Abstract
In order to develop a series of novel compounds with antiplatelet aggregation activities, 3,15-disuccinate-12-coumarin substituted derivatives were designed and synthesized based on the natural product andrographolide. In vitro antiplatelet aggregation activities were evaluated by the turbidimetric method with thrombin, adenosine diphosphate (ADP), arachidonic acid (AA), and collagen as inducers. The biological evaluation revealed that compound
Keywords

Thrombus is a quite common cardiovascular disease and seriously threatens human health and life quality. 1,2 Platelet aggregation is one of the important factors leading to thrombosis. Antiaggregation drugs are often used as the top-priority drugs to treat thrombosis. 3,4 Currently, a variety of antiplatelet aggregation drugs have been successfully marketed. According to different mechanisms, there are thromboxane A2 inhibitor (aspirin), 5 phosphodiesterase inhibitor (cilostazol), 6 5-hydroxytrypamine receptor antagonist (sarpogrelate), 7 platelet membrane glycoproteins Ⅱb/Ⅲa receptor inhibitor (tirofiban), 8 adenosine diphosphate (ADP) induction inhibitor (clopidogrel), 9 Ca2+ channel antagonist (flunarizine), 10 -12 and protease active receptor-1 (PAR-1) antagonist (vorapaxar sulfate). 13
Andrographolide
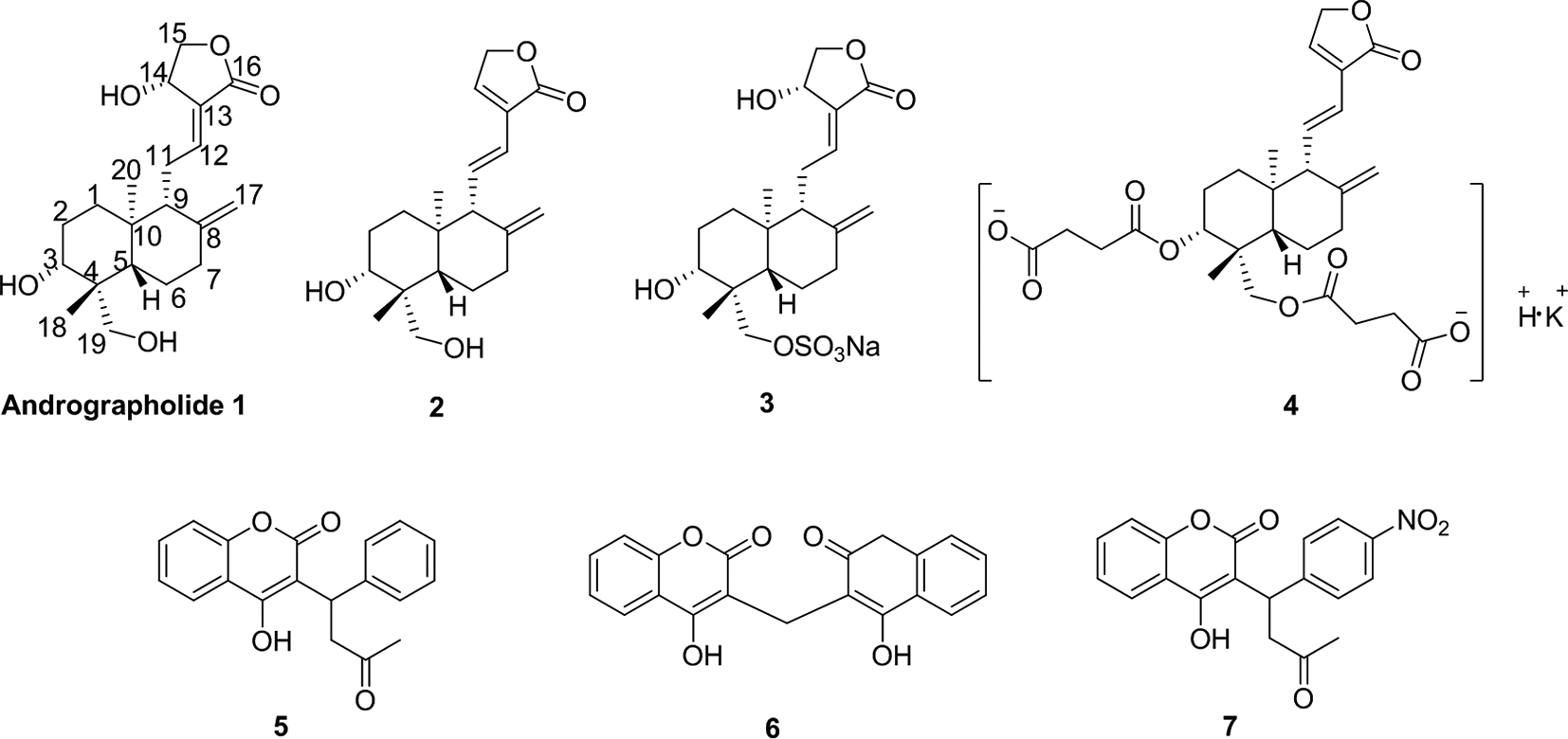
Figure
The well-known Coumarins, easily available natural compounds, exhibit a wide range of biological activities such as acetylcholinesterase inhibitor,
36
antioxidant,
37
antitumor,
38
anti-inflammatory,
39
alpha-chymotrypsin inhibitor,
40
and anti-HIV.
