Abstract
To date, the monohexosylceramides present in the eukaryotic alga Euglena gracilis has not been reported. In this study, we extracted and purified a lipid fraction that eluted similarly to other reported monohexosylceramides. The structural determination of the lipid fraction revealed a monohexosylceramide (m/z = 889.5 and a loss of m/z = 162), corresponding to the formula C54H99O8N having moieties corresponding to a monohexose (C6H12O6), a 9-methyl-4,8-sphingadienine (C19H37O2N), and a nonacosanoic acid with 2 double bonds (C29H54O2). This is the first report of the isolation of monohexosylceramides from E. gracilis and will promote its utilization in functional foods and cosmetics.
Euglena gracilis Z (family Euglenaceae) is a microscopic, unicellular, photosynthetic, green alga, which is a member of the phylum Euglenozoa in the Excavata supergroup and part of the eukaryote domain. 1,2
Sphingolipids are lipids composed of sphingoid bases and their derivatives are ceramides, sphingosine, sphingosine-1-phosphate, glycosphingolipids, glycosylinositolphosphoceramides, sphingomyelins, ceramide phosphates, and gangliosides. 3 -5 Sphingolipids are composed of 3 structural parts, namely, a polar head group, a sphingoid base, and a carboxyalkyl group, and there are many structural variations in the long-chain bases and fatty acids and hydrophilic functional groups. Because a high concentration of ceramide has been detected in the stratum corneum of the skin, and its concentration decreases in patients with atopic dermatitis 6 and psoriasis vulgaris, 7 the topical administration of ceramide to the skin in moisturizers and cosmetics is considered to be beneficial in preventing transepidermal water loss. In addition, glycosylceramides increase the expression of genes involved in tight junctions and ceramide delivery in normal human epidermal keratinocytes. 8 Moreover, the dietary intake of glucosylceramide has been shown to improve atopic dermatitis, 9 -11 and topical administration of glucosylceramide improves ultraviolet A-induced wrinkle formation, 12 as well as preventing tumor metastasis in mice. 13 Thus, the utilization of sphingolipids as functional molecules has recently expanded. Therefore, natural sources of monohexosylceramides are increasingly in demand. 14
Several sphingolipids have been reported in marine organisms. For example, a monohexosylceramide containing 9-methyl-4,8-sphingadienine as the sphingoid base and hydroxylignoceric acid as the fatty acid moiety have been identified in haptophytes. 15 In addition, gangliosides containing [4E,8E]-sphinga-4,8-dienine and 2-hydroxy-Δ3-unsaturated fatty acid chains have been identified in Tetraselmis phytoplankton. 16 Also, several studies have reported that sphingolipids are obtained from members of the Euglenozoa phylum. For example, a glycosphingolipid containing mannose and galactose (1:2) as the sugar moieties; C18-dihydrosphingosine and 17-methyl sphingosine as the sphingoid bases; and normal and 2-hydroxy fatty acids as the fatty acid moieties have been reported in Trypanosoma cruzi. 17 In addition, a glycosphingolipid containing fucose and galactose (1:1) as the sugar moieties; sphingosine and trace dihydrosphingosine as the sphingoid bases; and C16:0, C18:0, and 2‐hydroxy C18:0 as fatty acids have been reported in Trypanosoma mega. 18 Furthermore, ceramides containing 19-methyl-C20-phytosphingosine and C20-phytosphingosine as sphingoid bases and stearic acid and α-hydroxystearic acid as fatty acids have been reported in Crithidia fasciculata (a member of the Trypanosomatida order). 19 Finally, in 1995, GM1α (Galβ1–3(NeuAcα2–6)GalNAcβ1–4Galβ1–4Glcβ-Cer) containing sphingosine or dihydrosphingosine was detected in E. gracilis. 20 However, since this report, the chemical structures of the sphingolipids contained in E. gracilis have not been reported. Therefore, in this study, the monohexosylceramides obtained from E. gracilis were investigated using electrospray ionization-mass spectrometry (ESI-MS).
