Abstract
Alzheimer's disease (AD) is the most common neurodegenerative disease associated with aging. Bupleurum smithii Wolff. is a Chinese folk medicine used to reduce fever and inflammation. Regarding the key role of neuroinflammation in AD pathogenesis, it was speculated that B. smithii may be the source of compounds that treat AD through anti-inflammatory effects. This study aimed to investigate the effects of saikogenin F, a natural active ingredient from B. smithii, on cognition impairment and neuroinflammation in AD mice induced by amyloid β (Aβ). The AD mice model was established by intracerebroventricular (i.c.v.) injection of Aβ, and different doses of saikogenin F (10, 20, and 40 mg/kg) were intragastrically administrated once daily. Results of behavioral experiments, including the novel object recognition (NOR) test, Y-maze test, and Morris water maze (MWZ) test, showed that saikogenin F could ameliorate Aβ-induced cognition impairment in AD mice. Enzyme linked immunosorbent assay (ELISA) results showed that tumor necrosis factor α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), and reactive oxygen species (ROS) levels in hippocampal tissue increased after Aβ injection, while saikogenin F could significantly reduce the concentrations of these inflammatory factors. Western blotting results revealed that the Aβ-induced reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase subunits protein expression in mice hippocampus was remarkably downregulated by saikogenin F. Results of Iba-1 immunohistochemical staining showed that saikogenin F could effectively inhibit Aβ-induced activation of microglia in vivo. These results suggested that saikogenin F could relieve Aβ-induced cognitive impairment via inhibiting neuroinflammation and microglial activation. These effects may be achieved by inhibiting the expression of the NADPH oxidase subunits gp91phox and p47phox.
Introduction
Alzheimer's disease (AD) is the most common neurodegenerative disease associated with aging, which is characterized by progressive dementia with memory loss, cognitive impairment and deterioration of mental functions. 1 The deposition of amyloid β (Aβ) in the brain and blood vessels constitutes the main pathological sign of AD. 2 In recent years, microglia-mediated neuroinflammation has been recognized to play a key role in the pathogenesis of AD. Microglia are resident immune cells in the brain and play a key role in host prevention and tissue repair in the central nervous system. 3 When Aβ and lipopolysaccharide (LPS) cause either brain damage or immune stimulation, microglia are overactivated and release various pro-inflammatory factors, such as reactive oxygen species (ROS), nitric oxide (NO), tumor necrosis factor α (TNF-α), and interleukin-1β (IL-1β).4,5 These pro-inflammatory mediators contribute to the damage of neurons and further aggravate the deterioration of AD. Therefore, inhibiting microglia-mediated neuroinflammation may be a therapeutic option for AD treatment.
The genus Bupleurum includes more than 200 species, which are mainly found in Eurasia and North Africa. 6 The dried root of Bupleurum smithii Wolff. is a Chinese folk medicine used to reduce fever and inflammation, and is commonly used as a substitute for Chai Hu in northwest China. 7 Studies in recent years have shown that B. smithii has anti-inflammatory, antiviral, antitumor, immune regulation, anti-depressant, liver-protecting, and other biological activities. 8 Regarding the key role of neuroinflammation in AD pathogenesis, we speculated that B. smithii may be the source of compounds that treat AD through anti-inflammatory effects.
In our previous study, 10 compounds were isolated from the ethyl acetate extraction layer of B. smithii 9 ; these were saikogenin A, saikogenin C, saikogenin D, saikogenin E, saikogenin F, saikogenin Q, 35,7-trihydroxy-3′,4′-dimethoxyflavone, kaemferol, quercetin, and isorhamnetin. Previous studies found that saikosaponins showed a potent therapeutic effect in AD animal and cellular models.10–12 Considering the similar chemical structure and high content of saikogenin F, this compound may be one of the main anti-AD active ingredients in the ethyl acetate fraction of B. smithii. Here, we report the isolation of saikogenin F from B. smithii, and its in vivo effect on memory impairment and neuroinflammation in AD mice induced by Aβ intracerebroventricular (i.c.v.) injection. The effects of saikogenin F on activation of microglia and the expression of reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase subunits were further investigated to clarify the related mechanisms.
