Abstract
The antihypertensive effect of Kolaviron (KV) has been demonstrated in ethanol- and sucrose-model of hypertension in our previous studies. However, there remains a need to further confirm and validate this effect in other models of hypertension. This study was devised to appraise the antihypertensive action of KV in N(G)-nitro-L-arginine methyl ester (L-NAME) induced experimental format of hypertension. Thirty-six (36) male Wistar rats were divided into 6 groups of 6 animals each. Group I represents the control group while Group II animals received L-NAME 40 mg/kg only. Groups III to V animals received L-NAME 40 mg/kg and 50, 100, and 200 mg/kg KV, respectively. Group VI animals received L-NAME 40 mg/kg and 0.14 mg/kg Lisinopril (LIS). Treatment was done orally for 28 days after which blood pressure was determined via the invasive method. After this, vital organs and blood were retrieved for analysis. KV and LIS elicited significant contraction (P < .01-.001) in blood pressure producing up to 27%, 24%, and 22% reduction in systolic blood pressure, diastolic blood pressure, and mean arterial pressure, respectively. In addition, KV elicited a notable rise (P < .05-.001) in catalase, reduced glutathione, and superoxide dismutase in the blood and vital organs. Results from this study further demonstrate and confirm that KV possesses notable blood pressure-lowering effect possibly through its well-documented antioxidant effect. Additional studies are advocated to validate the results from this study and determine the precise mechanism for the antihypertensive action of KV.
Introduction
Hypertension (HTN) remains a huge global public health concern owing to the large contribution it makes to the total disease burden of the world.1–3 This large contribution of HTN to the disease burden of the world may be attributed to a large number of debilitating diseases and complications often associated with it. HTN has been identified as the most frequent, potent, and principal avoidable risk factor for cardiovascular disease (CVDs).4–6
Additionally, HTN is strongly, independently, and linearly correlated with the danger of chronic kidney disease (CKD), CVDs, and all-cause mortality. 2 This means that by effectively reducing or managing elevated blood pressure (BP), the risk of mortality or morbidity associated with most CVDs can be reduced. However, the prevalence of HTN continues to rise especially in economically developing countries (EDCs) with the effective control of BP remaining abysmally low. 2
The prevalence of HTN in EDCs currently stands at 31.5% versus 28.5% in high-income countries. In Africa, the prevalence of HTN is as high as 46%. 7 This high prevalence of HTN in EDCs and the African continent is a silent cause for alarm partly due to the sometimes unavailable or broken health systems. 2 This is further compounded by the intolerable side effects and huge financial burden of western pharmacotherapeutic medications on patients.2,8 It is therefore important that cost-effective but equally therapeutic alternatives for BP control is available for patients.
Studies have shown that there is an inverted correlation between the risk of cardiometabolic events and the consumption of flavonoids.9,10 Furthermore, Maaliki et al 9 reported that increased flavonoid consumption has antihypertensive benefits, exerts cardiovasuloprotective effects, and reduces the progression of CVDs.
Kolaflavanone, Kolafavone, and Garcinia bioflavonoids I & II are flavonoids present in Kolaviron which is an extract of Garcinia kola11–13 Kolaviron (KV) is the defatted ethanol extract of Garcinia kola seeds which is widely available in the Southern part of Nigeria. 14 Proven pharmacological effects of KV include its anti-inflammatory and hepatoprotective,15–17 analgesic, 17 antioxidant,18–21 hypoglycaemic,22,23 anti-ulcer, 24 anti-malarial, 25 anti-viral, 12 cholesterol lowering,26–28 alpha-glucosidase inhibitory, 29 aromatase inhibitory, 29 antiplasmodial, 29 and antihypertensive26,27 effects. KV has also been proven to be safe. 30
While the antihypertensive effect of KV has been demonstrated in ethanol- and sucrose-induced models of HTN from our previous study, 26 there remains a need to further confirm this effect in other models of HTN with different mechanisms of induction of HTN. The effectiveness of KV to ameliorate HTN in different models could give an indication of the likely mechanism through which it exerts its antihypertensive effect.
This research was therefore designed to evaluate the BP lowering effect of Kolaviron in N[G]-nitro-L-arginine methyl ester (L-NAME) model of HTN. Experimental induction of HTN with L-NAME is a well-established and accepted model of HTN. 31
Experimental Results and Discussion
Effect of KV on Systolic BP (SBP), Diastolic BP (DBP), Mean Arterial Pressure (MAP), Heart Rate (HR), Pulse Pressure (PP), and Rate Pressure Product (RPP)
The administration of L-NAME produced significant elevation (P < .01-.001) of SBP, DBP, MAP, and RPP in test animals. The concurrent administration of L-NAME and 100 mg/kg KV produced a notable decrease (P < .01-.001) in SBP (27.40% reduction), DBP (23.89% reduction), and MAP (22.06% reduction), respectively. Similar significant results were obtained with 200 mg/kg KV which achieved a 23.70% reduction in SBP, 18.78% reduction in DBP, and 19.72% reduction in MAP, respectively. Additionally, significant BP reduction (P < .05-.001) was achieved with the concurrent administration of L-NAME and 0.14 mg/kg Lisinopril (LIS) (Table 1; Figures 1 to 6).
