Abstract
Objective
This study provides a comprehensive overview of galactomannans, focusing on their chemical structure, physicochemical properties, primary sources, and diverse industrial applications.
Method
The structural composition of galactomannans was analyzed, along with their physicochemical properties, including solubility, gelation potential, and viscosity. The study examined galactomannans extracted from leguminous seeds such as guar, carob, fenugreek, tara, and cassia. Their functional roles across various industries were also assessed.
Results
Galactomannans consist of a mannose backbone with galactose side chains, with their properties influenced by factors such as mannose content, molecular weight, and branching structure. These characteristics enable their widespread application across multiple industries. In the food sector, galactomannans function as thickening and stabilizing agents, enhancing texture. In pharmaceuticals, they serve as excipients and drug delivery components. Additionally, they play crucial roles in the paper, textile, petroleum, cosmetic, and explosives industries due to their binding, thickening, and water-resistant properties.
Conclusion
The unique structural and physicochemical properties of galactomannans make them valuable across various industries. Ongoing research continues to expand their applications, promoting sustainable and innovative industrial developments.
Introduction
During the differentiation of certain plant cells, significant quantities of polyosidic materials are synthesized to form mucilage. This substance is relatively hydrophilic. 1 Mucilage is a gelatinous, high molecular weight substance produced by almost all plants. It is primarily composed of polysaccharides but also contains proteins, minerals, and lipids. While the monosaccharide composition and polysaccharide structure of mucilage have been studied, only recently have initial connections been established between mucilage chemistry and its functional properties. 2 Due to its considerable variability in terms of chemical constituents, mucilage likely assumes a multitude of physiological functions in plants. It is found in rhizomes, roots, and seed endosperms, where it may primarily act as an energy reserve. Foliar mucilages also play a role in wound responses, plant-host-pathogen interactions, water transport, and responses to abiotic stress. The high water-fixing capacity of hydroxyl groups in the polysaccharide core allows mucilage to hydrate and store significant water reserves, providing plants with the ability to withstand physiological drought. It is increasingly evident that extracellular mucilages, in particular, can play a crucial role in the drought resistance of certain plant species. 3 While previous studies have examined the monosaccharide composition and structure of mucilage polysaccharides, it is only recently that the functional properties of mucilage have been linked to its underlying chemistry. 2 Plant-derived mucilage, due to its unique health and nutrition-related properties (including anticancer activity, enzymatic inhibition, immune stimulation, and anti-diabetic activity), is widely used as an active ingredient in the production of pharmaceutical, functional, and nutraceutical products. 4 By hydrolysis, mucilages yield a large number of different monosaccharides. Based on the hydrolysis products obtained, they can be classified into pentosans (such as xylan) or hexosans (like starch or cellulose). Gums and mucilages are closely related to hemicelluloses in terms of composition. 5 Structurally, mucilage is a complex of polymeric polysaccharides, mainly composed of highly branched carbohydrate structures consisting of monomeric units of L-arabinose, D-xylose, D-galactose, L-rhamnose, and galacturonic acid. Additionally, they contain glycoproteins and a variety of bioactive compounds such as tannins, alkaloids, and steroids. 4 Galactomannans from seeds, also known as seed gums, constitute the second major group of storage polysaccharides in terms of distribution in the plant kingdom and are associated with the cell wall of seed endosperms belonging to various botanical families. 6 Galactomannans are polysaccharides with a mannose backbone and galactose side branches, mainly sourced from Leguminosae seeds and microbial sources. 7 Galactomannans are valued for their viscosity retention, emulsifying, and stabilizing properties, with significant applications in the food and textile industries. Initially used in printing pastes for their thickening ability, their biodegradable and biocompatible nature has broadened their textile applications. While guar galactomannan remains dominant, other types are gaining industrial interest. Research highlights their potential for diverse textile applications, making them a promising alternative. 8
The objective of this review is to comprehensively examine and synthesize the current state of knowledge regarding galactomannans as principal reserve polysaccharides in plants. This review provides a detailed analysis of their distribution, structure, biosynthesis, and physiological roles across various botanical families. Additionally, it explores the diverse functions of galactomannans in plant metabolism, emphasizing their significance as energy storage compounds in seed endosperms. By critically evaluating existing research, this paper aims to identify knowledge gaps and propose directions for future investigations. Ultimately, it serves as a comprehensive resource to enhance the understanding of the biological importance and potential industrial applications of galactomannans in plant physiology, pharmaceuticals, and other sectors.
Methods
To provide a comprehensive and up-to-date overview of the topic, we conducted a narrative literature review using several reputable databases, including PubMed, Scopus, ScienceDirect, and Google Scholar. The search covered the period from October 1, 2018, to June 2024. We focused on identifying relevant peer-reviewed articles, academic books, and dissertations. Keywords such as galactomannans, seeds, and applications were used to guide the search in alignment with the objectives of this review.
