Abstract
There is no effective method for treating pulmonary fibrosis (PF) until now. This study investigated the anti-fibrotic effect of schisantherin A (SCA) extracted from Schisandra chinensis and its potential molecular mechanism in PF. A bleomycin-induced PF mouse model in vivo and transforming growth factor (TGF)-β1-induced A549 epithelial-mesenchymal transition (EMT) cell model in vitro were used for assessing the anti-fibrotic effect of SCA. Histopathological examination was conducted after hematoxylin and eosin and Masson staining. The level of TGF-β1 was tested by ELISA. The expression levels of α-smooth muscle actin, E-cadherin, and inflammatory cytokines (COX2, IL-1β, IL-6, and TNF-α) were determined by quantitative reverse transcription polymerase chain reaction and Western blot. The expression of extracellular signal-regulated kinase (ERK) was tested in lung tissues and cells by Western blot. The in vivo experiments revealed that SCA treatment markedly improved body weight and pulmonary index and reformed the destruction of the lung tissue structure. We observed that SCA inhibited the process of TGF-β1-induced EMT in the in vitro experiments. Inflammatory cytokines were reduced greatly in lung tissues and cells by SCA. Our study also indicated that SCA decreased phosphorylated ERK. It was concluded that SCA can attenuate PF by regulating the ERK signaling pathway, which suggests that SCA may be used as a potential therapeutic drug for PF.
Pulmonary fibrosis (PF) is a chronic, progressive, and lethal lung disease, characterized by the destruction of the pulmonary parenchyma, deposition of extracellular matrix, and obvious changes in the phenotype of both fibroblasts and alveolar epithelial cells. 1,2 Although the pathogenesis of PF is not fully understood, inflammation is known to play a crucial role in its initiation and progression. Therefore, the regulation of chronic inflammation and fibrogenesis is a key event in inducing fibrosis. 3 In the lower airways, some proinflammatory cytokines are released by accumulating inflammatory cells so as to regulate the proliferation and secretory activity of alveolar fibroblasts. Mitogen-activated protein kinases (MAPKs), including extracellular signal-regulated kinases (ERK), c-Jun N-terminal kinases, and p38, exert important effects on the immune responses in the lungs. Among MAPKs, ERK is the major mediator of cellular transcriptional activities, including inflammatory responses. 4 A previous study has indicated that pretreatment with silymarin could inhibit lung inflammation in mice, possibly relating to ERK pathways. 5
Although many anti-inflammatory drugs are currently being used for treating PF, side effects are widely observed with their chronic use. 6 For instance, dexamethasone (DXM) may show skin echymosis , high blood pressure, and other adverse effects, although it is effective in treating PF. 7 Therefore, herbal medicine treatment for PF is being sought to substitute conventional and chemical drugs that have deleterious side effects. 8 Previous studies demonstrated that schisantherin A (SCA), an active ingredient extracted from the fruit of Schisandra chinensis, can protect against liver fibrosis, 9 ischemia/reperfusion-induced neuronal injury, 10 6-OHDA-induced dopaminergic neuron damage in zebrafish, 11 and lipopolysaccharide-induced acute respiratory distress syndrome (ARDS) in mice. 12 It could also suppress inflammation, 13,14 alleviate liver injury, 15,16 and improve learning and memory of mice. 17 However, whether SCA affects lung fibrosis by intervention of the ERK pathway remains unknown.
In this study, we investigated the protective effects of SCA against bleomycin (BLM)-induced PF and transforming growth factor (TGF)-β1-induced epithelial-mesenchymal transition (EMT) in A549 cells and revealed its underlying mechanism.

Materials and Methods
Cell Culture and Treatment
A549, human alveolar epithelial cells, were purchased from the Cell Bank of the Institute of Biochemistry and Cell Biology and cultured in Dulbecco’s modified Eagle’s medium (Hyclone, Los Angeles, USA) supplemented with 10% fetal bovine serum (Sijiqing, Tianhang Biotechnology Co., Ltd, Hangzhou, China), incubated in a 5% CO2 atmosphere at 37 ℃. Cells were then treated with 5 ng/mL TGF-β1 (PeproTech, Suzhou, China ).