41
In addition, coumarin derivatives have obvious antiplatelet aggregation activities.
42
-46
Among them, warfarin
Based on the above, it is highly worthwhile to develop accessible and effective methods to construct novel antiplatelet aggregation compounds with andrographolide and coumarins.
Results and Discussion
Preparation of Target Compounds 11
By analyzing the structure, andrographolide had many reaction sites such as 3,19-OH, C-8 = C-17, C-12, C-15. Based on our previous work, to improved aqueous solubility, 2 molecules of carboxylates were still introduced at 3,19-OH. To improve bioactivities, coumarins tried to be introduced at C-12. As expected, we successfully designed the synthetic route to prepare the new skeleton compounds

The synthetic route of derivatives 11. Reagents and conditions: (a) pyridine, succinic anhydride, reflux, 12 hours, then, 4 M hydrochloric acid, 60°C, 30 minutes; (b) potassium permangante, tetrahydrofuran, −5°C; (c) potassium tert-butoxide, 4-methylcoumarins 10, dimethyl sulfoxide , room temperature, 5 hours, then, acetic acid, 1 hour, then potassium bicarbonate.
Biological Properties
In vitro antiplatelet aggregation activities of
The IR and IC50 of Target Compounds 11 In Vitro.

ADP, adenosine diphosphate; AA, arachidonic acid; IC50, half-maximal inhibitory concentration.
*P < 0.05 vs aspirin. **P < 0.01 vs aspirin.
aIRs were expressed as the means ± standard error of 6 experimental replicates (n = 6).
b“–”, not tested.

Antiplatelet aggregation activities of compounds 11 at different concentrates (final concentration: 1.7, 3.4, and 6.8 µM). (a) Thrombin, (b) adenosine diphosphate (ADP), (c) arachidonic acid (AA), and (d) collagen. Data were expressed as the means ± standard error of 6 experimental replicates (n = 6).
Compared Entry
For thrombin, AA, and collagen as inducers,
For ADP as inducer, only
To sum up, for thrombin, AA, and collagen as inducers,
Cytotoxicity Assay In Vitro
Mouse fibroblast cells (L929) were selected to evaluate cell toxicity of target derivatives
The Absorbance and Survival Rates of Target Compounds 11 In Vitro.

*P < 0.05 vs aspirin. **P < 0.01 vs aspirin.
aSurvival rates were expressed as the means ± standard error of 6 experimental replicates (n = 6).