Physicochemical Properties and Functional Applications of Galactomannans
Seed Anatomy
In the seeds of Trigonella foenum-graecum (fenugreek), the endosperm cells are entirely filled with galactomannans. Despite the apparent presence of a primary cell wall between the storage walls, the endosperm has been found to be non-living. The aleurone layer present in the seeds of T. foenum-graecum, Trifolium incarnatum (crimson clover), and Medicago sativa (alfalfa) is the cellular layer responsible for producing galactomannan hydrolases. In guar seeds, the endosperm is also non-living, and the cells are almost entirely filled with galactomannans. Conversely, in the seeds of Ceratonia siliqua (carob), the endosperm cells are living, and galactomannans can be considered as a thickening agent of the wall. In this case, there is no clear distinction between the endosperm and the aleurone layer, and enzymes are likely produced and released into the cell walls by the endosperm cells. The endosperm cell cytoplasm is filled with protein bodies, serving as reserves during germination. While a unicellular aleurone layer is present between the testa and endosperm, there is no direct evidence that it produces galactomannan hydrolases. In contrast to carob, in Sesbania marginata seeds, an aleurone layer is clearly visible between the testa and the endosperm, but simultaneously, most endosperm cells contain protein bodies. A completely different anatomy occurs in Schyzolobium parahybum seeds. Instead of an aleurone layer, long haustorium-like cells are observed, seemingly immersed in a glassy galactomannan endosperm. 9 Dicotyledonous fenugreek seeds consist of a wrinkled brown-yellow seed coat, enclosing two halves of a translucent whitish endosperm primarily composed of water-soluble polysaccharides (galactomannans). The seeds contain proteins (25%-30%), an ethanolic extract (7%-9%), steroid saponins (5%-7%), galactomannans (25%-30%), insoluble fibers (20%-25%), and ash (3%-4%). Between the two halves of the endosperm is a sandy yellowish germ portion. This germ is mainly composed of good-quality edible proteins and also contains an ethanolic extract (5%-7%) consisting of diosgenin, yellow pigments, trigonelline, free amino acids, B-complex vitamins, and flavonoids. The fibrous material mainly comprises insoluble cellulose fibers and water-soluble galactomannans. 10
Storage Polysaccharides in Higher Plants
The plant cell wall constitutes an extracellular matrix surrounding each plant cell. This matrix is composed of a complex mixture of polysaccharides and glycoproteins with varied osidic structures and compositions. The polysaccharides in the plant cell wall can be neutral or acidic, linear or branched, and may be substituted with methyl or acetyl groups. Neutral polysaccharides include arabinogalactans, arabinans, galactans, β-glucans, glucomannans, xyloglucans, xylans, mannans, and galactomannans. Acidic polysaccharides include homogalacturonans, pectic polysaccharides, and glucuronoxylans. 11 Plant cells can also store reserve polysaccharides, typically during periods of intense photosynthetic activity. Starch is the sole polysaccharide stored in plastids, while non-starch polysaccharides are stored either in vacuoles or outside the plasmalemma. Vacuolar polysaccharides can be used as reserve materials, while extra-plasmalemmal polysaccharides can serve as structural macromolecules or reserve substances. Non-starch reserve polysaccharides are primarily found in seeds, roots, rhizomes, tubers, bulbs, shoot stems, and to a lesser extent, in leaves. They may occur concurrently with starch or be the sole carbohydrate reserves in a given plant organ. 12 Cell wall polysaccharides are chemically inert and exhibit varying degrees of water solubility. This characteristic imparts similar advantages to starch, such as low reactivity and the ability to form gels, allowing the existence of a cellular compartment—the cell wall—that permits water flow with considerable freedom. Storage polysaccharides of the cell wall have generally been classified into three groups: mannans, xyloglucans, and galactans. 9 Mannans serve as the primary energy reserve polysaccharides in seeds of various plant families (eg, legumes, palms) and can also be found as structural components in the secondary cell wall of conifers. Mannans can be classified into linear mannans, galactomannans, glucomannans, and galactoglucomannans. 13 Among these, galactomannans are the most extensively studied due to their economic importance. They are used as thickening and stabilizing agents in various industries. Moreover, they can be isolated from seeds with relatively high purity and yield, are water-soluble, non-toxic, biocompatible, and suitable for various biological applications.
Overview of Polysaccharides
Natural biopolymers can be classified into eight major families, including nucleic acids, polyamides, polyoxoesters, polythioesters, inorganic polyesters, polyisoprenoids, and polysaccharides. The latter have been identified across a myriad of organisms, ranging from bacteria to animals, including fungi, micro- and macro-algae, as well as higher plants. 14 Plants employ diverse strategies to accumulate substantial amounts of these compounds, facilitating their adaptation to different environments. These substances are mobilized during development, with their products serving various purposes such as energy generation and raw material production. Among the key substances stored by plants, numerous carbohydrate polymers have been evolutionarily selected. 9 Approximately 99% of total natural polysaccharides are found in plants and vegetables, making them a major renewable source of these polymers, owing to their useful physicochemical properties such as emulsifying power, viscoelasticity, polyelectrolyte conformation, adhesion, bio-compatibility, and stabilizing power. They are commonly exploited in various application domains. Natural plant polysaccharides are non-toxic, biocompatible, biodegradable, and water-soluble. 15 Polysaccharides are composed of numerous monosaccharide residues linked together by O-glycosidic bonds. The vast structural diversity of polysaccharides, arising from differences in monosaccharide composition, types and patterns of linkage, chain forms, and degree of polymerization, dictates their physical properties, including solubility, flow behavior, gelation potential, and surface/interface characteristics. Structural diversity also dictates the unique functional properties exhibited by each polysaccharide. Commercially available polysaccharides used in food and non-food industries as stabilizers, thickeners, gelling agents, crystallization inhibitors, and encapsulating agents, among others, are also termed hydrocolloids. 16 In recent years, plant polysaccharides have emerged as a significant class of bioactive natural products. A wide range of polysaccharides has been reported to exhibit anti-tumor, immunostimulatory, anti-complementary, anti-inflammatory, and anticoagulant activities. Most polysaccharides derived from higher plants do not induce significant side effects, a major concern associated with bacterial polysaccharides and synthetic compounds. 17
