Abstract
Recently, additional therapeutic potentials of classical antibiotics are gaining considerable attention. The discovery of penicillin in the 1920s had a major impact on the history of human health. Penicillin has been used for the treatment for fatal microbial infections in humans and has led to the discovery of several new antibiotics.
Inflammation is a protective mechanism of host defense in response to invading pathogens or tissue damage. 1 When the body is infected by pathogens, the macrophages remove the pathogens through phagocytosis and produce various inflammatory mediators such as nitric oxide (NO), prostaglandin E2 (PGE2), tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-6 to recruit other immune cells. 2,3 Although these inflammatory processes are important to maintain homeostasis in vivo, prolonged or excessive inflammation induces pathogenesis of diseases such as asthma, atopic dermatitis, atherosclerosis, and inflammatory bowel disease. 4 Therefore, it is necessary to control the level of inflammation. Lipopolysaccharide (LPS) is a fundamental component of the outer membrane of gram-negative bacteria that acts as an endotoxin. 5 Macrophages are activated by recognizing the pathogen-associated molecular pattern of LPS through pattern recognition receptors on the cell surface. 6 Activated macrophages are characterized by the production of several inflammatory mediators such as NO and PGE2, and pro-inflammatory cytokines including TNF-α, IL-1β, and IL-6. 7 The released pro-inflammatory molecules are regulated by inducible gene expression that is mediated by the activation of nuclear factor kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathway. 8,9 Nuclear factor kappa B is a transcription factor that exists in the form of homodimers or heterodimers. 10 In the unstimulated state, NF-κB is located in the cytoplasm bound by the inhibitory protein kappa B (IκB). 11 Inhibitory protein kappa B is phosphorylated by the MAPK and phosphoinositide 3-kinase/Akt (PI3K/Akt) pathways. 12 Phosphorylation of IκB triggers its ubiquitination and subsequent degradation by the proteasome. The free NF-κB dimers rapidly translocate from the cytoplasm to nucleus and bind to the promoter of responsive genes and activate the transcription of inducible target genes including inducible nitric oxide synthase (iNOS), cyclooxygenase type-2 (COX-2), and pro-inflammatory mediators. 13 Mitogen-activated protein kinases are well-conserved serine/threonine kinases that play an important role in cellular activities such as gene expression, mitosis, differentiation, and cell survival/apoptosis. 14 The MAPK family is composed of 3 protein kinases including extracellular signal-regulated kinase 1/2 (ERK 1/2), c-jun N-terminal kinase 1/2 (JNK 1/2), and p38. 15 Mitogen-activated protein kinases are activated by phosphorylation and induce activation of NF-κB and activator protein-1 (AP-1) that modulate gene expression. 16,17 Thus, NF-kB and MAPK signaling pathways are regarded as major targets for anti-inflammatory effect.
We first evaluated the cytotoxicity of DCS in RAW 264.7 macrophages. The cells were treated with various concentrations (2, 4, 6, 8, and 10 mM) of DCS for 24 hours (Figure 1). Cell viability was assessed by measuring 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) formazan crystals which are converted by tetrazolium salt via mitochondrial dehydrogenase in RAW264.7 cells.
20

(a) The chemical structure of
NO is a molecular messenger that plays an important role in cellular signaling in vivo.
21
NO molecules are produced by several types of nitric oxide synthases (NOSs), including neuronal NOS (nNOS), endothelial NOS (eNOS), cytokine-iNOS, and bacterial NOS (bNOS), and are involved in various physiological functions such as neurotransmission, vasodilation, oxidative stress, antibacterial activity, and immune defense against pathogens.
22
In inflammatory responses to pathogenic stimuli, iNOS induces the overproduction of NO.
23,24
In previous studies, it has been reported that the excessive NO production leads to inflammatory and autoimmune diseases.
25
Therefore, downregulating NO production and iNOS expression may be an appropriate strategy for anti-inflammatory therapies. To investigate whether DCS could inhibit NO production in LPS-stimulated RAW264.7 cells, we measured NO production in the form of nitrite in the cell culture media using the Griess reagent. As shown in Figure 2A, the LPS-only treated group showed markedly increased level of NO production compared to the LPS nontreated group.

