Abstract
Capsaicin, an ingredient of green and red bell peppers, shows anticancer activity in several malignant cell lines. Thymidylate synthase (TS) is a well-validated anticancer drug target in non-small cell lung cancer (NSCLC) cells. However, whether capsaicin and 5-fluorouracil (5-FU) induce synergistic cytotoxicity in NSCLC cells by regulating TS expression is unclear. This study investigated the cytotoxicity of capsaicin and 5-FU co-treatment on two hoursuman lung adenocarcinoma cell lines, H520 and H1703, and the underlying mechanisms. Capsaicin decreased TS expression in a p38 mitogen-activated protein kinase (MAPK) inactivation–dependent manner in H520 and H1703 cells. Enhancement of p38 MAPK activity by transfection with constitutive active mitogen-activated protein kinase kinase six vectors increased TS expression and cell survival. In addition, capsaicin and 5-FU co-treatment enhanced synergistic cytotoxicity and inhibited cell growth associated with TS downregulation and p38 MAPK inactivation in H520 and H1703 cells. Capsaicin and 5-FU co-treatment did not affect the cellular content of capsaicin. These results show that capsaicin may be combined with 5-FU to treat NSCLC.
Non-small cell lung cancer (NSCLC) accounts for most lung cancer–related deaths worldwide 1,2 and 80% of all lung cancer cases. 3 Thymidylate synthase (TS), a therapeutic target of 5-fluorouracil (5-FU) and capecitabine (a prodrug of 5-FU), catalyze the de novo synthesis of deoxythymidine monophosphate (dTMP) in cells. 4 5-FU is an antimetabolite chemotherapeutic drug, and its cytotoxicity inhibits TS expression. High TS expression is a primary cause of resistance to TS inhibitors. For example, increased TS expression leads to pemetrexed resistance in NSCLC 5 and 5-FU resistance in colon cancer. 6 High TS expression is also associated with poor prognosis in NSCLC patients. 7 Heat shock protein 90 (HSP90) inhibition downregulates TS expression and extracellular signal-regulated protein kinase (ERK)1/2 activation and sensitizes colorectal cancer cells to 5-FU-based chemotherapy. 8 In addition, 5-FU induces p38 mitogen-activated protein kinase (MAPK) activation and mediates apoptosis, while 5-FU inhibition promotes cell survival. 9 However, the role of p38 MAPK in regulating TS expression and in chemoresistance of NSCLC cells to 5-FU is still unclear.
Capsaicin (trans-8-methyl-N-vanillyl-6-nonenamide), the major pungent principle component found in red and chili peppers, 10 has pharmacological properties such as anticancer and anti-inflammatory activities. 11 -13 Capsaicin shows anticancer activity in animal models, inhibiting lung carcinogenesis. 14 In addition, capsaicin increases apoptosis and decreases benzo(a)pyrene-induced lung tumorigenesis in Swiss albino mice. 15 It also sensitizes hepatocellular carcinoma cells to sorafenib (a multikinase inhibitor) through protein kinase B (AKT) signal inhibition. 16 A recent study showed that the capsaicin-induced anticancer effects on osteosarcoma cell lines involve multiple MAPK signaling pathways, as indicated by inactivation of ERK1/2 and p38 pathways and activation of the c-Jun N-terminal kinase (JNK) MAPK pathway. 17
This study investigated the cytotoxicity of capsaicin and 5-FU co-treatment on NSCLC cells. We selected the human lung squamous cell carcinoma cell lines H520 and H1703 to investigate whether capsaicin has anticancer effects and to clarify further the underlying mechanisms. We also analyzed the molecular mechanism underlying regulation of TS expression by capsaicin to enhance the cytotoxic effect of 5-FU in H520 and H1703 cells. Our results may provide a rationale for combining capsaicin with 5-FU to treat NSCLC.
