Abstract
Endometrial cancer drug treatments often produce undesirable effects. Thus, discovering new drugs with fewer side effects is required. Cordycepin is a constituent of Cordyceps sinensis, which has been proven to inhibit tumor growth by stimulating the adenosine A3 receptor (A3R). However, cordycepin is rapidly degraded by adenosine deaminase (ADA) and has a clinically unacceptable short half-life. One of its derivatives, MRS5698, was predicted to exhibit antitumor effects with a poor affinity to ADA by our previous validated in silico experiments. The purpose of this study was to explore the possibilities of using MRS5698 as a novel antitumor agent through experiments on Ishikawa and HEC-1A cells. The detection of inhibition and apoptotic rate of MRS5698 and cisplatin, and their combination, on Ishikawa and HEC-1A cells were performed by MTT assays and flow cytometry, respectively. The inhibition rates of MRS5698 on Ishikawa and HEC-1A cells were both significantly higher than the control groups (P < 0.05). MRS5698 produced a higher inhibitory effect on HEC-1A cells than on Ishikawa cells with IC50 values of 20.55 and 27.25 μg/mL, respectively. MRS5698 had a stronger inhibitory effect than cisplatin on HEC-1A cells. The Annexin V-FITC/propidium iodide assays demonstrated that the total rate of apoptosis of MRS5698 on HEC-1A cells was higher than that on Ishikawa cells. The results of MTT assay and cellular apoptosis showed that the combined use of MRS5698 and cisplatin produces dose-independent antagonistic effects. MRS5698 produced antitumor effects on both cell lines, which were better than that of cordycepin. However, the combined use of MRS5698 and cisplatin produced an antagonistic effect. A further in vivo study could be considered for investigating the antitumor effects of either MRS5698 monotherapy or MRS5698 in combination with other nonplatinum-based chemotherapeutic drugs in treating endometrial cancer.
According to the World Cancer Research Fund International (WCRFI), 380 000 new cases of endometrial cancer were diagnosed in 2018, making it the sixth most common cancer in women worldwide. 1 NCCN Clinical Practice Guidelines in Oncology state that the preferred chemotherapeutic drugs used in treating endometrial cancer is a multiagent chemotherapy regimen such as paclitaxel combined with carboplatin and cisplatin combined with doxorubicin. These chemotherapeutic drugs often cause serious systemic side effects that may lead to discontinuation of the treatment. Therefore, designing novel drugs that are less toxic with comparable antitumor effects is crucial. 2 Many novel drug targets have been discovered and one of the most promising is the adenosine A3 receptor (A3R). 3
A3R is a G protein-coupled receptor that belongs to the family of adenosine receptors. It is highly expressed in both primary and metastatic tumors. 4 Several studies have revealed that the activation of A3R results in tumor suppression, including endometrial cancer cells. The mechanism behind it is that when A3Rs are stimulated, they will activate intracellular glycogen synthase kinase-3b (GSK-3b) resulting in the suppression of cyclin D1, a protein that is crucial to cell cycle G1/S transition. 5
Recent research revealed that some Chinese natural products, such as Cordyceps, contain compounds that exhibit antitumor effects, one of which is cordycepin or 3′-deoxyadenosine, which is an adenosine analog proven to suppress tumor growth. 6,7 Cordycepin is a constituent of Cordyceps sinensis which has antitumor effects by incorporating into RNA and inhibiting transcription elongation and RNA synthesis. In vivo studies using C. sinensis extract was found to be effective in hematogenous lung metastasis in mice when coadministered with methotrexate. The body weight of the mice did not decrease significantly and the survival time was longer than that of the controls. 8 A further in vivo study revealed that when cordycepin was administered orally, the weight of the cancer lump inoculated in the thigh of mice was reduced by 36% when compared with the control, without body weight loss or any systemic toxicity. 9 Cordycepin exhibits its antitumor effects by stimulating adenosine A3R, which results in cell growth inhibition. However, direct application of cordycepin to treat tumors is limited due to its poor pharmacokinetic properties. A study 5 revealed that cordycepin is largely degraded by adenosine deaminase (ADA), an enzyme expressed in a variety of tissues, which rapidly degrades cordycepin into an inactive compound, 3′-deoxyinosine. An in vivo study revealed that the half-life (t 1/2) of cordycepin in rats was 1.6 minutes. 10 Coadministration of an ADA inhibitor such as pentostatin and erythro-9-(2-hydroxy-3-nonyl) adenine can significantly extend the t 1/2 of cordycepin. However, these ADA inhibitors can induce several rare but serious side effects such as hemolytic uremic syndrome, leukopenia, thrombocytopenia, and anemia when applied clinically in humans. 11 To prevent rapid degradation by ADA, it would be desirable to find a compound with a similar chemical structure to cordycepin that may theoretically exhibit similar pharmacological effects, but, at the same time, is either less degradable or even undegradable by ADA.