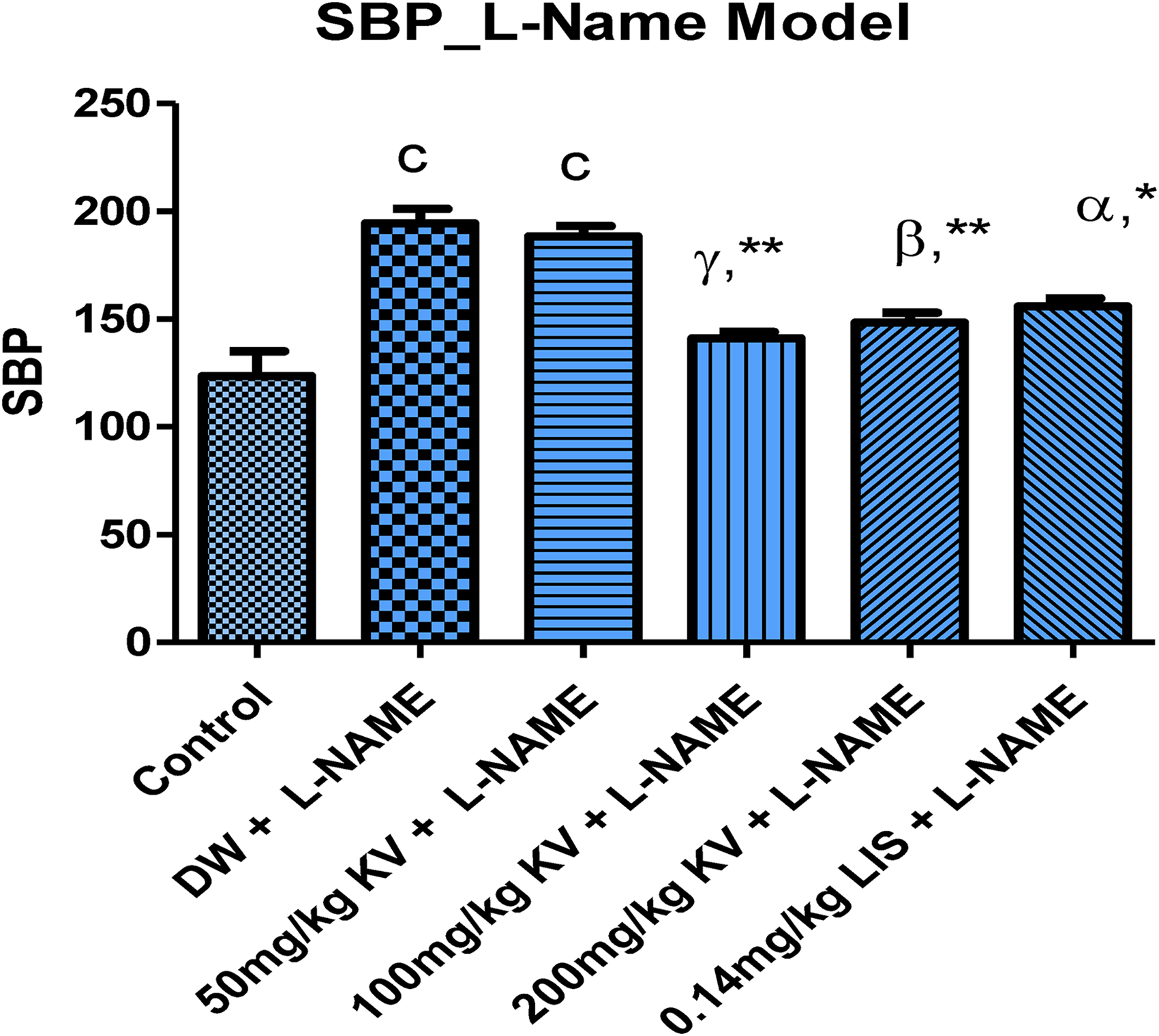
Systolic blood pressure. aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P <.01, ***P < .001 versus KV 50 mg/kg + L-NAME. Abbreviations: LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester; SBP, systolic blood pressure.

Diastolic blood pressure. aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01, ***P < .001 versus KV 50 mg/kg + L-NAME. Abbreviations: LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester; DBP, diastolic blood pressure.

Mean arterial blood pressure. aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01, ***P < .001 versus KV 50 mg/kg + L-NAME. Abbreviations: LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester; MAP, mean arterial pressure.