Materials and Methods
Cell Lines and Chemicals
We obtained human lung squamous cell carcinoma cell lines H520 and H1703 from the American Type Culture Collection (Manassas, VA, USA) and cultured them at 37 °C in a 5% CO2 humidified atmosphere in Roswell Park Memorial Institute (RPMI) 1640 complete medium supplemented with 2.2% (w/v) sodium bicarbonate, 0.03% (w/v)
We purchased capsaicin with 90% purity from Fluorochem (Pamplona, Spain), 5-FU from Bristol-Myers Squibb (New York City, NY, USA), cycloheximide (CHX) and actinomycin D from Sigma-Aldrich (St. Louis, MO, USA), and SB2023580 from Calbiochem-Novabiochem (San Diego, CA, USA). Actinomycin D and SB2023580 were dissolved in dimethyl sulfoxide (DMSO), and CHX was dissolved in Milli-Q-purified water (Millipore, Billerica, MA, USA).
Western Blot Analysis
After different treatments, we subjected equal amounts of proteins from each set of experiments to Western blot analysis, as previously described. 18
Plasmid and Transfection
We plated exponentially growing H520 and H1703 cells (106) for 18 hours and then obtained the MAPK kinase 6E (MKK6E; a constitutively active vector form of MKK6), as previously described. 19 The sense-strand sequences of short interfering RNA (siRNA) duplexes were as follows:
TS: 5′-GCACAUAUUUACCUGAAUC-3′
Scrambled (control): 5′-GCG CGC UUU GUA GGA TTC G-3′ (Dharmacon Research, Lafayette, CO, USA)
We purchased the p38α MAPK siRNA (sc-29433) from Santa Cruz Biotechnology (Santa Cruz, CA, USA). H520 and H1703 cells were transfected with 200 nM siRNA duplexes using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) for 24 hours.
Quantitative Real-Time Polymerase Chain Reaction
We performed polymerase chain reaction (PCR) using ABI Prism 7900HT according to the manufacturer’s instructions. Specific PCR products were amplified using the SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA). For each sample, we normalized the data to the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The designed primers were as follows:
TS forward primer: 5'- ACTGCAAAGAGTGATTGACACC-3'
TS reverse primer: 5'-CACTGTTCACCACATAGAACTGG-3'
GAPDH forward primer: 5'-CATGAGAAGTATGACAACAGCCT-3'
GAPDH reverse primer: 5'-AGTCCTTCCACGATACCAAAGT-3'
We performed analysis using the comparative Ct value method. For each sample, data were normalized to GAPDH.
MTS Assay
We performed in vitro 3-(4,5-dimethylthiazol-2-yl)−5-(3-carboxymethoxyphenol)−2-(4- sulfophenyl)−2H-tetrazolium (MTS) assay. Briefly, we cultured H520 and H1703 cells in 96-well tissue culture plates at a density of 5000 cells/well. To assess cell viability, we added drugs after plating. At the end of culture, we added 20 µL of MTS solution (CellTiter 96 Aqueous One solution Cell Proliferation Assay; Promega, Madison, WI, USA), incubated the cells for a further 2 hours, and measured absorbance at a wavelength of 490 nm using an ELISA plate reader (Biorad Technologies, Hercules, CA, USA).
Trypan Blue Dye Exclusion Assay
We treated H520 and H1703 cells with capsaicin and/or 5-FU for 24, 48, and 72 hours. Post-treatment, we harvested 500 cells and measured the proportion of dead cells using a hemocytometer by counting the number of cells stained with trypan blue, which can penetrate dead cells, but is excluded from living cells, as follows:
Colony Formation Assay
Post-treatment, H520 and H1703 cells were immediately washed with phosphate-buffered saline (PBS) and trypsinized to quantify cell numbers. We plated the cells at a density of 500 cells per 60 mm Petri dish in triplicate for each treatment, cultured them for 7‐14 days, and stained cell colonies with 1% crystal violet solution in 30% ethanol. Finally, cytotoxicity was determined as follows 20 :
Electrochemical Measurement of Cellular Capsaicin Content
We performed electrochemical measurements using a CHI 830 electrochemical workstation. Briefly, we connected an integrated three-electrode strip to the electrochemical workstation equipped with a specially designed electrochemical cell. For measurements, we placed a 20 µL sample drop on the electrochemical cell; an exposed part of the electrode system was not covered with insulating film, creating an electrochemical reaction zone for forming a reaction layer. Each experiment utilized a new electrode strip in a non-deaerated and unstirred solution.
