Abstract
Fluoride is an essential trace element, but its beneficial range is narrow, and excess fluoride may have negative health effects. The objective of this study was to investigate the potential cytoprotective effects of epigallocatechin-3-gallate (EGCG) in cultured neuro-2a neuroblastoma cells exposed to sodium fluoride (NaF)-induced oxidative stress. Isolated Neuro-2a cells were exposed to increasing concentrations of NaF (0, 1, 2, 4, 6, and 8 mM) for 24 hours to induce oxidative stress. Moreover, to determine the concentration of EGCG necessary for protective effects, we exposed isolated Neuro-2a cells to increasing concentrations of EGCG (0, 0.5, 1, 5, 10, 20, 40, 60, 80, and 100 μg/mL) for 24 and 48 hours. Pretreatment with EGCG at various doses (0, 0.5, 1, 5, 10, 20, and 40 μg/mL) was evaluated in Neuro-2a cells for 24 hours, followed by an NaF (4 mM per culture well) challenge for 24 hours. As shown in this study, EGCG can protect Neuro-2a cells from NaF-induced apoptosis. This effect may be due to the reactive oxygen species scavenging activity of EGCC.
Diabetes mellitus is a common chronic metabolic disease. Fluorine is widely dispersed in nature, almost entirely in the form of fluorides. The daily exposure of humans to fluorine mainly depends on the intake of this element in drinking water. 1 Fluorides occur naturally in the environment, and daily exposure occurs through drinking water and is linked to geographical region. 2 In some regions, such as various areas of China, India, and Bengal, the drinking water contains dangerously high levels of fluoride (>1.2 mg/L), which lead to serious health problems. Green tea, originally cultivated in China, is considered one of the most popular beverages worldwide. 3 Epigallocatechin-3-gallate (EGCG; Figure 1) is a natural component of green tea, representing approximately 43% of its total phenolic content. 4 This compound is thought to be responsible for most of green tea’s therapeutic benefits, and has received considerable attention due to its antioxidant effects. 5 In recent years, EGCG has been investigated as a potential therapeutic target in different areas of study, including neuronal diseases, cancer, and type 2 diabetes. 6 -8 The protective effect of EGCG on the damage induced by NaF is seldom studied. This paper discusses the damage of NaF to Neuro-2a cells (mouse-derived neuroblastoma cells), and the potential protective effect of EGCG.

Chemical structure of epigallocatechin-3-gallate.
We first studied the cytotoxicity of NaF. Neuro-2a cells were treated with (0-8 mM) NaF for 24 hours and subjected to a cell counting kit8 (CCK-8) assay to assess cell viability. The Neuro-2a cells were treated with increasing concentrations of NaF for 24 hours, and dose-dependent inhibition of the cell proliferation rate was observed (Figure 2). Compared with the control group, the cells treated with 4 mM NaF for 24 hours showed a decrease in cell proliferation to 44% (P < 0.05). The Neuro-2a cell viability decreased significantly (P < 0.01) to 13.5% when the concentration exceeded 6 mM and reached 8 mM compared with that of the control group. This finding indicated that NaF was toxic to the Neuro-2a cells and that the toxicity was positively correlated with the concentration. Thus, 4 mM NaF and 24 hours of incubation were chosen for subsequent experiments.

Dose-dependent effects of NaF on the viability of Neuro-2a cells. **Significant difference (P < 0.05); ***Very significant difference (P < 0.01).
The effects of EGCG on the Neuro-2a cells were further investigated to determine the optimal treatment concentration. Pretreatment of the Neuro-2a cells with 0-100 µg/mL EGCG for 24 and 48 hours elicited time- and dose-dependent inhibition of the cell proliferation rate (Figure 3). The Neuro-2a cell proliferation rate reached a maximum (90%-95%) when the concentration of EGCG was 0-40 µg/mL, and this value was significantly greater than that in the 60-100 µg/mL EGCG group (P < 0.01). Additionally, compared with the cells treated with 0-40 µg/mL for 24 hours, the Neuro-2a cells pretreated with 0-40 µg/mL EGCG for 48 hours showed a significantly reduced cell proliferation rate (P < 0.05). Therefore, 0-40 µg/mL and 24 hours of incubation were considered safe to use in the subsequent experiments.