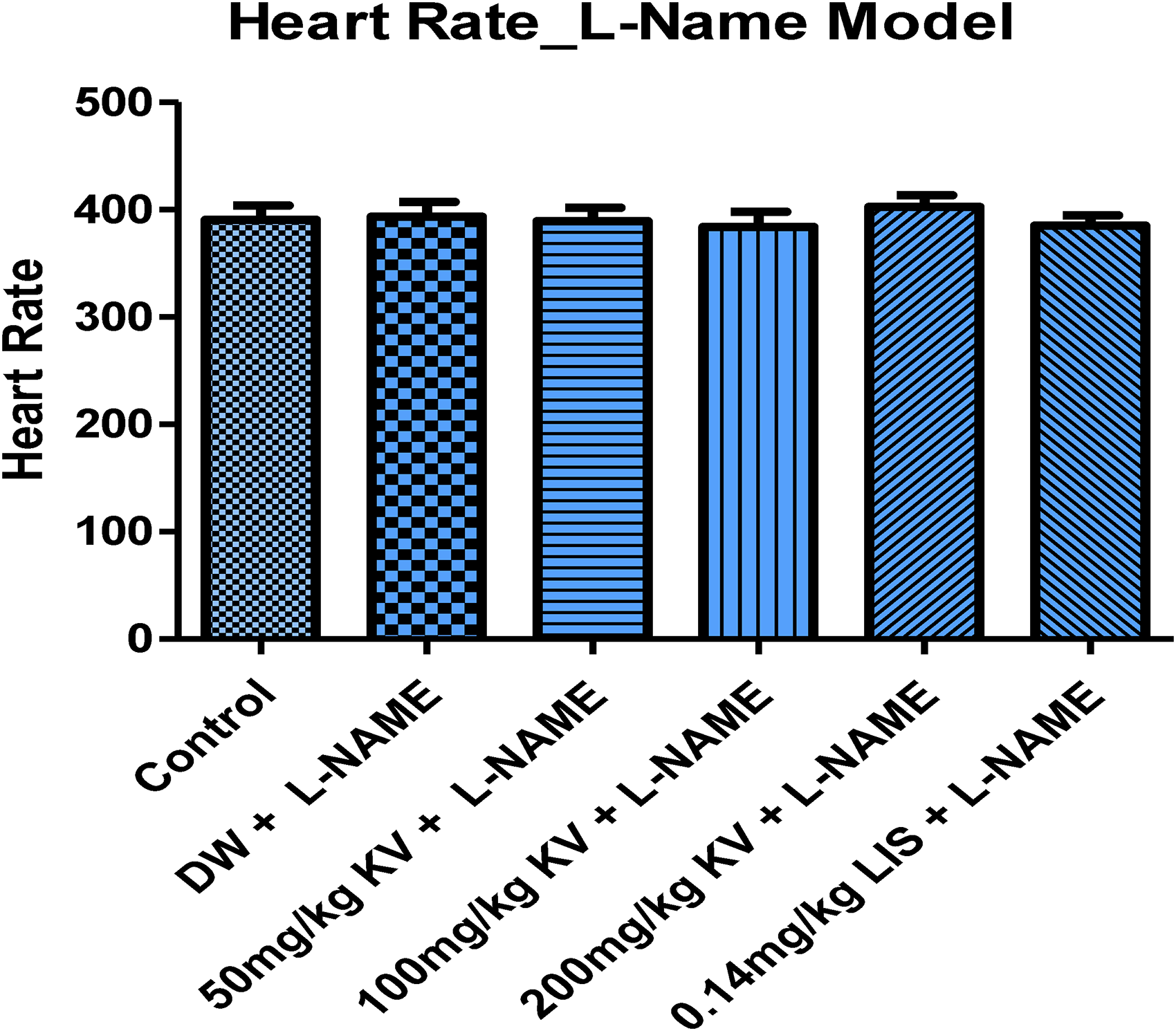
Heart rate. aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01, ***P < .001 versus KV 50 mg/kg + L-NAME. Abbreviations: LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester; HR, heart rate.

Pulse rate. aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01, ***P < .001 versus KV 50 mg/kg + L-NAME. Abbreviations: LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester.
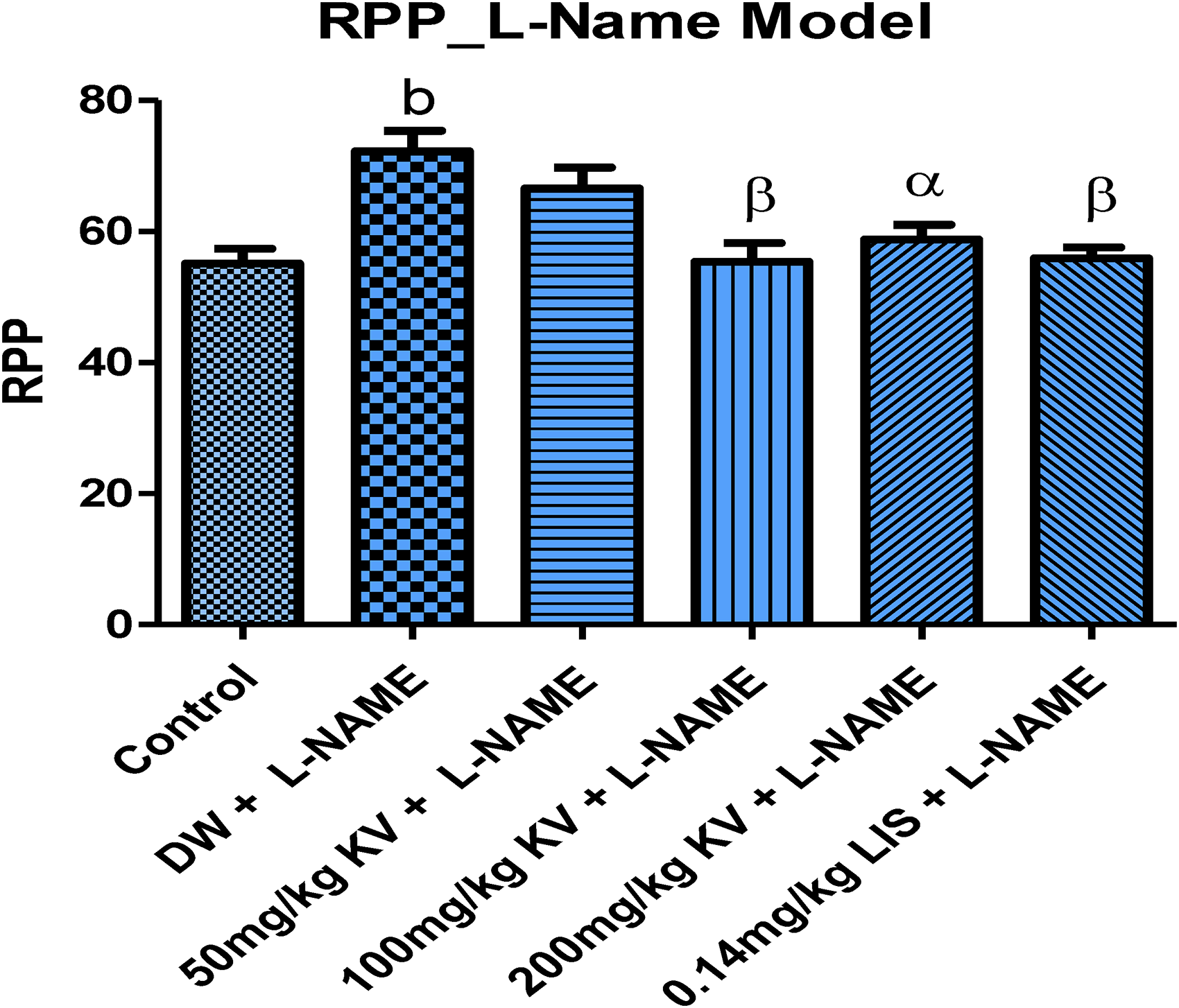
Rate pressure product. aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01, ***P < .001 versus KV 50 mg/kg + L-NAME. Abbreviations: LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester; RPP, rate pressure product.