Statistical Analysis
For each protocol, we performed three or four independent experiments. Results were expressed as the mean ± standard error of the mean (SEM). Statistical calculations were performed using SigmaPlot 2000 software (Systat Software, San Jose, CA, USA), and differences in measured variables between experimental and control groups were assessed using the unpaired t test. P < .05 was considered statistically significant.
Results
TS mRNA and Protein Levels Decreased After Capsaicin Exposure
Capsaicin (12.5, 100 µM) inhibits NSCLC-induced endothelial cell migration. 21 To determine whether TS expression is associated with the effects of capsaicin, we treated either H520 or H1703 cells with either 100 µM capsaicin for 4‐24 hours or different concentrations of capsaicin (5, 10, 20, 40, 60, 120 µM) for 24 hours, and then used real-time PCR and Western blot analysis to determine TS mRNA and protein expression, respectively. Capsaicin decreased TS mRNA and protein expression in a time- and dose-dependent manner (Figure 1(A–D)). Capsaicin also decreased phospho-p38 MAPK expression. To determine the role of p38 MAPK in regulating TS expression, we investigated whether an increase in p38 MAPK activity inhibits TS downregulation in capsaicin-treated H520 and H1703 cells. H520 and H1703 cells were transiently transfected with a plasmid carrying MKK6E (Figure 1(A and D)). Compared to transfection with the control vector pcDNA3, MKK6E transfection restored the capsaicin-induced decrease in TS mRNA and protein expression. However, once the H520 and H1703 cells were pretreated with p38 MPAK inhibitor (SB2023580) or transfected with si-p38 RNA, the TS mRNA and protein expression in capsaicin-treated H520 and H1703 cells further decreased (Figure 2(C–F)). Therefore, capsaicin-induced TS downregulation is associated with p38 MAPK inactivation.

Capsaicin decreased TS expression in a dose- and time-dependent manner. (A and B) (Left) H520 and H1703 cells (106) were cultured in complete medium for 18 hours and treated with 50 µM capsaicin for 4, 8, 12, 16, and 24 hours. (Right) Different concentrations (5, 120 µM) of capsaicin were added to H520 and H1703 cells in complete medium for 24 hours. Total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression. Results (mean ± SEM) were from three independent experiments. *P < .05 and **P < .01 using Student’s t test for comparison between treated and untreated cells. (C and D) Post-treatment, cell extracts were examined by Western blot analysis to determine TS, phospho-p38 MAPK, actin, and p38 MAPK expression.

Capsaicin decreased TS expression via p38 MAPK inactivation. (A and B) H520 and H1703 cells (5 × 105) were transfected with MKK6E for 24 hours prior to capsaicin treatment in complete medium for 24 hours. Results (mean ± SEM) were from three independent experiments. **P < .01 using Student’s t test for comparison between capsaicin-treated cDNA3- and MKK6E-transfected cells. (C and D) SB2023580 (10 µM) was added to H520 and H1703 cells for 1 hour before 60 µM capsaicin treatment for 24 hours. Results (mean ± SEM) were from four independent experiments. **P < .01 using Student’s t test for comparison between cells treated with either capsaicin-DMSO or capsaicin-SB2023580. (E and F) H520 and H1703 cells (5 × 105) were transfected with p38 MAPK siRNA for 24 hours prior to 60 µM capsaicin treatment in complete medium for 24 hours. **P < .01 using Student’s t test for comparison between capsaicin-treated si-p38 RNA-or si-scrambled RNA-transfected cells. Post-treatment, cell extracts were examined via real-time PCR (A, C, E) and Western blot analysis (B, D, F) to determine TS mRNA and protein expression, respectively.
Capsaicin Decreased TS mRNA and Protein Stability via P38 MAPK Inactivation
We examined the possible mechanisms underlying post-transcriptional regulation of TS transcripts after capsaicin and SB2023580 co-treatment. In the presence of actinomycin D, capsaicin-treated H520 and H1703 cells showed decreased TS mRNA expression compared to untreated cells (Figure 3(A)). In addition, TS expression progressively decreased with time in the presence of CHX (an inhibitor of de novo protein synthesis) (Figure 3(B)). However, capsaicin-treated H520 and H1703 cells showed significant TS degradation after CHX treatment compared to untreated cells (Figure 3(B)). Therefore, TS in H520 and H1703 cells is less stable after capsaicin treatment. Interestingly, SB2023580 co-treatment enhanced TS mRNA and protein instability in capsaicin-treated H520 and H1703 cells (Figure 3(A and B)). Therefore, capsaicin decreases TS expression by enhanced TS mRNA instability via p38 MAPK inactivation.

