Abstract
Jaltomata procumbens is a semidomesticated plant species used as food, and the presence of several types of secondary metabolites are detected in one of its forms. Growth conditions, the electrical conductivity (EC) of the nutrient solution, and the pruning management may influence the secondary metabolite content. The objective was to evaluate the effects of EC and pruning on the metabolite content of 2 J. procumbens forms, in greenhouse and hydroponics. Treatments included 2 forms (erect and decumbent), 3 EC levels (1, 2, and 3 dS m−1), and 2 pruning levels (with and without pruning). The contents of phenolic acids, flavonoids, and terpenoids were evaluated. EC and pruning management differentially influenced the contents of each group of metabolites in both forms in a combined and individual manner, with EC exerting a stronger effect than pruning. A total of 7 phenolic acids, 4 flavonoids, and 5 terpenoids were detected: the erect form contained significant amounts of syringic acid, rutin, and carnosol at 33.44, 36.43, and 0.26 mg 100 g−1 of dry weight, respectively, whereas the decumbent form had high contents of gallic and rosmarinic acids and morin at 59.03, 28.31, and 61.54 mg 100 g−1 of dry weight, respectively. Considering the variability observed depending on the metabolite group of interest, the specific management of EC and pruning is required for each form.
Keywords
Introduction
The Jaltomata procumbens species, in domestication process and native to Mexico that grows erect and bears a dark-blue fruit, is viable for future food purposes. Different forms (Fms) in specific regions of the country exhibits decumbent growth and bears green fruit in the mature stage. Fruits from these Fms are collected and consumed as food. 1
J. procumbens contains important nutrients,2,3 and beneficial components, such as carotenoids. In addition, the Fm bearing dark-blue fruit exhibits antioxidant activity. 4 The Fms show considerable potential for study in controlled environments, 1 wherein technologies such as greenhouses and hydroponics are used. 5
The aforementioned technologies are useful for determining the viability of new plant genetic resources for agricultural purposes; they facilitate studies on the effects of changing environmental factors on the growth and development of plant because they allow for experiments wherein the species are subjected to different conditions from their natural environment. 6 One important aspect in these studies is fruit quality, which is a characteristic that has been virtually ignored in breeding programs. 7 While studying fruit quality, the analysis of secondary metabolites is useful to evaluate the response of the plant to environmental factors, 8 which are modified by cultivation conditions. Ultimately, this response is reflected by the fruit quality.
Among the factors that affect the contents of secondary metabolites under cultivation conditions are salinity and pruning. Salinity is related to the number of ions available in the root zone of the plant and is determined using electrical conductivity (EC): high EC values can generate salinity stress, which affects nutrient solution uptake and plant growth,9,10 while a low EC affects growth and yields due to nutrient deficiency. 11 Pruning is another factor that affects metabolite synthesis12,13; this agricultural practice also allows regulating vegetative and reproductive growth, and facilitating plant management in controlled environments, thereby promoting the removal of reserve substances and improving fruit quality. 1 These factors influence the antioxidant system by inhibiting or promoting enzymatic activity or gene expression involved in the biosynthesis of metabolites.10,14
Therefore, since the secondary metabolite content is affected differentially by growth conditions, and under the hypothesis that EC and pruning management influence secondary metabolite content, this research aimed to evaluate the effect of EC and plant pruning in 2 J. procumbens Fms, erect and decumbent, on the contents of phenolic acids, flavonoids, and terpenoids under greenhouse and hydroponic conditions.
Materials and Methods
The research was performed in a greenhouse at Campus Montecillo, Colegio de Postgraduados, Mexico, from July to December 2019. Water and nutrients were supplied via drip irrigation in an open hydroponic system.
Erect and decumbent Fms of J. procumbens from Tlaxcala, Mexico (19°15´ N and 97°53´ W, at 2500 m altitude) were evaluated. The region experiences sub-humid template climate C(w1) y C(w2) and has an average annual temperature between 12°C and 18°C. Ripe fruit seeds recollected in 2015 were used; the seeds were rinsed off with tap water, dried under shadow at room temperature (22°C), stored in 30 mL plastic bottles, and refrigerated at 4°C.