Results
First, the lipids were extracted from E. gracilis using chloroform/methanol, treated with a mild alkali, purified by silica gel chromatography, and divided into 50 fractions. Fractions 6-17 were collected as candidate sphingolipids and further purified by high-performance liquid chromatography (HPLC). Fraction 32 obtained using HPLC purification was subjected to ESI-MS analysis (Figure 1) followed by fragment analysis. Subsequently, ESI-MS was used to analyze the structure of fraction 32. An ion at m/z 889.5 was detected and considered to be a monohexosylceramide. The structure was further analyzed using fragment ion analysis using tandem MS measurements (MS/MS). The m/z 889.5 ion generated a fragment ion with m/z 727.5 (Table 1), indicating the loss of a hexose moiety (m/z 162). From the MS data, the formula C54H99O8N was obtained, and the structure of a monohexosylceramide having a 9-methyl-4,8-sphingadienine (C19H37O2N) sphingoid moiety, and a nonacosanoic acid with 2 double bonds (C29H54O2) as the fatty acid moiety (Figure 2).
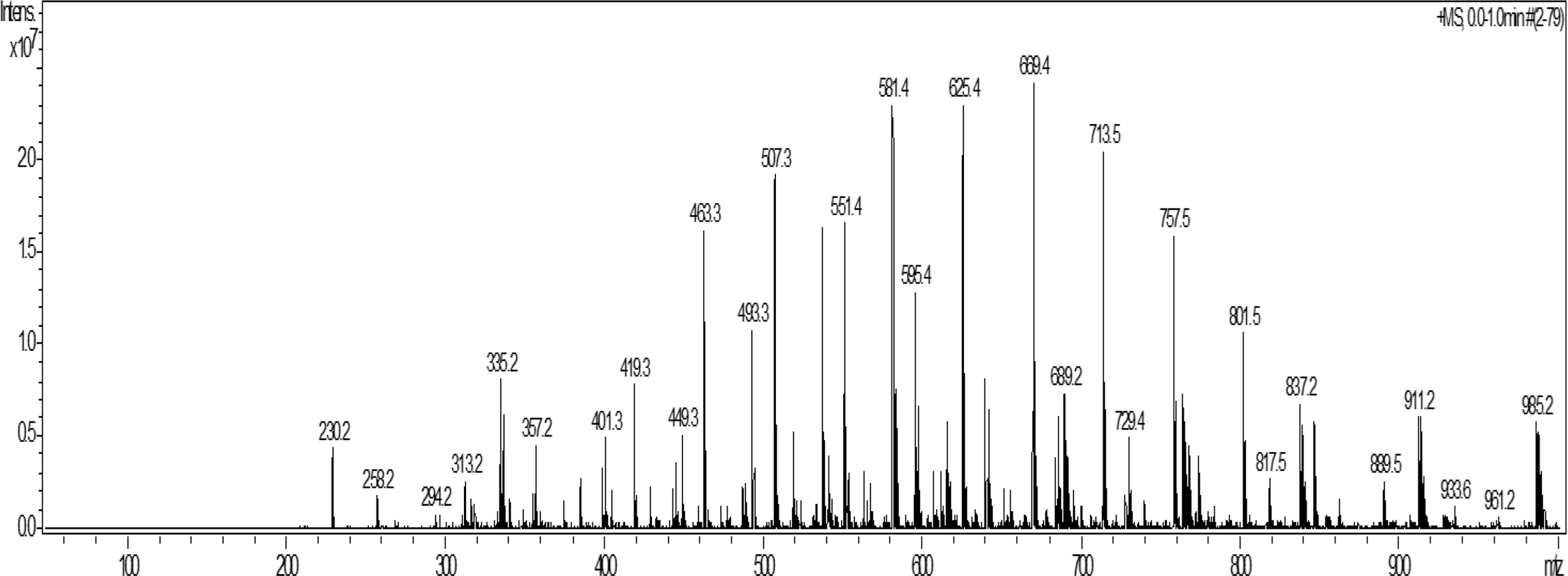
Positive-ion-mode electrospray ionization-mass spectrometry (ESI-MS) of fraction 32. ESI-MS (using HC ultra, Bruker Daltonics) was applied to fraction 32 purified from Euglena gracilis.

Deduced structure of the ion at m/z = 889.5. From the m/z information, the chemical composition formula was estimated to be C54H99O8N. Fragment-ion analysis suggested that it contains monohexose (C6H12O6), 9-methyl-4,8-sphingadienine (C19H37O2N) as the sphingoid moiety and nonacosanoic acid with 2 double bonds (C29H54O2) as the fatty acid moiety.
Ratio of the Intensities of Product Ions Generated From Ion m/z 889.5 by Electrospray Ionization-Mass Spectrometry (MS)/MS.

