Abstract
Owing to the side effects of current drugs for treating atopic dermatitis (AD), a chronic disease in the skin, traditional herbal medicine is receiving much attention as an alternative treatment. Asarum sieboldii has traditionally been used to treat colds, fevers, headaches, coughs, neuralgia, chronic bronchitis, asthma, and allergies. In this study, 6 compounds (echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin) in A. sieboldii were analyzed simultaneously using high-performance liquid chromatography (HPLC), and the proposed analytical method was validated. In addition, the inhibitory effects of the A. sieboldii extract and the 6 analyzed compounds on the expression of chemokine were evaluated in HaCaT cells. The HPLC method showed high linearity, with a correlation coefficient of ≥0.9999. The limits of detection for the 6 compounds ranged from 0.00 to 0.02 µg/mL, and the limits of quantification ranged from 0.01 to 0.05 µg/mL. The intraday and interday precisions were 0.15%-1.90%; the accuracy was 97.36%-103.23%, and the recoveries of the 6 compounds were 97.45%-103.93%. The content of each compound in A. sieboldii, as determined using the corresponding calibration curve, was in the range of 0.380-12.062 mg/g. This optimized simultaneous quantification method will be suitable for improving the quality control of A. sieboldii. Moreover, the 6 compounds in A. sieboldii showed an inhibitory effect on the production of chemokines, which suggests that A. sieboldii has an antiallergic effect.
Keywords
Atopic dermatitis (AD), a chronic disease, can lead to eczematous and itchy lesions due to ecological and environmental factors, as well as changes in various immune systems. 1 AD shares common characteristics with other inflammatory diseases such as psoriasis and allergic contact dermatitis, but genetic, immune, and environmental factors specific to AD have also been identified. 2 Patients with AD often have or are at a very high risk of developing other allergic diseases such as asthma and allergic rhinitis. 3,4 Current drugs for AD, such as corticosteroids, may be effective, but have serious side effects, including the systematic side effects associated with the use of topical corticosteroids. 5,6 Owing to the chronic nature of AD and a lack of response to steroids, many people are seeking alternative treatments. 7 In particular, traditional herbal medicine has recently received considerable attention for the treatment of AD and psoriasis. 8 Many studies have reported the effectiveness of traditional herbal medicines in treating AD in terms of reducing the severity of the problem and having beneficial effects on patients, such as improving the quality of life. 9 -12
Asarum sieboldii is a perennial herb belonging to the Aristolochiaceae family, which is mainly distributed across Korea, China, and Japan. 13,14 The roots of A. sieboldii have traditionally been used as a cure for colds, fevers, chills, headaches, acute toothaches, sinusitis, coughs, neuralgia, chronic bronchitis, asthma, and allergies. 15,16 Asarum sieboldii has also been shown to have various pharmacological actions, such as antiallergic, 17,18 anti-inflammatory, 19 antitussive, 20 antifungal, 13 neuroprotective, 21 and liver-protective 22 effects. The major components of A. sieboldii are essential oil, lignans, and alkaloids, as reported in previous studies. 19,23 -25 The study of the components of traditional herbal medicines is very important to ensure the reliability of pharmacological and clinical research. 26 Because traditional herbal medicines have different chemical compositions, it is necessary to develop accurate and effective simultaneous quantification methods. 27,28 Simultaneous determination of the major active components is also critical to understanding the material basis for pharmacological effects. 29 Simultaneous analytical studies of A. sieboldii have been performed, but these are limited to the degree of chemical profiling of the components, 30,31 and the simultaneous determination of active components has not been reported.
In this study, to realize a simple and reliable analytical method for the quantitative determination of A. sieboldii, a high-performance liquid chromatography-photodiode array (HPLC-PDA) method for the simultaneous quantification of 6 compounds in A. sieboldii was developed and validated. In addition, the antiallergic activities of the 6 marker compounds in A. sieboldii were measured using HaCaT keratinocyte cells.
Materials and Methods
Plant Materials
Asarum sieboldii (Aristolochiaceae) roots were purchased from Omniherb Co. (Yeong Cheon, Korea), and the plant material was identified at the Korea Institute of Oriental Medicine (KIOM). A voucher specimen (No. KIOM 29) has been deposited at the Herbal Medicine Research Division, Korea Institute of Oriental Medicine.