Experiment Management
The plant material was obtained from a previous experiment to break species dormancy. A germination chamber was used under diurnal (12 h at 30°C) and night conditions (12 h at 20°C). The seedlings were transferred to 24 mL plastic cups with peat moss as substrate and watered with distilled water; when the first true leaves appeared, the plants were placed in a greenhouse with 40% shade mesh and watered with tap water (pH 7.7 and EC 0.5 dS m−1). Thereafter, the plants were transplanted into 265 mL styrofoam cups containing red tezontle as substrate (4-10 mm particle diameter) and irrigated with Steiner 15 nutritive solution at 1 dS m−1 EC until they reached a height of 15 cm. After 40 days of sowing, the plants were transplanted into black 40 × 40 polyethylene bags (13L) containing the substrate red tezontle (diameter = 12 mm, density = 0.78 g cm−3, and humidity retention = 21.87%). The main stems and branches were moored. Steiner nutritive solution of 3 concentrations were applied with macronutrients and micronutrients supplied proportionality to each EC level: 1, 2 and 3 dS m−1, and was dosed starting from transplantation. The irrigation volume applied daily ranged between 0.23 and 1 L per plant, depending on the growth stage. The pH was maintained within the range of 5.5 to 6.5. The pH and EC were monitored using a portable equipment (Combo Waterproof HI-98129). For pruning management, treatments with and without pruning (WP and WoP, respectively) were applied. In the pruned plants, basal and lateral shoots were eliminated, thereby allowing normal plant ramification. Pests, such as potato flea beetle (Epitrix cucumeris), thrips (Thysanoptera sp.), green (Acrosternum hilare) and brown (Euschistus servus) stink bugs, beet armyworm (Spodoptera exigua), and aphid (Aphididae sp.) were presented, and pathogens, such as powdery mildew (Oidium sp.) and Cercospora leaf spot (Cercospora sp.), were also present. Beet armyworms were eliminated manually, whereas other pathogens and pests were eliminated using mancozeb (3.5 g L−1), abamectin (3 mL L−1), and sodium bicarbonate (20 g L−1).
Experimental Design
The arrangement of the treatments was a factorial design with the following factors and levels: 2 Fms (erect and decumbent), 3 EC levels (1, 2, and 3 dS m−1), and 2 pruning managements (WP and WoP), providing 12 treatments in total. The treatments were distributed in a completely randomized design with 4 replications. The experimental unit was one plant per pot, yielding 48 experimental units in total.
Sample Collection and Preparation
From each experimental unit, 100 g of physiologically mature fruit were harvested, ground, stored in 250 g capacity polyethylene bags at −70°C, and then lyophilized.
Extraction
From each lyophilized sample, 0.5 g of material was weighed and placed in 15 mL capacity conical centrifuge tubes. Thereafter, 5 mL of 80% methanol was added to the sample, and the mixture was stirred with a vortex for 10 s (G560 model, Scientific Industries) and subjected to ultrasound-assisted extraction (Branson B-220) for 30 min, in 10 min intervals, with 5 min rest between each interval. Subsequently, the sample was centrifuged at 5000 rpm for 5 min (centrifuge 3765 model, 291840 series, Thermo Scientific). Finally, using a 5 mL syringe, 1 mL of the extract was taken and placed in a 1 mL capacity vial. These samples were stored at 4°C until analysis using high-performance liquid chromatography (HPLC).
Secondary Metabolite Identification Using HPLC
The phenolic acids and flavonoids were identified based on the method of Aguiñiga-Sánchez et al 16 with modifications. A liquid chromatograph (agilent 1100 model) with an automatic injector (1200 model) and diode array detector (1100 model) was used. A Hewlett Packard Hypersil ODS column (125 × 40 mm) was employed with a gradient of (A) H2O at pH 2.5 with trifluoroacetic acid and (B) acetonitrile (ACN). The settings were as follows: flow velocity, 1 mL min−1; temperature, 30°C; variable injection volume; analysis time, 25 min; wavelengths of λ1 = 254, λ2 = 280, λ3 = 330, and λ4 = 365 nm. The absorption spectra of the analyzed samples were obtained and compared with those of the standards. Calibration curves were created at different wavelengths.
To identify the terpenoids, a Zorbax Eclipse XDB C-8 column (125 × 4.0 mm, 5 µm) was used with a gradient of (A) ACN: 80% and (B) H2O: 20%, flow velocity of 1 mL min−1, and temperature of 40°C. The wavelength used was 220 nm, and the injection volume was 20 µL. The absorption spectra of the analyzed samples were obtained and compared with those of the standards. Calibration curves were created at different wavelengths. The reference standards were from Sigma-Aldrich, USA: for flavonoids rutin, morin, and quercetin for λ = 254 nm, catechin, naringenin, hesperidin, phloretin, and phloridzin for λ = 280 nm, apigenin for λ = 330 nm, and myricetin, kaempferol, and isorhamnetin for λ = 365 nm; for phenolic acids protocatechuic, 3,5-di-hydroxybenzoic, p-hydroxybenzoic, vanillic or caffeic and b-resorcylic for λ = 254 nm, gallic, syringic, and p-coumaric for λ = 280 nm, and chlorogenic, ferulic, sinapic, and rosmarinic for λ = 330 nm; for terpenoids carnosol, stigmasterol, ursolic acid, alpha-amyrin, oleanolic acid, and β-sitosterol for λ = 220 nm.
Statistical Analysis
The obtained results were analyzed using the statistical program SAS 17 ; the analysis of variance was performed based on a completely random 2 × 3 × 2 factorial model and Tukey's test (P ≤ .05).
Results
The detected phenolic acids were sinapic, syringic, ferulic, rosmarinic, vanillic, gallic, and p-coumaric acids. The detected flavonoids included myricetin, rutin, morin, and quercetin. Finally, the detected terpenoids were carnosol, stigmasterol, β-sitosterol, and ursolic and oleanolic acids.
