Abstract
Spina Gleditsia has been traditionally used as a lead ingredient of many formulas for cancer treatment in Traditional Chinese Medicine. This work aimed to reveal the phytochemical foundation of Spina Gleditsiae and underpin a fundamental understanding of its anticancer activities. The growth inhibition activities were determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay of 7 solvent extracts of Spina Gleditsia in the human liver cancer cell line HepG2. The activities were then mapped onto the secondary metabolite profile of crude extracts by principal components analysis (PCA) of High Performance Liquid Chromatography-Ultraviolet (HPLC-UV) data. The top 5 PCA components of the map discriminated extract activities, mainly based on the differential content of 3 compounds. The identified compounds were then tested individually using cytotoxic assay on cancer cells in vitro. Principal components analysis constructed secondary metabolite mapping quickly identified functional compounds from the crude herbal extracts. (2R,3R)-7,3′,4′-Trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin inhibited the growth of human liver cancer cell line growth with activities of 38.6 ± 1.9, 40.7 ± 0.9, and 41.5 ± 3.1 μg/mL, respectively, comparable with that of cisplatin (IC50: 41.6 ± 2.9 μg/mL). (2R,3R)-7,3′,4′-Trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin are, for the first time, identified as the anticancer constituents of Spina Gleditsia. It thus strongly underpins a fundamental understanding of the anticancer activities of Spina Gleditsia and supports its ongoing clinical uses in China.
Spina Gleditsiae, the dry thorns of Gleditsia sinensis Lam, used as herbal decoctions, is an important herbal medicine with various therapeutic applications in traditional and folk medicinal systems of East Asian countries, such as China, Korea, and Japan. 1 In the Chinese Pharmacopoeia, Spina Gleditsiae has been officially recorded as a source of drugs with detoxication, detumescence, apocenosis, and antiparasite effects. 2 Moreover, a hot water infusion of Spina Gleditsiae, either alone or as a part of the Traditional Chinese Medicine (TCM) formula, is claimed to be used for the treatment of cancer-related symptoms in clinics of China.1 However, the clinical evidence base for this traditional use is sparse. One of the fundamental variables in evaluating such integrated therapy is the chemical profile of plant materials, a point usually represented through identification and quantification of functional compounds within complex natural products or clinical formulations.
Concerning the pharmacological aspect of Spina Gleditsiae, scientific evidence for its anticancer effects have recently been reported in the literature. In the rat hepatocellular carcinoma model implanted with cancerous Walker-256 cells, the n-butanol fraction of G. sinensis significantly restored liver morphology and dramatically induced apoptosis of cancer cells. Its pharmacological effects were determined to be regulatory effects on miRNAs, including miR-21/181b/183. 3 In both in vitro and in vivo colon cancer HCT116 models, the ethanol extract of Spina Gleditsiae inhibited the growth of cancer cells with an IC50 at 600 μg/mL and significantly reduced tumor size in HCT116-xenografted tissues. These activities were associated with the regulation of expression of ERK phosphorylation, p27, and matrix metalloproteinase-9 (MMP-9). 4 The ethanol extract of Spina Gleditsiae also led to growth inhibition, with G1 phase cell cycle arrest at a concentration of 400 μg/mL in human SNU-5 gastric cancer cells through activating p38 MAP kinase, inducing p21WAF1, and downregulating cyclin D1/CDK4 and cyclin E/CDK2 complexes. 5 Its water extract presented a similar inhibitory effect on HCT16 cells through increasing p53 levels; reducing cyclins and cyclin-dependent kinases; and phosphorylating ERK, p38 MAP kinase, and JNK. 6 Moreover, oral administration of the water extract of Spina Gleditsiae (25 mg/(kg·day)) significantly reduced the tumor size of the animal xenografted with PC3 prostate cancer cells. 7
The functional compounds, belonging to 5 major groups of secondary metabolites (triterpenes, sterols, flavonoids, phenolic, and alkaloids), have been isolated and elucidated from Spina Gleditsiae. 8,9 Previously, triterpenoid saponins and polysaccharides had been found to possess great anticancer activities on cancer cell lines in vitro. 9,10 However, the chemical foundation underlying the anticancer activities of Spina Gleditsiae has not been systematically investigated as yet, and an overall perspective on its anticancer phytochemical foundation still remains an ongoing challenge. The multivariate data analysis (MVA)-guided method, developed recently and characterized as minimum consuming of time, raw materials, and solvents, could provide an advanced technique for the fast screening of bioactive agents of complex medicinal products. 11,12 Therefore, a MVA-guided method was employed in this study to identify the anticancer components of Spina Gleditsiae.