MTT Assay for Cell Proliferation
The cell viability effect of SCA on A549 cells was estimated by measuring the reduction of MTT to formazan. A549 cells were seeded into 5 × 103/wells of a 96-well plate and incubated for 24 hours at 37 ℃. The cells were then treated with varied concentrations of SCA (0.625, 10 μm) and 5 ng/mL TGF-β1, and incubated for 48 hours at 37 ℃. Twenty microliters MTT (5 mg/mL) was then added to each well for 4 hours. The supernatant was discarded, and 150 µL dimethyl sulfoxide was added to each well. The plate was shaken at room temperature for 10 minutes, and the absorbance measured using an ELISA reader (TECAN, Salzburg, Austria) at 490 nm. Detections were repeated at least 3 times.
Animals and Treatment
Adult male ICR mice (22 ± 2 g, 3-4 weeks) were obtained from Changchun Yixin Animal Co.(Changchun, China), Ltd. All mice used in the study were kept in an autocontrolled room (25 ℃ ± 3 ℃, 40%-65% humidity) in a 12-hour light/dark cycle. Mice were allowed free access to food and water. All experiments were approved by the Beihua University Committee on Ethics in the Care and Use of Laboratory Animals (YUCE No. 32/309). All the experimental mice were randomly divided into 6 groups (10 mice): control group (CON); BLM-treated group (MOD); BLM plus DXM-treated group (DXM used as a positive control drug); BLM plus SCA-treated low-dose group (SCA-L, 1 mg/kg); BLM plus SCA-treated medium-dose group (SCA-M, 2 mg/kg); and BLM plus SCA-treated high-dose group (SCA-H, 4 mg/kg). All mice were anesthetized with 8% chloral hydrate. Then, they were administered a single intratracheal instillation of BLM dissolved in saline (5 mg/kg), while an equal volume of saline was instilled into the mice from the CON. The mice of SCA-L, SCA-M, and SCA-H received an intraperitoneal injection of SCA of different doses, while the CON and MOD mice were injected with a saline solution, every day for a total of 28 days, and the DXM group with DXM (1 mg/kg) for 3 weeks. The mice were sacrificed at day 28 after the first BLM treatment. The serum was collected and the lungs excised for further study.
Histopathological Assessment of Lung Tissues
Hematoxylin and eosin (H&E) staining and Masson’s trichrome staining were performed on paraffin-embedded lung tissue sections (4 µm). Lung tissue morphology was determined through light microscopy at a magnification of 200×. The severity of alveolitis, inflammation score, and scoring criteria were graded according to a previous report. 18 Masson’s staining was analyzed by Image-ProPlus 6.0 software.
RNA Extraction and Quantitative Real-Time PCR
An RNA Isolation Mini Kit (Vazyme, Nanjing, China) was used to extract the total RNA following the manufacturer’s protocols. HiScript III 1st Strand cDNA Synthesis Kit (Vazyme) was used for evaluating the production of cDNA. Real-time polymerase chain reaction (PCR) was carried out by utilizing AceQ Universal SYBR qPCR Master Mix (Vazyme) using sense and anti-sense primers, including β-actin, α-smooth muscle actin (α-SMA), E-cadherin, IL-1β, IL-6, TNF-α, and COX2. In accordance with the published gene sequences in GenBank, PCR primers were designed with Primer 5.0 software (Table 1). The reliability of the results was evaluated with a standard curve with β-actin as an internal reference. The CT value (amplification power curve inflection point) was obtained: ΔCt = CT (target gene) – CT (internal reference), ΔΔCt = ΔCt (treatment group) – ΔCt (control group); the relative expression of target genes was calculated using 2−ΔΔCt.
qRT-PCR Primers and Products.

qRT-PCR, quantitative reverse transcription polymerase chain reaction; α-SMA, α-smooth muscle actin.