The Fm factor affected the contents of flavonoids, terpenoid carnosol, and phenolic acids except for those of sinapic and vanillic acids. The EC influenced the rosmarinic acid content (P ≤ .05). Fm × pruning interaction presented statistical differences in sinapic acid, whereas EC × pruning presented statistical differences in rosmarinic and vanillic acids, which indicated that the effect of pruning on these metabolites was not the same within each Fm and EC level. The highest values of variation coefficients were presented in rosmarinic acid (30.42%), sinapic acid (33.51%), ferulic acid (37.15%), quercetin (40.26%), morin (40.54%), stigmasterol (46.5%), rutin (47.74%), β-sitosterol (50.64%), p-coumaric acid (54.58%), and carnosol (74.76%).
Phenolic Acids
The erect Fm presented higher contents of syringic, ferulic, and p-coumaric acids than those of decumbent by 32.12%, 34.61%, and 67.14%, respectively. Meanwhile, the decumbent exhibited higher contents of rosmarinic (30.46%) and gallic (25.57%) acids than those of the erect Fm (Figure 1).

Content of phenolic acids of fruits of 2 Jaltomata procumbens forms, cultivated at 3 electrical conductivity levels and 2 pruning treatments. Different letters among bars indicate statistical difference (Tukey, P ≤ .05). Abbreviations: Fer, ferulic; Gal, gallic; p-cum, p-coumaric; Ros, rosmarinic; Sin, sinapic; Syr, syringic; Van, vanillic.
The EC affected the rosmarinic acid content, a decrease in content was observed with increasing EC, with a difference of 38.91% between 1 and 2 dS m−1 EC, which registered the lowest value and then the content increased at 3 dS m−1 EC, but without statistical differences. The contents of vanillic and p-coumaric acids showed a similar behavior, but the highest values were obtained at 3 and 1 dS m−1 EC, respectively. The contents of ferulic and sinapic acids showed an increase at 2 dS m−1 and then decreased with increasing EC. Syringic acid content recorded a decrease with increasing EC, while an opposite trend was observed in gallic acid, but without statistical differences. WP management favored the contents of sinapic, syringic, ferulic, rosmarinic, and gallic acids, while WoP management favored vanillic and p-coumaric acid contents, but there were no statistical differences (Supplemental Material, Table S1).
EC × Pruning Combination
Plant growth with 3 dS m−1 EC × WP registered the highest rosmarinic acid content, which was higher by 81.91% than the lowest value, which was obtained with 2 dS m−1 EC × WP. Notably, the result obtained with 3 dS m−1 EC × WP was better than those obtained individually for EC (3 dS m−1) and WP, thereby demonstrating the positive effect of the combination. Within the results obtained from combinations of WoP management and EC levels, no statistical differences in rosmarinic acid content were observed; however, its content in the fruit decreased with increasing EC (Table 1). Meanwhile, the positive effect of 1 dS m−1 EC × WoP was observed, obtaining a higher value than those obtained with WoP and 2 and 3 dS m−1 EC, individually.
Effect of EC x Pruning Combination on Phenolic Acids Content of Fruits of 2 Jaltomata procumbens Fms.

Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviations: 1 x WP, 1 dS m−1 x with pruning; 2 x WP, 2 dS m−1 x with pruning; 3 x WP, 3 dS m−1 x with pruning; 1 x WoP, 1 dS m−1 x without pruning; 2 x WoP, 2 dS m−1 x without pruning; 3 x WoP, 3 dS m−1 x without pruning; EC, electrical conductivity; Fms, forms.
For other acids, 1 dS m−1 EC × WP favored the contents of syringic and p-coumaric acids, whereas with 2 dS m−1 EC favored sinapic and ferulic acids and that with 3 dS m−1 EC favored vanillic acid; meanwhile, with 3 dS m−1 EC × WoP management favored the gallic acid content, but without statistical differences (Table 1).
Fm x EC Combination
In the erect Fm, growth at 1 dS m−1 EC favored the syringic acid content, which was 39.76% higher than that in the decumbent, which registered the highest value with 2 dS m−1 EC. In the other acid contents in the erect Fm, 1 dS m−1 EC favored the contents of rosmarinic and p-coumaric acids, while 2 dS m−1 EC favored those of ferulic, vanillic, and gallic acids, and 3 dS m−1 EC favored the sinapic acid content. In the decumbent Fm, 2 dS m−1 favored the sinapic and p-coumaric acid contents, while 3 dS m−1 EC favored ferulic and gallic acids, and 1 dS m−1 EC favored rosmarinic and vanillic acid contents, but without statistical differences (Table 2).
Effect of Fm x EC Combination on Phenolic Acids Content of Fruits of 2 Jaltomata procumbens Fms.