To underpin a fundamental understanding of the anticancer activities of Spina Gleditsiae, this study constructed an HPLC secondary metabolite map using the MVA-guided method to clarify the anticancer compounds of Spina Gleditsiae on a human liver cancer cell line. It was thus expected to reveal the phytochemical foundation of Spina Gleditsiae and support its ongoing clinical uses in China.
Materials and Methods
Preparation of Extract and Standards
Spina Gleditsiae was purchased from Hao Yi Sheng Chinese herbal medicine store, Jinghua Street, Luoyang. The original plants were collected in Funiu Mountain (33 N latitude and 111 E longitude), Henan province, China (May 2018) and the identity kindly confirmed as G. sinensis Lam by Associate Professor Xinsheng Wang at the School of Chemical Engineering and Pharmaceutics, Henan University of Science and Technology. Voucher specimens have been deposited in the Natural Products Research Laboratory, School of Chemical Engineering and Pharmaceutics (access number: JGG2018-072).
Spina Gleditsiae was minced and portions of the minced material (10 g) were each mixed with 100 mL of different solvents: 10%, 25%, 45%, or 70% EtOH and extracted by shaking for 24 hours at room temperature. The solvents were completely removed at 35°C under reduced pressure and the residue freeze-dried overnight. Minced materials (200 g) were also extracted twice with 500 mL methanol and agitated at room temperature for 24 hours. The extracts were filtered, the solvent was completely removed at 35°C under reduced pressure, and the residue lyophilized overnight. The dry residues were then redissolved in 100 mL 10% methanol and sequentially extracted twice with 200 mL of dichloromethane, ethyl acetate, and n-butanol.
Isovitexin (≥98% purity) and cisplatin were purchased from Solarbio (Beijing, China). (2R,3R)-7,3′,4′-Trihydroxy-5-methoxy-flavanonol and (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol were prepared as reported before. 13 The standards were dissolved in 0.01% dimethylsulfoxide (DMSO, Solarbio, Beijing, China) in PBS solution.
Cancer Cell Culture
The human hepatoma cell line HepG2 was purchased from Beinglay Biotech (Wuhan, Hubei, China). Cells were grown in Dulbecco modified Eagle medium (DMEM) medium (Solarbio, Beijing, China) supplemented with 10% fetal bovine serum, 100 µg/mL penicillin, and 100 µg/mL streptomycin in a 5% carbon dioxide humidified incubator at 37°C. Experiments were performed when cells were approximately 80% confluent.
Growth Inhibition as Measured by 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Assay
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Solarbio, Beijing, China) assay was performed as follows. Briefly, 200 µL cell suspensions were seeded at a concentration of 1.5 × 104 cells/mL in a 96-well plate. After overnight incubation, Spina Gleditsiae extracts (200-75, 150-25, or 100-5 μg/mL), (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin (50-1.5625 μg/mL) were added in 10 µL aliquots. In this work, the systematic dilution of compounds was not used as different compounds presented greatly different levels of cytotoxicity against cancer cells. The dilution ranges for each compound were determined through several preliminary experiments. The current dilution could present the best cytotoxic tendency and thus gave precise IC50 values. The negative control was 10 µL of 0.01% DMSO in Phosphate Buffered Saline (PBS) solution. After 24 hours incubation, 20 µL MTT solution was added and incubated at 37°C for 4 hours. The optical density (O.D.) was measured at 590 nm using a Multiskan Spectrophotometer. Growth inhibition was determined using the following formula: Growth inhibition percentage = ((control O.D. − sample O.D.)/control O.D.) × 100%.