Effect of Kolaviron on Blood Pressure Parameters in L-NAME Hypertension Model in Rats.
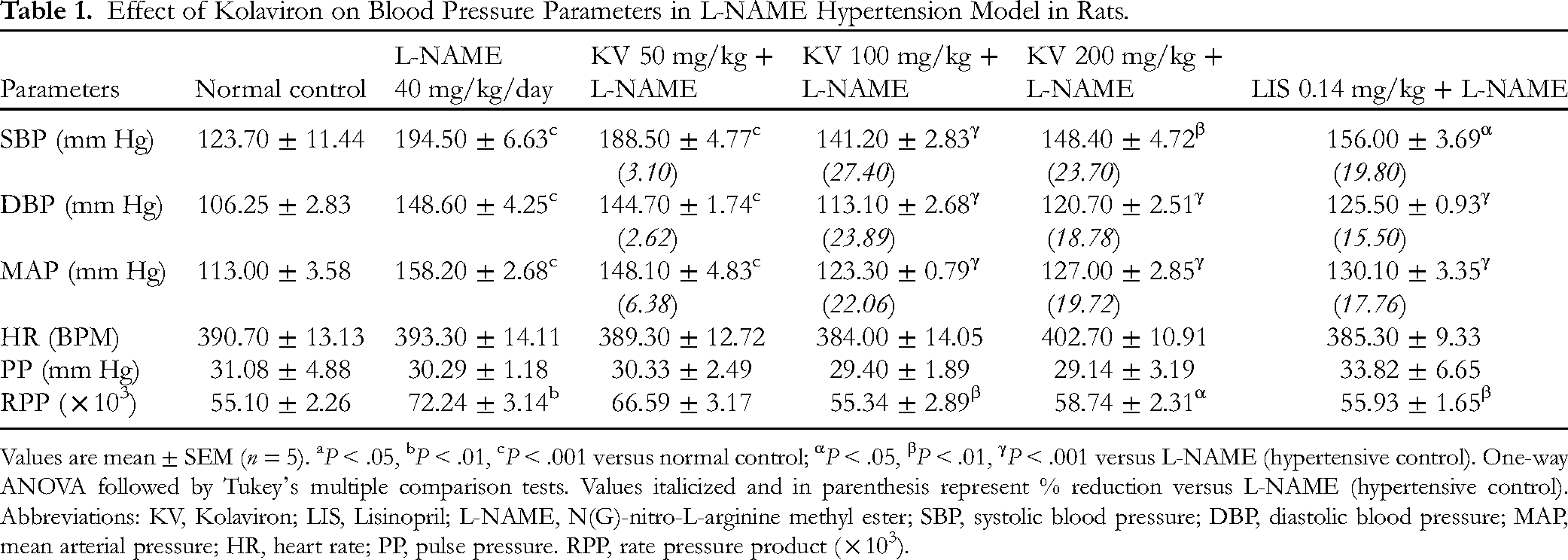
Values are mean ± SEM (n = 5). aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control). One-way ANOVA followed by Tukey's multiple comparison tests. Values italicized and in parenthesis represent % reduction versus L-NAME (hypertensive control). Abbreviations: KV, Kolaviron; LIS, Lisinopril; L-NAME, N(G)-nitro-L-arginine methyl ester; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; HR, heart rate; PP, pulse pressure. RPP, rate pressure product ( × 103).
