Abstract
Two cultivated varieties of Gleditsia sinensis Lam, Shuo Spina Gleditsiae (SSG) and Mi Spina Gleditsiae (MSG), are currently used in China as a mixture of the herb Spina Gleditsia. This work evaluates, for the first time, the difference in cytotoxicity, antioxidant activity, and flavonoid contents between SSG and MSG. Quantification of bioactive flavonoids in the samples was performed using high-performance liquid chromatography. Total antioxidant activity and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid free radical scavenging assays were used, and the growth inhibitory effects on cancer cells were determined using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide assay. Epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin were qualified and quantified; much higher amounts were found in the extract of SSG than that of MSG. In antioxidant assays, SSG extracts presented stronger effects than those of MSG. The half-maximal inhibitory concentration values of SSG were 46.2 and 47.8 μg/mL, compared with 70.5 and 73.0 μg/mL for MSG against EC109 and HepG2 cancer cells, respectively. It thus brings concern about potential quality issues regarding efficacy, safety for healthy food production, and potential medicinal uses.
Spina Gleditsiae, the dry thorns of Gleditsia sinensis Lam (G. sinensis), is currently used as a source of health products, cosmetics, and even medicines in the healthcare for amygdalitis, dermatocyst, dermapostasis, parasitosis, and cancer in China. Flavonoids, the major bioactive components of Spina Gleditsiae, exhibit a wide range of pharmacological properties, especially anticancer and antimicrobial activities. 1 From Spina Gledisiae, the flavonoids fusin, (2R,3R)-5,3′,4′-trimethoxyl-7-hydroxyl-flavanonol, 5,7,3′,4′-tetrahydroxyl-flavanonol, and 5-methoxyl-3′,4′,7-trihydroxylflavanonol presented significant cytotoxic effects against human esophageal (EC109), human lung (A549), and liver cancer cell lines (HepG2) with a half-maximal inhibitory concentration (IC50) values lower than 20 µg/mL, 2 whereas butein exhibited inhibitory effects on MCF7 cancer cells with an IC50 value of 28.5 µg/ mL. 3 Moreover, quercetin and 5,7,3′,5′-tetrahydroxy-flavanonol demonstrated antibacterial effects against Bacillus subtilis with minimal inhibitory concentrations (MIC) of 0.5 and 0.5 mg/mL, and against Xanthomonas vesicatoria with MIC of 0.75 and 0.75 mg/mL, respectively. 4
As a result of the wide use and medicinal value of Spina Gleditsiae, G. sinensis has great market demands and has become a popular cash crop in many regions of Asia. However, a key issue that both planters and users face is the plant varieties developed during the cultivation; 2 varieties of G. sinensis grow together symbiotically. They are characterized by the different size and growth density of the thorns on the stem. The one with huge but sparse thorns is known as Shuo Spina Gleditsiae (SSG, Shuo means huge in Chinese), the other as Mi Spina Gleditsiae (MSG, Mi means dense in Chinese), because it has rather small and dense thorns. The 2 spina Gleditsiae varieties may present different levels of bioactivities, thus leading to potential problems regarding quality control and medicinal uses.
To eliminate this confusion, the aim of this study was first to develop a method for the characterization and quantification of 6 flavonoid compounds in the 2 cultivated varieties of Spina Gleditsiae, and second, to evaluate their cytotoxic/antioxidant activities in vitro, thus providing a preliminary judgment on the quality of SSG and MSG.
Materials and Methods
Chemicals and Cell Lines
Epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin were purchased from Sigma (Solarbio, Beijing, China), and EC109 and HepG2 from Beinglay Biotech (Wuhan, Hubei, China). Cells were grown in Dulbecco’s modified Eagle medium supplemented with 10% fetal bovine serum, 100 µg/mL penicillin, and 100 µg/mL streptomycin in a 5% carbon dioxide humidified incubator at 37 °C. Experiments were performed when cells were approximately 80% confluent. Cisplatin was purchased from Sigma-Aldrich (Sternhein, Germany).