HPLC Instrumentation
The chemical profiles of samples and standards were obtained using a Waters series HPLC system comprising a Waters e2695 Separations Module with degasser, a variable wavelength 2489 UV/Vis detector, and an autosampler. The UV detector was set at 270 nm and the data were acquired using Empower 3 software. The column used was a Thermo C18, 5 µm (250 × 4.6 mm) maintained at ambient room temperature. Gradient elution of the samples and standard were performed using 0.01% formic acid (eluent A) and methanol (eluent B). The separation conditions for HPLC were developed earlier in this study. The initial conditions of gradient elution were 95% eluent B with a linear gradient to 66% from 1 to 15 minutes, to 44% from 15 to 33 minutes, to 40% from 33 to 45 minutes, and to 95% from 45 to 50 minutes, with this proportion being maintained for 5 minutes. The column was then returned to the initial condition at 55 minutes. The flow rate was 1 mL/min. Dry residues of Spina Gleditsiae extracts were dissolved in methanol at 5 mg/mL. The sample injection volume was 10 µL. To validate the constancy of data acquired and machine operation conditions in the following PCA and partial least squares (PLS) analysis, 3 injections were performed for each sample and standard.
HPLC Quantitation of (2R,3R)-7,3′,4′-Trihydroxy-5-Methoxy-Flavanonol, (2R,3R)-5,7,3′-Trihydroxy-4′-Methoxy-Flavanonol, and Isovitexin
The calibration curves of (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were obtained by preparing stock solutions (500 µg/mL) in methanol. Subsequent dilutions were made to give 500, 400, 300, 200, 100 and 50 µg/mL. Ten microliters of each final concentration was injected in triplicate. The linearity of the calibration curves was determined (R 2 > 0.991). The intraday precision was calculated by comparing the area obtained for 3 injections of standards within the day. For the interday precision, the areas of standard in the calibration curve for 3 days were summed and compared.
Multivariate Data Analysis
Principal components analysis was adopted and performed using SIMCA-P 16.0 MVA software (Umetrics, Sweden). HPLC chromatograms of Spina Gleditsiae extracts at 270 nm comprised 3301 discrete regions by data acquisition every 0.017 seconds from 0.00 to 55.00 minutes. The resulting data were formatted into Microsoft Excel 2010 and then exported into SIMCA-P 16.0 for analysis. Five principal components were chosen for PLS analysis. The process of generating a PLS model on randomly chosen samples followed by validation was repeated in order to ensure all extracts were excluded once.
Statistical Analysis
Each sample was tested in triplicate and each experiment was repeated in triplicate again. The data are shown as mean ± standard deviation (SD). Statistical comparison among treatments was carried out using one-way analysis of variance (ANOVA). The statistical significances between control and sample groups were calculated by the Student’s t-test. Data were taken as significant where P < 0.05.
Results
Growth Inhibitory Effects of Spina Gleditsiae for Human Hepatoma Cells In Vitro
The cytotoxic effects of different Spina Gleditsiae crude extracts, including 10% ethanol, 25% ethanol, 45% ethanol, 70% ethanol, dichloromethane, ethyl acetate, and n-butanol extracts, were determined on human hepatoma 7721 cancer cell line in vitro using MTT assay. All Spina Gleditsiae extracts presented growth inhibitory effects on cancer cells in a dose-dependent manner. According to the IC50 values presented in Table 1, the cytotoxic levels could generally be divided into 3 groups. The ethyl acetate extract showed the strongest activity (IC50: 78.9 ± 11.4 µg/mL) against cancer cells. The 45% ethanol, 70% ethanol, and dichloromethane extracts presented moderate effects with IC50 values from 136.5 to 171.9 µg/mL. The least cytotoxic were the 10% ethanol, 25% ethanol, and n-butanol extracts of Spina Gleditsiae with IC50 values around 190 µg/mL.