Materials and Methods
General Procedures
Electrospray ionization mass spectrometry (ESI-MS) data were recorded with a TSQ Quantum Access Max spectrometer (Thermo Scientific). Nuclear magnetic resonance (NMR) spectra were obtained in DMSO-d6 on an Avance DRX 400 MHz spectrometer (Bruker) at 400 MHz and 100 MHz, yielding 1H-NMR and 13C-NMR data, respectively. For thin-layer chromatography (TLC), precoated silica gel plates (GF254, Merck) were used. For column chromatography, SiO2 (100-200 mesh, Qingdao Marine Chemical Factory) and Sephadex LH-20 (GE Healthcare) were used. High performance liquid chromatography (HPLC) purifications were performed with a QuikSep-50IID HPLC system (H&E Technology) on a Pack Pro C18 prep-HPLC column (5 μm, 250 mm × 20 mm, YMC).
Materials
The dried root of B. smithii Wolff. was purchased from the Bozhou medicinal material market (Anhui province, China), and identified by Prof. Yunsheng Zhao (School of Pharmacy, Ningxia Medical University). A voucher specimen (No. 2018031501) was deposited in the Department of Medical Chemistry, Ningxia Medical University.
Preparation of Saikogenin F
Saikogenin F was obtained by the following procedures, as previously reported. 9 The dried roots (10.0 kg) were crushed and extracted with ethanol/water (7:3, reflux extraction for 3 times for 1 h each). The extract was evaporated under reduced pressure to obtain a crude extract, which was dissolved in 1L distilled water, followed by extraction with light petroleum, EtOAc, and n-BuOH (4 × 1L), successively. To obtain 3 different crude extracts, these extracts were evaporated under reduced pressure. The EtOAc extract (200 g) was subjected to a silica gel column eluted with a gradient of CH2Cl2-MeOH to obtain 12 components (Fr.A to Fr.L). Fr.D (6.5 g) was chromatographed on a Sephadex LH-20 gel column with CH2Cl2:CH3OH (4:6), and further purified by prep-HPLC to obtain saikogenin F (0.87 g), which was identified by spectroscopic and ESI-MS data. 9
Animal Experimental Procedure
Male ICR mice were used in this experiment and their body weights were 22 to 25 g. The mice were provided by the Experimental Animal Center of Ningxia Medical University. All mice operations complied with the regulations and general recommendations of the Chinese Laboratory Animal Management Regulations and were approved by the Ethics Committee of Ningxia Medical University (NXMU2016-098). After 7 days of adaptive feeding, ICR mice were randomly divided into 6 groups with 10 mice each, which were sham operation group (normal saline injection with normal saline gavage), model group (Aβ injection with normal saline gavage), positive control group (Aβ injection with gavage of donepezil 5 mg/kg/d), and high, middle, and low-dose saikogenin F groups (Aβ injection with gavage of saikogenin F 40, 20, and 10 mg/kg/d, respectively). 13 After minor chloral hydrate anesthesia, animals, except for the sham operation group, received i.c.v. Aβ injection (2.5 μg/μL, 4 μL each), as described before, 14 while the sham operation group mice were i.c.v. injected with the same volume of saline. After 24 h of modeling, the mice were given intragastric administration according to their body weight (0.2 mL/10 g), and the sham operation and model group mice were given the corresponding volume of normal saline. After Aβ injection, the novel object recognition (NOR) test was performed on the 14th day, the Y-maze experiment on the 17th day, and the Morris water maze (MWZ) experiment on the 21st day. The levels of inflammatory factors, including ROS, TNF-α, IL-1β, and interleukin-6 (IL-6), in hippocampal tissue were assessed by enzyme linked immunosorbent assay (ELISA) assay, and the expression of NADPH oxidase subunits gp91phox and p47phox was assessed by Western blotting assay. Iba-1 immunohistochemical staining was conducted to assess activation of microglia.