Conclusion
Through our efforts, 3,15-disuccinate-12-coumarin substituted derivatives
Experimental
General
Infrared spectra were determined on a Nicolet Avatar-370 spectrometer in potassium bromide (ν in cm−1). Melting points were measured on a Büchi B-540 capillary melting point apparatus and uncorrected. Electrospray ionization-mass spectra (ESI-MS) were determined on a Thermo Finnigan LCQ-Advantage. High-resolution MS (HRMS) was determined on an Agilent 6210 TOF instrument. 1H nuclear magnetic resonance (NMR) and 13C NMR spectra were recorded on Varian Mercury Plus-400 spectrometer (400 and 100 MHz) in dimethyl sulfoxide (DMSO)-d
6 or D2O, δ in parts per million, J in Hertz, using tetramethylsilane as an internal standard. Platelet aggregation rates were measured on LG-PABER Platelet aggregation apparatus (made in Beijing Shidi Scientific Instrument Co. LTD, Beijing). Column chromatography was carried out on silicagel (200-300 grading). Andrographolide
Synthesis of Derivatives
Synthesis of intermediate 8
Andrographolide
Synthesis of intermediate 9
Intermediate
Synthesis of product 11 (11a was selected as example)
Compound
Monopotassium 3,15-disuccinate-12-coumarin andrographolide 11a
Pale yellow solid, 1.18 g, yield 42.5%, purity 96.2%. 1H NMR (400 MHz, D2O): δ = 7.84-7.81 (1H, m, 5′-H), 7.65-7.45 (3H, m, 6′,7′,8′-H), 6.47 (1H, s, 3′-H), 6.19 (1H, d, J = 15.8 Hz, 12H), 5.72 (1H, m, 11H), 5.03 (1H, s, 13Ha), 4.87 (1H, s, 13-Hb), 4.13 (1H, d, J = 11.8 Hz, 15-Ha), 3.93-3.78 (2H, m, 3H, 15-Hb), 2.83-2.52 (9H, m, 9,18,19,22,23H), 2.12-1.62 (9H, m, 1,2,5,6,7H), 1.05 (3H, s, 14H), 0.97 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1 (C-21), 162.2 (C-4′), 159.3 (C-2′), 150.1 (C-9′), 148.8 (C-8), 135.5 (C-11), 129.1 (C-5′), 128.3 (C-7'), 128.0 (C-12), 127.2 (C-10′), 125.4 (C-6′), 111.2 (C-8′), 109.1 (C-13), 104.7 (C-3′), 75.0 (C-3), 65.9 (C-15), 60.9 (C-9), 47.5 (C-5), 40.9 (C-4), 39.8 (C-10), 36.7 (C-7), 33.6 (C-1), 29.5 (C-18), 29.2 (C-22), 29.1 (C-19), 29.0 (C-23), 24.3 (C-2), 24.0 (C-6), 21.9 (C-14), 14.7 (C-16) ppm. MS (ESI): m/z (%) =633.2 ([M + H]+, 100%). HRMS (ESI) calcd for C33H38KO10 [M + H]+: 633.2102; found 633.2116.
Monopotassium 3,15-disuccinate-12-(3′-methylcoumarin) andrographolide 11b
White solid, 1.16 g, yield 40.9%, purity 95.7%. 1H NMR (400 MHz, D2O): δ = 7.86-7.83 (1H, m, 5′-H), 7.65–7.42 (3H, m, 6′,7′,8′-H), 6.15 (1H, d, J = 15.8 Hz, 12H), 5.75 (1H, m, 11H), 5.04 (1H, s, 13Ha), 4.87 (1H, s, 13-Hb), 4.11 (1H, d, J = 11.8 Hz, 15-Ha), 3.92-3.79 (2H, m, 3H, 15-Hb), 2.83-2.55 (9H, m, 9,18,19,22,23H), 2.43 (3H, s, 3′-CH3), 2.12-1.68 (9H, m, 1,2,5,6,7 H), 1.01 (3H, s, 14H), 0.90 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.4 (C-24), 173.4 (C-17), 173.1 (C-21), 162.4 (C-4′), 159.4 (C-2′), 150.2 (C-9′), 148.7 (C-9), 135.4 (C-11), 129.2 (C-5′), 128.2 (C-7′), 128.0 (C-12), 127.3 (C-10), 125.4 (C-6′), 120.2 (C-3′), 111.1 (C-8′), 109.2 (C-13), 75.1 (C-3), 65.8 (C-15), 60.9 (C-9), 47.6 (C-5), 40.8 (C-4), 39.8 (C-10), 36.9 (C-7), 33.5 (C-1), 29.4 (C-18), 29.2 (C-22), 29.0 (C-19), 28.8 (C-23), 24.3 (C-2), 24.0 (C-6), 21.4 (C-14), 14.7 (C-16), 10.9 (3′-CH3) ppm. MS (ESI): m/z (%) =647.2 ([M + H]+, 100%). HRMS (ESI) calcd for C34H40KO10 [M + H]+: 647.2259; found 647.2273.