L-NAME and its concurrent administration with KV or LIS did not elicit any significant change in heart rate and pulse pressure across all groups. However, concurrent administration of 100 and 200 mg/kg KV and 0.14 mg/kg LIS resulted in a significant reduction (P < .05-.001) in RPP for groups IV to VI. RPP is a measure of the heart rate and the SBP the heart is pumping against (Table 1).
Effect of KV on Antioxidant Parameters in Blood and Vital Organs
In the blood, KV produced a notable increase in superoxide dismutase (SOD), reduced glutathione (GSH), and catalase (CAT) versus the control group, thereby reversing the reduction of the antioxidant indices by L-NAME. A similar effect was observed with LIS. There was a nonsignificant change (reduction) in the lipid peroxidation parameter, malondialdehyde (MDA) in the blood.
In the aorta, liver, and kidney tissues, KV and LIS significantly elevated the levels of SOD, CAT, and GSH (P < .05-.001), thereby improving antioxidant indices lowered by L-NAME. MDA levels in the liver were significantly reduced (P< .05-.01) by KV and LIS in the liver tissues. Additionally, there was a nonsignificant change in the levels of GSH, SOD, CAT, and MDA in the heart tissues (Tables 2 to 4).
Effect of Kolaviron on Blood Level GSH, SOD, CAT, and MDA in L-NAME Hypertension Model in Rats.

Values are mean ± SEM (n = 5). aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control). One-way ANOVA followed by Tukey's multiple comparison tests. Abbreviations: KV, Kolaviron; LIS, Lisinopril; GSH, reduced glutathione; SOD, superoxide dismutase; CAT, catalase; MDA, malondialdehyde.
Effect of Kolaviron on Aorta and Heart Levels of GSH, SOD, CAT, and MDA in L-NAME Hypertension Model in Rats.
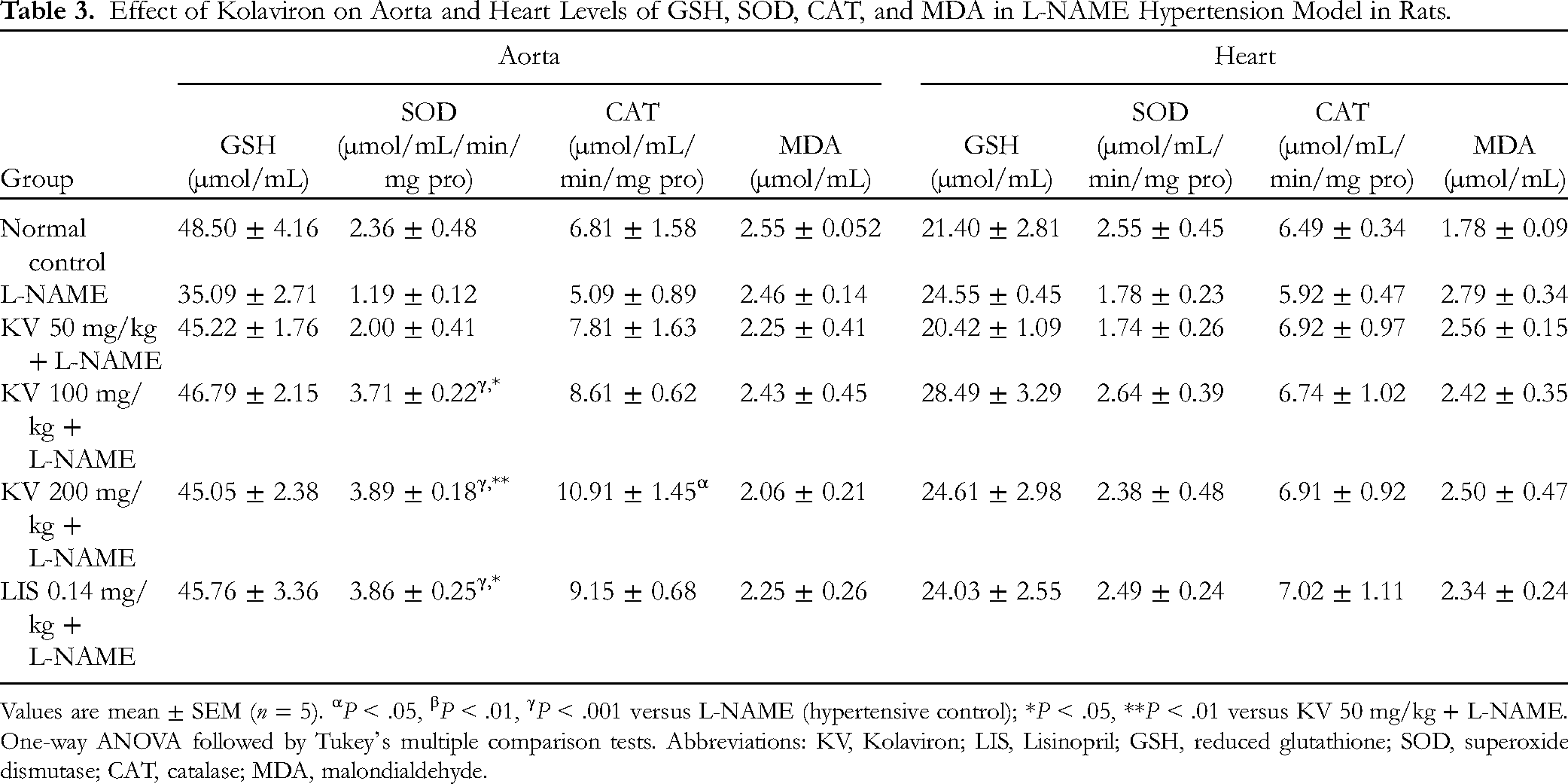
Values are mean ± SEM (n = 5). αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01 versus KV 50 mg/kg + L-NAME. One-way ANOVA followed by Tukey's multiple comparison tests. Abbreviations: KV, Kolaviron; LIS, Lisinopril; GSH, reduced glutathione; SOD, superoxide dismutase; CAT, catalase; MDA, malondialdehyde.