Galactomannans
Overview of Galactomannans
Galactomannans represent a significant group of polysaccharidic hydrocolloids produced in certain plants, occurring both in the form of cell wall polysaccharides and storage polysaccharides. They are primarily synthesized in the seeds of leguminous plants. A comprehensive study conducted by 10 examined 163 legume seeds, revealing that 119 endosperms contained galactomannans as the principal polysaccharide reserve component. However, galactomannans have also been identified in non-leguminous sources, including plants such as Cocos nucifera, Coffea arabica, Elaies guineensis, Phoenix dactylifera, and Phytelephas macrocarpa. In exceptional cases, such as Glycine max, Gymnocladus dioica, or Mucuna (Figure 1), galactomannans are found in the husk, inner tegument, and kernel, respectively. 18 Galactomannans have been recognized for their binding properties since ancient times, with Egyptians using carob gum to adhere mummy bandages. However, it was only in the twentieth century that galactomannans became industrial products. They find numerous applications, serving as thickening agents in the food industry for products like ice creams, mayonnaises, sauces, and bakery items. Additionally, they are utilized in non-food industries such as printing, photography, concrete production, explosives, oil drilling fluids, paint, ink, wax, textiles, beauty products, and anti-diarrheal products. 19 Studies on the structure, characterization, modifications, biogenesis, and applications of galactomannans are up-to-date, evidenced by the continuous growth of scientific data on these biopolymers. 20

Fenugreek, Guar, Tara, and Carob Seeds and Flours. 21
Molecular Structure of Galactomannans
Galactomannans share a common general structure, characterized by a backbone consisting of β-(1→4)-D-mannose to which a single branch of D-galactose is attached via an α-(1→6) linkage. The variations among them are primarily observed through the mannose/galactose (M/G) ratio, which varies depending on the plant source (Figure 2). The average molecular weight of galactomannans ranges from 1 to 2 × 106 g/mol, although this may vary based on extraction and solubilization conditions. 22

Molecular Structure of Galactomannans. 23
Primary Sources of Galactomannans
The four main plant sources of galactomannans are carob gum (Ceratonia siliqua L.), guar (Cyamopsis tetragonolobus), tara (Caesalpinia spinosa), and fenugreek (Trigonella foenum-graecum). 24 The mannose-to-galactose (M/G) ratio, indicating the number of mannose residues per galactose residue, is approximately 4:1 for carob gum (LBG), around 3:1 for tara gum (TG), about 2:1 for guar gum (GG), and roughly 1:1 for fenugreek gum (FG) (Figure 3; Table 1). Studies have demonstrated that molecular characteristics of galactomannans, such as M/G ratio and molar mass, can vary depending on the plant variety, growth conditions (climate, soil), and the extraction or polymer preparation method, even from the same source of galactomannans. 19

the Structures of Repetitive Units in the Four Main Galactomannans Involve Mannose Residues Linked by β-(1/4) Glycosidic Bonds, with Galactose Attached Through α-(1/6) Glycosidic Linkages. 21
Mannose/Galactose Ratio in Key Legumes.

Fenugreek (Trigonella foenum-graecum) is a significant plant containing various known phytochemicals and is harvested in different regions, particularly in western Asia. Galactomannans obtained from fenugreek seeds exhibit high carbohydrate content and low levels of proteins and fats. Previous research has demonstrated that fenugreek seed galactomannans have the ability to form higher quality and more stable emulsions compared to other galactomannans. Consequently, the production of emulsions containing this hydrocolloid can be beneficial for researchers in the food industry. 25
Carob gum galactomannans have the lowest galactose content. Carob gum galactomannans provide relatively stable and highly viscous solutions, minimally affected by pH changes. They also form edible gels as they interact with other polysaccharides such as carrageenan, agarose, and xanthan. The molecular size and structure (Man/Gal ratio) of galactomannans are crucial molecular features that strongly influence the functional properties of carob gum in pure aqueous dispersions or in food products. These molecular features are also significant determinants of their solution rheology, self-association capacity (during freezing and thawing), and interactions with other polysaccharides. However, there is still limited knowledge regarding intervarietal differences in the molecular characteristics of carob seed galactomannans. Due to the interest in physiological benefits such as mitigating glycemic and insulin responses, reducing serum cholesterol levels, etc, studies characterizing the structures and solution properties of plant polysaccharides are crucial for bridging structure-function gaps in these materials and aiding in the development of new health-beneficial food products. 26
Tara gum is a commercial galactomannan derived from the endosperms of the spiny tree Caesalpinia. It is considered a significant alternative to guar gum and carob gum due to its similar functional properties and the scarcity of supply for both common galactomannans. It is typically used as a thickening and stabilizing agent in the food and chemical industries. However, its application is limited by its rheological properties, which depend on the molecular weight of the polysaccharides. Tara gum with a high molecular weight exhibits high viscosity even at low concentrations, which can lead to issues of partial hydration and chemical modifications in products. To meet various processing demands in different industries, it is necessary to decrease its viscosity and prepare tara galactomannans with different molecular weights. Relevant research is needed to explore its potential as a dietary supplement, biomaterial, and biomedicine. 27
Guar gum is the most commonly used galactomannan in the industry and has an M/G ratio of approximately 2.2. Its popularity stems mainly from its structural properties that enable the production of highly viscous polymer solutions. Guar can also be chemically or enzymatically modified to extend its industrial applications. The various forms of guar and its modifications have generated a variety of commercial uses in fields such as hydraulic fracturing, food, pharmaceuticals, textiles, cosmetics, detergents, as well as healthcare and personal care. Furthermore, guar is considered safe for human consumption and is often combined with other biopolymers to thicken or bind food products. Since guar has the lowest M/G ratio among natural galactomannans available on the market, modified guars with a higher M/G ratio can be obtained by guar debranching. These modified guars should exhibit properties similar to other natural galactomannans. 28