The effect of
Prostaglandin E2, synthesized by COX-2, is associated with a wide range of pathological conditions, and drugs that inhibit PGE2 production are being studied for therapeutic purposes.
27
To examine whether DCS could inhibit LPS-induced PGE2 production in RAW264.7 macrophages, we evaluated the amount of PGE2 in cell culture using enzyme-linked immunosorbent assay (ELISA). As shown in Figure 3A, N-[2-(cyclohexyloxy)-4-nitrophenyl] methanesulfonamide (NS-398), a specific inhibitor of COX-2 protein, was used as a positive control.
28
Lipopolysaccharide induced an increase in PGE2 production. The group treated with the COX-2 inhibitor showed a 59.5% reduction in PGE2 production compared to the group treated with LPS alone.
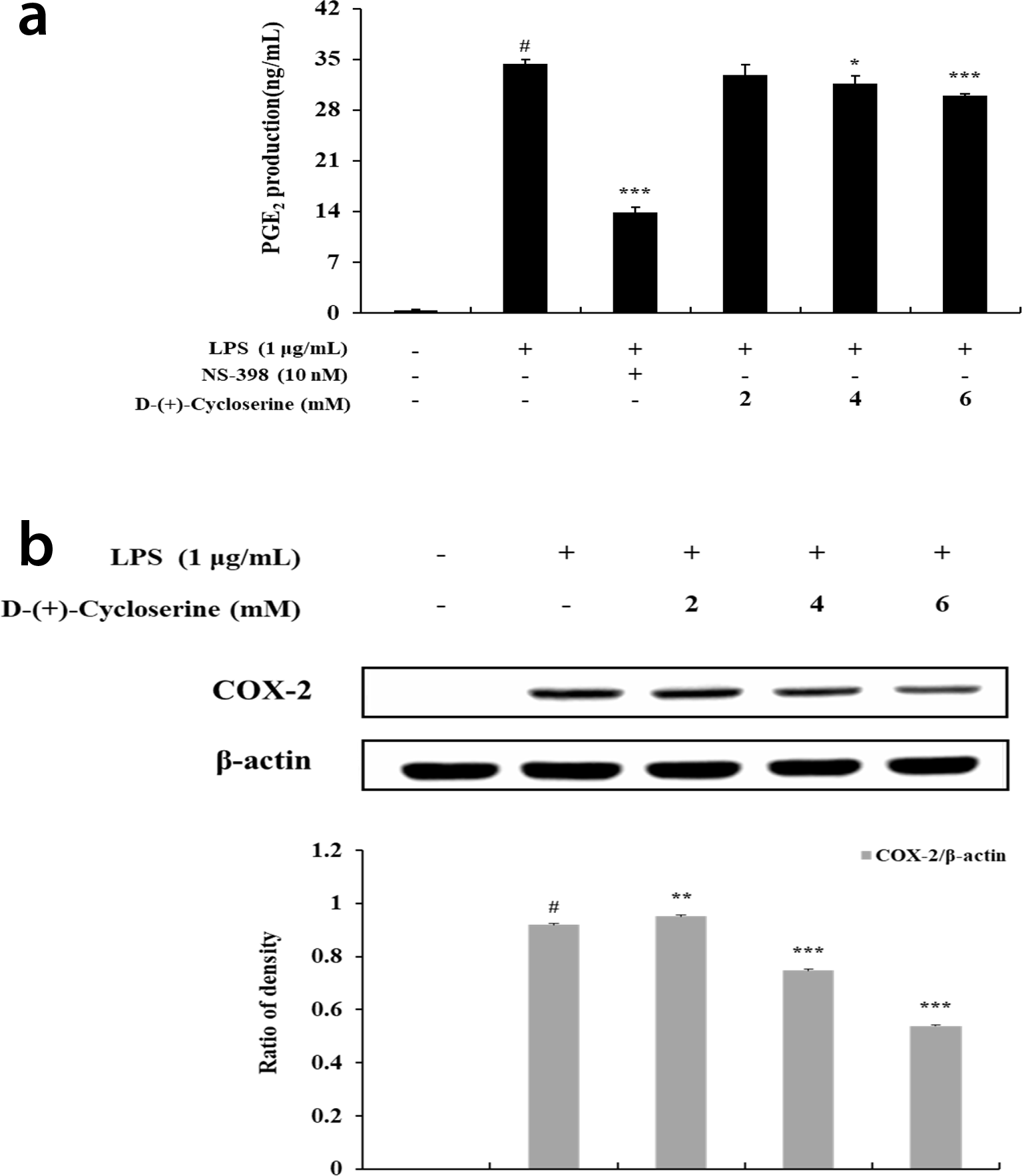
The effect of
Western blot analysis was used to investigate whether the inhibitory effect on PGE2 production was due to the regulation of COX-2 protein. The production of COX-2 was significantly increased in the group stimulated with LPS-only compared to the nontreated group. The expression of COX-2 protein reduced to some extent in the LPS-treated group in the presence of DCS.
Well-known pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 are activated by pathogenic stimuli and mediate inflammatory responses that have been linked to a variety of diseases. 29 -31 To investigate the anti-inflammatory potential of DCS, the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 was measured in LPS-stimulated RAW264.7 cells using ELISA. Lipopolysaccharide stimulation induced TNF-α, IL-1β, and IL-6 production in the cell culture media. At the concentrations tested, DCS inhibited LPS-induced production of IL-1β (49.3%, 80.1%, and 86.6%) and that of IL-6 (41.6%, 71.9%, and 81.6%) in a concentration-dependent manner (Figure 4). However, DCS did not influence the LPS-induced TNF-α production.