Effect of Kolaviron on Liver and Kidney Levels of GSH, SOD, CAT, and MDA in L-NAME Hypertension Model in Rats.

Values are mean ± SEM (n = 5). aP < .05, bP < .01, cP < .001 versus normal control; αP < .05, βP < .01, γP < .001 versus L-NAME (hypertensive control); *P < .05, **P < .01, ***P < .001 versus KV 50 mg/kg + L-NAME. One-way ANOVA followed by Tukey's multiple comparison tests. Abbreviations: KV, Kolaviron; LIS, Lisinopril; GSH, reduced glutathione; SOD, superoxide dismutase; CAT, catalase; MDA, malondialdehyde.
Effect of KV on Biochemical Parameters
In this study, L-NAME, KV, or LIS did not produce any significant change in all serum biochemical parameters assessed (Table 5).
Effect of Kolaviron on Serum Biochemical Parameters in L-NAME Hypertension Model in Rats.

Values are mean ± SEM (n = 5). P > .05 versus normal control and hypertensive control. One-way ANOVA followed by Tukey's multiple comparison tests. Abbreviations: KV, Kolaviron; LIS, Lisinopril; CHOL, cholesterol; HDL, high-density lipoprotein; TRIG, triglycerides; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; ALB, albumin; CREAT, creatinine.
Effect of KV on Hematological Parameters
There was no significant change in all the hematological parameters assessed in this study except for a significant elevation (P < .05) in MID# (other types of WBC that are not classified as lymphocytes or granulocytes) produced by L-NAME in the hypertensive control group. This effect was, however, reversed by KV 100 mg/kg as evidenced by a significant decline in MID# (P< .05) (Table 6).
Effect of Kolaviron on Hematological Parameters in L-NAME Hypertension Model in Rats.

Values are mean ± SEM (n = 5). aP < .05 versus normal control; αP < .05 versus L-NAME (hypertensive control). One-way ANOVA followed by Tukey's multiple comparison tests. Abbreviations: KV, Kolaviron; LIS, Lisinopril; WBC, white blood cells; LYM, lymphocytes; MID, other types of WBC that are not classified as lymphocytes or granulocytes; GRANL, granulocytes; MID%: other types of WBC differentials; LYM%, lymphocytes differentials; GRAN%, granulocytes differential; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; PLT, platelet; RDW, red cell distribution width; MPV, mean platelet volume; PDW, platelet distribution width; PCT, plateletcrit.
Discussion
While the ameliorative and antihypertensive effect of KV has been evaluated in ethanol- and sucrose-model of HTN, this current study seeks to confirm the antihypertensive action of KV through a different model of HTN. Based on our literature review, it seems that there are still insufficient studies confirming the antihypertensive effect of KV in experimental models. In this study, we examined the antihypertensive action of KV in the L-NAME induced experimental model of HTN in rats.
L-NAME elicited a significant elevation in SDP, DBP, and MAP leading to HTN in test animals and producing BP above baseline in nonhypertensive animals (control group). The chronic treatment of test animals with L-NAME for 4 weeks (28 days) is a well-established and well-known model of experimental HTN.31–34 The results obtained in this study were consistent with literature.
The concurrent administration of L-NAME with KV significantly reversed the elevation of BP elicited by L-NAME, producing up to 24%, 27%, and 22% reduction in DBP, SBP, and MAP, respectively, in experimental animals. These results are consistent with literature in which KV elicited a marked reduction in BP which was elevated in the sucrose- and ethanol-induced models of HTN. 26 Additionally, KV elicited a significant reduction in rate pressure product (RPP), a measure of the heart rate, and the SBP the heart is pumping against. This further demonstrates the ability of KV to reduce SBP in test animals.
KV's antioxidant ability is well-researched and documented in literature. There are several pharmacological effects of KV documented in literature which are associated with its antioxidant capacity and ability to reduce oxidative stress. Our findings in this study are consistent with literature in which KV significantly elevated antioxidant parameters (SOD, GSH, and CAT) in the blood, liver, aorta, and kidney of test animals. Additionally, KV reduced the lipid peroxidation biomarker, malondialdehyde, in the liver of test animals consistent with literature.