IC50 Values (μg/mL) of Crude Extracts of Spina Gleditsiae on Human Hepatoma HepG2 Cancer Cell Line.

HPLC-PCA Map
As shown in Figure 1, each point represents 1 HPLC dataset for a given extract and each extract was tested in triplicate. On supervision, as illustrated by the ellipses superimposed in Figure 1, all 21 samples of crude Spina Gleditsiae extracts could be discriminated into 3 groups. This map demonstrates good correlation between composition and inhibition. Crude extracts with IC50 values >186 µg/mL, and 10% ethanol, 25% ethanol and n-butanol extracts are considered as poor inhibitors and located in the top of the map. Those of 45% ethanol, 70% ethanol, and dichloromethane show intermediate inhibition (136 < IC50 < 171 µg/mL) and located in the left lower part. The more potent inhibition (IC50 = 78.9 µg/mL) is located in the crude ethyl acetate extract in the right lower part of the map. Furthermore, as the variance was mainly due to the top 5 components (PC1-5, 97.8%), it could determine the contributing variables using the PCA Loading and Contribution Plot. According to the location of correlated peaks in the Biplot combining the score and loading plots, HPLC peaks with retention times of 19.8, 21.7, and 24.6 minutes, in particular, had the highest contribution to the most active ethyl acetate extracts. Under the same HPLC operation condition, the standard compounds (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were gradient eluted from the column at retention times of 19.835, 21.749, and 24.557 minutes, respectively. The identities of these contribution compounds were thus confirmed by spiking samples with standards isolated and reported previously. 