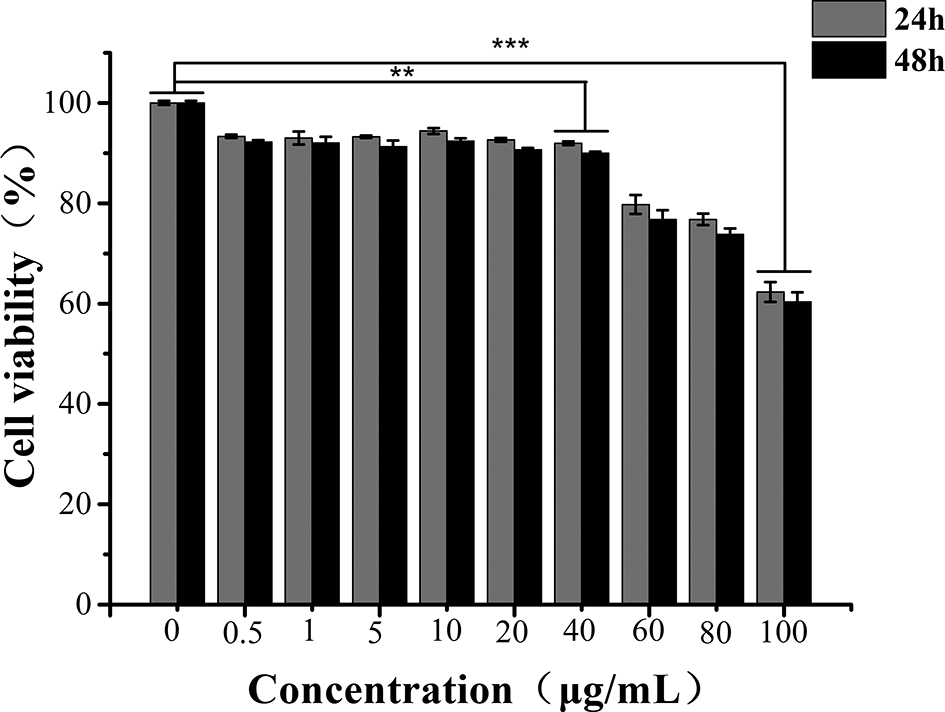
Dose-dependent effects of epigallocatechin-3-gallate on the viability of Neuro-2a cells. **Significant difference (P < 0.05); ***Very significant difference (P < 0.01).
Next, experiments were carried out to examine the potential protective effects of EGCG (Figure 4) against a lethal dose of NaF. The effects of EGCG on the Neuro-2a cell morphology induced by NaF injury were observed by inverted microscopy. The cell proliferation rate increased to a maximum (78.7%) when the concentration of EGCG increased to 10 µg/mL and was significantly greater than that in the NaF-treated group (42.2%) (P < 0.05). These results indicated that EGCG had a significant protective effect on the oxidative damage caused by NaF.
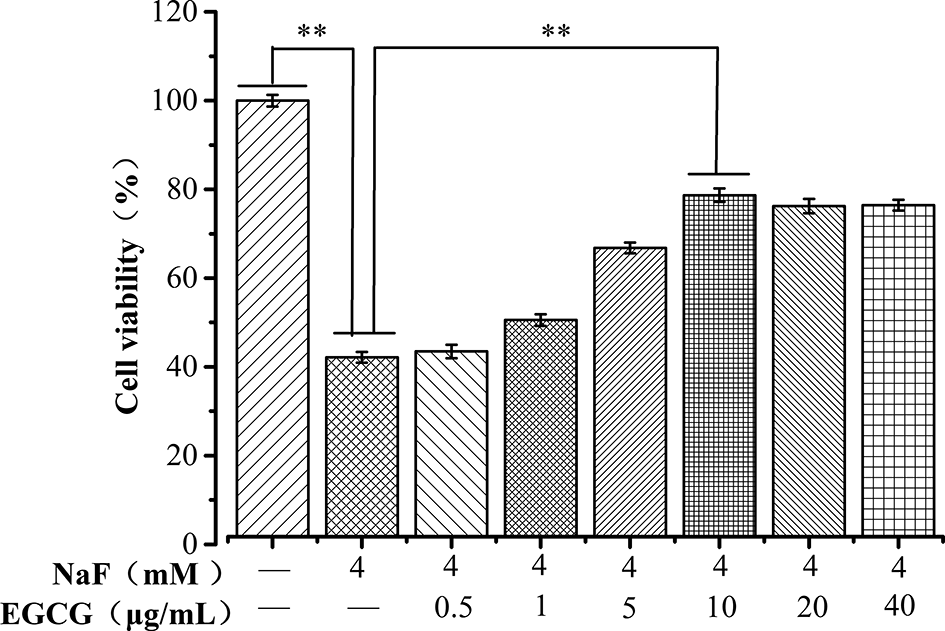
Effects of tea polyphenols on the viability of Neuro-2a cells induced by NaF. **Significant difference (P < 0.05).
The intracellular reactive oxygen species (ROS) levels were analyzed by dichloro-dihydro-fluorescein diacetate (DCFH-DA), which is cell permeable and sensitive to oxidation inside cells. After 24 hours of NaF incubation with or without EGCG at different doses, intracellular ROS generation was tested in Neuro-2a cells. As shown in Figure 5, when the cells were not treated with drugs, the ROS level was low, and the relative fluorescence intensity was 1.75. Compared with the control group, the Neuro-2a cells treated with 4 mM NaF showed significantly elevated intracellular ROS production from 1.75 to 2.66 (P < 0.05). These findings indicate that NaF could significantly damage the cells. Pretreatment with EGCG at different concentrations in the NaF-induced Neuro-2a cells significantly reduced the intracellular ROS levels, causing a decrease in ROS production (P < 0.01) compared with that of the NaF group.