The effect of
Nuclear factor kappa B is a regulator of genes including inflammation and immunity. 32 Inactive NF-κB complex is controlled strictly by binding to IκB-α in the cytoplasm. 33 Activation by LPS leads to ubiquitination and subsequent degradation of IκB-α through the proteasome. 34 The released NF-κB complex then translocates from the cytoplasm to the nucleus and encodes genes which are involved in the expression of inflammatory cytokines such as TNF-α, IL-1β, and IL-6. 35 To confirm the mechanism whether DCS regulates NF-κB activation via phosphorylation of IκB-α, we evaluated the protein level of IκB-α and phosphorylated IκB-α in LPS-induced RAW264.7 using Western blot analysis. As shown in Figure 5, DCS inhibited the phosphorylation of IκB-α in a concentration-dependent manner. Consistent with these results, DCS suppressed the degradation of IκB-α in the cytoplasm in a concentration-dependent manner. In a previous study, it has been reported that NF-κB mediates the expression of pro-inflammatory mediators such as TNF-α, IL-1β, and IL-6, and induces the expression of inflammatory enzymes such as iNOS and COX-2, thereby enhancing the inflammatory response. 36 This study showed that DCS inhibits the degradation of IκB-α by regulating phosphorylation of IκB-α, thereby inhibiting NF-κB activation.

The effect of
We found that DCS inhibited the phosphorylation of IκB-α. Previous studies have reported that PI3K/Akt signaling pathways induce NF-κB activation by enhancing the degradation of IκB-α. 12 To determine whether DCS regulates NF-κB activation through the PI3K/Akt pathway, we checked the phosphorylation of Akt using Western blot analysis. As shown in Figure 6, DCS inhibited the phosphorylation of Akt compared to LPS-only treated group.

The effect of
Mitogen-activated protein kinases, the well-conserved serine/threonine protein kinases, play an important role in cellular activities.
37
In particular, MAPKs are known as inflammation-associated signaling pathways.
38,39
Lipopolysaccharide activates the phosphorylation of MAPKs.
40,41
The phosphorylated MAPKs, in turn, induce the expression of iNOS and COX-2 to mediate overproduction of inflammatory mediators such as TNF-α, IL-1β, and IL-6 through NF-κB activation.
42
Therefore, the phosphorylation of ERK, JNK, and p38 reflects the progression of inflammation. In the present study, to investigate whether DCS regulates the phosphorylation of MAPK, we examined the expression of phosphorylated MAPKs by Western blot analysis at various concentrations (2, 4, and 6 mM) of DCS in LPS-induced RAW264.7 macrophages. As shown in Figure 7, the LPS treatment group showed a marked increase in the phosphorylated ERK, JNK, and p38 compared to the LPS nontreated group.

The effect of
To further analyze the effect of DCS on the viability of HaCaT human keratinocytes, we evaluated the cytotoxicity of DCS using MTT assay. The cells were incubated in 24-well plates (5.0 × 104 cells/well) for 24 hours, and then treated with various concentrations of DCS (2, 4, 6, 8, and 10 mM) for an additional 24 hours. As shown in Figure 8, treatment with DCS at concentrations below 6 mM showed no cytotoxicity (% of cell survival ≥85%). However, cytotoxicity of DCS was detected at concentrations above 8 mM (31.7% and 11.9% at 8 and 10 mM DCS, respectively) in the HaCaT keratinocytes.