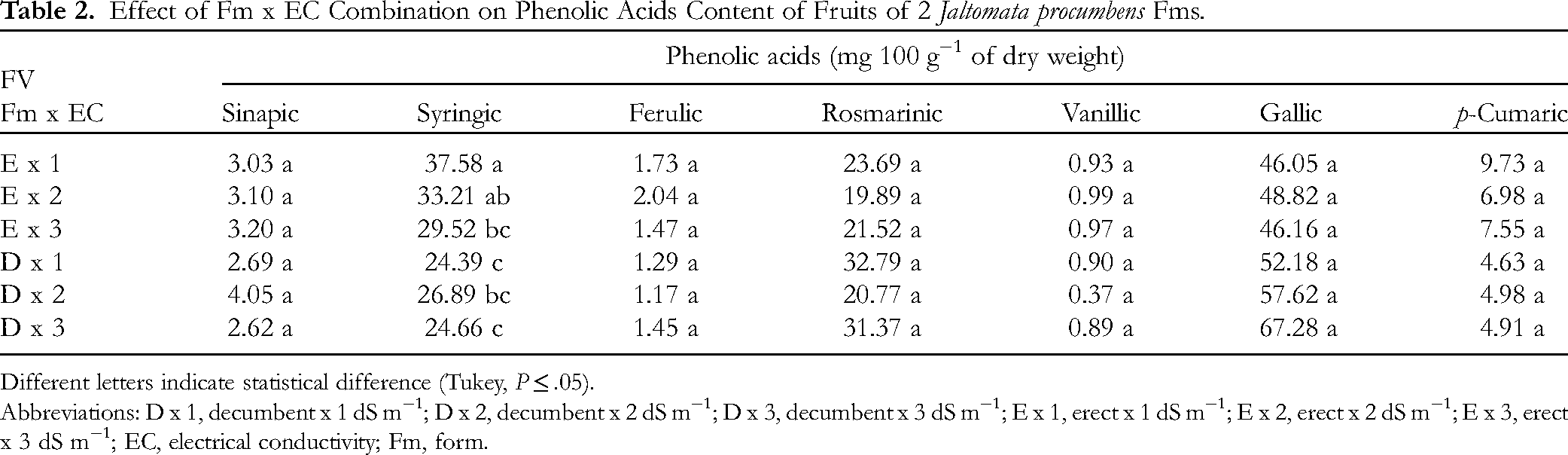
Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviations: D x 1, decumbent x 1 dS m−1; D x 2, decumbent x 2 dS m−1; D x 3, decumbent x 3 dS m−1; E x 1, erect x 1 dS m−1; E x 2, erect x 2 dS m−1; E x 3, erect x 3 dS m−1; EC, electrical conductivity; Fm, form.
Fm x Pruning Combination
WP management favored the contents of syringic, rosmarinic, and gallic acids in both Fms. For sinapic and vanillic acids, in the decumbent the WP management and in the erect the WoP management favored their contents; WoP management in both Fms positively affected p-coumaric acid, but there were no statistical differences (Table 3).
Effect of Fm x Pruning Combination on Phenolic Acids Content of Fruits of 2 Jaltomata procumbens Fms.

Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviations: D x WoP, decumbent x without pruning; D x WP, decumbent x with pruning; E x WoP, erect x without pruning; E x WP, erect x with pruning; Fm, form.
Flavonoids
The erect Fm had a 161.33% higher rutin content compared to that of decumbent Fm. By contrast, the decumbent showed superior quercetin, myricetin, and morin contents to those of the erect Fm by 37.16%, 91.19%, and 977.76%, respectively (Figure 2).
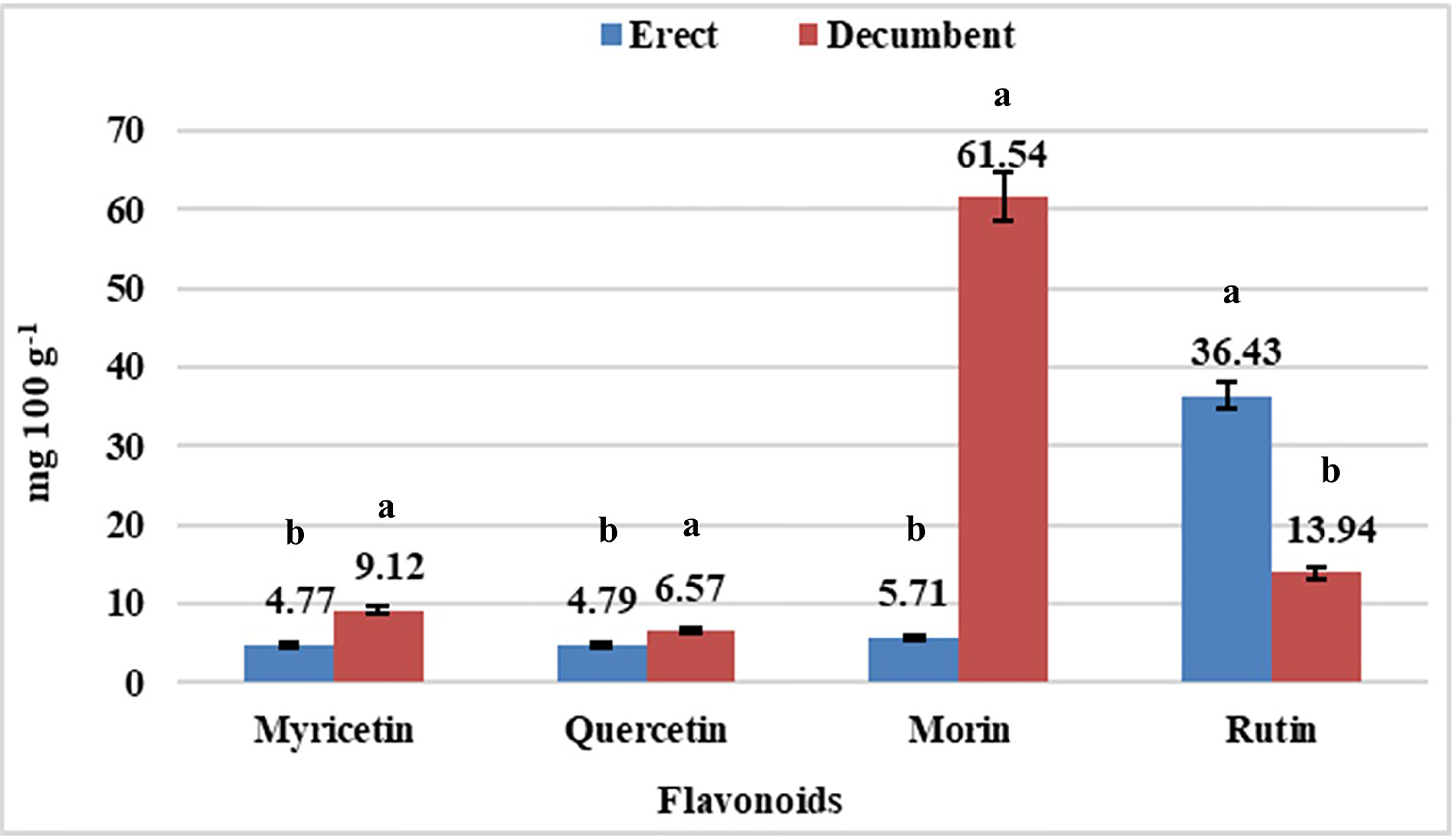
Content of flavonoids of fruits of 2 Jaltomata procumbens forms, cultivated at 3 electrical conductivity levels and 2 pruning managements. Different letters among bars indicate statistical difference (Tukey, P ≤ .05).
Considering EC, the contents decreased at 2 dS m−1 and then increased at 3 dS m−1 of EC, at which the highest contents were recorded for all flavonoids. For pruning management, WP yielded an increase in the contents of myricetin and quercetin, whereas WoP positively affected the contents of rutin and morin; however, no statistical differences were observed in both factors (Supplemental Material Table S2).
EC x Pruning Combination
Within each group of combinations of EC × WP and EC × WoP, a decrease in values was observed at 2 dS m−1 and then increased at 3 dS m−1 in myricetin, quercetin, and rutin; these trends were similar to their behavior to the EC factor individually, which possibly indicate the stronger effects of EC on their contents than those of pruning. An increase in the morin