Capsaicin decreased TS mRNA and protein stability via p38 MAPK inactivation. (A) H520 and H1703 cells were treated with 60 µM capsaicin and/or 10 µM SB2023580 for 12 hours with or without 2 µg/mL of actinomycin D for 4, 8, 12, and 24 hours, and total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression. (B) Cells were treated with 60 µM capsaicin and/or 10 µM SB2023580 for 12 hours, followed by co-treatment with 0.1 mg/mL of CHX for 8, 12, and 24 hours. Whole-cell extracts were collected for Western blot analysis.
TS Downregulation Regulated Capsaicin-Induced Cytotoxicity and Growth Inhibition in H520 and H1703 Cells
We next examined the effect of siRNA-mediated TS knockdown on capsaicin-induced cytotoxicity and cell growth inhibition in H520 and H1703 cells. At 24 hours post-transfection, real-time PCR analysis showed a further decrease in TS mRNA expression in capsaicin-treated H520 and H1703 cells (Figure 4(A)). In addition, inhibition of TS expression by si-TS RNA increased the cells’ sensitivity to capsaicin compared to si-control transfected cells (Figure 4(B)). The combination of TS siRNA and capsaicin induced more inhibition of cell growth in H520 and H1703 cells compared to capsaicin alone (Figure 4(C)). In contrast, forced expression of the Flag-TS vector decreased capsaicin-induced cytotoxicity and growth inhibition (Figure 4(D and E)). Therefore, TS downregulation enhances capsaicin-induced cytotoxicity and growth inhibition in H520 and H1703 cells.

TS downregulation expression enhanced capsaicin-induced cytotoxicity and growth inhibition. (A) H520 and H1703 cells were either transfected with siRNA duplexes (200 nM) specific to TS or scrambled (control) in complete medium for 24 hours prior to capsaicin treatment in complete medium for 24 hours; total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression. (B) Post-treatment, cytotoxicity was determined by MTS assay. (C) Cells were either transfected with si-TS or si-scrambled RNA and then treated with 60 µM capsaicin for 24, 48, and 72 hours; living cells were determined by MTS assay. Results (mean ± SEM) were from three independent experiments. **P < .01 using Student’s t test for comparison between capsaicin-treated si-TS RNA-or si-scrambled RNA-transfected cells. (D) Cells were either transfected with pcDNA3 or Flag-TS expression vector for 24 hours and then treated with capsaicin for 24 hours. Cytotoxicity was determined by MTS assay. (E) After transfection with either pcDNA3 or Flag-TS, the cells were treated with 120 µM capsaicin for 24, 48, and 72 hours; living cells were determined by MTS assay. **P < .01 using Student’s t test for comparison between capsaicin-treated pcDNA3- or Flag-TS-transfected cells.
The P38 MAPK Signaling Pathway Mediated Capsaicin-Induced Cytotoxicity
Forced MKK6E expression restored the capsaicin-induced decrease in H520 and H1703 cell viability (Figure 5(A)) and also reversed capsaicin-induced growth inhibition (Figure 5(B)). In contrast, p38 MAPK inactivation by SB2023580 or knockdown of p38 MAPK expression further significantly decreased cell viability in capsaicin-treated H520 and H1703 cells compared to capsaicin alone (Figure 5(C and E)), and more effectively inhibited cell growth than either capsaicin or SB2023580 alone (Figure 5). Therefore, TS downregulation and p38 MAPK inactivation affect capsaicin-induced cytotoxicity and growth inhibition in H520 and H1703 cells.

