Abstract
The stratum corneum tryptic enzyme kallikrein 5 (KLK5) is a serine protease that is involved in the cell renewal and maintenance of the skin barrier functions. The excessive activation of KLK5 causes an exacerbation of dermatoses, such as rosacea and atopic dermatitis. Nasturtium officinale R. Br. (Brassicaceae) is a hardy perennial plant native to Europe and commonly called “Watercress” or “Cresson,” which is one of the brassica vegetables. We aimed to investigate whether the bioactive sulfur-containing compound inhibits the KLK5 protease. The sulfur-containing compounds were evaluated using an enzymatic assay to measure the anti-KLK5 activity. Our study revealed that the sulfur-containing compound inhibits the KLK5 and 7 protease activity. Indole-3-acetonitrile-4-methoxy-2-S-β-
Keywords
Stratum corneum tryptic enzyme kallikrein 5 (KLK5) is a serine protease, expressed in the epidermis and involved in cell renewal and maintenance of the skin barrier function.
1
KLK5 plays a central role in the degradation of corneodesmosomes, which are the main adhesive structures in the cornified cell layer. KLK5 is also involved in the activation of other epithelial serine proteases such as kallikrein-related peptidase.
2
Although KLK5 is important for the maintenance of skin homeostasis, its overactivation can impair the skin barrier function and contribute to the discovery of various dermatoses.
3,4
Several studies have reported certain triterpenoids that are bioactive natural products and suppress the serine protease activity. Plant-derived pentacyclic triterpenoids, such as oleanolic acid, ursolic acid, and β-boswellic acid, have been reported to inhibit the esterase activity in human neutrophils.
5
Nasturtium officinale R. Br. (Brassicaceae) is a hardy plant native to Europe and commonly called “Watercress” or “Cresson,” which is a species of the brassica vegetables, such as broccoli, cabbage, Cauliflower, Kale, wasabi, and horseradish. The plant has aroused interest because of the presence of sulfur-containing compounds and their important role in protecting human health. The characteristics mentioned above are speculated to be due to some kinds of sulfur compounds deriving from myrosinase-catalyzed hydrolysis of glucosinolates in the Brassicaceae family.
5
Glucosinolates could exert neuroprotective activity by regulating inflammatory responses in the central nervous system.
6,7
Although N. officinale is widely known for its actions in dietary supplements and cosmetics, its effects on atopic dermatitis are yet to be investigated. Therefore, we investigated the efficacy of a sulfur-containing compound from N. officinale on KLK5 and KLK7 protease activity. The sulfur-containing compound (Figure 1) was obtained as an amorphous white powder, and the structure was determined to be indole-3-acetonitrile-4-methoxy-2-S-β-
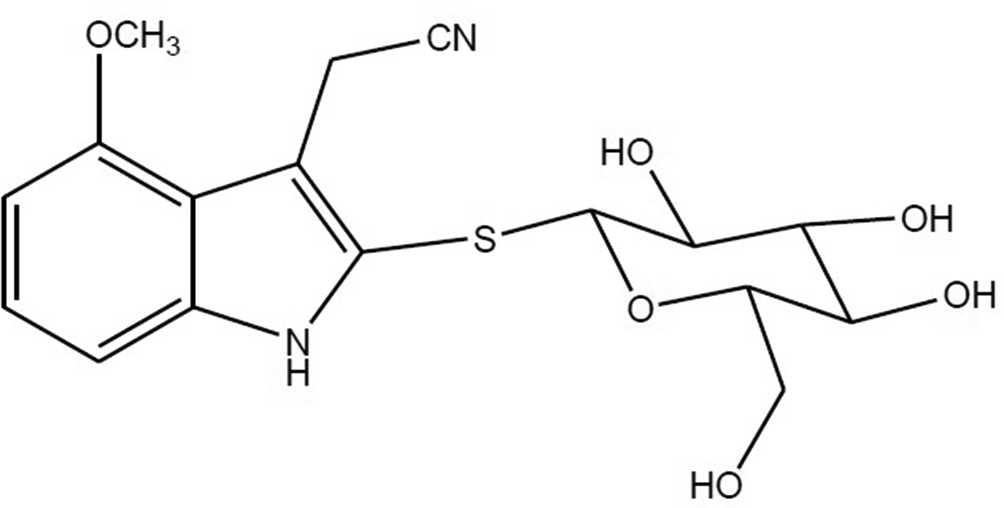
Chemical structures of indole-3-acetonitrile-4-methoxy-2-S-β-
Assay to Indole-3-Acetonitrile-4-Methoxy-2-S-Β-

RFU, relative fluorescent unit.
Recombinant human kallikrein 5 (KLK5, 8.1 nM) was mixed with 10 µM test compounds and Boc-Val-Pro-Arg-AMC fluorogenic peptide (100 µM). After incubating for 5 minutes, the RFU was measured at Ex 380 nm/Em 460 nm. A serine protease inhibitor, leupeptin hemisulfate (42.1 µM), was used as positive control. Indole-3-acetonitrile-4-methoxy-2-S-β-
Comparative Activities of Ursolic Acid and Indole-3-Acetonitrile-4-Methoxy-2-S-Β-

IC50, half-maximal inhibitory concentration; RFU, relative fluorescent unit.