NOR Test
The NOR test was operated as described 15 in a clean and odorless mouse rearing box with a size of 30 × 30 × 35 cm. The test procedure consisted of 3 stages: adaptation period, training period, and retention period. In the adaptation period, each mouse was put into the box without objects individually, to explore freely for 10 min. In the following training period, 2 identical objects with no odor were placed in the box. Each mouse was placed in the middle of the box, and allowed to explore freely for 5 min. The time the mouse spent in touching or sniffing the objects was recorded as exploring time. Then the mouse was returned to its home cage immediately. The retention period was 24 h after the training period, in which one of the identical objects was replaced with a similar size novel object different in shape and color. The mice were gently placed in the middle of the box and allowed to explore for 5 min. The time spent exploring each object was recorded, and the preference index (PI, %) was calculated as the percentage of novel object exploring time in total exploring time.16,17
Y-Maze Test
The Y-maze test 18 was performed to investigate the effect of saikogenin F on the working memory impairment of mice caused by Aβ. The Y-maze is composed of 3 black-painted iron quadrangular prism arms with no cover. The 3 arms are interconnected by a middle triangle symmetrically at an angle of 120° and marked with A, B, and C. The quadrangular prism body is 40 cm long, 15 cm high, and 12 cm wide at the top, and 8 cm wide at the bottom. During the experiment, a mouse was placed at the end of one arm and allowed to explore freely in the maze within 8 min sessions, and the sequences of arm entries were recorded. The spontaneous alternation rate was calculated as the proportion of arm choices that differed from the last 2 choices. Before each trial, scent cues were eliminated by spraying the interior of the maze with 70% ethanol solution. The spontaneous alternation rate of each mouse was calculated through the recorded data.
MWM Test
The MWZ test was operated as described 19 to evaluate the effects of saikogenin F on Aβ-induced spatial cognition impairment. The experimental device consists of a circular pool with a diameter of 1 m and with supporting camera equipment. The pool was filled with an appropriate amount of warm water at 25 °C and divided into 4 quadrants. In the IV quadrant, a hidden transparent platform with a diameter of 6.5 cm is fixed at 0.5 cm below the water surface. In a training session, mice were released from the edge of each quadrant into the water, and allowed to swim in the pool until they found the hidden platform. The mouse was placed and forced to stay on the platform for 30 s if it could not find the platform within 90 s. Mice were trained for 3 consecutive days and 2 times each day. The place navigation test was performed 24 h after the last training, and escape latency was recorded as the time required for the mice to find the platform. Results were recorded and analyzed with the WMT-100S analysis management system (Techman Software).
ELISA Experiments
After the behavioral experiment, the mice were sacrificed and the whole hippocampi were rapidly collected on ice. Each hippocampus was homogenized in phosphate-buffered saline and centrifuged (10 000 r/min, 15 min, 4 °C). TNF-α, IL-1β, IL-6, and ROS levels were detected with the corresponding mouse ELISA kits, following the manufacturer’s procedure.
Western Blotting Analysis
Hippocampus tissues of mice were homogenized with protein extraction buffer, lysed on ice with lysis buffer for 60 min, then centrifuged (15 000 r/min, 15 min) to obtain the supernatant. The supernatant fluid protein concentration was determined by BCA protein assay (Pierce, Rockfold). An equal amount of protein (60 μg) was loaded onto 12% sodium dodecyl sulfate (SDS)-polyacrylamide gel (Ameresco) for electrophoresis, followed by transferring to a polyvinylidene fluoride (PVDF) membrane (Millipore). At room temperature, the membrane was blocked for 1 h with 5% nonfat dried milk in Tris-buffered saline Tween-20 (TBST). After thorough washing with TBST, the membrane was incubated overnight at 4 C with primary antibodies (mouse monoclonal antibodies against gp91phox and p47phox, both 1:500, Cayman Chemical). The blots were then incubated with the corresponding conjugated anti-mouse immunoglobulin G-horseradish peroxidase (1:4000, Santa Cruz Biotechnology). BM Chemiluminescence blotting substrate (Roche Applied Science) was used to detect the proteins. β-Actin was used for standardization. ImageJ version 1.44 software (National Institutes of Health) was used for band pattern analysis.
Immunohistochemistry Staining
Mice brains were fixed in 4% paraformaldehyde, embedded in regular paraffin, and serially sectioned (5 μm). After deparaffinization and hydration, free floating sections were washed with phosphate buffered saline (PBS) and treated with 1% hydrogen peroxide for 15 min. Sections were placed in the blocking buffer for 15 min, then incubated separately overnight at 4 C with a rabbit anti-Iba-1 (1:800, Wako) antibody in normal goat serum and triton X-100. After washing with PBS, they were incubated for 90 min with biotinylated anti-rabbit (1:200, Vector, Burlingame), followed by another incubation for 1 h at room temperature with avidin-biotin complex (1:100). Diaminobenzidine (DAB) in 0.05M tris-buffered saline was used to visualize peroxidase activity. A BX51 optical light microscope (Olympus) at 200 × magnification was used to capture images to quantify the Iba-1 immunoreactivity in the hippocampus. The average area fractions of Iba-1 stained regions were measured by ImageJ software. Iba-1 positive regions were selected by manually adjusting the threshold. The selected area divided by total area captured was calculated as the fraction of immunoreactive region, and the data were expressed as ratios of the value compared to the sham operation group.