We employed in silico experiments with 31 A3R agonists exhibiting a similar chemical structure to cordycepin in our previous study, 12 and we predicted that MRS5698 had a high affinity to A3R but a low affinity to ADA, and it has an appropriate ADMET (absorption, distribution, metabolism, excretion, and toxicity) profile. MRS5698 is a highly selective A3R agonist with a low affinity to other adenosine receptor subtypes such as A2, A2a, and A2b 13 ; a high selectivity means a low possibility of side effects. A study in alleviating persistent pain in rats showed that when MRS5698 was given orally, the nonlethal tolerance dose was up to 150 mg/kg, which is 400 times that of the ED50 dose. 14 The effectiveness of MRS5698 in suppressing endometrial cancer cells has not been studied, but since stimulating A3R has demonstrated antitumor effects, theoretically MRS5698 should also exhibit these effects. Further investigation of MRS5698 on tumor cells may lead to a novel drug for cancer treatment. In this study we performed MTT assays and flow cytometry on 2 types of endometrial cancer cell lines, the Ishikawa and HEC-1A cell lines, as the target tumors to explore the possibility of using MRS5698 as an antitumor agent.
The inhibition rates of MRS5698, cisplatin, and their combination on Ishikawa cells and HEC-1A were appraised through the MTT (tetrazolium) assay. Table 1 illustrated that the effects of 7.06 and 14.12 µg/mL MRS5698 were similar, the difference being insignificant (P = 0.582). The total rate of inhibition caused by both MRS5698 and cisplatin on Ishikawa cells was dose-dependent. Table 2 shows that the half maximal inhibitory concentration (IC50) values of cisplatin and MRS5698 were 6.80 and 27.25 µg/mL, respectively. This indicated that a higher concentration of MRS5698 is needed to provide the same inhibition rate as cisplatin.
Inhibition Rates of MRS5698, Cisplatin, and Their Combinations on Ishikawa and HEC-1A Cells.