Discussion
In this study, we identified monohexosylceramides in E. gracilis. We extracted and purified alkali-tolerant lipids and determined their chemical structures to be monohexosylceramides. Our analysis using ESI-MS combined with fragment-ion analysis provided abundant information on the chemical composition. We identified a monohexosylceramide containing a monohexose, 9-methyl-4,8-sphingadienene, and nonacosanoic acid with 2 double bonds. However, because of technical limitations, further structural analysis is required to determine the exact structures of the sphingolipids contained in E. gracilis. For example, ESI-MS analysis cannot reveal the positions of the double bonds or methyl group in the sphingoid base and fatty acid. Moreover, this analysis cannot discriminate between glucose and galactose or the configuration of the sugar. To determine the chemical structures precisely, other techniques, such as nuclear magnetic resonance analysis, should be applied. Moreover, we identified the reported sphingolipids by utilizing retention values (R f) from thin-layer chromatography (TLC) analysis based on our previous studies. However, other spots might contain other sphingolipids with different structures. Therefore, analysis of the other TLC spots should be pursued in subsequent studies.
Because the topical application of ceramide to the skin decreases transepidermal water loss, 7 the application of monohexosylceramide to human primary keratinocytes increases the skin barrier function genes, 8 and the dietary intake of glucosylceramide decreases transepidermal water loss 9,10 ; thus, the detection of these sphingolipids will pave the way for the utilization of E. gracilis lipids in moisturizers, cosmetics, and functional foods. However, to date, there have been no reports of the chemical structures of the monohexosylceramides in Euglenozoa until this study.
In summary, we have proposed the identities of monohexosylceramides identified from the lipid fraction of E. gracilis. We hope that this report will encourage further investigation of the sphingolipids in E. gracilis and their use in cosmetics and functional foods.
Conclusion
A candidate monohexosylceramide was purified and analyzed from E. gracilis. By fragment-ion analysis, the compound was revealed to have a structure having a monohexose (C6H12O6) moiety, a 9-methyl-4,8-sphingadienine (C19H37O2N) sphingoid moiety, and a nonacosanoic acid with 2 double bonds (C29H54O2) as the fatty acid moiety.
Experimental
Extraction of Mild Alkali-Tolerant Lipids
Nine grams of E. gracilis was used for the extraction of lipids. Euglena gracilis was divided into 0.3 g aliquots, mixed with 1 mL of methanol, 0.5 mL of chloroform, and vortexed for 1 minute. Chloroform (0.5 mL) was added and vortexed for 1 minute. The solution was sonicated for 5 minutes, mixed with 0.8 M potassium hydroxide dissolved in methanol (2 mL), and incubated at 42 °C with shaking at 160 rpm for 30 minutes. To the solution, chloroform (5 mL) and distilled water (2.25 mL) were mixed and vortexed until saponification. The solution was centrifuged at 700 g for 10 minutes, and the lower phase collected.
Purification of Mild Alkali-Tolerant Lipids
The extracted lipids were concentrated using a rotary evaporator. The concentrated lipids were solubilized in chloroform. The solubilized lipids were applied to a chloroform-saturated 30 g silica gel column, which was washed with chloroform (600 mL) 3 times. The purified lipids were eluted with ethyl acetate/methanol (9:1) at 10 mL intervals, followed by a methanol wash. Fractions 6-17 were collected, adjusted to 2 mg/mL, and subjected to HPLC (LC-20AT, CTO-20AC, Shimadzu, Kyoto, Japan). Glycosylceramide purification by HPLC was performed using an Inertsil SIL 100 A column (5 µm, 4.6 mm diameter × 250 mm length, GL science Inc., Tokyo, Japan). The mobile phase consisted of buffer A (chloroform) and buffer B (95% methanol), and separation was achieved using the following gradient program: 00.00 minutes of A 100%/B 0%, 30.00.60.00 minutes of A 92.5%/B 7.5%, 80.00-90.00 minutes of A 10%/B 90%, 90.01-100.00 minutes of A 100%/B 0%, and 100.01 minutes STOP. The flow rate was 0.7 mL/min and the volume of the sample injection loop was 500 µL. The fractions were spotted onto TLC plates and visualized by orcinol/sulfate staining.
Electrospray Ionization-Mass Spectrometry
The dried samples were solubilized in 50 µL of chloroform/methanol (1:1), diluted with 950 of µL methanol, and subjected to ESI-MS (HC Ultra, Bruker Daltonics, Bremen, Germany), as previously described. 21,22
Footnotes
Acknowledgements
We are grateful to Mr. Keisuke Tsuge for providing technical support for using ESI-MS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the authors received financial support from euglena, Co., Ltd.