content in the fruit was observed when increasing EC in combination with WP, whereas an opposite behavior was observed in combination with WoP, but without statistical differences (Table 4). However, the positive effect of EC (3 dS m−1) × WP combination on myricetin, morin, and rutin was recorded with higher values than those recorded individually for the same level of EC and pruning. For quercetin, although the highest value was obtained individually at 3 dS m−1 EC (Supplemental Material Table S2); with 1 dS m−1 EC × WP combination, the content was higher.
Effect of EC x Pruning Combination on the Content of Flavonoids of Fruits of 2 Jaltomata procumbens Forms.

Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviations: 1, 1 dS m−1; 2, 2 dS m−1; 3, 3 dS m−1; EC, electrical conductivity.
Fm x EC Combination
Quercetin was favored by growth at 3 dS m−1 EC for the decumbent Fm and at 2 dS m−1 for the erect Fm; however, no statistical differences were observed within and between Fms. Considering the other flavonoids, a 3 dS m−1 EC favored their contents for both Fms, with statistical differences between Fms (Figure 3).

Effect of Fm x EC combination on the content of flavonoids of fruits of 2 Jaltomata procumbens Fms. Different letters among bars indicate statistical difference (Tukey, P ≤ .05). Abbreviations: D x 1, decumbent x 1 dS m−1; D x 2, decumbent x 2 dS m−1; D x 3, decumbent x 3 dS m−1; E x 1, erect x 1 dS m−1; E x 2, erect x 2 dS m−1; E x 3, erect x 3 dS m−1; EC, electrical conductivity; Fm, form.
Fm x Pruning Combination
Statistical differences between Fms were recorded in myricetin, morin, and rutin. WP management promoted the contents of myricetin and morin in the erect Fm, while WoP management enhanced the effects in the decumbent Fm. For rutin, WoP favored its content in both Fms. For quercetin, WP management promoted its content, but without statistical differences within and between Fms (Figure 4).

Effect of Fm x pruning combination on the content of flavonoids of fruits of 2 Jaltomata procumbens Fms. Different letters among bars indicate statistical difference (Tukey, P ≤ .05). Abbreviations: D x WoP, decumbent x without pruning; D x WP, decumbent x with pruning; E x WoP, erect x without pruning; E x WP, erect x with pruning; Fm, form.