Sample Preparation
Plant samples of SSG and MSG were collected in Xiaoantou Village, Songxian County, Luoyang, Henan Province, China (January 2018). The identity was kindly confirmed as G. sinensis Lam by Professor Zhiming Gao at the Agricultural School, Henan Agricultural University. Two voucher specimens (access number: JGS2018-007A and B) have been deposited in the Natural Products Research Laboratory, School of Chemical Engineering and Pharmaceutics, University of Science and Technology.
The dry plant samples of SSG (100 g) and MSG (100 g) were minced in a grinder and then extracted with ethanol (1 L) twice for 24 hours at room temperature (25 °C). The supernatants were filtered and collected. The solvent was completely removed at 35 °C under reduced pressure, and the residue dried in a fume cupboard. The dried residues were redissolved in distilled water (1 g: 10 mL) and extracted with the same volume of ethyl acetate, twice. The ethyl acetate extracts were collected, and the solvents completely removed to produce either SSG or MSG residues. Three replicates were performed for each analysis.
HPLC Instrumentation
For the quantification of the samples and preparation of the calibration curves, a Waters series high-performance liquid chromatography (HPLC) system was used comprising a Waters e2695 Separations Module with degasser, a variable wavelength 2489 ultraviolet (UV)/visible detector and an autosampler. The UV detector was set at 270 nm, and data were acquired using Empower 3 software. The column used was a Thermo C18, 5 µm (250 × 4.6 mm) maintained at ambient room temperature. Gradient elution of the samples and standards was performed using 0.1% formic acid (eluent A) and methanol (eluent B). The initial conditions of gradient elution were 10% of eluent B with a linear gradient to 90% from 1 to 35 minutes, and this proportion is maintained for 1 minute. The column was then returned to the initial condition at 37 minutes and maintained until the end of the run at 40 minutes. The flow rate was 1 mL/minute. Dry residues of SSG and MSG extracts were dissolved in methanol at 5 mg/mL. The injection volume of each sample was 10 µL. Three injections were performed for each sample and standards.
Quantitation of Epicatechin, Isovitexin, Fisetin, Fustin, Quercetin, and Aromadendrin
Epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin calibration curves (n = 6) were obtained by preparing stock solutions (1000 µg/mL) in methanol. Subsequent dilutions were made to give 500, 400, 300, 200, 100, and 50 µg/mL. The linearity of the calibration curves was determined (r 2 > 0.998). The recovery of samples was over 0.961. The limit of detection was calculated based on a signal-to-noise ratio of 3.3. The limit of quantitation was then calculated based on a signal-to-noise ratio of 10 using the lowest concentration in the calibration and the highest noise observed when injecting a blank. The intraday precision was calculated by comparing the area of epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin (in triplicate) at the midpoint of the calibration curve (0.2 mg/mL) within the day, and the resulting coefficients of variation obtained (% CV) were 0.12%, 0.25%, 0.08%, 0.13%, 0.12%, and 0.11%, respectively. For the interday precision, the areas of epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin for the midpoint in the calibration curve (0.2 mg/mL) for 3 different days were added up and compared. The resulting % CVs obtained were 1.23%, 0.89%, 1.09%, 2.81%, 2.19%, and 0.99%, respectively. All standard concentrations are quoted in μg/mL, mean ± standard deviation, n = 3.
Total Antioxidant Activity Test
SSG and MSG extracts were prepared at concentrations of 0.24, 0.48, 0.72, 0.96, and 1.20 mg/mL. A stock solution of phosphorus molybdenum reagent (0.6 mol/L sulfuric acid, 28 mmol/L sodium phosphate, and 4 mmol/L ammonium molybdate) was prepared. Each sample (1 mL) was mixed with 3.0 mL phosphorus molybdenum reagent solution. The mixtures were placed in 95 °C water baths for 90 minutes. The samples were cooled down to room temperature (25 °C), and then the absorbance at 625 nm was measured. Distilled water was used as a blank with butylated hydroxytoluene (BHT) as positive control.