While many plants can be sources of galactomannans, only carob gum, guar, tara, and cassia gum are commercially manufactured. Among these sources of galactomannans, guar gum, readily available at a lower cost, is extensively studied by various researchers. 29 Therefore, galactomannans cannot always be replaced by others in a specific application. This is why galactomannans from several different plant sources must be used for commercial production, despite their variable availability and price differences. Molecular, chemical, and structural variations leading to differences in their functional behavior and occurrence have also resulted in the existence of a broad spectrum of these plant polysaccharides. 30 In general, natural galactomannans pose challenges in water solubility due to their high molecular weights and elevated viscosity in aqueous solutions. Additionally, these galactomannans exhibit lyotropic and thermotropic liquid crystallinities at specific concentrations in water, establishing them as a significant group within the realm of branched polysaccharides. 31
Distribution of Galactose Units on Mannose Chains
Galactomannans exhibit similar overall structures but differ in their mannose/galactose (M/G) ratio. A study conducted by 32 revealed that the distribution of galactose units along the mannose chain does not follow a simple statistical pattern. Enzymes such as β-mannanase derived from Aspergillus niger or germinating guar seeds can degrade galactomannan polymers. These enzymes cleave the β(1-4) linkages in the mannose chain at positions devoid of galactose. Analysis of the monosaccharide composition of the degradation products (oligosaccharides) has proposed three types of galactose unit distribution: an ordered or regular arrangement, a random arrangement, and a uniform block arrangement (Figure 4) (where “smooth regions,” completely devoid of galactose branching, alternate with “bristled regions,” where galactose residues are present on all mannose units). 19
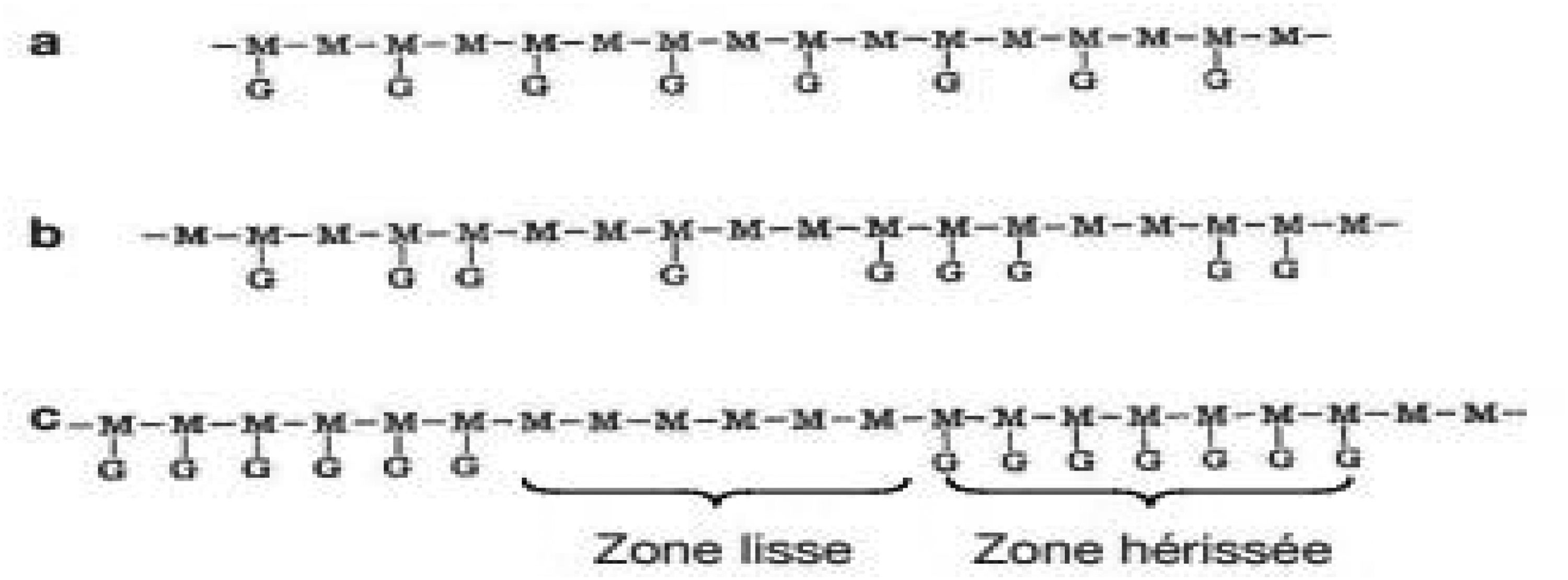
Different Types of Distribution of Galactose Residues Along the Mannose Chain: Regular Arrangement (a), Random Arrangement (b), and Uniform Block Arrangement (c). 19
Conformation of Galactomannans
Galactomannans have similar overall structures but vary in the mannose-to-galactose ratio (M/G). A study conducted by 32 revealed that the distribution of galactose units along the mannose chain does not follow a simple statistical distribution. Enzymes such as β-mannanase from Aspergillus niger or germinating guar seeds can degrade galactomannan polymers by cleaving the β (1-4) linkages in the mannose chain at sites devoid of galactose. Analysis of the monosaccharide composition of degradation products (oligosaccharides) suggested three types of galactose unit distribution: an ordered or regular arrangement, a random arrangement, and a uniform block arrangement (where “smooth zones,” entirely devoid of galactose branching, alternate with “spiky zones” where galactose residues are present on all mannose units). 19 Monosaccharide units in galactomannans have a cis pair of hydroxyl groups on their pyranosyl cyclic structure (positions 2,3 in the case of mannose and positions 3,4 in the case of galactose). In contrast, glucose, cellulose, and starch have trans-hydroxyl groups. The presence of a cis-hydroxyl pair with an essentially linear chain configuration leads to numerous unique interactions in galactomannans. The hydrogen bonding property of galactomannan polymers is much stronger than that of glucan. In solution, galactomannans adopt an elongated rod-like conformation, occupying a large volume of gyration. Fenugreek polysaccharides have a mannose skeleton that is almost completely substituted, leaving little chance for inter-chain association. The rotating molecules of these linear polysaccharides colloide with each other and with clusters of solvent molecules to produce high-viscosity solutions, dependent on molecular weight. 10
Physicochemical Properties of Galactomannans
When comparing different galactomannans, it is evident that increased substitution leads to higher solubility, but additional cross-linking through hydrogen bonds is associated with lower solubility. The M/G ratio varies considerably depending on the source of galactomannans and typically ranges between 1.1 and 5.0. Variations in the structure of galactomannans, especially in the M/G ratio and fine structure, result in significant changes in solubility, viscosity, and interactions with other polysaccharides. 33
− Solubility: In practice, the solubility of galactomannans in aqueous solutions is generally measured as a percentage, evaluating the proportion of the product that remains in solution after centrifugation. Solubility depends on factors such as the physicochemical properties of molecules, the balance of interactions between molecules and the solvent, as well as the physical state of the material. 34 Galactomannans with high galactose content are generally more soluble in water and tend to form weaker gels compared to those with a lower M/G ratio. This higher solubility is attributed to the presence of a greater number of side chains, which keep the main mannose chains sufficiently apart to prevent effective intermolecular interactions. 33 The level of galactose substitution is also a key factor in galactomannan solubility. A galactose substitution rate of at least 10% is necessary for adequate solubility in water. Galactomannans with galactose content below 10% tend to form precipitates at room temperature due to the association of non-substituted regions of the polymer. Solubility at lower temperatures is limited to chains with galactose content higher than the average value of the whole gum, and fractions with different M/G ratios can be obtained by dissolving at progressively higher temperatures. 35 In conclusion, the solubility of galactomannans depends on various factors, including concentration, solution temperature, structural characteristics of polymers, and molecular weight. 34
− Stability: The depolymerization of mannose chains leads to a significant reduction in viscosity. This occurs, for example, when galactomannan solutions at very low pH are heated. At room temperature, these solutions are stable in a pH range from 3 to 11, but they can undergo degradation by microorganisms. Therefore, sterilization is recommended before storage. 36 Solutions of guar and tara gum exhibit very good stability during freeze-thaw treatments, while carob gum becomes partially insoluble after freezing. 37