ELISA Assay
TGF-β1 levels in the collected serum of mice were detected using a TGF-β1 ELISA Kit (Proteintech, Wuhan, China) according to the manufacturer’s instructions. All the samples were analyzed in triplicate. The concentrations of TGF-β1 in the samples were determined from a standard curve.
Measurement of Protein Expressions
The contents of protein prepared from the cells were analyzed by utilizing a bicinchoninic acid assay kit (Beyotime, Shanghai, China). Total protein extracts from lung tissues and cells were separated by SDS-PAGE, then transferred to polyvinylidene fluoride membranes. The membranes were incubated with primary and secondary antibodies (ABclonal Technology, WuHan, China). After incubation, membranes were washed in Tris-buffered saline containing 0.5% Tween 20, followed by ECL for luminescence generation. The image and gray level were analyzed with Image J software. The protein expression level was assessed by the β-actin ratio.
Statistical Analysis
All the data were expressed with mean ± SD. Comparisons between groups were performed by one-way analysis of variance, followed by a 2-tailed unpaired t-test. All statistical analyses were performed on SPSS 22.0 software. A P value <0.05 was considered statistically significant.
Results
Changes in Body Weight and Pulmonary Index After SCA Administration
A considerable decline in the body weight was detected in the BLM-induced group in comparison with the control group (Table 2, P < 0.01). The loss of body weight associated with BLM administration was significantly relieved by SCA-M (P < 0.05) and SCA-H (P < 0.01) compared with only BLM administration to the MOD group. Furthermore, as shown in Table 2, the pulmonary index of the BLM-induced group clearly increased as a result of the significantly increased weight of wet lung and decreased body weight compared with the CON group (P < 0.001). However, after SCA-M (P < 0.05) and SCA-H (P < 0.01) administration for 28 days, the pulmonary index remarkably declined as a result of the decreased weight of wet lung and increased body weight.
The Effect of SCA on Body Weight and Pulmonary Index of Mice Among Different Experimental Groups.

BLM, bleomycin; CON, control group; DXM, BLM plus dexamethasone-treated group; MOD, BLM-treated group; SCA, schisantherin A; SCA-H, BLM plus SCA-treated high-dose group; SCA-L, BLM plus SCA-treated low-dose group; SCA-M, BLM plus SCA-treated medium-dose group.
**P < 0.01, *** P < 0.001 vs CON group; # P < 0.05, ## P < 0.01, ### P < 0.001 vs MOD.
SCA Attenuated Histological Changes in Pulmonary Fibrosis Induced by BLM
Histopathological evaluation of the pulmonary sections revealed that significant tissue damage was caused by BLM (Figure 1(A)). H&E staining indicated that normal lung tissue structure, a complete alveolar cavity, and a few inflammatory cells were preserved in the CON group. In the MOD group, there was significant infiltration of inflammatory cells in the alveolar space and pulmonary interstitium, along with thickened alveolar septum, and hyperplastic capillary and extensive lesions. The lungs of the animals treated with SCA-M and SCA-H revealed significantly reduced inflammatory lesions compared with the model group. The pathology index showed the same result (Figure 1(B)).