Enhancement of p38 MAPK activity by MKK6E transfection decreased capsaicin-induced cytotoxicity. (A) After either pcDNA3 or MKK6E transfection, H520 and H1703 cells were treated with capsaicin for 24 hours. Cytotoxicity was determined by MTS assay. Results (mean ± SEM) were from three independent experiments. (B) After either pcDNA3 or MKK6E transfection, cells were treated with 50 µM capsaicin for 24, 48, and 72 hours; living cells were determined by MTS assay. *P < .05 and **P < .01 using Student’s t test for comparison between capsaicin-treated MKK6E or pcDNA3 vector–transfected cells. (C) Cells were pretreated with 5 µM SB2023580 for 1 hour and then co-treated with different concentrations of capsaicin for 24 hours. Cytotoxicity was determined by MTS assay. **P < .01 using Student’s t test for comparison between capsaicin-treated cells either with or without SB2023580 pretreatment. (D) Cells were treated with 60 µM capsaicin and/or 2.5 µM SB2023580 for 1‐3 days; living cells were determined by trypan blue dye exclusion assay. **P < .01 using Student’s t test for comparison between capsaicin-treated and capsaicin +SB2023580-treated cells. (E) After either si-p38 MAPK or si-scrambled RNA transfection, cells were treated with capsaicin for 24 hours; cytotoxicity was determined by MTS assay. Results (mean ± SEM) were from three independent experiments. (F) After either si-p38 MAPK or si-scrambled RNA transfection, cells were treated with capsaicin for 24, 48, and 72 hours; living cells were determined by MTS assay. a,b*P < .05 and a,b**P < .01 using Student’s t test for comparison between capsaicin-treated (40 or 120 µM) si-p38 MAPK-or si-scrambled RNA-transfected cells. Abbreviations:MAPK, mitogen-activated protein kinase; PBS, phosphate-buffered saline.

Capsaicin and 5-FU co-treatment did not affect the capsaicin content of H520 and H1703 cells. (A and B) Cells were treated with 60 µM capsaicin and/or 50 µg/mL of 5-FU; Screen-printed electrodes (SPEs) were used to analyze the cellular content of capsaicin. Cyclic voltammograms of treatment were observed at a bare SPE in 0.2 M phosphate-bufferedsaline (PBS) at a scan rate of 0.02 V/s.
Capsaicin and 5-FU Exerted a Synergistic Cytotoxic Effect on H520 and H1703 Cells
We analyzed whether capsaicin enhances the cytotoxic effect of 5-FU by downregulating TS expression in H520 and H1703 cells. We determined the effect of capsaicin and 5-FU co-treatment on cell viability using MTS assay. Capsaicin and 5-FU co-treatment for 24 hours resulted in a higher loss of cell viability compared to capsaicin or 5-FU treatment alone (Figure 6(A and B)). Next, H520 and H1703 cells were treated with capsaicin and/or 5-FU, and cell proliferation was determined 1‐3 days post-treatment. Capsaicin and 5-FU co-treatment had a higher cell growth inhibition effect compared to either capsaicin or 5-FU treatment alone (Figure 6(C)). In addition, the colony-forming ability of capsaicin and 5-FU co-treatment was lower compared to 5-FU treatment alone in H520 (5.36%, 17.50%) and H1703 cells (3.50%, 14.67%) (Figure 6(D)). Therefore, capsaicin and 5-FU co-treatment has a synergistic cytotoxic effect on H520 and H1703 cells.