12 This indicated that the variance of growth inhibitory effect of Spina Gleditsiae extracts on HepG2 cancer cells depended on the concentration of these compounds, the chemical structures of which are shown in Figure 2.

Principal component analysis scores plot for Spina Gleditsiae crude extracts. Each point on the plot represents 1 HPLC dataset of an extract, and points with the same name indicate replicates (n = 3); the percentage variation contributed by PC1 and PC2 are 53.3% and 26.5%, respectively.
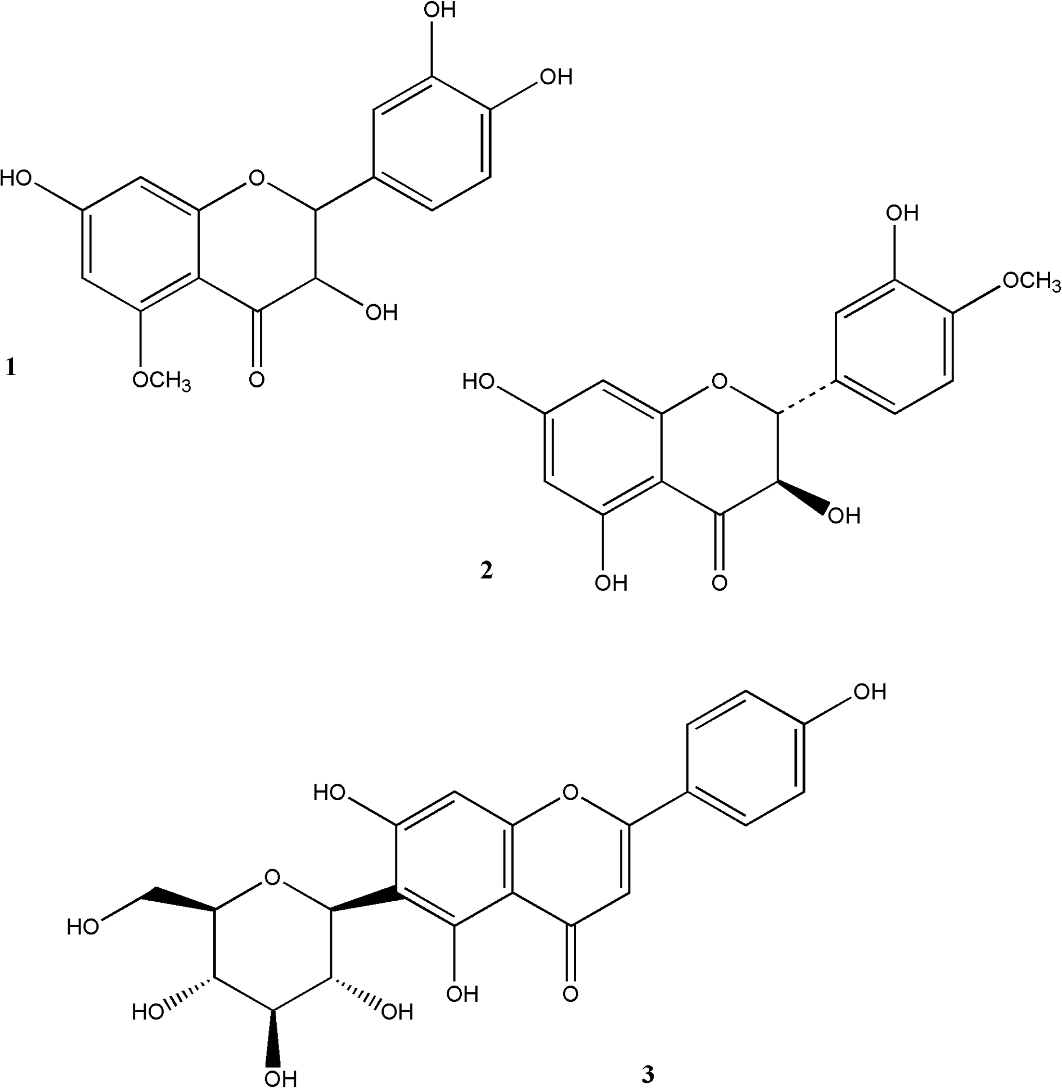
The chemical structures of (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol (1), (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol (2), and isovitexin (3).
Partial Least Squares Prediction of Inhibition by Spina Gleditsiae Crude Extracts
The supervised classification and PLS regression model was further employed to provide a correlation between complex multivariate chemical datasets and the experimental IC50 of Spina Gleditsiae extracts. The IC50 values of the crude extracts against HepG2 cells were imported as the y-matrix in the corresponding HPLC-PLS regression model. Five components were used for the model, with variance (R2 ) values at 0.988 and cross-validated variance (Q 2) values at 0.965. The value of Q 2 above 0.5 was considered as a good prediction of the constructed model. The overall predictions for each extract are summarized in Table 1. The correlation coefficient between predicted vs actual values was 0.999.