Statistical Analysis
Results were expressed as the mean ± SEM SPSS16.0 statistical software was used for data processing. A one-way analysis (ANOVA) followed by Dunnett's t-test and the Student's t-test were applied to assess the statistical significance of the differences among treatment groups and control group. P < .05 was considered to be statistically significant.
Results
Purification and Structure Elucidation of Saikogenin F
Dried roots of B. smithii were extracted under reflux with ethanol/water (7:3). The crude extract was extracted successively using light petroleum, EtOAc, and n-BuOH. The EtOAc extract was subjected to chromatographic separation on normal SiO2 and Sephadex LH-20 columns, and prep-HPLC to provide saikogenin F (Figure 1A), which was identified by comparison of its MS and NMR spectroscopic data with those in the literature. 9
Saikogenin F Ameliorates Cognitive Impairment in AD Mice
In the NOR test, after Aβ injection, the index values of preference for new objects (PI) in the Aβ model group were remarkably lower than those in the sham operation group, suggesting that Aβ injection could impair NOR ability in mice. Saikogenin F could significantly increase the PI value (Figure 1B).

Saikogenin F ameliorates cognitive impairment in amyloid β (Aβ)-injected Alzheimer’s disease (AD) mice. Mice were intragastrically administrated with saline, saikogenin F (10, 20, and 40 mg/kg), or donepezil (5 mg/kg). (A) The chemical structure of saikogenin F. (B) Preference index (PI) in novel object recognition (NOR) test. (C) Spontaneous alternative response rate in the Y-maze test. (D) The escape latency in Morris water maze (MWM) test. Each bar represents the mean ± SEM of data (n = 10).
In the Y-maze test, after Aβ injection, the spontaneous alternative response rates in the Aβ model group significantly decreased compared with those in the sham operation group, suggesting that Aβ injection could significantly impair the memory ability of mice. Treatment with saikogenin F could significantly increase the rate of spontaneous alternation (Figure 1C).
In the MWZ test, the escape latency of the model group mice was remarkably increased compared with the sham operation group, while saikogenin F treatment could significantly reduce the escape latency (Figure 1D). No significant difference was observed in swimming speed among all groups. These results showed that saikogenin F could effectively ameliorate cognitive impairment in AD mice induced by Aβ injection.
Saikogenin F Inhibits Neuroinflammation in Aβ-Induced AD Mice
The effect of saikogenin F on Aβ-induced production of pro-inflammatory cytokines in the hippocampus of AD mice was investigated. Compared with the sham operation group, the release of ROS, TNF-α, IL-1β, and IL-6 increased significantly after i.c.v. injection of Aβ, suggesting that the Aβ-induced learning and memory impairment in AD mice may be associated with neuroinflammation. Compared with the model group, treatment with saikogenin F could remarkably decrease the contents of ROS, TNF-α, IL-1β, and IL-6 (Figure 2A-D). The results showed that saikogenin F could effectively inhibit Aβ-induced neuroinflammation in AD mice.

Saikogenin F reduces the levels of amyloid (Aβ)-induced pro-inflammatory mediators. Aβ-injected mice were intragastrically administrated with saline, saikogenin F (10, 20, and 40 mg/kg), or donepezil (5 mg/kg) for 21 days. Concentrations of pro-inflammatory mediators reactive oxygen species (ROS) (A), tumor necrosis factor α (TNF-α) (B), interleukin-1β (IL-1β) (C), and IL-6 (D) in mice brain hippocampal tissues were measured with ELISA kits. Each bar represents the mean ± SEM of data from 3 independent experiments (n = 3).