Chemical and Reagents
The 6 reference compounds (echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin) and internal standard (ferulic acid) used in the present study were purchased from ChemFaces Biochemical Co., Ltd. (Wuhan, China). The chemical structures of the compounds are shown in Figure 1. The purities of the 6 compounds were >98%, as evaluated by HPLC. HPLC-grade solvents (methanol, acetonitrile, and water) were purchased from JT Baker Chemical Co. (Phillipsburg, NJ, USA), and analytical-grade trifluoroacetic acid was purchased from Samjeon Chemical Co. (Seoul, Korea).

Chemical structures of the 6 marker compounds in Asarum sieboldii.
Preparation of Standard Solutions
Each of the 6 reference compounds was weighed accurately and dissolved in methanol to prepare individual stock solutions at appropriate concentrations. The stock solutions were initially prepared by dissolving the commercially obtained standard materials (2.5 mg echinacoside, 2.0 mg vanillic acid and sesamin, 5.0 mg kakuol and methyl eugenol, and 4.0 mg asarinin) in 10 mL methanol. To construct the calibration curves, a series of working standard solutions were prepared by diluting the stock solutions with methanol. The working solution concentrations were 3.91, 7.81, 15.63, 31.25, 62.5, 125, and 250 µg/mL for echinacoside, kakuol, and methyl eugenol, 1.56, 3.13, 6.25, 12.5, 25, and 50 µg/mL for vanillic acid, 3.13, 6.25, 12.5, 25, 50, 100, and 200 µg/mL for sesamin, and 6.25, 12.5, 25, 50, 100, 200, and 400 µg/mL for asarinin. Ferulic acid was used as an internal standard with a concentration of 25 µg/mL for all analytes.
Preparation of 70% Ethanol Extract and Sample Solutions
Asarum sieboldii was extracted twice with 70% ethanol (100 g/L) for 2 hours at 70 °C and the extract was then filtered (Whatman No. 1 filter paper). The extract was concentrated using a rotary evaporator under reduced pressure at 40 °C, lyophilized, and then stored at −20 °C until use. The yield of the dried extract from the initial crude materials was 15.45% (w/w).
For the simultaneous determination of the 6 compounds, the A. sieboldii extract was dissolved in methanol (10 mg/mL) by sonication for 10 minutes. Before HPLC analysis, the sample solution was filtered through a syringe filter (0.2 µm).
Instrumentation and Chromatographic Conditions
The sample was analyzed using a Waters Alliance e2695 liquid chromatography system (Waters Co., Milford, MA, USA) equipped with a Waters 2998 PDA detector. All chromatographic data were acquired and processed with Empower software (Waters Co.). Chromatographic separation was performed using a Phenomenex Luna C18 column (250 mm × 4.6 mm, particle size 5 µm; Phenomenex, Torrance, CA, USA), and 0.1% (v/v) trifluoroacetic acid aqueous solution (A) and acetonitrile (B) were used as the mobile phase. The gradient profile was as follows: 10%-35% B for 0-10 minutes, 35%-55% B for 10-12 minutes, and 55%-90% B for 12-30 minutes. Subsequently, a wash with 100% B was applied for 8 minutes, followed by equilibration with 90% A/10% B for 8 minutes. The flow rate was 1.0 mL/min, and the injection volume was 20 µL. The components were quantified based on the peak area at 290 nm. The peaks were identified by comparison of the retention times and ultraviolet spectra with those of the standard compounds.
Calibration Curves, LOD, and LOQ
The calibration curves were constructed by plotting the peak area versus the concentration of each compound. The linear regression equations were calculated in the form y = ax ± b, where x is the concentration and y is the peak area of each compound. The linearity was established by the coefficient of determination (r 2). The limit of detection (LOD) and limit of quantification (LOQ) of each standard were determined by injecting the diluted standard solutions, at signal-to-noise ratios of approximately 3 and 10, respectively.
Precision, Accuracy, and Recovery
The intraday and interday precisions were adopted to determine the repeatability and reproducibility of the developed method. To evaluate the intraday precision and accuracy, a mixed standard solution was analyzed 5 times on a single day. The analysis of the mixed standard solution was repeated on 3 consecutive days to evaluate the interday precision and accuracy. The precision was expressed as the relative SD (RSD, %), and the accuracy was determined as the percentage of the observed concentration relative to the nominal concentration. Recovery experiments were used to evaluate the accuracy of the developed method. The A. sieboldii extract was spiked with each analyte at recovery levels of 80%, 100%, and 120%. The percent recovery of each compound in the spiked sample was calculated as follows: recovery (%) = (detected concentration − original concentration)/spiked concentration × 100.