Monopotassium 3,15-disuccinate-12-(3′-ethoxyformylcoumarin) andrographolide 11c ,
Pale yellow solid, 1.04 g, yield 33.5%, purity 96.4%. 1H NMR (400 MHz, D2O): δ = 7.85-7.82 (1H, m, 5′-H), 7.66–7.48 (3H, m, 6′,7′,8′-H), 6.19 (1H, d, J = 15.8 Hz, 12H), 5.73 (1H, m, 11H), 5.04 (1H, s, 13Ha), 4.88 (1H, s, 13-Hb), 4.21 (2H, q, J = 8.2 Hz, 3′-COOCH 2 CH3), 4.14 (1H, d, J = 11.8 Hz, 15-Ha), 3.91-3.88 (2H, m, 3H, 15-Hb), 2.83-2.56 (9H, m, 9,18,19,22,23H), 2.12-1.62 (9H, m, 1,2,5,6,7 H), 1.29 (3H, t, J = 8.2 Hz, 3′-COOCH2 CH 3 ), 1.03 (3H, s, 14 H), 0.97 (3H, s, 16 H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.4 (C-17), 173.1 (C-21), 165.0 (C-3′-COOCH2CH3), 159.2 (C-2), 150.1 (C-9′), 148.6 (C-8), 144 (C-4′) 135.5 (C-11), 129.2 (C-5′), 128.3 (C-7′), 128.0 (C-12), 127.1 (C-10′), 125.4 (C-6′), 115.1 (C-3′), 111.2 (C-8′), 109.2 (C-13), 75.1 (C-3), 65.9 (C-15), 61.4 (C-3′-COOCH 2 CH3), 60.8 (C-9), 47.5 (C-5), 40.8 (C-4), 39.9 (C-10), 36.8 (C-7), 33.4 (C-1), 29.5 (C-18), 29.3 (C-22), 29.1 (C-19), 29.0 (C-23), 24.2 (C-2), 24.0 (C-6), 21.8 (C-14), 14.7 (C-16), 14.2 (C-3′-COOCH2 CH 3) ppm. MS (ESI): m/z (%) =705.2 ([M + H]+, 100%). HRMS (ESI) calcd for C36H42KO12 [M + H]+: 705.2313; found 705.2330.
Monopotassium 3,15-disuccinate-12-(3′-chlorocoumarin) andrographolide 11d
Pale yellow solid, 1.33 g, yield 45.5%, purity 96.2%. 1H NMR (400 MHz, D2O): δ = 7.86-7.82 (1H, m, 5′-H), 7.65-7.42 (3H, m, 6′,7′,8′-H), 6.17 (1H, d, J = 15.8 Hz, 12H), 5.79 (1H, m, 11H), 5.04 (1H, s, 13Ha), 4.88 (1H, s, 13-Hb), 4.13 (1H, d, J = 11.8 Hz, 15-Ha), 3.93-3.84 (2H, m, 3H, 15-Hb), 2.80-2.52 (9H, m, 9,18,19,22,23H), 2.12-1.68 (9H, m, 1,2,5,6,7H), 1.05 (3H, s, 14H), 0.96 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.6 (C-20), 174.5 (C-24), 173.2 (C-17), 173.1 (C-21), 159.7 (C-2′), 153.4 (C-4′), 150.1 (C-9′), 148.6 (C-8), 135.5 (C-11), 129.2 (C-5′), 128.3 (C-7′), 128.0 (C-12), 127.3 (C-10′), 125.4 (C-6′), 116.7 (C-3′), 111.1 (C-8′), 109.2 (C-13), 75.1(C-3), 65.9 (C-15), 60.8 (C-9), 47.5 (C-5), 40.8 (C-4), 39.7 (C-10), 36.7 (C-7), 33.7 (C-1), 29.4 (C-18), 29.2 (C-22), 29.1 (C-19), 29.0 (C-23), 24.2 (C-2), 24.0 (C-6), 21.7 (C-14), 14.9 (C-16) ppm. MS (ESI): m/z (%) = 667.2 ([M + H]+, C33H37 35ClKO10, 100%), 669.2 ([M + H]+, C33H37 37ClKO10, 33%). HRMS (ESI) calcd for C33H37 35ClKO10 [M + H]+: 667.1712, found 667.1734; for C33H37 37ClKO10 [M + H]+: 669.1683, found 669.1698.