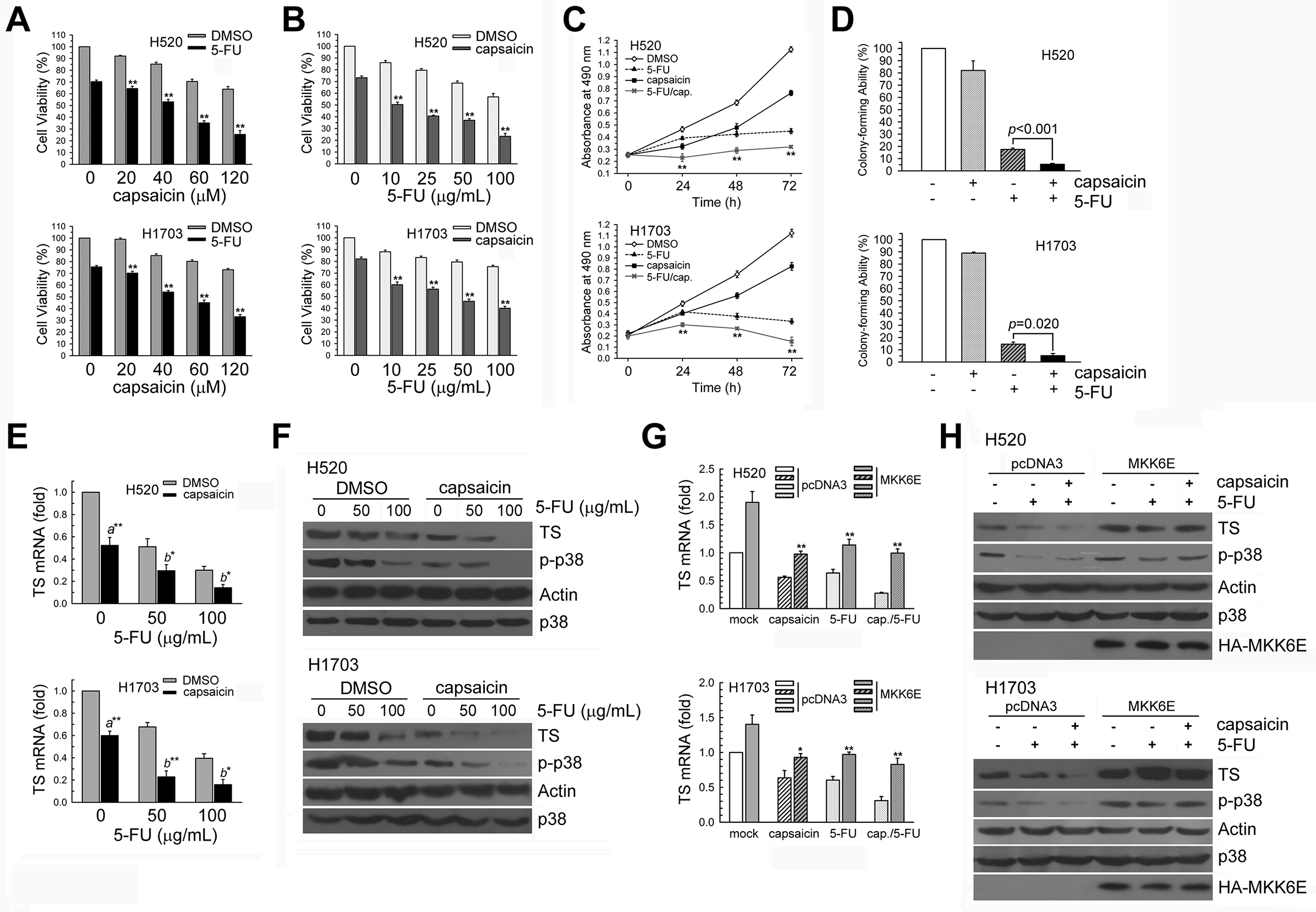
Capsaicin and 5-FU co-treatment synergistically enhanced cytotoxicity. (A and B) H520 and H1703 cells were treated with 60 µM capsaicin and/or 50 µg/mL of 5-FU; cell viability was analyzed by MTS assay. (C) Cells were treated with 60 µM capsaicin and/or 10 µg/mL of 5-FU for 1‐3 days; living cells were determined by MTS assay. **P < .01 using Student’s t test for comparison between cells treated with either capsaicin or 5-FU alone or both. (D) Cells were treated with 40 µM capsaicin and/or 10 µg/mL of 5-FU for 24 hours, and the colony-forming ability of cells was determined by Colony formation assay (CFA). **P < .01 using Student’s t test for comparison between cells treated with either 5-FU alone or with capsaicin and 5-FU. (E) Cells (106) were cultured in complete medium for 18 hours and then treated with 60 µM capsaicin and 50 and 100 µg/mL of 5-FU for 24 hours. Post-treatment, total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression. Results (mean ± SD) were from four independent experiments. a**P < .01 using Student’s t test for comparison between cells treated either with or without capsaicin. b**P < .01 and b*P < .05 using Student’s t test for comparison between cells treated with either capsaicin alone or capsaicin and 5-FU. (F) Post-treatment, cell extracts were examined by Western blot analysis to determine TS, phospho-p38 MAPK, actin, and p38 MAPK expression. (G and H) Either MKK6E (3 µg) or pcDNA3 (3 µg) expression plasmids were transfected into cells using lipofectamine. After expression for 24 hours, the cells were treated with 60 µM capsaicin and 50 µg/mL of 5-FU for an additional 24 hours; total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression and Western blot analysis to determine TS expression. Abbreviations:MAPK, mitogen-activated protein kinase; PCR, polymerase chain reaction.