Tea polyphenols suppressed NaF-induced generation of ROS in Neuro-2a cells. **Significant difference (P < 0.05); ***Very significant difference (P < 0.01).
We further investigated the effects of NaF on cell apoptosis in Neuro-2a cells and the potential protective effects of EGCG. The Neuro-2a cells were incubated for 24 hours, and apoposis was observed with a fluorescence microscope and flow cytometry. The Annexin V-fluorescein isothiocyanate (FITC) fluorescence signal is green and the propidium iodide (PI) fluorescence signal is red under a fluorescence microscope, which shows the apoptosis of the Neuro-2a. Enhanced cell green fluorescence was observed after exposure to NaF (Figure 6). Flow cytometry analysis and fluorescence microscope confirmed the decrease in green fluorescent intensity by EGCG treatment (Figure 6). The results showed that EGCG protects the Neuro-2a cells from NaF-induced apoptosis. In addition, no cell death was observed by PI staining. Therefore, NaF can mainly induce Neuro-2a cells apoptosis at an early stage.

(a) Apoposis of Neuro-2a detected by flow cytometer. (b) Apoposis of Neuro-2a observed under a fluorescence microscope.
A previous study found that tea polyphenols can alleviate motor impairments, DAergic neuronal injury, and α-syn aggregation induced by N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine in nonhuman primates. 9 Yang et al. also found that EGCG has neuroprotective effects on outer retinal degeneration after sodium iodate insult. 10 Our study confirmed that EGCG could attenuate the Neuro-2a cytotoxicity induced by NaF. This finding may be due to the ROS scavenging activity of EGCC. Excessive fluoride intake can consume antioxidants and free radical scavengers in the body, causing free radical production that disrupts the antioxidant defense system. The body’s oxidative stress level increases, causing cell damage. 11,12 EGCG has attracted much attention because of its strong antioxidant activity. However, the related molecular biological mechanisms need to be further investigated. Our findings provide a theoretical basis for the intervention treatment of fluorosis.
Experimental
Chemicals
NaF was obtained from Tianjin Jinbei Fine Chemical Co., Ltd. (Tianjin, China), EGCG (>98% purity) from Aladdin (Shanghai, China), minimum essential medium (MEM) and fetal bovine serum (FBS) from Gibco (Grand Island, New York State, USA), and penicillin-streptomycin solution from Wolcavi (Beijing, China). CCK8 and dimethyl sulfoxide were purchased from Solarbio (Beijing, China), and Annexin V-FITC/PI Apoptosis Detection Kit from Beyotime (Beijing, China).
Cell Culture
Neuro-2a cells were provided by the Institute of Cell Research, Institute of Life Sciences, Chinese Academy of Science. Cells were then cultured in MEM supplemented with 10% FBS and 1% penicillin and streptomycin. Isolated primary cells were incubated at 37°C in 5% CO2 at 95% humidity. The medium was replaced twice a week in all experiments. The Neuro-2a cells were cultured in serum-free medium with MEM for the proper time for drug treatment, and then, the cells were harvested and extracted for analysis.
Determination of ROS
A dichlorofluorescein (DCF) staining assay was used to detect intracellular ROS. 13 DCFH-DA is a fluorescent probe that specifically combines with ROS. DCFH-DA was diluted to a final concentration of 10 µM in serum-free medium to make a 1:1000 dilution. Briefly, the Neuro-2a cells were washed with PBS and incubated with fresh MEM containing 10 µM DCF at 37°C for 35 minutes. Then, 1 × 106 cells were harvested and resuspended in PBS. The mean fluorescence intensity of the intracellular ROS after treatment was determined by using a fully automatic fluorescence enzyme labeling instrument at an excitation wavelength of 488 nm and an emission wavelength of 525 nm. In the control group, the positive control substance Rosup was added at the same 1:1000 dilution.
Determination of Cell Apoptosis
Cell apoptosis was detected with an Annexin V-FITC/PI apoptosis detection kit. The cell medium was collected, and the cells were washed with PBS, harvested with trypsin without EDTA in a 1.5 mL Eppendorf (EP) tube and washed. The cells and medium were collected in an EP tube and centrifuged at 1000 rpm for 5 minutes. The medium was removed and the cells were resuspended in Annexin V-FITC buffer. The cells were incubated with 5 µL of V-FITC and 10 µL of PI in the dark at 25°C for 15 minutes and detected with an FC 500 MPL flow cytometer.
Statistical Analysis
The results are expressed as the mean ± standard deviation of triplicate measurements. Student’s t-test was applied for the statistical comparison. A value of P < 0.05 was considered significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangxi, China (No.2019JJB130122 and 2019JJA130116).