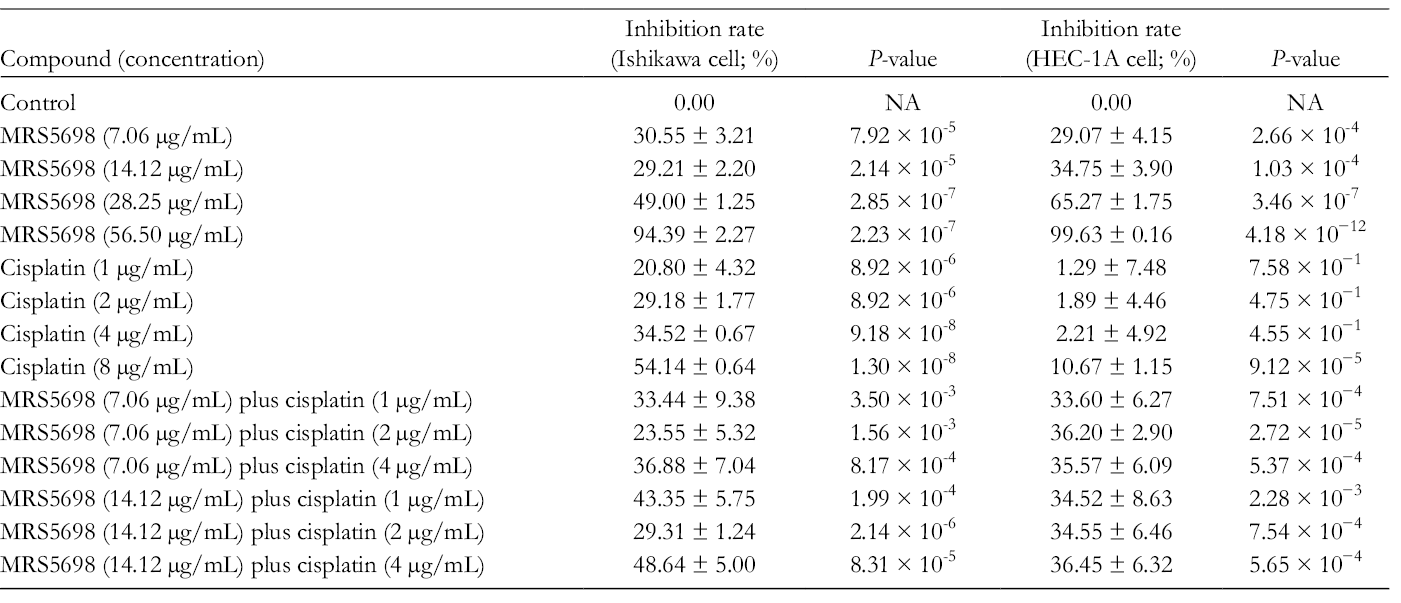
IC50 Values for MRS5698, Cisplatin, and Their Combinations on Ishikawa and HEC-1A Cells.

NA, the inhibition rates did not reach 50%.
Table 3 shows that the combination index (CI) values of the combination (MRS5698 plus cisplatin) were larger than 1, which predicted that the interaction between MRS5698 and cisplatin was an antagonism. Table 1 illustrates that the inhibition rates of the combination groups were dose-independent. Another point worth mentioning is that the inhibition rates of Ishikawa cell caused by the combination of MRS5698 (7.06 and 14.12 µg/mL, respectively) with cisplatin 2 µg/mL were the lowest among the combination groups. The exact cause of this interesting phenomenon is still unknown. A 7.06 µg/mL MRS5698 single treatment produced a statistically insignificant effect, and also with various concentrations of cisplatin (P = 0.156), which indicated that the addition of cisplatin to MRS5698 did not produce any extra inhibitory effects.
Combination Index (CI) of MRS5698 and Cisplatin on Ishikawa Cells.

CI larger than 1 represents antagonism, CI equal to 1 represents an addictive effect, CI smaller than 1 represents synergism.
According to our previous study, 12 100 µg/mL of cordycepin had an inhibition rate of about 57.1%, which was approximately 37% lower than that of 56.50 µg/mL of MRS5698 (Table 1). In addition, the IC50 value of cordycepin was 89.03 µg/mL, which was approximately 62 µg/mL higher than that of MRS5698. This result indicated that the inhibition effect of MRS5698 surpassed that of cordycepin.
On HEC-1A cells, the inhibition rates of MRS5698 were dose-dependent as well. The highest dose of MRS5698 produced an inhibition rate of 99.63% (Table 1). The IC50 value of MRS5698 was 20.55 µg/mL, which was lower than that on Ishikawa cells. MRS5698 produced a better effect on HEC-1A cells than Ishikawa cells (Table 2). The inhibition rates of different concentrations of cisplatin on HEC-1A cells were much lower than those on Ishikawa cells; they decreased by about 20% to 45% (P = 6.16×10-3) (Table 1). Comparing the results of the MRS5698 groups with the cisplatin groups on HEC-1A cells, the inhibition rate of 7.06 µg/mL of MRS5698 was 3 times greater than that of 8 µg/mL of cisplatin. Compared with MRS5698 alone, the inhibition rates of the combination groups with 7.06 µg/mL of MRS5698 were slightly higher. The inhibition rates increased from 5% to 7% (P = 0.363) (Table 1). However, the combination groups with 14.12 µg/mL of MRS5698 produced a similar effect compared with MRS5698 single-drug treatment, which had no statistical difference (P = 0.979).