Chronic administration of L-NAME is documented to induce HTN through its ability to block the L-arginine – nitric oxide (NO) pathway, resulting in less production of NO, NO deficiency and reduced bioavailability of NO.31,32 L-NAME does this by chronically blocking nitric oxide synthase (NOS), an enzyme that catalyzes the L-arginine – NO pathway. This pathway plays an important role in the physiological regulation of BP and is a well-established regulator of the cardiovascular system31,35–37 Besides this, induction of HTN through the chronic administration of L-NAME involves other mechanisms, such as sympathetic nervous system,38,39 the renin-angiotensin-aldosterone system,40,41 and the oxidative stress system.31,41,42 These mechanisms are similar to the mechanisms through which ethanol and sucrose from our previous study seem to have induced HTN.
The chronic ingestion of ethanol is known to lead to HTN through an increase in the activation of the sympathetic nervous system accompanied by the release of sympathetic amines which leads to stimulation of oxidative reactions, vasoconstriction, and an increase in heart rate.43–45 Literature suggests that active endogenous vasoconstrictor agents, such as angiotensin II, noradrenaline, endothelin-1, as well as nitric oxide, a very active vasodilator, play a crucial role in ethanol-induced experimental HTN. 46 Similarly, ingestion of sucrose is associated with an elevation of the peripheral sympathetic nervous system activity, hyperinsulinemia, and increased stickiness of blood platelets with a potential to lead to thrombosis.47,48 Furthermore, chronic ingestion of ethanol and sucrose is associated with an increase in reactive oxygen species (ROS) and oxidation; both of which play a crucial part in the development and onset of arterial HTN. 49
Based on all of this, it is, therefore, possible that KV is able to ameliorate elevated BP caused by L-NAME in a way similar to its effect on ethanol- and sucrose-model of HTNs as reported in our previous study. This involves the possible mitigatory effect of KV on oxidative stress, NO bioavailability, and attenuation of angiotensin-II-induced vascular smooth muscle cells (VSMC) proliferation.
The involvement of ROS, NO and Angiotensin II in the etiology and development of HTN is well documented in literature. 26 In a hypertensive state, there is heightened oxidative stress, leading to lower bioavailability of NO which leads to constriction of VSMC. This eventually leads to elevated BP. By virtue of its antioxidant capacity, KV is able to reduce the oxidative stress associated with the hypertensive state and possibly increase the bioavailability of NO in the endothelium and VSMC. The increased availability of NO, therefore, leads to vasodilation of the VSMC and thus, BP is reduced.
KV has been reported to attenuate Angiotensin II-induced VSMC proliferation and constriction 50 which may possibly lead to a reduction in BP in a hypertensive state. Additionally, KV has been reported to reduce the activity of the Angiotensin converting enzyme and improve the levels of endothelial NOS (eNOS). 27 eNOS is the enzyme required for the production of NO. An increase in the level of eNOS will therefore lead to an increase in the production and bioavailability of NO. This consequently leads to vasodilation of the VSMC. Furthermore, KV has been documented to elicit relaxation of smooth muscle in the blood vessels of the aorta of rabbits. 51
Based on all of this, it is possible that KV elicits its antihypertensive effect through its documented antioxidant effects, its documented ability to improve lipid profile, its ability to improve NO production and finally its ability to attenuate VSMC proliferation and constriction. All of these consequently lead to vasodilation and reduction in BP. Additionally, many flavonoids including those obtained from Kolaviron have been shown to exhibit both cardioprotective and antihypertensive actions that prevent endothelial dysfunction.9,10
It is important to state that while the lipid profile improving the ability of KV was reported in our previous studies,26,30 KV only improved lipid profiles nonsignificantly in this study. This may, however, be connected with the duration of treatment of KV in this present study. In this study, KV was administered for 4 weeks based on the documented duration for induction of HTN using L-NAME. However, in our previous studies, KV was administered for 8 and 12 weeks, respectively.
Conclusion, Limitation of Study, and Future Direction
In this study, we further confirmed the antihypertensive effect of KV through the L-NAME model of HTN. It can thus be said that KV has notable antihypertensive action, which may possibly be due to its antioxidant improvement capacity, its lipid profile improvement ability, and its ability to enhance the production of NO. Additional studies are recommended to validate the findings of this study and determine the precise mechanism of the antihypertensive action of KV.
One limitation of this study is that NO and NOS levels were not evaluated. We recommend that further studies should evaluate and measure NO and NOS levels in the test animals.
Studies show that difference exists between men and women in terms of the awareness, prevalence, control, and treatment of HTN.52,53 Men are known to have a higher incidence of HTN than women. 54 Potential future research is recommended to evaluate the gender-based response to Kolaviron in a hypertensive state. Indeed, gender differences to Kolaviron have been demonstrated by Olatoye and Akindele. 30
Experimental Materials and Method
Approval by Local Ethics Committee
The Health Research Ethics Committee, College of Medicine, University of Lagos approved the experimental procedures for this study (Approval number: CMUL/HREC/10/19/656). Experimental procedures utilized in this study followed the provisos of the US National Academy of Sciences Guide for the Care and Use of Laboratory Animals. 55
Drugs and Chemicals
Ethyl acetate, acetone, petroleum ether (BDH Lab Supplies), Normal saline (Albert David Limited), Lisinopril (AstraZeneca), and L-NAME (Sigma-Aldrich). Heparin (Pfizer), Urethane, chloralose (Sarex Chemicals), and Formalin (BDH Lab Supplies).