Saikogenin F Inhibits Microglial Activation in Aβ-Induced AD Mice
Iba-1 immunohistochemical staining was performed to examine the effect of saikogenin F on activation of microglial cells in mice brain. Results showed that the Iba-1 positive area significantly increased in the model group mice brain compared with those in the sham operation group; saikogenin F treatment could remarkably reduce the Iba-1 positive areas (Figure 3), suggesting an inhibitory effect of saikogenin F in Aβ-induced microglial activation in vivo.

Effect of saikogenin F on amyloid β (Aβ)-induced microglial activation in vivo. After intragastrically administrated with saline, saikogenin F (10, 20, and 40 mg/kg), or donepezil (5mg/kg) for 21 days, the brains of mice were collected and microglial cells were visualized by Iba-1 immunohistochemistry staining (Scale bar = 100 μm; a: control, b: Aβ, c: Aβ + saikogenin F 10 mg/kg, d: Aβ + saikogenin F 20 mg/kg, e: Aβ + saikogenin F 40 mg/kg, f: Aβ + donepezil 5 mg/kg). The average area fractions of Iba-1 stained regions were measured by ImageJ software (version 1.44), and the data were expressed as ratios of the value compared to the sham operation group. Each bar represents the mean ± SEM.
Saikogenin F Suppresses NADPH Oxidase Subunits Protein Expression in Aβ-Induced AD Mice
NADPH oxidase plays a key role in microglia-mediated neuroinflammation. Activation of NADPH oxidase requires cytosolic subunits (p47phox, p67phox, and p40phox) and catalytic subunits gp91phox and p22phox. 20 Results of Western blot analysis showed that Aβ could increase gp91phox and p47phox protein expression in AD mice brain, while this increase was remarkably suppressed by saikogenin F treatment (Figure 4). Thus, the inhibitory effects of saikogenin F in Aβ-induced microglial activation and neuroinflammation may be associated with inhibiting the expression of NADPH oxidase subunits gp91phox and p47phox.
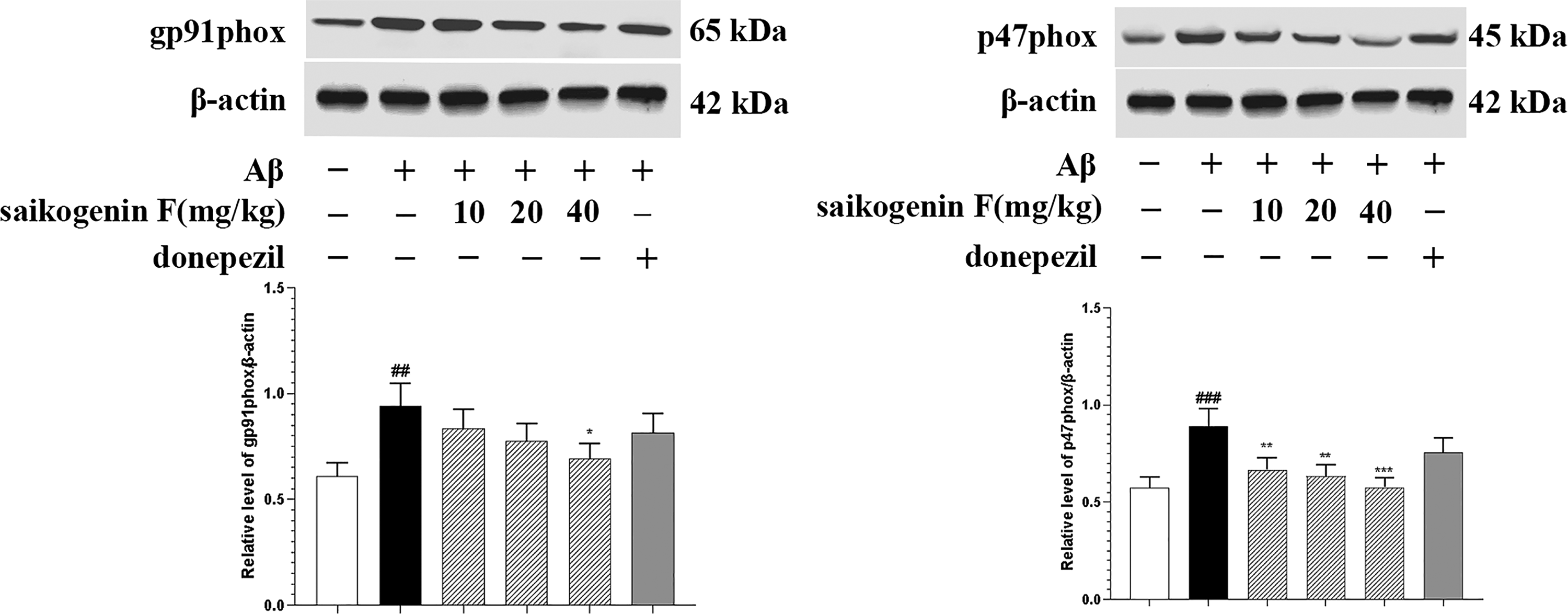
Effects of saikogenin F on amyloid β (Aβ)-induced protein expression of NADPH oxidase subunits gp91phox and p47phox in Alzheimer's disease (AD) mice brain. After being intragastrically administrated with saline, saikogenin F (10, 20, and 40 mg/kg), or donepezil (5 mg/kg) for 21 days, hippocampal tissues of the mice were collected and gp91phox and p47phox protein expressions were measured using Western blot analysis. Expression levels of β-actin were used as loading control. ImageJ software was used in densitometry analysis of specific bands. Each bar represents the mean ± SEM.