Monopotassium 3,15-disuccinate-12-(6′-methylcoumarin) andrographolide 11e
white solid, 1.14 g, yield 40.2%, purity 95.3%. 1H NMR (400 MHz, D2O): δ = 7.59-7.56 (1H, m, 5′-H), 7.31–7.26 (2H, m, 7′,8′-H), 6.46 (1H, s, 3′-H), 6.19 (1H, d, J = 15.8 Hz, 12H), 5.73 (1H, m, 11H), 5.03 (1H, s, 13Ha), 4.86 (1H, s, 13-Hb), 4.14 (1H, d, J = 11.8 Hz, 15-Ha), 3.93-3.83 (2H, m, 3H, 15-Hb), 2.83-2.53 (9H, m, 9,18,19,22,23H), 2.34 (3H, s, 6′-CH3), 2.12-1.65 (9H, m, 1,2,5,6,7H), 1.02 (3H, s, 14H), 0.98 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1 (C-21), 162.2 (C-4′), 159.3 (C-2′), 148.8 (C-8), 147.2 (C-9′), 135.6 (C-11), 135.4 (C-6′), 132.3 (C-7′), 128.0 (C-12), 127.2 (C-10′), 127.1 (C-5′), 116.9 (C-8′), 109.1 (C-13), 104.7(C-3′), 75.0 (C-3), 65.9 (C-15), 60.7 (C-9), 47.5 (C-5), 40.9 (C-4), 39.8 (C-10), 36.9 (C-7), 33.4 (C-1), 29.5 (C-18), 29.2 (C-22), 29.1 (C-19), 29.0 (C-23), 24.3 (C-2), 24.1 (C-6), 21.9 (C-14), 21.6 (6′-CH3), 14.5 (C-16) ppm. MS (ESI): m/z (%) =647.2 ([M + H]+, 100%). HRMS (ESI) calcd for C34H40KO10 [M + H]+: 647.2259; found 647.2273.
Monopotassium 3,15-disuccinate-12-(6′-methoxylcoumarin) andrographolide 11f
Pale yellow solid, 1.22 g, yield 41.8%, purity 96.0%. 1H NMR (400 MHz, D2O): δ = 7.79-7.76 (1H, m, 8′-H), 7.20-7.16 (1H, m, 7′-H), 6.90-6.86 (1H, m, 5′-H), 6.47 (1H, s, 3′-H), 6.17 (1H, d, J = 15.8 Hz, 12H), 5.71 (1H, m, 11H), 5.03 (1H, s, 13Ha), 4.88 (1H, s, 13-Hb), 4.16 (1H, d, J = 11.8 Hz, 15-Ha), 3.90-3.78 (5H, m, 3H, 15-Hb, 4′-CH3), 2.83-2.56 (9H, m, 9,18,19,22,23H), 2.12-1.65 (9H, m, 1,2,5,6,7H), 1.04 (3H, s, 14H), 0.98 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.6 (C-20), 174.5 (C-24), 173.5 (C-17), 173.1 (C-21), 162.2 (C-4′), 159.3 (C-2′), 157.4 (C-6′), 148.8 (C-8), 142.4 (C-9′), 135.5 (C-11), 128.2 (C-10′), 128.1 (C-12), 124.2 (C-8′), 117.8 (C-7′), 110.9 (C-5′), 109.1 (C-13), 104.7 (C-3′), 75.0 (C-3), 65.8 (C-15), 60.9 (C-9), 55.9 (6′-OCH3), 47.5 (C-5), 40.9 (C-4), 39.8 (C-10), 36.9 (C-7), 33.5 (C-1), 29.5 (C-18), 29.2 (C-22), 29.1 (C-19), 29.0 (C-23), 24.3 (C-2), 24.1 (C-6), 21.8 (C-14), 14.5 (C-16) ppm. MS (ESI): m/z (%) = 663.2 ([M + H]+, 100%). HRMS (ESI) calcd for C34H40KO11 [M + H]+: 663.2208; found 663.2219.