The cellular apoptosis rates of MRS5698, cisplatin, and their combination on Ishikawa cells and HEC-1A cells were examined by staining the cells with Annexin V-FITC/propidium iodide (PI) and flow cytometry. On Ishikawa cells, the average apoptotic rate of the control group was 4.49% ± 0.93% (Figure 1) while the experimental (MRS5698) groups produced a directly proportional rate of apoptosis (Figure 1) and were higher than the control samples (Table 4). The total rates of apoptosis in the cisplatin groups were not significantly different from the control group (ie, P > 0.05) (Table 4 and Figure 1). Most of the differences between the total apoptotic rate of the combination groups and control groups were not statistically significant except for the combination groups with a higher concentration of MRS5698 (MRS5698 14.12 µg/mL plus cisplatin 4 µg/mL, MRS5698 28.25 µg/mL plus cisplatin 1, 2, and 4 µg/mL) (Table 4).

The apoptosis rate of (a) Ishikawa cells (control group). The apoptotic rate of (b) 14.12 µg/mL and (c) 28.25 µg/mL of MRS5698 on Ishikawa cells. The apoptotic rate of (d) 2 µg/mL and (e) 4 µg/mL cisplatin on Ishikawa cells.
Rates of Apoptosis of Ishikawa Cells Caused by MRS5698, Cisplatin, and Their Combinations.
With HEC-1A cells, the average apoptotic rate of the control groups was 5.24% ± 1.36% (Table 5). All samples in the experimental (MRS5698) groups reached a higher total apoptotic rate than the control groups, and the total rates of apoptosis in the experimental groups were dose-dependent and statistically different from those of the control groups (Table 5). Besides this, the apoptotic rates of low and middle concentration MRS5698 on both Ishikawa and HEC-1A cell lines were comparable, but the total rate of apoptosis of the high concentration MRS5698 on HEC-1A cells was double that on Ishikawa cells (Tables 4 and 5). Compared with MRS5698 single-drug groups, the combination groups exhibited a slightly higher rate of apoptosis (Table 5). However, the differences among the combination groups were not significant (P = 0.455).
Rates of Apoptosis of HEC-1A Cells Caused by MRS5698, and Combination of MRS5698 and Cisplatin.

Our previous in silico study has already shown that MRS5698 is superior to cordycepin with regard to Ishikawa cell inhibition, 12 and the aim of this experimental study was to further investigate the inhibitory effects of MRS5698 in comparison with cisplatin, a platinum compound, that is commonly used for the treatment of metastasis and relapses in endometrial cancer in combination with other cytotoxic agents. 15 It was found that at the same concentration, MRS5698 produced a favorable inhibitory effect on both Ishikawa and HEC-1A cell lines. However, the combination of MRS5698 and cisplatin had no significant advantage over the application of MRS5698 alone, which suggests that simultaneous administration of both agents resulted in no synergistic effect in either cell line.
Two endometrial cancer cell lines were used in this study, Ishikawa and HEC-1A. At a dose of 56.50 µg/mL the inhibition rate was 94.39% ± 2.27% for Ishikawa cells and 99.63% ± 0.16% for HEC-1A cells, which showed that MRS5698 is highly effective in inhibiting cell proliferation in both cell lines. According to a previous study, 12 the IC50 value of cordycepin on Ishikawa cells was 89.03 µg/mL, which is much higher than that of MRS5698 on the same cell line. It indicated that MRS5698 had a better inhibitory effect on Ishikawa cells. In addition, in the results from the docking simulation, MRS5698 had a poor affinity for ADA, which suggested that MRS5698 might have a longer half-life due to its less rapid metabolism by ADA. These results have been encouraging enough to merit further investigation for its antitumor effects in vivo and its actual metabolism by ADA and other enzymes in the human body.