ABTS Free Radical Scavenging Assay
SSG and MSG extracts were prepared at concentrations of 0.24, 0.48, 0.72, 0.96, and 1.20 mg/mL. Then, 0.8 mL of 7.4 mmol/L ABTS solution and 0.8 mL of 2.6 mmol/L potassium persulfate were mixed and reacted in the dark for 12 hours. The prepared mixture was then diluted 100 times using absolute ethanol to produce the ABTS+ working solution. ABTS+ working solution (4 mL) was mixed with 1 mL sample and 1 mL absolute ethanol. The mixtures were then shaken for 1 minute, and the absorbance was measured at 734 nm. The ABTS free radical scavenging rate = (A0 − A)/A0 × 100%. A0 is the absorbance of ABTS+ working solution + absorbance of absolute ethanol; A is the absorbance of ABTS+ working solution + absorbance of samples.
Cytotoxic Activity Assay
The 3-(4,5-dimethylthiazol-2-yl)−2,5-diphenyl tetrazolium bromide (MTT) assay was performed as follows: briefly, 200 µL cell suspension was seeded at a concentration of 1.5 × 104 cells/mL in a 96-well plate. After overnight incubation, serial doubling dilutions of SSG and MSG extracts (100, 3.125 µg/mL) were added in 10 µL aliquots. The negative control was 10 µL 0.01% dimethyl sulfoxide in Hank’s solution. Cisplatin (100, 3.1 μg/mL) was used as a positive control. Each concentration was tested three times. After 24 hours of incubation, 20 µL MTT solution (5 mg/mL, Sigma-Aldrich Ltd, Gillingham, Dorset, UK) was added and incubated at 37 °C for 4 hours to develop color. The optical density (OD) was measured at 590 nm using a Multiskan Spectrum Reader. The growth inhibition was determined using cytotoxicity percentage = ((control OD−sample OD)/control OD) × 100%.
Statistical Analysis
The data are shown as mean ± standard deviation. Statistical comparison among treatments was carried out using a one-way analysis of variance by Microsoft Excel 2017. The statistical significance between control and sample groups was calculated by the Student’s t-test. Data were taken as significant where P < 0.05.
Results and Discussion
The constituents of Spina Gleditsiae include triterpenes, sterols, flavonoids, phenols, and alkaloids. 1 To date, the separation and analysis of its active flavonoid compounds have been barely reported. Based on analytical techniques reported for flavonoids of medicinal plants, reversed-phase C18 columns and mobile phases consisting of an acidic buffer and an organic solvent were employed in this study. 5 Formic acid (0.1%) combined with an optimized gradient elution was used to obtain sufficient resolution between the targeted flavonoids and an acceptable gradient time for the HPLC analysis of SSG and MSG. Moreover, the low standard deviation of intraday and interday precision observed supported the accuracy of the analytical method.
According to the literature, flavonoid compounds are the major bioactive components of Spina Gleditsiae. 1 These include epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin; these were eluted from the column using the methodology developed in this study, with retention times of 14.003, 19.674, 20.539, 21.639, 24.492, and 30.966 minutes, respectively. Moreover, compared with the retention time of a standard in parallel experiments, a major compound eluted at 16.330 minutes could be determined as isoscopoletin (6-hydroxy-7-methoxycoumarin). The quantitation of epicatechin, isovitexin, fisetin, fustin, quercetin, and aromadendrin was achieved using peak area. The resulting concentrations (μg/mL, mean ± SD) of these 6 flavonoids in SSG and MSG extracts are shown in Table 1. Among them, epicatechin demonstrated the highest concentration with 795.5 and 666.3 µg/mL in SSG and MSG, respectively. The contents of isovitexin and aromadendrin were relatively low in both of SSG and MSG. Briefly, the tendency of the contents of the 6 flavonoids showed similar profiles in both extracts. However, their concentrations demonstrated an obvious difference, as presented in Table 1. Except for isovitexin, which presented a similar level (184 µg/mL), all the other flavonoids exhibited statistically higher amounts in the extract of SSG than that of MSG (P < 0.05). Aromadendrin showed the highest difference, which was nearly twice the level in SSG compared with MSG.
Concentrations (mg/mL, mean ± SD, N = 3 Measurements) of Epicatechin, Isovitexin, Fisetin, Fustin, Quercetin, and Aromadendrin in SSG and MSG Extracts.