Indole-3-acetonitrile-4-methoxy-2-S-β-
Experimental
General
Nasturtium officinale was purchased in November 2018 in a rural farmhouse, Youngin-si, Gyeonggi-do, South Korea, and the plant was identified by one of the authors (K.S. Bae). A voucher specimen (Skedrm20181108) has been deposited at the raw material room, SKEDERM cosmetic R&D center, South Korea. Nuclear magnetic resonance (NMR) spectra were recorded on a Varian Inova-400 FT-NMR spectrometer (CA, USA) using tetramethylsilane as an internal standard, δ in ppm, J in Hz. High-resolution electrospray ionization mass spectrometry (HR-ESI-MS) were measured on Bruker APEXII mass spectrometer in m/z. EI-MS were measured on a VG ZABHS mass spectrometer at 70 eV. Silica gel (200, 300 mesh) was obtained from Merck Co. Ltd. Ursolic acid was purchased from Wako Pure Chemical Industries (Osaka, Japan). Dexamethasone (Wako Pure Chemical Industries), leupeptin hemisulfate salt, and chymostatin (Sigma-Aldrich, St. Louis, MO, USA) were purchased for use as reference reagents.
Extraction and Isolation
The air-dried N. officinale (2 kg) was extracted with 70% EtOH, 3 × 20 L, 7 days each) at room temperature, and the EtOH was removed under reduced pressure to give a residue (315 g), which was suspended in distilled water and extracted with n-hexane and EtOAc, respectively. The EtOAc extract (24 g) was subjected to column chromatography over silica gel (200, 300 mesh, 2500 g) and eluted with CHCl3), CHCl3–MeOH (95/5), (90/10), (85/15), (80/20), (70/30), (50/50), (30/70), (10/90) and MeOH to yield 10 fractions (Fr.1–Fr.10). Fr.2-4 was subjected to a silica gel column eluting with CHCl3–MeOH (19/1). Indole-3-acetonitrile-4-methoxy-2-S-β-
Measurements of Enzymatic Activity
KLK5, KLK7, trypsin, and chymotrypsin C activities were evaluated according to the following methods supplied by R & D Systems Inc. (Minneapolis, MN, USA). The enzyme activity-dependent increase in the relative fluorescent unit (RFU) was measured, and the percentage of inhibition was calculated based on the formula: (1 − (A − B)/(C − B)) × 100, where A = RFU of test samples with enzymes, B = basal RFU without enzymes, and C = RFU of vehicle controls with enzymes.
To measure the KLK5 activity, the enzymatic reaction was performed at room temperature in 100 mM NaH2PO4buffer (pH 8.0) containing 0.25 µg/mL recombinant human KLK5 (R & D Systems Inc.), 100 mM of Boc-V-P-R-AMC Fluorogenic Peptide Substrate (R & D Systems Inc.), and 1.1% DMSO at final concentrations. KLK5 (final 8.1 nM) was preincubated with test samples for 5 minutes, followed by the addition of a peptide substrate. After incubating for 5 minutes, RFU was measured at Ex 380 nm/Em 460 nm. Leupeptin hemisulfate (42.1 µM) was used as a positive control.
To measure the KLK7 activity, recombinant human pro-KLK7 (R & D Systems Inc.) was activated by bacterial thermolysin at 37°C for 2 hours just before the enzyme assay. Thereafter, the enzymatic reaction was performed at room temperature in 50 mM Tris, 150 mM NaCl buffer (pH 8.5) containing 1 µg/mL activated-KLK7, 10 µM Mca-R-P-K-P-V-E-Nval-W-R-K (Dnp)-NH2 Fluorogenic Peptide Substrate II (R & D Systems Inc.), 150 mM NaCl, and 1.1% DMSO at final concentrations. Activated KLK7 (final 38.5 nM) was preincubated with test samples for 5 minutes, followed by the addition of a peptide substrate. After incubating for 60 minutes, RFU was measured at Ex 320 nm/Em 405 nm. Chymostatin (10 µM) was used as a positive control.
To measure the trypsin activity, the enzymatic reaction was performed at room temperature in 100 mM NaH2PO4 buffer (pH 8.0) containing 0.25 µg/mL recombinant human trypsin (Wako Pure Chemical Industries), 100 µM of Boc-V-P-R-AMC Fluorogenic Peptide Substrate, and 1.1% DMSO at final concentrations. KLK5 was preincubated with test samples for 5 minutes, followed by the addition of a peptide substrate. After incubating for 1 minute, RFU was measured at Ex 380 nm/Em 460 nm. Leupeptin hemisulfate (42.1 µM) was used as a positive control.
To measure the chymotrypsin C activity, recombinant human pro-chymotrypsin C (R & D Systems Inc.) was activated by trypsin at 37°C for 1 hour just before the enzyme assay. Thereafter, the enzymatic reaction was performed at room temperature in 25 mM Tris, 0.5 mM CaCl2 buffer (pH 8.0) containing 1 µg/mL activated chymotrypsin C, 10 µM Suc-A-A-P-F-AMC (Bachem AG, Bubendorf, Switzerland) used as a fluorogenic substrate, and 1.1% DMSO at final concentrations. Activated chymotrypsin C (final 34.7 nM) was preincubated with test samples for 5 minutes, followed by the addition of a peptide substrate. After incubating for 60 minutes, RFU was measured at Ex 380 nm/Em 460 nm. Chymostatin (10 µM) was used as a positive control.
Statistical Analysis
All of the data are presented as means ± standard deviation. Statistical analyses used the one-way analysis of variance followed by Dunnett’s test. P < 0.05 was considered to be statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was carried out with the support of Classys INC. (Project title: Construction of high purity, concentration extracts techniques from natural materials).