Terpenoids
In Fm factor, statistical differences in the carnosol content were registered. The erect Fm had 85.71% higher carnosol content than the decumbent Fm. Considering the other terpenoids, the erect Fm registered higher contents of ursolic acid and stigmasterol, while the decumbent Fm showed higher contents of oleanolic acid and β-sitosterol, but without statistical differences (Table 5).
Effect of the Form on Terpenoids Content of Fruits of 2 Jaltomata procumbens Forms, Cultivated at 3 Electrical Conductivity Levels and 2 Pruning Managements.

Different letters indicate statistical difference (Tukey, P ≤ .05).
No statistical differences were observed between the different EC levels and pruning management, however high EC decreased the contents of carnosol, oleanolic acid, and β-sitosterol, whereas an EC of 2 dS m−1 resulted in increased contents of ursolic acid and stigmasterol, and then decreased with increasing EC. WP management favored the contents of terpenoids (Supplemental Material Table S3).
EC × Pruning Combination
EC × WP management promoted the content of terpenoids. The combination of 1 dS m−1 EC × WP favored the contents of carnosol, oleanolic acid, and β-sitosterol, but their values decreased with increasing EC. The combination of 2 dS m−1 EC × WP favored the contents of ursolic acid and stigmasterol, which increased with respect to 1 dS m−1; however, the contents decreased at 3 dS m−1 EC, but without statistical differences (Table 6). In addition, the positive effect of EC (1 and 2 dS m−1) × WP combinations on the content of these metabolites was observed, yielding higher values than those obtained for individual treatments of EC and WP management (Supplemental Material Table S3).
Effect of EC x Pruning Combination on Terpenoids Content of Fruits of 2 Jaltomata procumbens Forms, Cultivated at 3 EC Levels and 2 Pruning Managements.

Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviations: 1, 1 dS m−1; 2, 2 dS m−1; 3, 3 dS m−1; EC, electrical conductivity.
Fm x EC Combination
In the erect Fm, growth at 1 dS m−1 EC favored the contents of carnosol and β-sitosterol, whereas that at 2 dS m−1 favored the contents of ursolic and oleanolic acids and stigmasterol. In the decumbent Fm, 1 dS m−1 EC promoted the contents of oleanolic acid and β-sitosterol, whereas 2 dS m−1 EC enhanced those of carnosol and ursolic acid, and 3 dS m−1 EC favored that of stigmasterol. However, no statistical differences were observed between combinations (Table 7).
Effect of Fm x EC Combination on Terpenoids Content of Fruits of 2 Jaltomata procumbens Fms, Cultivated at 3 EC Levels and 2 Pruning Treatments.

Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviations: 1, 1 dS m−1; 2, 2 dS m−1; 3, 3 dS m−1; EC, electrical conductivity; Fm, form.
Fm x Pruning Combination
WP management favored the contents of stigmasterol and β-sitosterol in the erect Fm and those of oleanolic and ursolic acids in the decumbent Fm; moreover, the same management promoted the carnosol content in both Fms. WoP management favored the contents of oleanolic and ursolic acids in the erect Fm and those of stigmasterol and β-sitosterol in the decumbent, but without statistical differences (Table 8).
Effect of Fm x Pruning Combination on Terpenoids Content of Fruits of 2 Jaltomata procumbens Fms, Cultivated at 3 Electrical Conductivity Levels and 2 Pruning Managementes.

Different letters indicate statistical difference (Tukey, P ≤ .05).
Abbreviation: Fm, form.
Discussion
Phenolic compounds, which include phenolic acids (non-flavonoids) and flavonoids, 18 are specific to plant species, with their levels varying according to cultivar or variety. 19 In wheat crop (genus Triticum) ferulic acid is present as the main and most abundant phenolic acid, 20 but without the presence of gallic acid. In 200 wheat cultivars with different genetic background and geographical origins, a ferulic acid content of 28.4-65.1 mg 100 g−1 of dry weight was reported. 21 Herein, differences in the levels of these acids were recorded, with gallic acid presenting the highest content in both Fms (decumbent = 59.03 mg 100 g−1; erect = 47.01 mg 100 g−1), followed by syringic and rosmarinic acids; ferulic acid, which was also present in J. procumbens, was one of the compounds with the lowest content in the fruits from both Fms (decumbent = 1.30 mg 100 g−1; erect = 1.75 mg 100 g−1). Gallic acid has an important antimicrobial, antifungal, anticancer, and anti-inflammatory properties, 22 while syringic acid shows therapeutic applications in the prevention of diabetes, cancer, and cerebral ischemia. 23 Rosmarinic acid has anti-inflammatory, neuroprotective, and antidepressant effects. 24
The presence of flavonoids has been reported in species such as Vaccinium vitis-idaea (quercetin = 6.3-64.3 mg 100 g−1 of dry weight; kaempferol = 3.4-4.6 mg 100 g−1 of dry weight), Fragaria ananassa (quercetin = 21.9-50.7 mg 100 g−1 of dry weight), and Vaccinium myrtillus (myricetin = 8.5-12.1 mg 100 g−1 of dry weight; quercetin = 7.3-28.9 mg 100 g−1 of dry weight). 25 In J. procumbens, myricetin and quercetin were recorded, but morin and rutin were present at higher values than those of the first two compounds. The decumbent Fm presented the highest morin content (61.54 mg 100 g−1), while the erect Fm had the highest rutin content (36.43 mg 100 g−1). Morin, which is a pentahydroxyflavone, is an important phytochemical with antioxidant, antiviral, and hepatoprotective effects 26 ; in addition, is reported morin acts against oxidative stress in the reproductive system of male rats before lymphoma treatment, therefore it may have therapeutic potential as a protective substance for treatment of patients with cancer. 27 Morin is present in strawberry (F. ananassa) at a content of 0.06 mg 100 g−1 of dry weight 28 and in Morus alba within a range of 0.5-1.2 mg 100 g−1, except in the fruit. 29 Rutin, which is a glycoside of flavonol, is an important compound in the pharmaceutical industry owing to its functions in reducing glucose levels and systolic and diastolic blood pressure as well as in the treatment of myocardial hypertrophy, colitis, and peritonitis. 30 In 6 Malus domestica cultivars, the rutin content in the fruit cuticle was reported with a range of 3.7–16.3 mg 100 g−1 of dry weight. 31 Considering the reported values of these metabolites in these species, the J. procumbens Fms under study, erect and decumbent, are important sources of rutin and morin, respectively.