Abbreviations: SSG, Shuo Spina Gleditsiae; MSG, Mi Spina Gleditsiae.
a Significant different where P < 0.05.
As shown in Figure 1, the antioxidant effects of SSG and MSG were much lower than that of BHT for total antioxidant activity. However, the scavenging effects increased gradually when the concentration of SSG and MSG increased, reaching BHT’s value (about 95%) at a concentration of 1.2 mg/mL. The SSG extracts demonstrated higher antioxidant and free radical scavenging effects than those of the MSG extracts in both tests. In Figure 2, SSG extracts exhibited much stronger cytotoxicity than MSG extracts in both cancer cell lines tested. The IC50 values of SSG were 46.2 and 47.8 µg/mL, compared with those of MSG at 70.5 and 73.0 µg/mL for EC109 and HepG2 cells, respectively. In both antioxidant (Figure 1) and cytotoxic (Figure 2) assays, SSG extracts presented stronger effects than those of MSG, which corresponded with their flavonoid profiles. These results are also consistent with the related activities of flavonoids reported in the literature. 6 Epicatechin isolated from Litchi pericarp aqueous extract showed great antioxidant and growth inhibitory activities on human lung cancer A549 cells. 7 Isovitexin could protect deoxyribonucleic acid (DNA) and erythrocytes from oxidative damage. It also triggered the process of apoptosis in HeLa cells by downregulating Bcl-2 level with the simultaneous upregulation of Bax and caspase-3 protein expression. 