Cell Culture
A spontaneously immortalized human HaCaT keratinocyte cell line (a gift from Dr H. K. Shin, KIOM, Daejeon, Korea) was cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco BRL, Gaithersburg, MD, USA) with 10% fetal bovine serum (Gibco BRL), 100 U/mL penicillin (Gibco BRL), and 100 µg/mL streptomycin (Gibco BRL) at 37 °C in a humidified 5% carbon dioxide (CO2) atmosphere.
Cell Viability
Cells were seeded in 96-well, flat-bottom plates (5 × 104 cells/well) and incubated with A. sieboldii extract, echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin for 24 hours. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide solution (0.5 mg/mL) was added to each well for 2 hours (5% CO2 at 37 °C), and 100 µL of dimethyl sulfoxide was added to solubilize the purple formazan crystals. Subsequently, the absorbance at 540 nm was recorded using a microplate reader (Thermo Fisher Scientific, Waltham, MA, USA).
Chemokine Assay
The HaCaT cells were stimulated with tumor necrosis factor-alpha (TNF-α, 10 ng/mL, R&D Systems, Minneapolis, MN, USA) and interferon (IFN)-γ (10 ng/mL, R&D Systems). The cells were subsequently incubated with A. sieboldii extract, echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin for 24 hours. The production of macrophage-derived chemokine (MDC) and regulated upon activation normal T-cell expressed and presumably secreted (RANTES) chemokine in the supernatant was measured using enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems) according to the manufacturer’s protocol.
Data Analysis
All values are expressed as means ± SEM. Statistical comparisons were performed using the unpaired Student’s t-test and analysis of variance for repeated measurements using Sigma Plot software version 13.0, 2016 (Systat Software Inc., San Jose, CA, USA). Differences were considered significant at P < 0.05.
Results
Optimization of HPLC Conditions
Typical chromatograms obtained for the A. sieboldii extract and the standard sample containing 6 compounds are presented in Figure 2. To achieve efficient separation of the 6 compounds, various mobile phase compositions (methanol and acetonitrile with acetic acid, formic acid, and trifluoroacetic acid) and wavelengths (200-400 nm) were evaluated. The separation was more efficient with the mobile phase of acetonitrile than that of methanol. Although the 3 types of acids did not differ significantly, we selected trifluoroacetic acid, which is a widely used mobile phase and is reported to have better resolution and peak shapes than other acids. 32 -34 The most suitable conditions for quantitative analysis (detection wavelength of 290 nm) were obtained using a gradient elution of the mobile phase consisting of 0.1% (v/v) trifluoroacetic acid (A) and acetonitrile (B). The 6 compounds were separated within 30 minutes, and the retention times of echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin were 8.2, 9.4, 19.7, 20.5, 21.4, and 22.6 minutes, respectively. Good baseline resolution was obtained in all analyses under the proposed analysis conditions.

HPLC analysis of Asarum sieboldii. HPLC of (A) the standard mixture and (B) the A. sieboldii extract (echinacoside [1], vanillic acid [2], kakuol [3], methyl eugenol [4], sesamin [5], asarinin [6], and the IS, ferulic acid). HPLC, high-performance liquid chromatography; IS, internal standard.
Calibration Curves, LOD, and LOQ
A calibration curve was constructed for each reference compound using the peak area versus the concentration. The correlation coefficients of all the calibration curves exceeded 0.9999. The LOD and LOQ are common variables for assessing the sensitivity of an analytical method. For the analyzed reference compounds, the LOD values were in the range of 0.00-0.02 µg/mL, and the LOQ values in the range of 0.01-0.05 µg/mL. The detailed data used for the determination of the linearity, range, LOD, and LOQ of each compound are given in Table 1.
Linear Regression Data, Linear Range, LODs, and LOQs of the 6 Compounds.