Terpenoids are metabolites of plant or fungi. 32 They are present in numerous flowering plants, and the vast majority are restricted to a given linage or even a single species. 33 In Solanum lycopersicum, more than 21 metabolites belonging to the class of carotenoids are reported 34 and lycopene, a type of terpenoids, is the most abundant. 35 In the species under study, J. procumbens, the terpenoid present in the highest content in both Fms was ursolic acid (erect = 28.29 mg 100 g−1; decumbent = 26.25 mg 100 g−1), followed by stigmasterol in the erect Fm (14.79 mg 100 g−1) and β-sitosterol in the decumbent (13.65 mg 100 g−1). Among the terpenoids, different functions have been reported as defense against pathogens and herbivores and as antioxidants or pigments. 33 Ursolic acid, which is a pentacyclic triterpene, has antioxidant, anti-inflammatory, antibacterial, and antifungal effects as well as protective effects against ethanol-induced hepatotoxicity. 26 Stigmasterol has anti-inflammatory, anticancer, antiosteoarthritic, and cholesterol-lowering activity. 36 For β-sitosterol, hypocholesterolemic, anti-inflammatory, antibacterial, and antifungal effects have been reported. 37 Natural sources of phytochemicals, such as the fruits of J. procumbens are important due to their health benefits, and can be incorporated in people diet to prevent or treat human disease.26,36
In addition to the genetic factor, growth conditions are other factors that affect the contents of phenolic compounds 19 and terpenoids. 38 Herein, the effects of the factors evaluated on the content of each metabolite, individual and combined factors (EC and pruning) behaved differently, registering an increase or decrease in values depending on the EC level, pruning management, and their combination. Considering the increase in contents of phenolic compounds, the combination of 2 dS m−1 EC × WP showed notable contents of sinapic, ferulic, and gallic acids, while 1 dS m−1 EC × WP enhanced the contents of syringic and p-coumaric acids, and 3 dS m−1 EC × WP positively affected the contents of rosmarinic and vanillic acids. For flavonoids, the 3 dS m−1 EC × WP combination showed notable effects on myricetin, morin, and rutin contents, while 1 dS m−1 EC × WP increased the quercetin content. For terpenoids, the 1 dS m−1 EC × WP combination yielded notably high carnosol, oleanolic acid, and β-sitosterol contents, while 2 dS m−1 EC × WP resulted in high ursolic acid and stigmasterol contents. This behavior is similar to that reported in S. lycopersicum, where the contents of phenolic compounds behaved differently under the effect of stress due to salinity generated by NaCl and temperature where the contents of 175, 865, and 568 metabolites were altered by salinity, heat, and by combination of both, respectively; this behavior was attributed to the effect of stress types on the plant antioxidant system, which inhibited or promoted the activity of enzymes such as DAHPS, PAL, C4H, 4CL, and C3H as well as the expression of genes such as SlF3H, SlFLS, SlF3GT, and SlF3RT. 14
When the results within each Fm subjected to various EC levels and pruning management were analyzed, the values of some metabolites were lower than those recorded under the individual effects of EC and pruning or in their combination; however, this behavior was attributed to the genotype of each Fm, which presented different values for their metabolite contents, where the enzyme activity and gene expression would be presenting differentially; therefore, the behavior of the antioxidant system of each Fm should be determined because antioxidant activity is an important tool to evaluate plant response to certain types of stress; for example, Brassica campestris L. ssp. Chinensis exhibited high activities of antioxidant enzymes superoxide dismutase (SOD) and peroxidase at low (0 and 0.3 dS m−1) and high EC (9.6 dS m−1). 10 This is also relevant because the content of each metabolite would be influenced under certain growth conditions, and a higher content of the metabolite of interest would be obtained.