8 Fisetin was reported to induce DNA damage in human oral cancer HSC3 cells. It eliminated cancer cells through enhancing the expression of B-cell lymphoma 2 (BCL2), antagonist/killer, and BCL2-associated X, reducing BCL2 and BCL-x and increasing the cleaved forms of caspase-3,caspase-8, caspase-9, cytochrome c, apoptosis-inducing factor, and endonuclease G in HSC3 cells. 9 Treatment with quercetin significantly attenuated cell migration and invasion in human osteosarcoma cells (HOS). Moreover, quercetin could downregulate hypoxia-inducible factor-1 alpha, vascular endothelial growth factor, matrix metalloproteinases (MMP)2, and MMP9 messenger ribonucleic acid and protein expression levels. Additionally, treatment with quercetin attenuated metastatic lung tumor formation and growth in the nude mouse model of osteosarcoma. 10 Aromadendrin was evidenced to have remarkable antilipid peroxidation efficacy and could inhibit cardiac hypertrophy through downregulating the nuclear factor of activated T-cells and mitogen-activated protein kinase pathways. 11
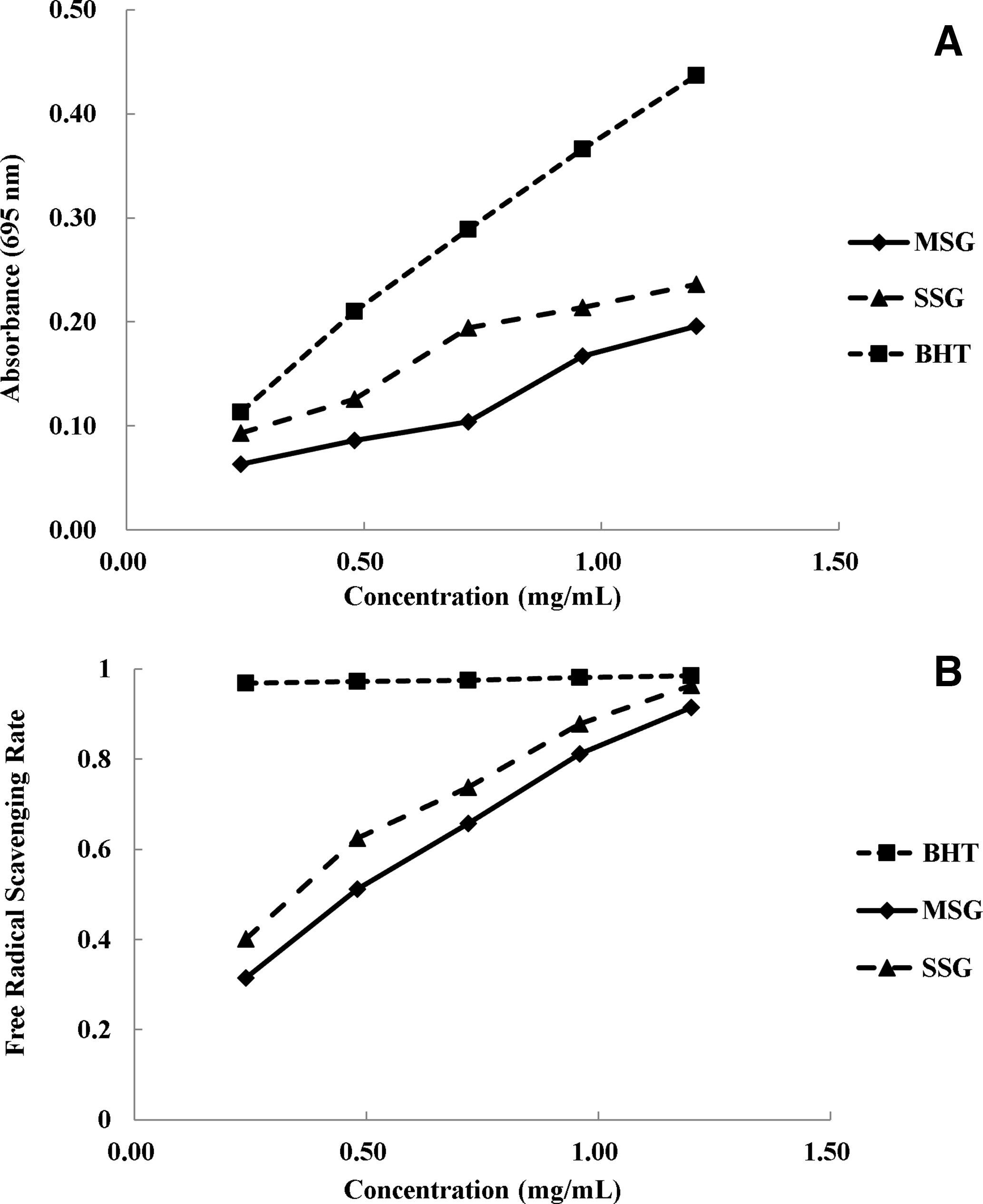
(A) Total antioxidant and (B) 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid free radical scavenging activities of SSG and MSG extracts in vitro. BHT, butylated hydroxytoluene; MSG, Mi Spina Gleditsiae; SSG, Shuo Spina Gleditsiae.
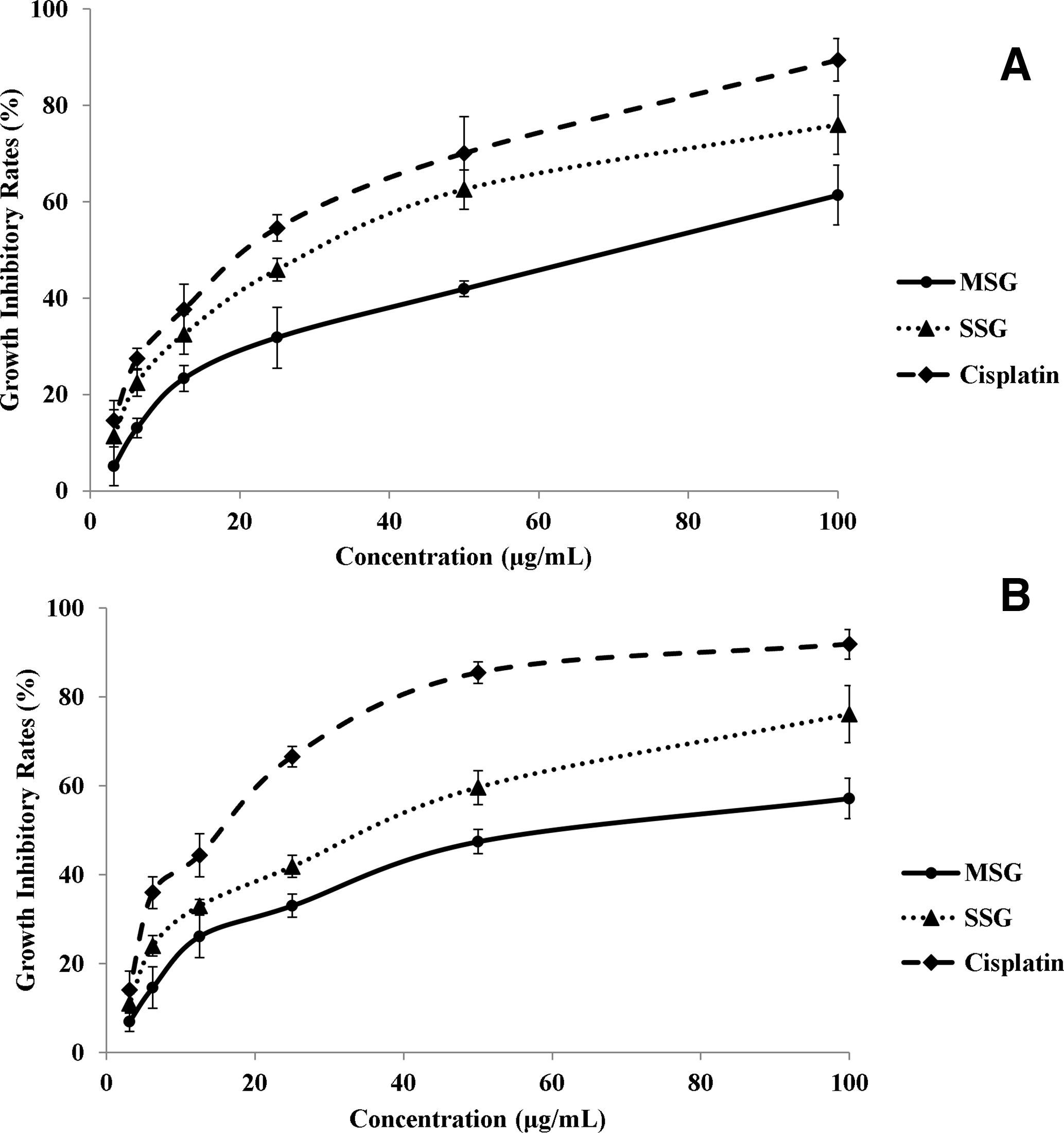
Cytotoxicity of SSG and MSG extracts on (A) human EC109 esophagus and (B) HepG2 liver cancer cells in vitro. BHT, butylated hydroxytoluene; MSG, Mi Spina Gleditsiae; SSG, Shuo Spina Gleditsiae.