Monopotassium 3,15-disuccinate-12-(6′-methanoylcoumarin) andrographolide 11g
Pale yellow solid, 1.02 g, yield 33.6%, purity 96.3%. 1H NMR (400 MHz, D2O): δ = 7.87-7.84 (1H, m, 8′-H), 7.17-7.12 (2H, m, 5′,7′-H), 6.47 (1H, s, 3′-H), 6.19 (1H, d, J = 15.8 Hz, 12H), 5.72 (1H, m, 11H), 5.03 (1H, s, 13Ha), 4.87 (1H, s, 13-Hb), 4.13 (1H, d, J = 11.8 Hz, 15-Ha), 3.93-3.78 (2H, m, 3H, 15-Hb), 2.83-2.52 (9H, m, 9,18,19,22,23H), 2.28 (3H, s, 6′-OCOCH3), 2.12-1.62 (9H, m, 1,2,5,6,7H), 1.05 (3H, s, 14H), 0.97 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.4 (C-17), 173.1 (C-21), 169.0 (6′-OCOCH3), 162.2 (C-4′), 159.3 (C-2′), 148.8 (C-8), 148.0 (C-6′), 146.9 (C-9′), 135.5 (C-11), 128.0 (C-12), 127.6 (C-10′), 123.9 (C-8′), 121.3 (C-7′), 118.1 (C-5′), 109.1 (C-13), 104.7 (C-3′), 75.1 (C-3), 65.8 (C-15), 60.9 (C-9), 47.4 (C-5), 40.7 (C-4), 39.6(C-10), 36.8 (C-7), 33.5 (C-1), 29.5 (C-18), 29.3 (C-22), 29.1 (C-19), 29.0 (C-23), 24.2 (C-2), 24.0 (C-6), 21.7 (C-14), 20.3 (6′-OCOCH 3), 14.6 (C-16) ppm. MS (ESI): m/z (%) =691.2 ([M + H]+, 100%). HRMS (ESI) calcd for C35H40KO12 [M + H]+: 691.2157; found 691.2172.
Monopotassium 3,15-disuccinate-12-(6′-chlorocoumarin) andrographolide 11h
pale yellow solid, 1.16 g, yield 39.5%, purity 96.4%. 1H NMR (400 MHz, D2O): δ = 8.04-8.01 (1H, m, 5′-H), 7.48-7.36 (2H, m, 7′,8′-H), 6.46 (1H, s, 3′-H), 6.18 (1H, d, J = 15.8 Hz, 12H), 5.75 (1H, m, 11H), 5.05 (1H, s, 13Ha), 4.87 (1H, s, 13-Hb), 4.17 (1H, d, J = 11.8 Hz, 15-Ha), 3.90-3.88 (2H, m, 3H, 15-Hb), 2.83-2.57 (9H, m, 9,18,19,22,23H), 2.14-1.65 (9H, m, 1,2,5,6,7H), 1.04 (3H, s, 14H), 0.93 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1 (C-21), 162.2 (C-4′), 159.3 (C-2′), 148.8 (C-8), 148.2(C-9′), 135.5 (C-11), 131.0 (C-6′), 129.3(C-7′), 128.6 (C-10′), 128.0 (C-12), 126.8 (C-5′), 122.9 (C-8′), 109.1 (C-13), 104.7 (C-3′), 75.1 (C-3), 65.9 (C-15), 60.8 (C-9), 47.5 (C-5), 40.8 (C-4), 39.8 (C-10), 36.8 (C-7), 33.4 (C-1), 29.5 (C-18), 29.4 (C-22), 29.2 (C-19), 29.1 (C-23), 24.3 (C-2), 24.0 (C-6), 21.9 (C-14), 14.8 (C-16) ppm. MS (ESI): m/z (%) = 667.2 ([M + H]+, C33H37 35ClKO10, 100%), 669.2 ([M + H]+, C33H37 37ClKO10, 33%). HRMS (ESI) calcd for C33H37 35ClKO10 [M + H]+: 667.1712, found 667.1736; for C33H37 37ClKO10 [M + H]+: 669.1683, found 669.1699.
Monopotassium 3,15-disuccinate-12-(7'-methylcoumarin) andrographolide 11i
Yellow solid, 1.07 g, yield 37.6%, purity 95.5%. 1H NMR (400 MHz, D2O): δ = 7.74-7.71 (1H, m, 5′-H), 7.03-7.00 (2H, m, 6′,8′-H), 6.48 (1H, s, 3′-H), 6.18 (1H, d, J = 15.8 Hz, 12H), 5.73 (1H, m, 11H), 5.03 (1H, s, 13Ha), 4.87 (1H, s, 13-Hb), 4.14 (1H, d, J = 11.8 Hz, 15-Ha), 3.92-3.78 (2H, m, 3H, 15-Hb), 2.83-2.61 (9H, m, 9,18,19,22,23H), 2.34 (3H, s, 7′-CH3), 2.12-1.62 (9H, m, 1,2,5,6,7H), 1.03 (3H, s, 14H), 0.98 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1 (C-21), 162.1 (C-4′), 159.3 (C-2′), 150.0 (C-9′), 148.8 (C-8), 143.1 (C-7′), 135.5 (C-11), 126.7 (C-5'), 128.0 (C-12), 124.2 (C-10′), 125.7 (C-6′), 117.2 (C-8′), 109.2 (C-13), 104.7 (C-3′), 75.0 (C-3), 65.9 (C-15), 60.8 (C-9), 47.5 (C-5), 40.8 (C-4), 39.8 (C-10), 36.6 (C-7), 33.4 (C-1), 29.5 (C-18), 29.3 (C-22), 29.1 (C-19), 29.0 (C-23), 24.3 (C-2), 24.0 (C-6), 21.9(C-14), 21.2 (7′-CH3), 14.7 (C-16) ppm. MS (ESI): m/z (%) =647.2 ([M + H]+, 100%). HRMS (ESI) calcd for C34H40KO10 [M + H]+: 647.2259; found 647.2276.