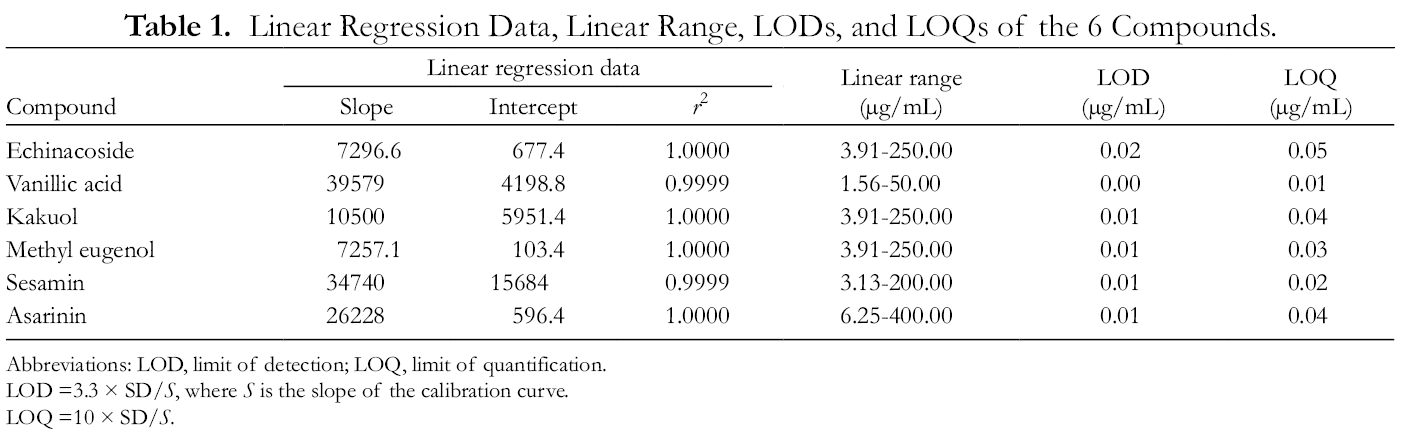
Abbreviations: LOD, limit of detection; LOQ, limit of quantification.
LOD =3.3 × SD/S, where S is the slope of the calibration curve.
LOQ =10 × SD/S.
Precision, Accuracy, and Recovery
The precision and accuracy of the instrument were assessed by analyzing the standard solution upon repeated injection (n = 5). Three different concentrations (low, middle, and high) were used for each standard solution. The coefficient of variation (CV) values were used as a measurement of the precision and the measured concentration relative to the nominal concentration of the standard solution was used as a measurement of the accuracy. As detailed in Table 2, the intraday and interday precisions were in the range of 0.15%-1.90% and the accuracy values in the range of 97.36%-103.23%. The intraday and interday coefficients of variation were found to be less than 5% and the accuracy values were within the range of 90%-110%, which indicates that this method is effective and has high accuracy. 35 -38
Intraday and Interday Precision and Accuracy for the 6 Reference Compounds.

Abbreviation: CV, coefficient of variation.
Precision was determined as CV; CV (%) = (SD/mean measured concentration) × 100.
Accuracy (%) = (mean measured concentration/nominal concentration) × 100.
Recovery tests were also carried out to assess the accuracy of the analytical method. The A. sieboldii extract was spiked with 3 different known amounts of the standard solutions. The recoveries of the 6 compounds ranged from 97.45% to 103.93%, with RSD values of 0.08% to 1.33% (Table 3). These results indicate that this analytical method is appropriate for the simultaneous quantification of the major compounds in A. sieboldii extract.
Recoveries of the 6 Reference Compounds.

Recovery (%) = (detected concentration − original concentration)/spiked concentration × 100.
Quantitative Analysis of A. sieboldii Extract
Chemical profiling and quantification of the major compounds in the A. sieboldii extract were performed using the developed HPLC method. The content of each compound was determined using the corresponding calibration curve (Table 4). The amounts of echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin were 6.706, 0.380, 1.969, 1.852, 2.816, and 12.062 mg/g, respectively. Among the 6 analyzed compounds, the content of asarinin in the A. sieboldii extract was found to be the highest.
Contents of the 6 Compounds in Asarum sieboldii Extract.
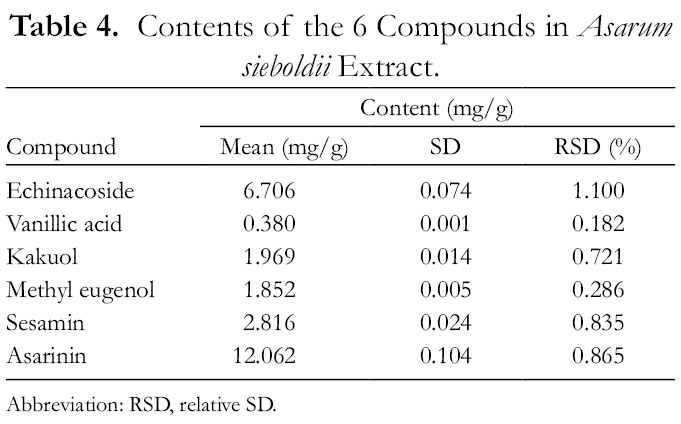
Abbreviation: RSD, relative SD.