− Viscosity: Galactomannans can form highly viscous solutions at low concentrations, which are slightly affected by pH, the addition of electrolytes, and heat treatment. These solutions typically show non-Newtonian behavior, with viscosity decreasing with increasing shear rate. Viscosity depends primarily on molecular weight, while synergistic interactions seem to be determined by the mannose/galactose (M/G) ratio and the fine structure of galactomannan chains. 38 In general, galactomannans with higher galactose content are easily soluble in water and exhibit a weaker tendency to form gels due to synergistic interactions compared to those with lower galactose content. 33 Hydrocolloids are used to modify the textural attributes of foods, and studying their rheological behavior is essential for calculating any process involving fluid flow (eg, pump sizing, extraction, filtration, extrusion, purification). They also play a crucial role in analyzing flow conditions in food processes such as pasteurization, evaporation, drying, and aseptic processing. 38
− Interaction with Other Polysaccharides: Galactomannans extracted from seeds of four different Leguminosae species, namely Adenanthera pavonina, Caesalpinia pulcherrima, Gleditsia triacanthos, and Sophora japonica, exhibit characteristics suitable for use in the food and biomedical industries. Synergistic interactions of the two unconventional galactomannans (G. triacanthos and S. japonica) with carrageenan and xanthan offer alternatives to traditional galactomannans, such as guar gum and carob gum, with the same polysaccharides. These interactions can be used to confer different rheological properties to food products. 39 Furthermore, galactomannans with fewer side chains (higher M/G ratio) can interact with other polysaccharides due to their long blocks of unsubstituted mannose units. Galactomannans are rich in hydroxyl groups, allowing them to bind and absorb water. They are also rich in cis-OH groups, allowing chain aggregation through hydrogen bonding, making hydration more complex. In this case, cross-linking through exchange may occur. Substitution with galactose introduces steric hindrance between molecules, thereby improving solubility in water. 33
− Stability and Degradation of Galactomannans: Galactomannans can undergo

General Structure of Galactomannan and Bonds Susceptible to Cleavage by Different Enzymes. 40
Biological Functions of Galactomannans
The biological role of galactomannans in legume seeds is intricate and challenging to comprehend. Beyond serving as reserve polysaccharides, they also create a conducive environment for seed germination and shield against water stress during periods of water scarcity. Additionally, they regulate water absorption by seeds from different plant species, fostering favorable conditions for growth in the prevailing climatic conditions of a geographical region. This high water affinity, shared to varying degrees by all galactomannans, has found numerous applications in industry and food production. 30
Biosynthesis of Galactomannans
Galactomannans are synthesized in developing endosperms by GDP Mann-dependent mannosyltransferase and UDP Gal-dependent galactosyltransferase (GalT). 35 Most carbon is transported as sucrose in plants due to its non-reducing nature. The α-1,2-glycosidic bond in sucrose can be irreversibly/reversibly cleaved by invertase/sucrose synthase, as illustrated in Figure 6. Invertase catalyzes sucrose cleavage into glucose and fructose, while sucrose synthase converts sucrose into UDP-glucose and fructose in the presence of UDP. Fructose is phosphorylated by fructose kinase to form fructose-6-phosphate, isomerizing into mannose-6-phosphate by phosphomannoisomerase. Subsequently, phosphomannomutase transfers the phosphate group from carbon 6 to carbon 1. Furthermore, GDP-mannose pyrophosphorylase uses this mannose-1-phosphate, producing GDP-mannose and eliminating inorganic phosphate. UDP-glucose is converted to UDP-galactose catalyzed by UDP-galactose 4-epimerase. Substrates like GDP-mannose and UDP-galactose are synthesized in the cytoplasm and then transported into the Golgi apparatus by sugar nucleotide transporters for galactomannan biosynthesis. Mannan synthase thus produces the polysaccharide mannane skeleton using GDP-mannose, and galactosyl residues are transferred from UDP-galactose to the mannane skeleton by galactosyltransferase to produce galactomannans. 41 In some species with a high degree of galactose substitution, ie, lower M:G ratios like fenugreek (M:G = 1.1) and guar (Cyamopsis tetragonolobus; M:G = 1.6), the M:G ratio is determined during synthesis. In Senna occidentalis seeds (M:G = 2.3-3.2), galactomannan synthesized is edited in the endosperm by a specific α-galactosidase (Figure 6). 35

Biosynthesis of Galactomannans. 41
Mobilization of Galactomannans
In fenugreek, during the mobilization of galactomannan from the endosperm, a transient accumulation of starch was quantitatively observed, along with electron microscopy studies in cotyledons and the axis of fenugreek. Starch is synthesized and rapidly mobilized from cotyledons with a concomitant increase in α-amylase activity. Galactomannans are decomposed by three hydrolases: α-galactosidase, endo-β-mannanase, and β-mannosidase. Polysaccharides are disassembled into their monosaccharide constituents (free mannose and galactose), simultaneously with sucrose production in the endosperm. Apparently, sucrose acts as the transport sugar that carries the products of storage mobilization (carbon and energy) to the growing embryo. Starch is produced transiently in cotyledons. 9
42 conducted in-depth studies on the fate of guar galactomannan products after mobilization. They detected the activities of phosphomannoisomerase and phosphoglucoisomerase in the endosperm and suggested that these enzymes are likely responsible for the epimerization of mannose into glucose, which will probably be used for sucrose synthesis in the endosperm. 43 highlighted that legume seed endo-β-mannanase acts as a genuine mannanase, with its hydrolytic action on galactomannans hindered by galactose substitution. This underscores the importance of prior depolymerization of the polymer and likely explains the very high levels of α-galactosidase activity found during in vivo galactomannan mobilization in species containing highly substituted polymers. Exo-β-mannanase (or β-mannosidase or β-D-mannoside mannohydrolase) is an enzyme showing specificity for manno oligosaccharides and no activity on the polymer. Based on these results, it can be suggested that this enzyme functions to hydrolyze manno oligosaccharides produced by the concerted action of α-galactosidase and endo-β-mannanase 9 (Figure 7).