Monopotassium 3,15-disuccinate-12-(7'-methoxylcoumarin) andrographolide 11j
Pale yellow solid, 1.07 g, yield 36.9%, purity 96.3%. 1H NMR (400 MHz, D2O): δ = 7.74-7.71 (1H, m, 5′-H), 6.96-6.92 (2H, m, 6′,8′-H), 6.45 (1H, s, 3′-H), 6.19 (1H, d, J = 15.8 Hz, 12H), 5.72 (1H, m, 11H), 5.02 (1H, s, 13-Ha), 4.86 (1H, s, 13-Hb), 4.12 (1H, d, J = 11.8 Hz, 15-Ha), 3.93-3.81 (5H, m, 3H, 15-Hb, 7′-OCH3), 2.83-2.64 (9H, m, 9,18,19,22,23H), 2.13-1.67 (9H, m, 1,2,5,6,7H), 1.04 (3H, s, 14H), 0.96 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1 (C-21), 162.1 (C-4′), 160.3 (C-7′), 159.3 (C-2′), 151.1 (C-9′), 148.8 (C-8), 135.5 (C-11), 130.8 (C-5′), 128.0 (C-12), 119.2 (C-10′), 111.2 (C-6′), 109.1 (C-13), 104.7 (C-3′), 98.2 (C-8′), 75.1 (C-3), 65.7 (C-15), 60.9 (C-9), 55.8 (7′-OCH3), 47.6 (C-5), 40.9 (C-4), 39.8 (C-10), 36.7 (C-7), 33.4 (C-1), 29.4 (C-18), 29.2 (C-22), 29.1 (C-19), 29.0 (C-23), 24.1 (C-2), 24.0 (C-6), 21.6 (C-14), 14.7 (C-16) ppm. MS (ESI): m/z (%) =663.2 ([M + H]+, 100%). HRMS (ESI) calcd for C34H40KO11 [M + H]+: 663.2208; found 663.2219.
Monopotassium 3,15-disuccinate-12-(7'-methanoylcoumarin) andrographolide 11k
Pale yellow solid, 1.19 g, yield 39.2%, purity 95.2%. 1H NMR (400 MHz, D2O): δ = 7.83–7.80 (1H, m, 5′-H), 7.41-7.38 (1H, m, 8′-H), 7.28-7.25 (1H, m, 6′-H), 6.46 (1H, s, 3′-H), 6.18 (1H, d, J = 15.8 Hz, 12H), 5.71 (1H, m, 11H), 5.03 (1H, s, 13Ha), 4.86 (1H, s, 13-Hb), 4.12 (1H, d, J = 11.8 Hz, 15-Ha), 3.93–3.78 (2H, m, 3H, 15-Hb), 2.83-2.52 (9H, m, 9,18,19,22,23H), 2.28 (3H, s, 7′-OCOCH3), 2.11-1.61 (9H, m, 1,2,5,6,7H), 1.03 (3H, s, 14H), 0.96 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1 (C-21), 169.1 (7′-OCOCH3), 162.4 (C-4′), 159.4 (C-2′), 152.7 (C-7′), 150.5 (C-9′), 148.7 (C-8), 135.5 (C-11), 130.1 (C-5′), 128.0 (C-12), 124.0 (C-10′), 118.4 (C-6′), 114.9 (C-8′), 109.1 (C-13), 104.8 (C-3′), 75.1 (C-3), 65.9 (C-15), 60.8 (C-9), 47.5 (C-5), 40.9 (C-4), 39.6 (C-10), 36.7 (C-7), 33.5 (C-1), 29.4 (C-18), 29.2 (C-22), 29.1 (C-19), 29.0 (C-23), 24.2 (C-2), 24.0 (C-6), 21.7 (C-14), 20.3 (7′-OCOCH 3), 14.5 (C-16) ppm. MS (ESI): m/z (%) =691.2 ([M + H]+, 100%). HRMS (ESI) calcd for C35H40KO12 [M + H]+: 691.2157; found 691.2174.