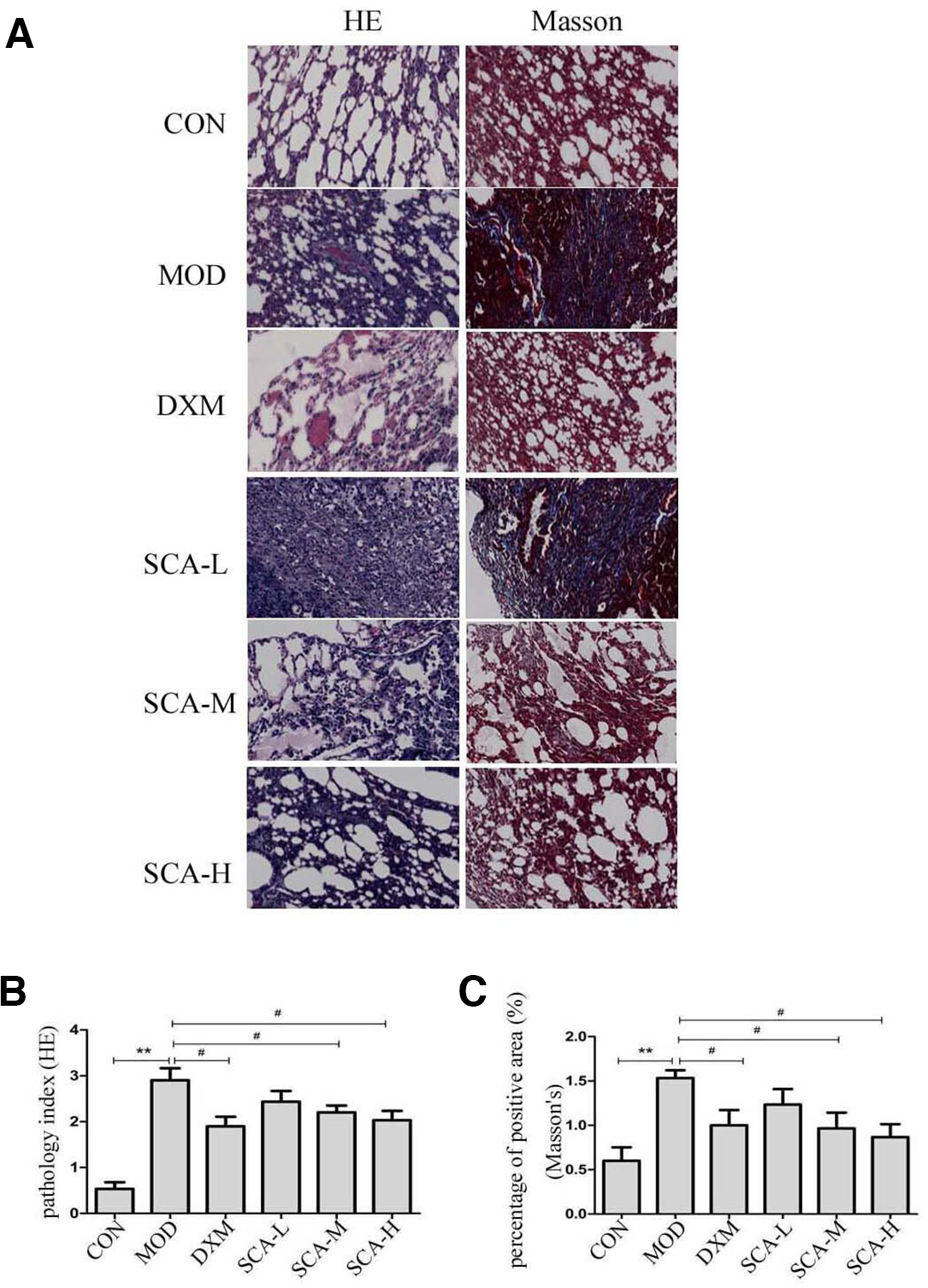
SCA alleviated BLM-induced lung injuries in mice. Following a single administration of 5 mg/kg BLM at day 0, the mouse received daily gavage of either SCA (1, 2, or 4 mg/kg) or DXM for 28 days and then was sacrificed, and lung tissue samples collected. (A) HE staining and Masson’s staining of lung tissues (×200); (B) alveolitis and inflammation score (pathology index); (C) fibrotic score (percentage of positive area). Data are taken from 3 slides from each mouse and 10 fields in each slide. ** P < 0.01 vs CON group; # P < 0.05 vs MOD group. BLM, bleomycin; CON, control group; DXM, dexamethasone; HE, hematoxylin and eosin; MOD, BLM-treated group; SCA, schisantherin A.
The collagen deposition was investigated by Masson’s staining (Figure 1(A)). The results indicated that the lung tissue of the control group had no obvious fibrous tissue. In the model group, there were large areas of collagen fiber bundles, which deposited in the bronchus and blood vessels. Moreover, the fibrosis score was significantly higher than that of the control group. These lesions were overtly ameliorated and only a small amount of collagen fiber deposition could be observed after treatment with SCA-M and SCA-H for 28 days. The scores for fibrosis by the percentage of positive area also indicated that SCA-M and SCA-H had been able to inhibit the progression of fibrosis (Figure 1(C)).