There is now a growing need to establish a scientific evidence base for the application of natural raw materials to guide the food/pharmaceutical industry or natural products consumers to be more discriminatory in their choice of raw materials or products. Additionally, with currently no clear identification between SSG and MSG, there is no guarantee of quality. Blind use of SSG and MSG may far reduce either the margin of safety or efficacy. There is thus an urgent need not only for review by pharmaceutical companies of the Spina Gleditsiae-contained products but also for educating users to relate quality and efficacy/safety. The data presented in this work provide valuable information as to which Spina Gleditsiae variety may be suitable for further investigation and for pilot trials as a valuable medicinal plant material.
Conclusion
This work firstly determined that the levels of flavonoid contents and cytotoxic and antioxidant activities of SSG were significantly higher than those of MSG. It thus can be beneficial for bringing concerns about potential quality issues regarding efficacy, safety, and medicinal uses. The work provides important information on the quality control process and raw material selection for the pharmaceutical industry. Also, this result may help to eliminate the confusion in the future cultivation, industrial, and medicinal choice of Spina Gleditsiae. As a result of different localities of growth, plant variety, and processing techniques, wide variability in the bioactive contents of raw materials undermines the applications of natural products. This study will help the researcher to uncover critical areas of quality control of natural products that many were not able to explore.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Project no. U1504830).