Biochemical Pathways Involved in Galactomannan Catabolism. 9
Biological Activities of Galactomannans
In recent years, polysaccharides of plant origin have emerged as a significant class of bioactive natural products. A wide range of polysaccharides have been reported to exhibit diverse biological activities such as anti-tumor, immunostimulatory, anti-complementary, anti-inflammatory, anti-coagulant, and fibrinolytic effects. Unlike bacterial polysaccharides and synthetic compounds, most polysaccharides derived from higher plants are relatively non-toxic and do not induce significant side effects. 17 Mannans, including galactomannans and glucomannans, have demonstrated prominent biological activities in mammals, particularly macrophage activation and stimulation of T lymphocytes. Consequently, they are regarded as potent immunostimulants with significant activity against infectious diseases and tumors. 44
Immunomodulatory Activity
The biological activity of galactomannans is diverse and complex, although their immunomodulatory potential is generally acknowledged. Galactomannans represent a heterogeneous class of polysaccharides concerning origin, structure, and biological activities. For instance, Aspergillus fumigatus conidia contain galactomannans that induce maturation of human Langerhans cells via the DC-SIGN receptor, leading to innate immune response activation. Galactomannans from Caesalpinia spinosa and Aspergillus fumigatus also bind to the pentraxin receptor, but the underlying molecular mechanism remains unknown. Studies have demonstrated that galactomannans from Ramalina celastri and Morchella esculenta stimulate macrophage activity and NF-κB transcription factor activation, primarily associated with inflammatory responses. Furthermore, galactomannans extracted from Mimosa scabrella and Leucaena leucocephala have been reported to protect against flavivirus infection after sulfation. 45 A galactomannan with an octasaccharide repetitive unit (ACP) was isolated from the Taiwanese medicinal fungus Antrodia cinnamomea. Its chemical structure was determined, and ACP exhibited significant activity in enhancing phagocytosis and bactericidal activity of murine J774A.1 macrophages against Escherichia coli. 46 These findings highlight the pharmacological potential of galactomannans and their ability to modulate the immune response to combat bacterial infections.
Antidiabetic Activities
Diabetes mellitus is a chronic metabolic disorder characterized by hyperglycemia and metabolic disturbances in proteins, carbohydrates, and fats. This condition results from the failure of insulin production, insulin action, or both. Managing type 2 diabetes remains complicated due to several factors inherent to the pathological process, including insulin resistance, hyperinsulinemia, altered insulin secretion, and decreased insulin-induced glucose absorption and utilization. Inhibitors of α-glucosidase and α-amylase, such as metformin and glibenclamide, are used to treat this type of diabetes. 47 Recently, viscous polysaccharides have garnered interest as potential treatments for diabetes mellitus. Carob gum is an example of a commonly used viscous polysaccharide in foods and as an adjuvant for diabetes treatment. Research has shown that carob gum can reduce blood glucose levels in rats, significantly inhibit glycogenesis, and increase liver glycogen content in mice. These results suggest that the regulation mechanism of blood glucose by carob gum could be related to promoting glucose absorption by the liver and peripheral tissues and inhibiting the hepatic glycogenesis pathway. Additionally, carob gum can reduce or delay glucose absorption in the intestinal tract, increase satiety, and decrease hunger. 48 Guar gum, a legume flour rich in galactomannans, has sparked considerable interest as an oral antidiabetic agent in the treatment of type 2 diabetes (non-insulin-dependent). The exact mechanisms by which it reduces postprandial blood glucose elevation are not well understood, but its presence increases the viscosity of gastrointestinal contents, affecting physiological functions such as gastric emptying and peristalsis. Increased viscosity and reduced water activity during starch hydrothermal treatment might influence α-amylase activity. Guar galactomannans have a direct non-competitive inhibitory effect on α-amylase. Their effects on lowering postprandial blood glucose involve not only modifications of intestinal physiology but also direct inhibition of the first step in starch biochemical degradation. 49
Effect on Hypercholesterolemia and Cardiovascular Diseases
Dyslipidemias are common lipid metabolism disorders worldwide, characterized by increased plasma levels of various lipid and lipoprotein fractions, including total cholesterol, low-density lipoproteins (LDL), very low-density lipoproteins (VLDL), triglycerides, and chylomicrons. These dyslipidemias have a significant impact on the development of cardiovascular diseases (CVD), especially hypercholesterolemia and LDL levels. Therefore, reducing these levels can aid in managing CVD. 50 The role of viscous polysaccharides in reducing blood cholesterol and triglyceride levels in humans and animals has been extensively studied. Guar gum (GG) is one of the most viscous galactomannans that is not digested in the mammalian small intestine and is particularly effective in lowering cholesterol levels. Its effect on glucose metabolism in humans with diabetes mellitus has also been reported. Streptozotocin-induced diabetic rats have been used in some studies to investigate the effects of dietary fibers on diabetic metabolic states. Xanthan gum (XG) and locust bean gum (LBG) are also viscous polysaccharides with hypocholesterolemic and hypoglycemic effects. Although GG has been successfully used for hypercholesterolemia treatment, its clinical application is limited due to its unpleasant taste. Attempts have been made to create palatable foods containing an effective amount of GG or to produce beverages containing hydrolyzed GG, which improves lipid and carbohydrate metabolism. Other polysaccharides or a combination of polysaccharides more effective than GG could be useful for the clinical treatment of hyperglycemia. The way in which viscous polysaccharides exert their hypocholesterolemic effects remains uncertain, but many studies have suggested that their high viscosity is an important factor in their potent activity. In rats and hamsters, high-viscosity GG has a strong hypocholesterolemic activity compared to its low-viscosity counterpart. Synergies observed with the rheological properties of XG and galactomannans mixtures are well-known in the agri-food industry. Gelation occurs by mixing two non-gelling gums XG and LBG. A mixture of XG and GG does not result in gel formation but increases viscosity to a value higher than expected by the simple addition of the two polysaccharide viscosities. The increased viscosity and gelling property obtained by mixing these polysaccharide combinations may be useful in inducing a powerful hypocholesterolemic effect. Only a few attempts have been made to study the hypocholesterolemic and hypoglycemic effects of highly viscous polysaccharide blends. 