Effects of SCA on BLM-Induced Pulmonary Inflammation Response
The entire pathogenetic process of PF is mainly driven by inflammation and fibrosis. 19 During inflammation, inflammatory cytokines are related to the molecular mechanisms of PF. 20 Therefore, the levels of inflammatory cytokines were evaluated to determine whether SCA would alter the inflammatory responses. TGF-β1 level in serum was detected using ELISA, and the levels of COX2, IL-1β, IL-6, and TNF-α in lung tissues were examined by qPCR and Western blot analysis. Levels of the inflammatory cytokines TGF-β1, COX2, IL-1β, IL-6, and TNF-α were increased in the model group compared with the control group (Figure 2, P < 0.01; P < 0.001). SCA-H significantly decreased the levels of these inflammatory cytokines in serum and lung tissues (Figure 2). Our assay results demonstrated that SCA obviously reduced the inflammatory response in BLM-injured mice.

Effects of SCA on BLM-induced pulmonary inflammation response. (A) Effect of SCA on the expression of TGF-β1; (B) mRNA levels of COX2, IL-1β, IL-6, and TNF-α; (C) protein levels of COX2, IL-1β, IL-6, and TNF-α. Each bar represents means ± SD of 3 independent experiments ** P < 0.01, *** P < 0.001 vs CON group; # P < 0.05, ## P < 0.01, ### P < 0.001 vs MOD group. BLM, bleomycin; CON, control group; MOD, BLM-treated group; SCA, schisantherin A; TGF, transforming growth factor.
Effects of SCA on TGF-β1-Induced EMT in A549 Cells
A previous study has reported the crucial role of EMT in pathogenesis of PF. 21,22 Hence, we examined the effects of SCA on TGF-β1-induced EMT in the A549 cell line, a kind of human type Ⅱ alveolar epithelial cell, which is regarded as a usual cell model of EMT. Figure 3(A) shows that cell viability was not affected by less than 5 µM SCA, whereas more than 10 µM SCA resulted in significant inhibition. A549 cells treated with 5 ng/mL TGF-β1 for 48 hours displayed a spindle-shaped, fibroblast-like morphology (Figure 3(B)). Pretreatment with SCA inhibited the elongated shape morphological alterations induced by TGF-β1, and fibroblast-like A549 cells recovered their normal epithelial morphologies. Then, we examined the levels of the mesenchymal phenotypic marker α-SMA and the characteristic epithelial phenotypic marker E-cadherin by qPCR and Western blot analysis. The results demonstrated that the expression of α-SMA was increased notably while the expression of E-cadherin was significantly decreased in the model group compared with the control group (Figure 3(C)–(D), P < 0.05; P < 0.01; P < 0.001). The expressions of marker proteins were obviously reversed compared with the model group after SCA-H treatment (Figure 3(C)–(D), P < 0.05; P < 0.01; P < 0.001). These results demonstrated that SCA could suppress the process of EMT, which in turn significantly decreased the occurrence of fibrosis.

Effects of SCA on the expression of α-SMA and E-cadherin in A549 cells among different experimental groups. (A) Viability of cells treated with different concentrations of SCA by MTT assay; (B) representative phenotypes of A549 in different groups; (C) mRNA levels of α-SMA and E-cadherin; (D) protein levels of α-SMA and E-cadherin. Each bar represents means ± SD of 3 independent experiments. * P < 0.05, ** P < 0.01, *** P < 0.001 vs CON group; # P < 0.05, ## P < 0.01, ### P < 0.001 vs MOD group. CON, control group; MOD, bleomycin-treated group; SCA, schisantherin A; α-SMA, α-smooth muscle actin.