Capsaicin-Induced TS mRNA and Protein Downregulation in 5-FU-Treated H520 and H1703 Cells
To determine the mechanism underlying the synergistic effect of capsaicin and 5-FU co-treatment, we hypothesized that capsaicin affects TS expression in 5-FU-treated NSCLC cells. To test this hypothesis, we exposed H520 and H1703 cells to capsaicin and/or 5-FU for 24 hours. Capsaicin decreased TS mRNA and protein expression in 5-FU-treated H520 and H1703 cells (Figure 6(E and F)). In addition, capsaicin decreased the phospho-p38 MAPK levels in 5-FU-treated H520 and H1703 cells (Figure 6(F)). Moreover, forced MKK6E expression increased TS mRNA and protein expression in 5-FU-treated and capsaicin +5-FU-co-treated H520 and H1703 cells (Figure 6(G and H)).
Capsaicin and 5-FU Co-Treatment Did Not Affect the Capsaicin Content of H520 and H1703 Cells
In recent years, screen-printed electrodes have attracted considerable attention in electrochemical sensing due to the inherent superiority of their manufacturing process, which is rapid, simple, inexpensive and easy to mass produce. Screen-printing technology characterizes a well-established technique for the fabrication of electrochemical sensors with high selectivity and sensitivity. 22 -25 Capsaicin has a functional base similar to that of norepinephrine, and the hydroxyl group (catechol) on the benzene ring can lose the electrochemical reaction of electrons being further oxidized into carbonyl (o-quinone derivative). In Figures 5 and 7, FU co-treatment did not affect the cellular amount of capsaicin in NSCLC cells.
MKK6E Transfection Increased TS Expression and Cell Survival Induced by Capsaicin and 5-FU Co-Treatment
We investigated whether capsaicin and 5-FU co-treatment–mediated TS downregulation is correlated with p38 MAPK inactivation in H520 and H1703 cells. Forced MKK6E expression increased TS mRNA and protein expression in capsaicin +5-FU-cotreated H520 and H1703 cells and also restored cell viability decreased by capsaicin and 5-FU co-treatment (Figure 8(A–C)). In contrast, p38 MAPK inactivation by si-p38 RNA transfection further decreased TS expression and cell viability in capsaicin +5-FU-cotreated H520 and H1703 cells (Figure 8(D–F)). Therefore, capsaicin enhances 5-FU-induced cytotoxicity by p38 MAPK inactivation–mediated TS downregulation in H520 and H1703 cells.

Enhancement of p38 MAPK activity restored the capsaicin +5-FU-induced inhibition of TS expression and cell survival in H520 and H1703 cells. (A and B) Either MKK6E (3 µg) or pcDNA3 (3 µg) expression plasmids were transfected into H520 and H1703 cells using lipofectamine. After expression for 24 hours, the cells were treated with capsaicin and 5-FU for an additional 24 hours; total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression and Western blot to determine TS expression. (C) Post-treatment as in (A), cytotoxicity was determined by MTS assay. Results (mean ± SD) were from four independent experiments. **P < .01 using Student’s t test for comparison between capsaicin +5-FU-treated MKK6E- and pcDNA3-transfected cells. (D) Cells were transfected with si-p38 RNA for 24 hours and then co-treated with 60 µM capsaicin and 10 µg/mL of 5-FU for 24 hours; total RNA was isolated and subjected to real-time PCR to determine TS mRNA expression. (E) Post-treatment, whole-cell extracts were collected for Western blot analysis. (F) Post-treatment as in (D), cytotoxicity was determined by MTS assay. *P < .05 and **P < .01 using Student’s t test for comparison between capsaicin +5-FU-treated in si-p38 MAPK- or si-scrambled RNA-transfected cells. Abbreviations:MAPK, mitogen-activated protein kinase; PCR, polymerase chain reaction.