Suppression of Chemokine Production in HaCaT Cells
To evaluate the effect of A. sieboldii and its constituent compounds on the suppression of AD-related chemokine production, we examined the effects of the extract on HaCaT cells by ELISA analysis. First, the cytotoxicity of A. sieboldii and its 6 components was evaluated. As shown in Figure 3 (left), the extract and 6 compounds did not affect the cell viability. However, the A. sieboldii extract and all 6 compounds (echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin) significantly reduced the production of the AD-related chemokines MDC and RANTES (Figure 3 middle and right).
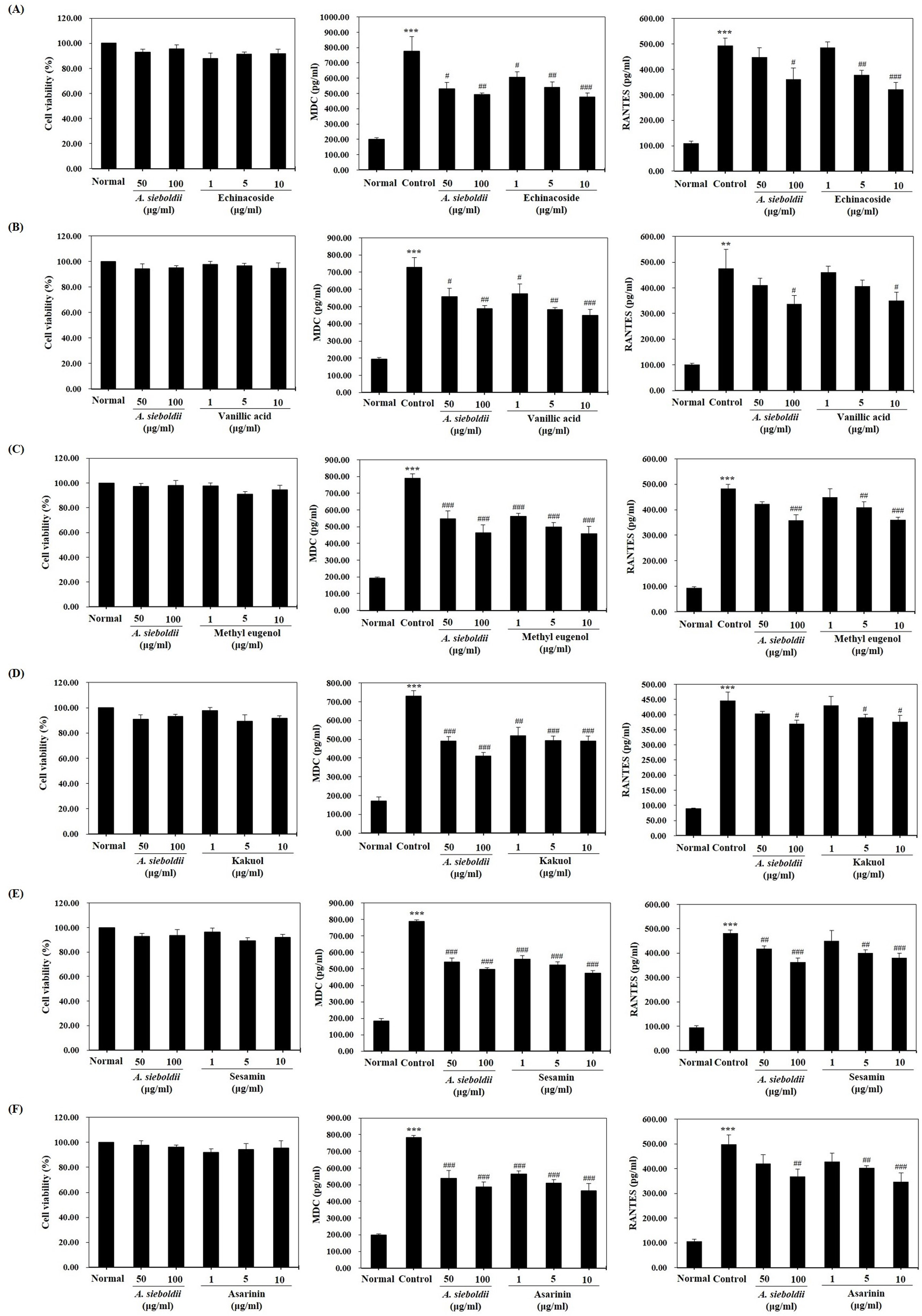
Suppression of the production of AD-related chemokines in HaCaT cells by compounds in A. sieboldii. Effects of (A) echinacoside, (B) vanillic acid, (C) kakuol, (D) methyl eugenol, (E) sesamin, and (F) asarinin on (left) cell viability, (middle) MDC expression levels in HaCaT cells, and (right) RANTES expression levels in HaCaT cells. Data are presented as mean ± SEM. **P < 0.01, ***P < 0.001 vs. normal; # P < 0.05, ## P < 0.01, ### P < 0.001 vs. control. AD, atopic dermatitis; MDC, macrophage-derived chemokine; RANTES, regulated uponactivation normal T-cell expressed and presumably secreted chemokine.