SCA Exhibited Anti-inflammatory Effects in TGF-β1-Induced A549 Cells
To evaluate the anti-inflammatory effects of SCA, we also examined pro-inflammatory cytokines, including COX2, IL-1β, IL-6, and TNF-α in TGF-β1-induced A549 cells through qPCR and Western blot analysis. The results showed that the mRNA and protein levels of COX2, IL-1β, IL-6, and TNF-α were increased in the model group compared with the control group (P < 0.05; P < 0.01; P < 0.001), while they were decreased after SCA-H treatment (Figure 4, P < 0.05; P < 0.01; P < 0.001). These results demonstrated that SCA treatment effectively inhibited the inflammatory response of the cells.
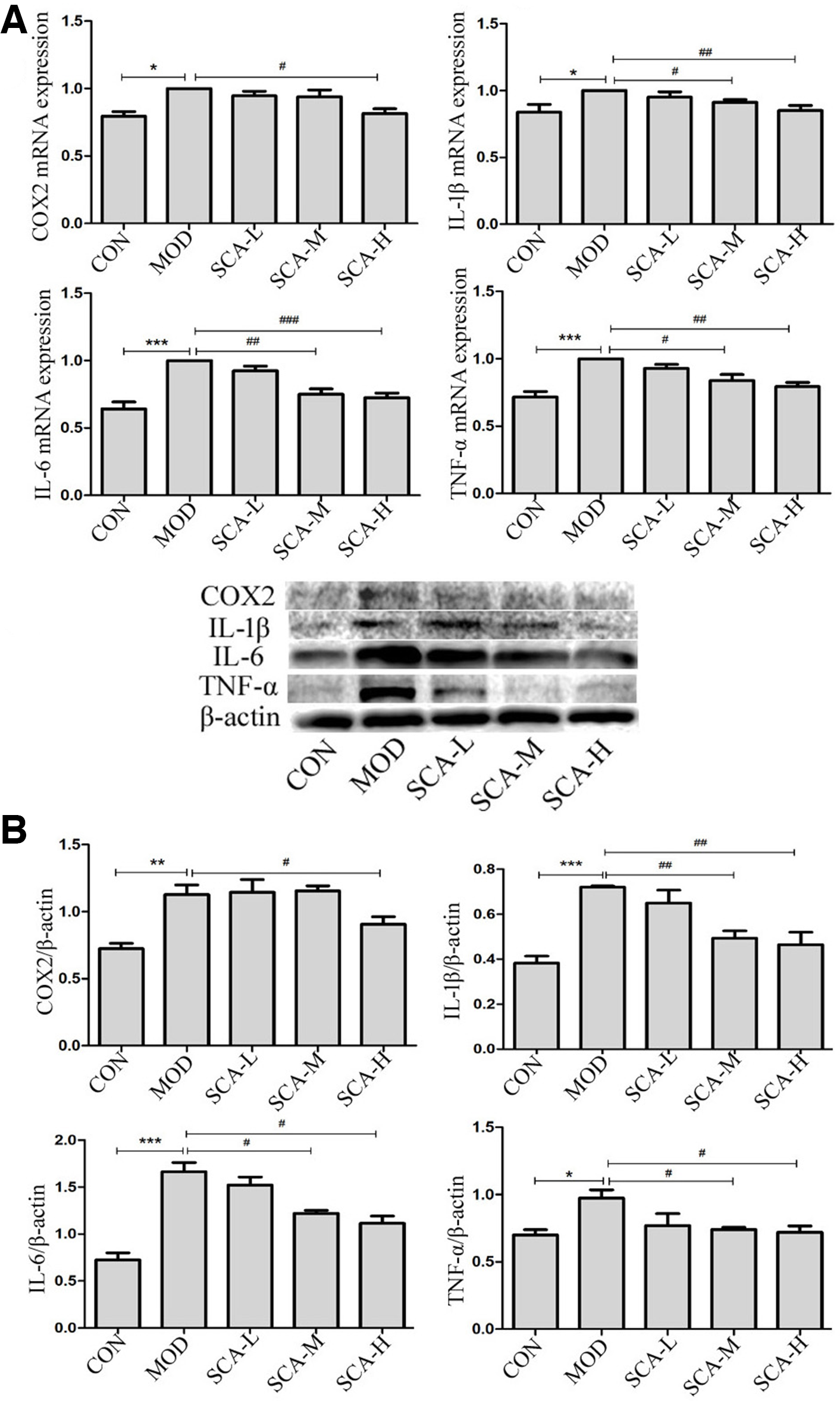
Effects of SCA on inflammatory mediators in TGF-β1-induced A549 cells. (A) mRNA levels of COX2, IL-1β, IL-6, and TNF-α; (B) protein levels of COX2, IL-1β, IL-6, and TNF-α. Each bar represents means ± SD of 3 independent experiments.* P < 0.05, ** P < 0.01, *** P < 0.001 vs CON group; # P < 0.05, ## P < 0.01 vs MOD group, ### P < 0.001 vs MOD group,. CON, control group; MOD, bleomycin-treated group; SCA, schisantherin A; TGF, transforming growth factor.