Determination of (2R,3R)-7,3′,4′-Trihydroxy-5-Methoxy-Flavanonol, (2R,3R)-5,7,3′-Trihydroxy-4′-Methoxy-Flavanonol, and Isovitexin in Spina Gleditsiae Extracts
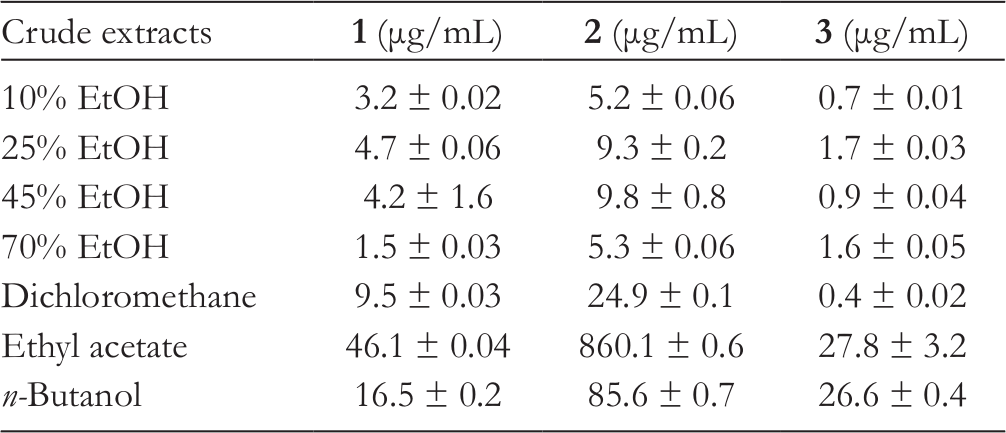
The concentrations of (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin in 7 extracts were determined by standard calibration curves and are summarized in Table 2. Among them, (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol (1.5-46.1 µg/mL) and (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol (5.2-860.1 µg/mL) presented relatively high amounts in all Spina Gleditsiae extracts, while the concentration of isovitexin (0.44-27.79 µg/mL) was lowest. The ethyl acetate extract contained the highest amount of 3 active compounds tested (46.1, 860.1, and 27.8 µg/mL), and their doses were higher in dichloromethane (9.8, 24.9, and 0.44 µg/mL) and n-butanol (16.5, 85.6, and 26.6 µg/mL) extracts than those of ethanol extracts (<4.67, <9.84, and <1.70 µg/mL) of Spina Gleditsiae.
The Concentration (μg/mL) of (2R,3R)-7,3′,4′-Trihydroxy-5-Methoxy-Flavanonol (1), (2R,3R)-5,7,3′-Trihydroxy-4′-Methoxy-Flavanonol (2), and Isovitexin (3) Within the Crude Extracts of Spina Gleditsiae.
Validation of Spina Gleditsiae Inhibitors of Human Cancer Cells
Based on the analysis above, (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were identified as the major anticancer compounds within Spina Gleditsiae. To validate further this conclusion, the cytotoxicity of these 3 compounds was tested on HepG2 cells in vitro using MTT assay. As shown in Figure 3, all the compounds could inhibit the growth of the cells in a dose-dependent manner. These results are comparable with that of the antineoplastic cisplatin (IC50 value: 41.6 ± 2.9 µg/mL). (2R,3R)-7,3′,4′-Trihydroxy-5-methoxy-flavanonol presented the strongest cytotoxicity among the compounds tested. (2R,3R)-5,7,3′-Trihydroxy-4′-methoxy-flavanonol and isovitexin presented a similar cytotoxic effect as the positive control cisplatin in the doses tested (50-1.56 µg/mL). The IC50 values for (2R,3R)−7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were found to be 38.6 ± 1.9 µg/mL (121.5 ± 5.9 µM), 40.7 ± 0.9 µg/mL (128.2 ± 2.7 µM), and 41.5 ± 3.1 µg/mL (128.2 ± 9.4 µM), respectively.

The growth inhibitory effects of (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin on human hepatoma HepG2 cancer cells.
Discussion
In this study, HPLC-PCA secondary metabolite maps revealed that (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin acted as three of the functional anticancer compounds within Spina Gleditsiae. Nevertheless, a HPLC-PLS regression model was developed which could successfully make the prediction of inhibition by Spina Gleditsiae crude extracts.
The complicated and similar chemical profiles of crude extracts from natural products raise the issues which always preclude visual and manual analysis to correlate certain compounds with the bioactive effects. 14 Thus, in recent years, MVA has been employed to model phytochemical data and to aid determination of discriminative bioactive constituents. 11,12 In this study, a HPLC-PCA map was constructed to model the metabolic profiling of secondary metabolites and successfully determine the anticancer compounds of Spina Gleditsiae. Compared with the traditional bioassay-guided fractionation, the identification and grouping of functional compounds from plant extracts was much quicker and more efficient through the multivariate analysis mediated secondary metabolite mapping method. 15 As shown in Table 2, the ethyl acetate extract with the most anticancer potency (IC50 = 78.9 µg/mL) had significantly higher concentrations of (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol (46.1 µg/mL), (2R,3R) -5,7,3′-trihydroxy-4′-methoxy-flavanonol (860.1 µg/mL), and isovitexin (27.8 µg/mL) than those of the other extracts. However, the dose levels of 3 compounds did not show significant difference in the Spina Gleditsiae extracts of intermediate and poor active groups, thus leading to the similar cytotoxic effects of those extracts. Overall, (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were predicted as the main inhibitors of Spina Gleditsiae against human liver cancer cells.