The effect of
In summary, our findings demonstrate that DCS treatment inhibits the production of NO, PGE2, IL-1β, and IL-6 and the expression of iNOS and COX-2 in LPS-stimulated RAW264.7 cells. Furthermore, DCS suppresses NF-κB activation by blocking degradation of IκB-α through the inhibition of ERK and Akt phosphorylation. Based on these anti-inflammatory functions of DCS, we suggest that DCS could be considered for therapeutic uses in inflammatory diseases.
Experimental
Chemicals and Reagents
Cell Culture and Sample Treatment
RAW264.7 macrophage cells were obtained from the Korean Cell Line Bank (Seoul, Korea). The cells were subcultured at 37°C using DMEM including 10% FBS, penicillin (100 U/mL), and streptomycin (100 µg/mL) in a humidified atmosphere of 5% CO2. The cells were subcultured every 2 days. For the DCS treatment, DCS was dissolved in distilled water and diluted with culture medium. The cells were pretreated at various concentrations (2, 4, and 6 mM) of DCS for 1 hour and then stimulated with LPS 1 µg/mL for the indicated durations of time.
Measurement of Cell Viability
Cell viability was determined using the MTT assay. Cells were plated at 1.0 × 105 cells/well in 24-well plates in DMEM with 10% FBS and 1% penicillin/streptomycin. The cells were pretreated with various concentrations of DCS (2, 4, and 6 mM) for 1 hour and then treated with 1 µg/mL LPS for 24 hours. A total of 100 µL of MTT solution (2 mg/mL) was added after incubation and the cells were further incubated for 4 hours. The formazan crystals in each well were dissolved in 1 mL of DMSO for 30 minutes on a microplate shaker. Optical density (OD) was measured by using a Microplate reader at 540 nm. Cell viability in the wells with control medium in the absence of any treatment was set at 100%.
NO Production
The accumulated nitrite in the culture media, an indicator of NO production, was determined using the Griess reagent. Cells were plated at 1.0 × 105 cells/well in 24-well plates in DMEM with 10% FBS and 1% penicillin/streptomycin. The cells were pretreated with various concentration of DCS (2, 4, and 6 mM) for 1 hour and then treated with 1 µg/mL LPS for 24 hours. After incubation, 100 µL of the culture supernatant was mixed with 100 µL of Griess reagent for 10 minutes. The absorbance of the mixture was measured at 540 nm with a microplate reader. The nitrite concentration in the supernatants was calculated from a standard curve of nitrite solution.
Measurement of Cytokines
RAW264.7 cells were plated at 1.0 × 105 cells/well in 24-well plates in DMEM with 10% FBS and 1% penicillin/streptomycin. The cells were pretreated with various concentration of DCS (2, 4, and 6 mM) for 1 hour and then treated with 1 µg/mL LPS for 24 hours. The supernatants were collected and the level of TNF-α, IL-1β, IL-6, and PGE2 was determined using ELISA kits following the manufacturer’s protocols.
Western Blot Analysis
RAW264.7 cells were plated in 60-mm cell culture dishes (6.0 × 105 cells/dish) for 40 hours. The cells were then pretreated with various concentrations of DCS (2, 4, and 6 mM) for 1 hour and then treated with 1 µg/mL LPS for the indicated durations of time. After treatment, the cells were washed with PBS. The cells were lysed on ice with RIPA buffer (50 mM Tris-HCl [pH 7.5], 2 mM ethylenediaminetetraacetic acid (EDTA), 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS), 1% Nonidet P-40, and 1% protease inhibitor cocktail) for 20 minutes. The harvested cell lysates were centrifuged at 15,000 rpm for 15 minutes. The protein concentrations in the supernatant were determined using a BCA protein assay kit. Equal amounts of total protein (30 µg) were separated by SDS-polyacrylamide gel electrophoresis on a 8% or 10% gel and transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were then blocked with 5% bovine serum albumin (BSA) and incubated for 2 hours. The membranes were washed with TBS buffer containing 0.1% Tween 20 (TBST) and then incubated with specific primary antibodies at 4°C overnight. The membranes were washed with TBST buffer and incubated with horseradish peroxidase-conjugated secondary antibodies in TBST buffer at room temperature for additional 2 hours. After washing the membranes, the proteins were detected using an ECL kit.
Statistical Analysis
All data are presented as the mean ± standard deviation from 3 independent experiments. Statistical analysis was conducted using the Student’s t-test. P-value of <.05 was considered to be statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the 2019 Scientific Promotion Program funded by Jeju National University, South Korea.