SCA Suppressed ERK Signaling Pathway In Vivo and In Vitro
As activation of the ERK pathway is involved in BLM-stimulated fibrotic injury and in TGF-β1-induced EMT, 23 -25 we examined whether SCA affected ERK signaling activation. Phosphorylated ERK (pERK) and ERK activities were detected in both BLM-treated lung tissues and TGF-β1-induced A549 cells by Western blot analysis. The data showed that both BLM (Figure 5(A), P < 0.01) and TGF-β1 (Figure 5(B), P < 0.05) treatments upregulated the phosphorylation of ERK. However, SCA-H treatment in vivo (Figure 5(A), P < 0.05) and in vitro (Figure 5(B), P < 0.01) caused a significant drop in the levels of pERK, although total ERK was constant in all samples. Together, these results revealed that SCA protected mice against BLM-induced PF and suppressed TGF-β1-induced EMT in A549 cells by inhibiting the ERK signaling pathway in vivo and in vitro.

SCA inhibited the ERK signaling pathway. (A) Protein levels of pERK and ERK in A549 cells; (B) protein levels of pERK and ERK in mouse lung tissues. Each bar represents means ± SD of 3 independent experiments.* P < 0.05, ** P < 0.01 vs CON group; # P < 0.05, ## P < 0.01 vs MOD group. CON, control group; ERK, extracellular signal-regulated kinase; MOD, bleomycin-treated group; pERK, phosphorylated extracellular signal-regulated kinase; SCA, schisantherin A.
Discussion
Although plenty of research effort has been made in experimental and clinical studies, there are not effective treatments for PF. 26 The development of efficient therapeutic interventions to ameliorate the pathogenic events in PF seems to gain significance. Since PF is closely associated with lung inflammation, anti-inflammatory medicines would be a new way of therapy for PF. Many studies have proved that SCA exerts potent anti-inflammatory activity. For example, SCA decreased the levels of pro-inflammatory cytokines by blocking LPS-induced activation of NF-κB and MAPK pathways in ARDS. 12 SCA inhibited the inflammatory response in LPS-activated BV-2 microglia, which relied on ERK phosphorylation. 13 SCA could inhibit the production of inflammatory cytokines by blocking NF-κB and MAPK pathways in RAW264.7 cells. 27 SCA had an anti-inflammatory influence on IL-1β-induced human osteoarthritis chondrocytes, 14 but whether it can influence PF by reducing inflammation remains largely unknown. We here explored the therapeutic effects of SCA on PF by conducting in vivo and in vitro experiments.
Bleomycin-induced lung fibrosis is a widely used animal model of human PF. 6,28 The current study indicated that BLM treatment not only decreased the weight of mice but also increased the wet weight of the lung and lung indices. However, the SCA-treated group exhibited significantly higher lung weight and lung indices and their weight loss was not evident. The histopathological results in this study showed a significant structure distortion of lung tissues in BLM-induced mice. Mice in the SCA-treated groups (SCA-M and SCA-H) had analogous presentations with less infiltration of inflammatory cells and less collagen deposition in comparison with the BLM group. The effects of SCA on histological alterations might be due to its anti-inflammatory and anti-fibrotic activities, which prevent the accumulation of collagen in BLM-induced lung tissues.