Plant Material Extraction and Drug Preparation
Freshly obtained Garcinia kola seeds were cut, dried, ground to a semipowdery state (6.2 g), and then extracted by the adjusted method of Iwu 18 to yield KV. The identity of Garcinia kola was authenticated in the Department of Botany, University of Lagos, Nigeria where a voucher specimen (LU H 3688) already exists in the University Herbarium.
First, the semipowdered seeds were extracted for 24 h with light petroleum ether (b.p. 40 °C-60 °C) in a Soxhlet apparatus. Next, the marc obtained was dried and extracted with methanol. Lastly, the extract obtained from this was concentrated, diluted to 200% of its volume, and extracted with ethyl acetate. This produced a dark golden yellow to brown semisolid with a yield of 6%. This semisolid is known as Kolaviron. Kolaviron was identified based on its physical and phytochemical properties as established in literature. This includes a golden yellow to brown color, a powdery crystalline structure, low to no solubility in water, and phytochemical constituents of tannins, cardiac glycosides, saponins, flavonoids, phenols, alkaloids, and carbohydrates.11-14,56-58.
Several working concentrations of Kolaviron (50, 100, and 200 mg/kg) were constituted using an equal mixture of propyl glycol and distilled water. Suitable liquid preparations of LIS were made to achieve a 0.14 mg/kg dose of LIS based on a 10 mg daily LIS dose. 59 L-NAME was dissolved in distilled water to achieve a 40 mg/kg working concentration. 31
Experimental Design and Treatment
Thirty-six (36) Wistar male rats with weights within a range of 150 to 180g were obtained from the College of Medicine's animal house, University of Lagos, Nigeria. The test animals were allowed to acclimatize under normal feeding, drinking, and standard conditions of 23 °C to 25 °C, 12-h light/12-h dark cycle. Test animals were then divided into 6 groups consisting of 6 animals each.
Group I was designated as the control group and received a 50–50 mixture of propyl glycol and distilled water. Group II animals (hypertensive control) were given only L-NAME 40 mg/kg. Group III animals were given only 50 mg/kg KV and L-NAME 40 mg/kg. Group IV animals were given 100 mg/kg KV and L-NAME 40 mg/kg. Group V animals were given 200 mg/kg KV and L-NAME 40 mg/kg. Lastly, Group VI animals received 0.14 mg/kg LIS and L-NAME 40 mg/kg. Treatment was done via oral gavage for 4 weeks 34 after which various BP parameters were measured and blood and organ were collected for analysis.
BP was determined using the invasive method 60 and the DBP, SDP, MAP, HR, PP, as well as RPP values, were recorded. After this, the animals were humanely sacrificed and about 5 mL of blood was collected via cardiac puncture for biochemical and hematological assessment.
Experimental Analysis
Biochemical Parameters
Sample blood collected was analyzed for levels of cholesterol (CHOL), high-density lipoprotein (HDL), triglycerides (TG), aspartate aminotransaminase (AST), alanine aminotransaminase (ALT), alkaline phosphate (ALP), urea, creatinine, and albumin using commercial kits produced by Roche and Cobas and an automated analyzer (Roche Diagnostics).
Hematological Parameters
Sample blood collected was used to estimate the levels of leucocytes (WBC), lymphocytes (LYM), differential from other white blood cells (MID), granulocytes (GRAN), lymphocyte differentials (LYM%), differential from other white blood cells (MID%), granulocyte differentials (GRAN%), hemoglobin (HGB), erythrocyte (RBC), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), platelet distribution width (PDW), mean corpuscular hemoglobin (MCH), red cell distribution width (RDW), mean platelet volume (MPV), hematocrit (HCT), platelet count (PLT), and plateletcrit (PCT) using automated hematology analyzer (Sysmex Haematology-Coagulation Systems, Model KX-21N, Sysmex Incorporation).
Estimation of Serum and Tissue In Vivo Antioxidants and MDA Levels
Malondialdehyde (MDA), catalase (CAT), SOD, and reduced glutathione (GSH) levels in harvested tissues (aorta, heart, liver, and kidney) and blood were estimated using established protocols as reported by Soon and Tan. 61
Data Presentation and Analysis
Results from this study are represented as mean ± SEM. Data analysis was conducted using GraphPad Prism 5 software (GraphPad Software Inc.). One-way ANOVA followed by Tukey's multiple comparison tests was conducted for each set of results. Statistical significance was represented by P-value < .05.
Footnotes
Acknowledgements
We acknowledge the support of Dr Khadijah Ismail-Badmus of the Department of Physiology, College of Medicine, University of Lagos, Lagos, Nigeria in the determination of BP measurement using the invasive methodology.
Author Contribution Statement
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
Not applicable.
Informed Consent
Not applicable.