Discussion
Capsaicin, the major pungent principle component found in bell peppers, has anticancer, anti-inflammatory, and antiproliferative activities. 26 -29 However, capsaicin inhibits the growth of leukemic cells, but not normal bone marrow mononuclear cells. 30 We found that p38 MAPK inactivation is correlated with TS mRNA and protein downregulation in capsaicin-exposed NSCLC cells. Enhancement of p38 MAPK activity restores the capsaicin-induced decrease in cell viability. Therefore, TS downregulation is involved in regulating capsaicin-induced cytotoxicity and growth inhibition in NSCLC cells.
Preclinical and clinical studies have shown a strong association between increased TS expression and development of resistance to 5-FU. 31,32 For example, B7-H3, a member of the B7 family, induces chemoresistance to 5-FU via TS upregulation through the phosphoinositide 3-kinase (PI3K)/AKT signaling pathway in colorectal cancer. 33 In addition, inhibition of the mammalian target of rapamycin (mTOR) acts synergistically with 5-FU by downregulating TS expression in gastric cancer. 34 Capsaicin and 5-FU co-treatment synergistically inhibits tumor growth via activation of the PI3K/AKT/mTOR pathway in the cholangiocarcinoma xenograft to a greater extent compared to 5-FU treatment alone. 35 These findings are consistent with our results. We found that capsaicin and 5-FU co-treatment inhibits NSCLC cell proliferation and viability associated with TS downregulation and p38 MAPK inactivation. However, capsaicin and 5-FU co-treatment does not affect the capsaicin content of NSCLC cells.
Capsaicin induces apoptosis by persistent disruption of the mitochondrial membrane potential. 36 In liver cells, capsaicin-induced apoptosis is involved in the increase in intracellular Ca2+, heme oxygenase-1 expression, 37 reactive oxygen species (ROS) generation, and then caspase-3 activation. 38 In hepatocellular carcinoma, capsaicin inhibits cell proliferation by inducing autophagy and apoptosis. 39 In addition, capsaicin activates p38 and JNK MAPK pathways, and p38 and JNK inhibitors protect against capsaicin-induced apoptosis by caspase-3, caspase-8, and caspase-9 downregulation. Capsaicin also significantly slows the growth of 786-O renal cancer xenografts in vivo 40 and induces antiproliferative effects in human small cell lung cancer and nude mice models via the E2F pathway. 41 In addition, capsaicin induces apoptosis via inactivation of p38 MAPK and ERK1/2 pathways and activation of the JNK MAPK pathway in human osteosarcoma. 17 In this study, capsaicin induced cytotoxicity in NSCLC cells via TS downregulation and p38 MAPK inactivation.
A previous study has shown that capsaicin treatment may be a useful adjunct therapy to improve chemosensitivity of 5-FU in cholangiocarcinoma via activation of the PI3K/AKT/mTOR pathway. 35 In addition, capsaicin inhibits AKT, providing a possible pathway to sensitize hepatocellular carcinoma cells to sorafenib via increased apoptosis, followed by caspase-9 activation. 16 In this study, capsaicin and 5-FU co-treatment enhanced p38 MAPK inactivation and downregulated TS in 5-FU-exposed H520 and H1703 cells, subsequently resulting in synergistic cytotoxicity in NSCLC cells.
To conclude, capsaicin enhances the sensitivity of NSCLC cells to 5-FU via p38 MAPK inactivation–mediated TS downregulation. Although further studies are required in order to evaluate the effects of capsaicin and 5-FU co-treatment in vivo, it represents a promising and attractive strategy for NSCLC treatment. Our results may provide additional insight into the potential synergistic effects of capsaicin and 5-FU co-treatment on NSCLC.
Footnotes
Acknowledgments
We thank Dr Jia-Ling Yang for providing us with the MKK6E plasmids. This study was funded by grants from the Ministry of Science and Technology, Taiwan, Grant MOST 108-2320-B-415-004 -, MOST 109-2314-B-002-175 -, and Ditmanson Medical Foundation Chia-Yi Christian Hospital Research Program R109-38(C-L. Tung).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