TGF-β1 is an attractant for fibroblasts and monocytes/macrophages and stimulates these cells to synthesize the pro-inflammatory and fibrogenic cytokines. 29 Airway remodeling of fibrotic lung diseases may be related to EMT of human A549 cells induced by TGF-β1. 30 EMT is a process through which epithelial cells are transformed into mesenchymal cells, which would constitute forming fibroblastic foci and excessive ECM accumulation. Therefore, EMT is characterized by changes in cell morphology, downregulating E-cadherin, and upregulating mesenchymal markers such as α-SMA. 31,32 Hence, an EMT inhibitor may serve as anti-PF agent that acted by targeting the EMT. In our study, TGF-β1 stimulation triggered EMT in A549 cells, and SCA inhibited the process by means of reversing changes in cell morphology, increasing the expression of E-cadherin, and decreasing the expression of α-SMA. These results demonstrated that SCA was able to block TGF-β1-induced EMT in A549 cells.
It has been revealed that PF is characterized by a loss of endothelial function and subsequent activation of the immune system, which is regarded as a direct result influenced by endocellular pro-inflammatory reactions. 33 Thus, inhibiting inflammation could be an effective and alternative approach for preventing the formation of PF. Increasing evidence demonstrated that pro-inflammatory cytokines TGF-β1, COX2, IL-1β, IL-6, and TNF-α promoted the development of lung fibrosis. 34,35 In our current study, it was shown that SCA notably downregulated pro-inflammatory cytokines, including COX2, IL-1β, IL-6, and TNF-α in both lung tissues and cells. In addition, SCA could suppress the level of TGF-β1 in BLM-induced mouse serum. These results indicated that SCA had a strong property to inhibit inflammatory mediators that alleviate BLM-induced and TGF-β1-induced inflammatory injury. According to our study, the anti-fibrotic effect of SCA was related to the amelioration of BLM-induced and TGF-β1-induced inflammatory response.
Despite some reports on the underlying target herbal medicines of anti-inflammation for PF, the potential mechanisms for the SCA-mediated regulation of inflammation in PF have not been clarified. The MAPK/ERK signaling cascade is a major pathway controlling cellular processes associated with fibrogenesis, including growth, proliferation, and survival. It has been proved that the anti-neuroinflammatory actions of SCA were reliant upon ERK phosphorylation-mediated Nrf2 activation. 13 Recently it has been indicated that activation of the ERK pathway is increased in BLM-induced PF. 36 Western blot of TGF-β1-induced A549 cells also demonstrates increased level of ERK phosphorylation. 37 Our results showed that the increased level of ERK phosphorylation after BLM and TGF-β1 stimulation was inhibited by SCA administration. Moreover, the decrease in ERK phosphorylation was correlated with the reduction of the expression of inflammatory cytokines. These findings suggested that SCA could inhibit ERK activation, thus reducing the expression of inflammatory cytokines in BLM-induced and TGF-β1-induced inflammation.
Conclusions
In summary, our study provided direct evidence to prove that in BLM-induced and TGF-β1-induced PF, the production of inflammatory mediators could be significantly decreased by SCA through inhibiting the ERK signaling pathway, which suggested that SCA might be a potential therapy option for PF. Further research is needed to clarify the target of SCA and its clinical applications.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Medical and Health Industry Development Special Foundation of Jilin Province Scientific Technology Development Project (20170311058YY), the Student's Training Program for Innovation and Entrepreneurship of Jilin Province (201811923144), and a project of the innovation plan for graduate students of Beihua University (2018041, 2019021, 2019027), Jilin Province Scientific Technology Project of Traditional Chinese Medicine (2019129), the Scientific Technology Research Project of the Education Department of Jilin Province (JJKH20191068KJ, JJKH20200076KJ), Jilin Science and Technology Innovation Development Program (20190601177).