51 Studies have shown that soluble fibers can lower plasma cholesterol concentrations due to two possible causes. The first is that soluble fibers increase the viscosity of digesta and augment the thickness of the unagitated layer in the small intestine, which may inhibit the absorption of cholesterol and bile acids. The second is that soluble fibers are an excellent substrate for microbial fermentation in the large intestine, producing volatile fatty acids that seem to suppress hepatic cholesterol synthesis. 52 A comparison of three types of galactomannans (guar gum, locust bean gum, and fenugreek gum) showed that all three reduce plasma cholesterol compared to a control diet based on purified cellulose. Additionally, the chemical structure influenced the extent of the effect. Fenugreek seeds are a good source of soluble dietary fiber (SDF), and their consumption has been demonstrated to lead to a significant reduction in serum and hepatic cholesterol levels. The factors responsible for this effect have not been established so far, but it has been suggested that total dietary fiber (TDF) may have played a key role in reducing cholesterol levels by increasing the fecal excretion of bile acids and salts as well as inhibitors of hepatic cholesterol biosynthesis by short-chain fatty acids produced by bacterial fermentation of SDF in the lower parts of the large intestine.53,54
Antioxidant Activity
Numerous purified polysaccharides derived from plants are now employed as functional antioxidant ingredients and nutraceuticals in various foods and pharmaceutical products. They act as scavengers of free radicals to protect living organisms against oxidative damage. Antioxidant polysaccharides from plant sources have garnered particular admiration due to their abilities to trap free radicals, block lipid peroxidation, and halt the induction of neuronal damage. Evaluating the in vitro antioxidant activity of plant-based polysaccharides involves assessing various antioxidant mechanisms, such as excess reactive oxygen species (ROS) (O2, OH, H2O2, etc), dysfunction of the antioxidant system (enzymes, SOD, CAT, non-enzymes: GSH), and oxidative stress-related signaling pathways such as AKT, MAPK, etc This is achieved through methods such as DPPH radical scavenging activity, hydroxyl radical scavenging activity, superoxide radical scavenging activity, anti-lipid peroxidation assay, and β-carotene-linoleic acid assay. The practical applications of antioxidant plant-based polysaccharides have now become a demanding area of research in biomedicine for the treatment of conditions such as obesity, atherosclerosis, liver injuries, neurodegenerative diseases, colitis, gastric cancers, breast cancer, etc 54
Applications of Galactomannans
Industrial use of Galactomannans
Galactomannans find diverse applications across several industries. In the paper industry, they serve as deflocculating and fiber-strengthening agents. The addition of moderate amounts of galactomannans to pulp enhances paper production, and carboxymethylated gum acts as a wet additive for producing lightweight papers with excellent ink-sealing properties. In the textile sector, galactomannans serve as binding agents when combined with starch and as thickeners in paints to create strongly patterned designs with thickened dye solutions. In the petroleum industry, galactomannans are used in hydraulic fracturing to break rocks under high pressure. Fracturing fluids, thickened with gum solutions, carry sand into fractured rocks, facilitating the smooth flow of oil or gas to the well. Cosmetic industries utilize galactomannans in hair shampoos, lotions, ointments, and toothpaste due to their non-toxic properties, slip during extrusion, and stabilizing capabilities. Additionally, galactomannans are employed in the explosives industry for water blocking, gel formation, and swelling, ensuring explosive properties even in humid conditions. 41 Also, Galactomannans have diverse industrial applications, including textile printing, wastewater treatment, and nanofiber production via electrospinning. In textiles, they enhance dyeing and printing processes by improving color fixation and pattern quality. Their biodegradable nature makes them valuable in wastewater treatment, aiding in the removal of dyes and heavy metals from industrial effluents, thus reducing environmental pollution. Additionally, galactomannans are utilized in electrospinning to produce nanofibrous webs, particularly for biomedical applications such as drug delivery and wound healing. Their versatility and eco-friendly properties position them as promising alternatives to synthetic materials across various sectors. 8
Food use of Galactomannans
Galactomannans can be utilized alongside other nutritional ingredients and enzymes in gluten-free bakery product production. They enhance the volume and moisture content of bread, serving as a substitute for gluten due to their viscoelastic properties. The addition of gum solution to wheat flour suspension increases viscosity, maximum viscosity, final viscosity, and setback values while lowering the gelatinization temperature. Wheat flour also experiences an increase in dough development time and water absorption capacity with the addition of a gum solution. Galactomannans, in combination with starch, effectively prevent shrinkage, cracking, and dehydration of frozen pie fillings. Gum solutions are used as thickeners, stabilizers, and extend the shelf life of beverages over a wide pH range due to their stability. Galactomannans also possess surfactant properties, utilized as oil-in-water emulsion stabilizers. Furthermore, in the production of stuffed meat products and sausages, galactomannans exhibit binding and lubricating capacities, binding meat pieces, preventing phase separation during heat treatments, and enhancing yield through water management. 41 Guar and carob gums are commonly used in ice cream formulation to regulate ice crystal formation and recrystallization and prevent lactose crystallization during frozen storage. They are also employed in other dairy products, such as spreadable cheeses, to enhance spreadability or yogurts for producing low-fat products with improved texture and stability. Carob galactomannans are extensively used in the food industry and have been classified by the European Codex as an approved human food additive (E410). Studies have also been conducted to enhance the textural properties of light and skimmed yogurts by incorporating galactomannans. 55 Galactomannans have also found applications as edible films due to their physical, chemical, and thermomechanical properties, such as moisture permeability and gas exchange (CO2 and O2) occurring in these materials. 56 Either, galactomannans are extensively utilized in the food industry for their functional properties, serving as binders, emulsifiers, stabilizers, thickeners, and fat replacers. Beyond their technological applications, they offer significant health benefits, including blood sugar regulation, improved gut health, cholesterol reduction, and potential roles in preventing constipation, diabetes, obesity, and cancer. 7