Monopotassium 3,15-disuccinate-12-(7'-chlorocoumarin) andrographolide 11l
Yellow solid, 1.19 g, yield 40.5%, purity 96.2%. 1H NMR (400 MHz, D2O): δ = 7.80-7.78 (1H, m, 5′-H), 7.25-7.20 (2H, m, 6′,8′-H), 6.48 (1H, s, 3′-H), 6.17 (1H, d, J = 15.8 Hz, 12H), 5.72 (1H, m, 11H), 5.02 (1H, s, 13-Ha), 4.88 (1H, s, 13-Hb), 4.12 (1H, d, J = 11.8 Hz, 15-Ha), 3.94-3.79 (2H, m, 3H, 15-Hb), 2.83-2.62 (9H, m, 9,18,19,22,23H), 2.12-1.79 (9H, m, 1,2,5,6,7H), 1.05 (3H, s, 14H), 0.94 (3H, s, 16H). 13C NMR (100 MHz, D2O): δ = 174.7 (C-20), 174.6 (C-24), 173.2 (C-17), 173.1(C-21), 162.4 (C-4′), 159.6 (C-2′), 151.6 (C-9′), 148.7 (C-8), 135.6 (C-11), 135.1 (C-7′), 128.2 (C-5′), 128.0 (C-12), 125.5 (C-6'), 125.2 (C-10′), 119.6 (C-8′), 109.1 (C-13), 104.7 (C-3′), 75.0 (C-3), 65.9 (C-15), 60.9 (C-9), 47.2 (C-5), 40.8 (C-4), 39.8 (C-10), 36.8 (C-7), 33.4 (C-1), 29.5 (C-18), 29.3 (C-22), 29.1 (C-19), 29.0 (C-23), 24.3 (C-2), 24.0 (C-6), 21.8 (C-14), 14.5 (C-16) ppm. MS (ESI): m/z (%) =667.2 ([M + H]+, C33H37 35ClKO10, 100%), 669.2 ([M + H]+, C33H37 37ClKO10, 33%). HRMS (ESI) calcd for C33H37 35ClKO10 [M + H]+: 667.1712, found 667.1738; for C33H37 37ClKO10 [M + H]+: 669.1683, found 669.1601.
Biological Assays In Vitro
Fresh arterial blood was taken from groin of SD male rats with 3.8% sodium citrate as anticoagulant (9:1 by volume). Then, whole blood samples were centrifuged at 1000 rpm/min for 10 minutes at room temperature to give the platelet-rich plasma (PRP). The residue continued to be centrifuged at 3000 rpm/min for 10 minutes to prepare platelet-poor plasma (PPP). PPP was employed to be the blank control. Thrombin, ADP, AA, collagen were employed as inducers. Aspirin was selected to be the positive control. Normal saline was selected as the negative control. The sample groups (5 µL) were prepared with target compounds dissolved in normal saline (1.7 μmol/L, 3.4μmol/L, 6.8μmol/L) and then was added into PRP-2 (200 µL) and incubated for 2 minutes, as well as positive control and negative control. Afterward, adding 20 µL thrombin (0.1 U/mL), ADP (5 mM/L), AA (20μM), collagen (1 mg/mL), respectively, induced platelet aggregation. IR could be calculated by the formula IR = [1 − (SG/NC)] × 100% (SG and NC represented the platelet aggregation rates of sample group and negative control, respectively)
Cytotoxicity Assay In Vitro
Cell toxicity of target derivatives
Statistical Analysis
Results were presented as the means ± standard error. Data were analyzed with one-way analysis of variance (SPSS software) to evaluate statistical significance of the differences. The level of significance was considered at P < 0.05 and P < 0.01.
Footnotes
Acknowledgements
We are grateful to the Natural Science Foundation of Jiangxi Province (No. 20171BAB215075), Traditional Chinese Medicine Research Key Projects of Health and Family Planning Commission of Jiangxi Province (No. 2017Z018 and No. 2018A314) for financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