Discussion
Asarum sieboldii has a centuries-long history as a folk medicine in Korea, China, and Japan and is also a traditional herbal medicine widely used to treat headaches, toothaches, and inflammatory diseases.21,26 However, there is a lack of scientific evidence to support these traditional practices, resulting in questions related to efficacy, quality control, and safety. 39,40
Previous studies have reported the chemical constituents of A. sieboldii, such as essential oils (eg, methyl eugenol, cineol, safrole, eucarvone, elemicin, and kakuol), lignans (eg, asarinin, xanthoxylol, and sesamin), and alkaloids (eg, aristolochic acid, aristolactam, and higenamine). 23 -25 The contents of the compounds were compared between different collections of A. sieboldii using HPLC, ultra HPLC-quadrupole time-of-flight/mass spectrometry (MS), and gas chromatography (GC)-MS. 30,41,42 In addition, the contents of the essential oils of A. sieboldii were compared using GC according to the growth conditions and years of cultivation. 43,44 Other studies have proposed methods for the quantitative analysis of the components of A. sieboldii for quality control. 31,45 However, previous studies were related to the chemical compounds of A. sieboldii, and the analysis of active components has not been reported yet.
In this study, a convenient and accurate HPLC-PDA method was established and verified for the simultaneous analysis of 6 components of A. sieboldii. According to the Chinese Pharmacopeia, essential oils and asarinin can be used as marker compounds for the quality control of A. sieboldii. 46 In the present study, asarinin (12.06 ± 0.10 mg/g) was found to be the most abundant component of A. sieboldii. In addition, kakuol, sesamin, and a major essential oil component, methyl eugenol, were detected simultaneously, as in other studies. 30,31 The phenolic compounds echinacoside and vanillic acid were also detected in A. sieboldii. It is essential to identify and quantify these components in order to identify any correlations between the major ingredients in herbal medicine and the biological effects. 47
In a previous study, Kim et al reported that A. sieboldii could be applied for the treatment of allergic diseases. 17 In another study, an A. sieboldii extract containing methyl eugenol, asarinin, and sesamin was reported to have antiallergic effects on various experimental models in vitro and in vivo. 18 Other studies have also reported antiallergic effects of echinacoside and vanillic acid. 48,49 The present study investigated the effects of A. sieboldii and 6 of its components on the chemokine expression in HaCaT cells. MDC and RANTES are representative inflammatory chemokines, predominantly expressed in various immune cells. Previous studies have shown that the chemokine levels in the serum and skin of AD patients are elevated, suggesting that chemokines are the key molecules that attract inflammatory lymphocytes to the skin. 50 -52 The results of our study show that A. sieboldii and all 6 components (echinacoside, vanillic acid, kakuol, methyl eugenol, sesamin, and asarinin) significantly reduced the production of MDC and RANTES in HaCaT cells in a dose-dependent manner. Therefore, the 6 components of A. sieboldii may have activity against skin inflammation by regulating the expression of chemokines.
Conclusions
In this study, an HPLC method for the simultaneous quantification of 6 marker compounds in A. sieboldii was developed and optimized. The method validation confirmed good linearity, LOD, LOQ, precision, accuracy, and recovery for each compound. Thus, our method could be suitable for application to the quality control of A. sieboldii. In addition, the 6 components in A. sieboldii reduced the expression of MDC and RANTES in HaCaT cells, indicating that the antiallergic activity of these compounds involves the inhibition of chemokine production. It is necessary for further studies to analyze the content of the 6 compounds of different batches, taking into account various factors affecting the quality of A. sieboldii, using the simultaneous determination method established in this study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government MSIP (NRF-2015M3A9E3052336) and the grant (KSN2012330) from the Korea Institute of Oriental Medicine, Republic of Korea.