The anticancer activities of (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were further confirmed in the in vitro validation tests, and these effects were comparable with that of the antineoplastic cisplatin. Claimed as an anticancer drug in the TCM system, the cytotoxic effects of Spina Gleditsiae have been reported in several in vitro and in vivo models. 3 -7 However, the phytochemical foundation of the anticancer activities of Spina Gleditsiae still needed further elucidation. Previously, 3 compounds identified from Spina Gleditsiae were known to inhibit the growth of human SK-Hep-1 liver cancer cells in vitro. p-Hydroxyl cinnamicaldehyde exhibited cytotoxicity with an IC50 value of 38.2 µM, while trans-coniferyl aldehyde and sinapaldehyde showed moderate effects with IC50 values of 58.1 and 54.6 µM, respectively. 16 In this study, (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were, for the first time, identified as the main anticancer constituents of Spina Gleditsiae. Among them, isovitexin, a glycosylflavonoid extracted from many plants, has been reported to exhibit cancer suppressive activities on various types of cancer. 17 Recently, Lv and Qiao reported that isovitexin could significantly suppress the growth of liver cancer cells HepG2 and SK-Hep1 in vitro, and the tumor growth of HepG2 tumor-bearing nude mice in vivo. The compound induced mitochondrial apoptosis through upregulation of Bax, cleaved Caspase-3, poly (ADP-ribose) polymerase (PARP), and cytoplasm Cyto-c, and also caused autophagy of cancer cells by the enhancement of LC3II, autophagyrelated protein (Atg) 3, Atg5, and Beclin1. 18 Isovitexin was also found to induce the apoptosis of human osteosarcoma U2OS and MG63 cells through decreasing the expression of CD133, CD44, ABCG2, and ALDH1. Moreover, it inhibited tumor growth of U2OS-SC xenografts in nude mice through decreasing CD133 protein levels and Bcl-2 levels; proliferating cell nuclear antigen (PCNA) expression, DNMT1 activity and expression; and increasing the apoptotic index and miR-34a levels. 19 Taken together, the anticancer function reported in the literature above confirmed the anticancer role of isovitexin for Spina Gleditsiae in this study.
The finding in the work first provided scientific evidence to support that (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were the major anticancer constituents of Spina Gleditsiae. However, their anticancer effects need to be further validated in other cancer models in vitro and in vivo. The underlying anticancer mechanisms also need to be elucidated in future studies.
Conclusion
In this study, the Spina Gleditsia flavonoid constituents (2R,3R)-7,3′,4′-trihydroxy-5-methoxy-flavanonol, (2R,3R)-5,7,3′-trihydroxy-4′-methoxy-flavanonol, and isovitexin were identified as the growth inhibitors of human liver cancer cells with activity comparable with that of the antineoplastic drug cisplatin. This is beneficial to underpin the phytochemical foundation of its anticancer activities, which expand the fundamental understanding of Spina Gleditsia and support its current medicinal uses in China. Moreover, PCA-mediated secondary metabolite mapping was employed to identify quickly the anticancer compounds from the crude plant extract. This approach could be an important first step to qualify herbal products required for medicinal uses. Further anticancer research of Spina Gleditsia may include the elucidation of detailed anticancer mechanisms at the molecular levels and purification of more phytochemicals, especially flavonoids, with the aim of obtaining powerful anticancer agents and lead compounds.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Project no. U1504830).