Growth conditions, such as salinity generated by the amount of ions available to the plants 10 and determined by the EC, influence the content of metabolites because salt stress experienced by the plant and affects the fruit quality. 11 Therefore, salt stress is considered one of the important types of stress in agricultural production. 14 For example, in Agastache rugosa, the high concentration of rosmarinic acid at lower EC levels (0.5, 1, and 2 dS m−1) and a decrease at 4 dS m−1 EC or higher was recorded; this effect was attributed to a low EC when is presented a nitrogen deficiency; in this state, genes involved in the rosmarinic acid biosynthetic pathway, such as phenylalanine ammonia-lyase, 4 coumarate, CoA ligase, and cinnamate 4-hydroxylase, are highly expressed. 11 An opposite effect was reported in Portulaca oleracea, where an increase in the flavonoid content was recorded at high salinity levels, which induced the cellular antioxidant mechanism and where the superoxide dismutase SOD has been suggested as an important enzyme in the tolerance of plants to stress. 39 In Licopersicum esculentum species, an increase in lycopene content was described when EC increased because of an over-regulation of genes that regulate or encode the enzymes involved in the biosynthesis of this metabolite triggered by osmotic stress experienced by the plant. 35
Regarding the effect of pruning on the contents of phenolic acids and flavonoids, an over-expression of genes involved in its biosynthesis, such as VvFLS1, VvF3’H, VvF3’5’H, VvANS, and VvMYBA1, was demonstrated in Vitis vinifera, influencing the increase in their contents in this species. 12 By contrast, in Licopersicum esculentum, a negative effect on lycopene synthesis was reported. 35 In J. procumbens, pruning effect had a positive and negative effect on the content of certain phenolic acids and flavonoids, while for terpenoids, pruning influenced the increase in their content. However, the pruning influence on the increase in the metabolite content was weaker compared to the influence of EC; a similar trend was reported for Mangifera indica L. in the evaluation of the effects of pruning and of a growth regulator on the phenolic acid content, where pruning had a lesser influence on the contents of phenolic acids. 13
This is the first work reported on the metabolite profile of J. procumbens, recognized as a semidomesticated species with potential for study in controlled environments. The results obtained are useful because knowing the possibility of influencing the metabolite content with the nutritive solution and pruning management would facilitate obtaining fruits with high fruit quality, an important characteristic in the population that has been virtually ignored in breeding programs, and because there is a need for natural sources of phytochemicals to prevent and treat human disease, J. procumbens could be considered to diversify the people diet.
Conclusions
Individually and in combination, the electrical conductivity of the nutrient solution and pruning management affected the contents of the metabolites evaluated in both, erect and decumbent, J. procumbens Fms.
The erect Fm showed noteworthy contents of syringic and p-coumaric acids, rutin, and carnosol, while the decumbent Fm recorded high contents of gallic and rosmarinic acids and morin.
The effects of combined EC and pruning on the content of metabolites exceeded their individual effects, with EC exerting a stronger effect than pruning.
The behavior of the antioxidant system in each plant population when subjected to these types of stress and the expression of genes linked to that system should be determined.
Owing to the observed variability, a specific electrical conductivity level and pruning management are required for each Fm to obtain the metabolite group of interest. The contents of terpenoids and phenolic acids (except rosmarinic, vanillic, and gallic acids) were promoted by low (1 dS m−1) and medium (2 dS m−1) electrical conductivity levels and pruning management, while the contents of flavonoids, except quercetin, were enhanced by high EC (3 dS m−1) and pruning management.
Other electrical conductivity levels should be evaluated so that the metabolite contents can be increased without affecting the yield of both Fms.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221150547 - Supplemental material for Effects of Electrical Conductivity and Pruning on Secondary Metabolite Contents in Fruits of Jaltomata procumbens (Cav.) J. L. Gentry
Supplemental material, sj-docx-1-npx-10.1177_1934578X221150547 for Effects of Electrical Conductivity and Pruning on Secondary Metabolite Contents in Fruits of Jaltomata procumbens (Cav.) J. L. Gentry by Ignacio Darío Flores-Sánchez, Manuel Sandoval-Villa and Ramón Marcos Soto-Hernández in Natural Product Communications
Footnotes
Acknowledgements
The authors acknowledge the Consejo Nacional de Ciencia y Tecnología (CONACYT, Mexico) for the scholarship given to the first author for his postgraduate studies and the Colegio de Postgraduados, Campus Montecillo for the materials provided to conduct this research. They also acknowledge the support of the M. C. Rubén San Miguel Chávez (Postgraduate in Botany, Colegio de Postgraduados, Campus Montecillo) during the metabolite determination using HPLC.
Author Contributions
IDF-S conducted literature research, experimental work-methodology, and analysis of results. RMS-H performed laboratory analysis-methodology, and MSV and RMS-H for resources. IDF-S, MS-V, and RMS-H were involved in original-draft preparation, and writing-review and editing, and MS-V and RMS-H in supervision. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the scholarship program of the Consejo Nacional de Ciencia y Tecnología of Mexico (CONACYT) and Colegio de Postgraduados.
Informed Consent
Not applicable because this article does not contain any studies involving human or animal subjects.
Trial Registration
Not applicable because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