Pharmaceutical use of Galactomannans
Galactomannans have a long history as excipients. Carob gum (ceratonia) was the first galactomannan introduced in 1936 as a cost-effective substitute for tragacanth gum and was quickly incorporated into various formulations for purposes like suspending excipient, thickening, and stabilizing in oral and topical liquid dosage forms, especially emulsions. In 1965, Merck Laboratories filed the first patent on the application of galactomannans in the pharmaceutical industry. This particular invention used galactomannans as binders to eliminate the granulation step before compression in tablet preparation. In 1969, Merck patented the first solid dosage form of oral vitamin compounds stabilized by preparing solutions of guar gum and carob gum. Although their incorporation was late, guar gum is one of the most commonly used galactomannans in the pharmaceutical industry, likely due to its regulatory status. Over the evolution of pharmaceutical literature on galactomannans, it can be established that while their use started early, it was only from the 1990s that interest in these materials substantially increased. 40 Galactomannans are mainly used in pharmaceutical and cosmetic technologies, in the form of crude flour rather than in pure form. Commercial flours contain approximately 85% pure galactomannan by mass and are widely used in many products. Galactomannans are often used in combination with other polysaccharides, such as xanthan gum, agar, and kappa-carrageenan, to form gels with novel properties. In recent years, the pharmaceutical use of galactomannans from various sources, both commercial and non-commercial, has been extensively studied. Galactomannans offer potential in the global trend toward using plant-based products for ecological reasons, and their production and application cause no pollution or disruption to the ecosystem. There is a variety of galactomannan sources and various pharmaceutical forms of application, such as tablets or capsules, hydrogels, and films. Besides their use as inert excipients, these polysaccharides also play a role in modifying drug release, particularly in the colonic environment, as a matrix or coating material. Galactomannans can, therefore, be used to develop extended or targeted drug release formulations. 57 Galactomannans from various plant sources, including guar gum, locust bean gum, fenugreek gum, tara gum, Delonix regia, and Leucaena leucocephala, have been extensively studied for their potential in nanocarrier development, particularly in drug delivery. Nanoprecipitation and crosslinking methods were commonly used for nanoparticle fabrication, while modified derivatives played a crucial role in green nanoparticle synthesis. These nanosystems have shown promise in oral immunization, cancer treatment, heavy metal removal, and antimicrobial applications. Additionally, structural variations in galactomannans influence their functionality, emphasizing the need for renewable sources and further pharmaceutical evaluation. Their potential as sustainable alternatives to synthetic polymers highlights their significance in biomedical and industrial applications, warranting further clinical studies. 58
The Table 2 provides an overview of various Galactomannans, including their physicochemical properties, biosynthesis, biological activities, and applications, along with relevant citations.
Overview of Galactomannans: Physicochemical Properties, Biosynthesis, Biological Activities, and Applications.

Conclusion
In conclusion, this comprehensive review highlights the multifaceted applications and diverse functionalities of galactomannans across various industries, emphasizing their significance in industrial, food, and pharmaceutical domains. The versatility of galactomannans, derived from plant sources, positions them as indispensable components in sectors ranging from paper and textile to petroleum and cosmetics. Their unique properties, such as binding, thickening, stabilizing, and water-blocking capabilities, contribute to their widespread utilization. In the food industry, galactomannans play a crucial role in enhancing the quality of bakery products, dairy items, and meat-based products, showcasing their adaptability as thickeners, stabilizers, and texture improvers. Moreover, the incorporation of galactomannans in edible films reflects their potential in addressing contemporary challenges in food packaging and preservation. The pharmaceutical industry benefits from the versatility of galactomannans, primarily in the form of excipients. From an industrial standpoint, galactomannans exhibit their efficacy in processes such as paper production, hydraulic fracturing in the petroleum sector, and the formulation of cosmetic products. Their role in enhancing the properties of explosives further underscores their versatility in varied industrial applications. In essence, this review underscores the paramount importance of galactomannans in contemporary industries, reflecting their adaptability, biocompatibility, and eco-friendly nature. As research continues to unravel their potential, galactomannans stand poised to play an increasingly vital role in addressing the dynamic challenges faced by diverse sectors, offering sustainable solutions for the future.
Footnotes
Acknowledgments
We express our sincere gratitude to everyone who contributed to the completion of this work, whether through research, analysis, or reviewing sources. We also extend our appreciation to previous researchers whose work formed the foundation of this review. Additionally, we thank all those who provided constructive feedback that helped improve this paper.
Ethical Considerations
This study is not applicable to both human and/oranimal studies that require ethical approval.
Author Contributions
Aicha Tedjani: Conception and design, methodology development, data acquisition, analysis and data interpretation, english revision
Aicha Mouane : conception revision; Manel Azzi: conception revision; Zakaria Boual : study supervision; Ayomide Victor Atoki: conception revision; Mohammed Messaoudi : conception revision. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that supports the findings of this study are available within the article.