Discussion
Bupleurum plants have been shown to have good anti-inflammatory effects. 21 In this study, we isolated saikogenin F from B. smithii, and revealed that saikogenin F could ameliorate the learning and memory impairment of AD mice induced by Aβ. Saikogenin F inhibited the levels of pro-inflammatory factors ROS, TNF-α, IL-1β, and IL-6, and microglial activation in mice brain. These effects may be associated with suppression of the NADPH oxidase subunits gp91phox and p47phox expression. Donepezil also showed an effect in inhibiting neuroinflammation, which is consistent with the previous report. 22
Previous studies have shown that neuroinflammation plays a key role in Aβ-induced cognitive impairment and neurotoxicity in the i.c.v. Aβ-injected mice model.23,24 Moreover, Aβ has been found to effectively induce activation of microglia, so as to improve the proliferation ability and increase the production of inflammatory cytokines, including ROS, TNF-α, IL-1β, and IL-6.24,25 These inflammatory factors are considered to be important substances for the activation of microglia. Recently it has been widely accepted that increased cytokines and chemokines lead to chronic neuroinflammation, which plays a key role in neuronal loss and neurodegeneration in AD pathogenesis. Therefore, we investigated the effect of saikogenin F on Aβ-induced neuroinflammation and microglia activation, and found that the inhibitory effect of saikogenin F on Aβ-induced pro-inflammatory factors release and microglial activation may contribute to its recognition protective effect in AD mice.
Evidence has shown that NADPH oxidase mainly exists in microglial cells in the brain. 26 NADPH oxidase, which is composed of catalytic subunit gp91phox, cytoplasmic subunit p47phox, and transmembrane subunits p22phox, is the main resource of ROS. Among various microglia-produced neurotoxic factors, NADPH oxidase-derived ROS plays a key role in neuroinflammation and AD pathogenesis. 27 The extracellular ROS released by microglia can cause direct neuron damage. Meanwhile, the intracellular ROS was found to act as a secondary messenger, enhancing gene expression by encoding lots of inflammatory factors. 28 In this study, we found that saikogenin F suppressed the protein expression of NADPH oxidase subunits gp91phox and p47phox, leading to a decrease in the production of ROS in mice brain, suggesting that the cognitive function protection and neuroinflammation inhibition effect of saikogenin F may be associated with suppression of the NADPH oxidase subunits protein expression.
In conclusion, this study demonstrates that a high dose of saikogenin F could ameliorate Aβ-induced cognitive impairment, which was mediated through inhibition of pro-inflammatory mediators and NADPH oxidase subunits gp91phox and p47phox. These results indicate that saikogenin F may be a valuable natural product with the potential to treat neurodegenerative diseases associated with neuroinflammation and cognitive impairment such as AD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fourth Batch of Ningxia Youth Talents Supporting Program, National Natural Science Foundation of China, Key R&D Program of Ningxia, and Natural Science Foundation of Ningxia (grant numbers TJGC2019091, TJGC2019100, 81660645, 81660673, 82060792, 82160759, 2018BFG02005, 2021BEG03100, 2020AAC03133, and 2021AAC03143).
