenin in DP cells. However, further studies using animal models and clinical trials are recommended to explore these effects in future evaluations for hair regenerative strategies.
Our study highlights that the combination of natural product extract mixture (ACGP) induces stemness properties via stabilization of β-catenin and supports hair follicle regeneration in DP cells (Figure 5). The targeting Akt/pAkt/β-catenin signaling pathway through enhancing stemness properties on this combination of extracts could lead to innovative advancements in hair rejuvenation research.
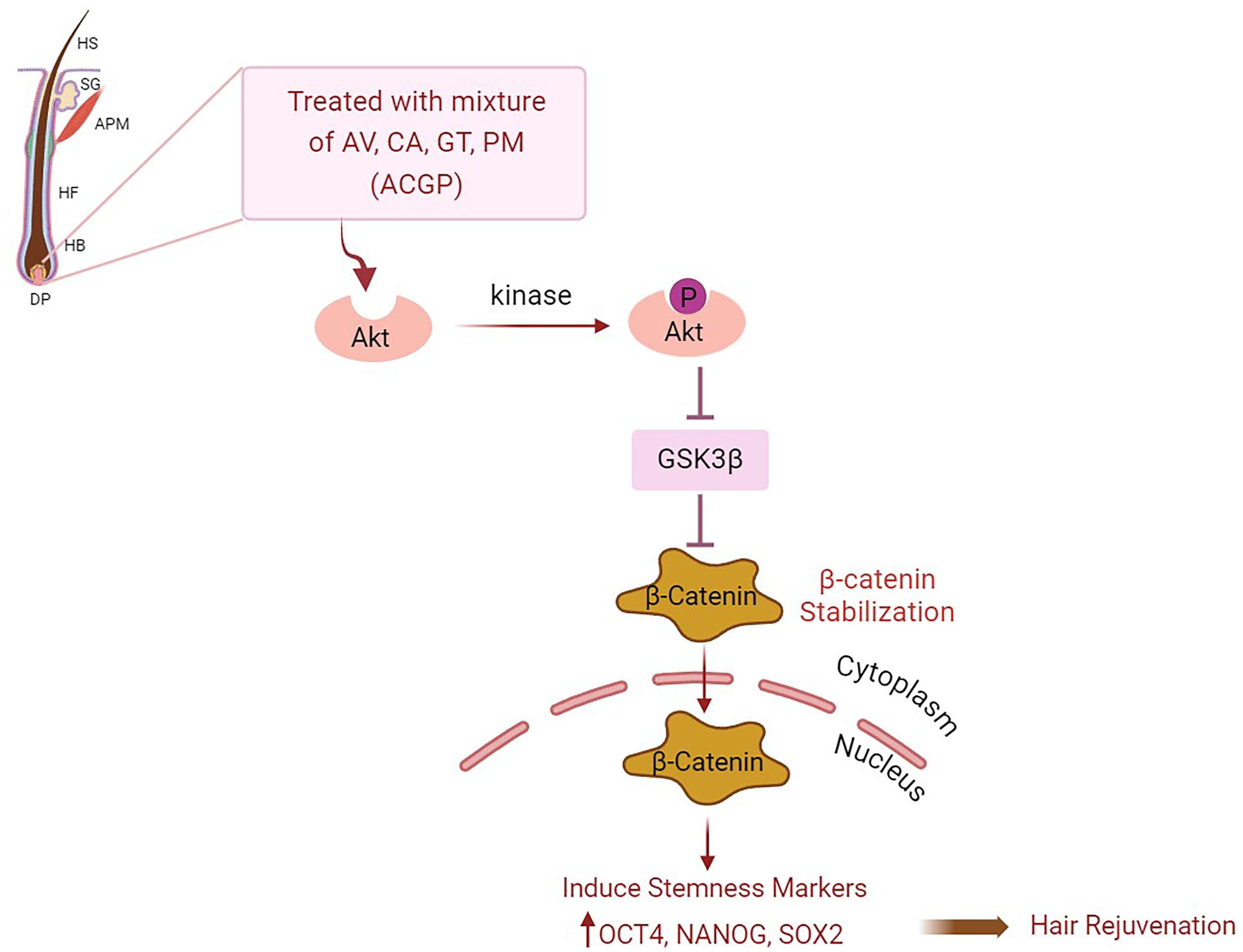
The combination of natural product extracts (ACGP) demonstrated the capacity to activated Akt can phosphorylate GSK3β at Ser9, leading to its inactivation and stabilization of β-catenin that stimulate stemness characteristics in hair follicles within DP cells. Treatment of DP cells with this combination of natural product extracts led to an elevation in stemness markers (OCT4, NANOG, SOX2), thus facilitating hair growth promotion. HS: Hair shaft: SG: sebaceous gland; APM: arrector pili muscles; HF: hair follicle; HB: hair bulb; DP: dermal papilla. Created with BioRender.com.
This study reveals that, in a cell-based model, extracts from a combination of ACGP enhance stemness characteristics for hair rejuvenation by stabilizing β-Catenin and stimulating the Akt signaling pathway in human dermal papilla cells. Cell-based experiments are the most commonly employed model for conducting preliminary in vitro studies.28,29 However, we suggest incorporating animal models and clinical trials in future research to further explore and confirm these activities.
Conclusion
In conclusion, our observation indicate that combination of natural product extract (ACGP) is more effective at inducing stemness characteristics in DP cells than individual extract. The synergistic effect of the ACGP mixture enhances stemness by stabilizing Akt/pAkt/β-catenin signaling pathway, along with its potentiating factors, which could support the development of novel hair regenerative strategies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251320146 - Supplemental material for Exploration of Aloe vera, Centella asiatica, Green Tea, and Pueraria mirifica Extracts in Modulating Stem Cell-Like Properties for Hair Rejuvenation Through β-Catenin Stabilization and Activation of the Akt Signaling Cascade in Human Dermal Papilla Cells
Supplemental material, sj-docx-1-npx-10.1177_1934578X251320146 for Exploration of Aloe vera, Centella asiatica, Green Tea, and Pueraria mirifica Extracts in Modulating Stem Cell-Like Properties for Hair Rejuvenation Through β-Catenin Stabilization and Activation of the Akt Signaling Cascade in Human Dermal Papilla Cells by Zin Zin Ei, Supaporn Barrameesangpet and Pithi Chanvorachote in Natural Product Communications
Footnotes
Acknowledgements
The Author (Z.Z.E.) is grateful to the Second Century Fund (C2F) for the postdoctoral fellowship, Chulalongkorn University.
Authors’ Contributions
Conceptualization, P.C.; methodology, P.C.; validation, P.C.; formal analysis, Z.Z.E., and P.C.; investigation, Z.Z.E., S.B., and P.C.; resources, P.C.; writing-original draft preparation, Z.Z.E.; and S.B., writing-review and editing, P.C.; supervision, P.C. All Authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Council of Thailand (NRCT), (grant number N42A670567).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