Interestingly, all of the apoptotic rates in the cisplatin groups did not reach 10% for Ishikawa cell lines. According to Fong et al, 12 cell cycle analysis with cisplatin was conducted on Ishikawa cells, the result of which showed that cisplatin arrested the S-phase in the cell cycle and inhibited the growth of Ishikawa cells. Cisplatin-induced apoptosis is a long and complicated procedure. 16 Regarding the HEC-1A cells, there were no significant differences between the total apoptotic rate of the control group and that treated with cisplatin within the first 24 hours. 17 This indicated that MRS5698 had a different and faster mechanism of apoptosis in Ishikawa cell lines compared with cisplatin.
As mentioned earlier, the concentration required to achieve a significant inhibitory rate was 56.50 µg/mL; this result could predict that the dose is likely to be safe, because according to another study in alleviating persistent pain in rats, the lethal dose of MRS5698 was 150 mg/kg (400 times more than the ED50 dose). 18 Assuming that the lethal dose for humans is at a similar ratio, the lethal dose would be 10 500 mg for a typical human weighing 70 kg. Although pharmacokinetic parameters for MRS5698 in humans are not yet available, the amount of MRS5698 needed to achieve an extracellular concentration of 56.50 µg/mL should be theoretically far less than the lethal dose.
The inhibition rate of cisplatin was significantly higher for Ishikawa cells than HEC-1A cells, which may be due to cisplatin resistance. According to research conducted by Rouette et al, 17 cisplatin upregulates B-cell-lymphoma-2 expression in HEC-1A cells by activating protein kinase C and Akt2. Unlike cisplatin, MRS5698 exhibits antitumor effects by activating A3R, which does not alter gene expression. Therefore, MRS5698 has a different antitumor mechanism that is less likely to interfere with gene expressions. This makes MRS5698 a better option for endometrial cancer caused by HEC-1A cells or other cisplatin-resistant carcinomas.
Both inhibitory and apoptosis experiments conducted in this study showed that there was no extra effect when cisplatin was combined with MRS5698. According to NCCN Clinical Practice Guidelines in Oncology, 19 the preferred treatment for endometrial cancer is a multiagent regimen. A study showed that the coadministration of paclitaxel and either cisplatin or another platinum-based chemotherapeutic drug exhibited a better effect in treating advanced and recurrent endometrial cancer. 15 Hence, the clinical value of MRS5698 may be reduced due to its antagonistic effect with cisplatin. However, the potential of MRS5698 in combination with other nonplatinum-based chemotherapeutic drugs should not be neglected, since MRS5698 has a different mechanism of action. A further study of the antitumor effect of MRS5698 with other chemotherapeutic drugs with a different mechanism from cisplatin should be considered.
Cisplatin is an effective chemotherapeutic drug used in the treatment of endometrial cancer. However, the toxicity, which includes ototoxicity and nephrotoxicity, often leads to discontinuation of treatment. 15 Moreover, human carcinomas including endometrial carcinoma often show resistance to cisplatin. 17 In this study, MRS5698 revealed its superiority when HEC-1A cells showed resistance to cisplatin. Furthermore, a previous study has also shown that it is well tolerated and of low toxicity in an animal experiment and at the same time alleviates chronic pain, 18 which could be beneficial to patients who need to take analgesic agents when they are under cancer treatment. As a general rule, less pill burden means better adherence to the treatment regimen.
MRS5698 has appropriate drug-like properties to be a potential oral chemotherapeutic drug and exhibits favorable antitumor effects on both Ishikawa and HEC-1A cell lines. The inhibitory effects of MRS5698 were better than those of cordycepin. However, the interaction between MRS5698 and cisplatin was not synergistic. A further in vivo study could be considered for investigating the antitumor effects of MRS5698 monotherapy or in combination with other nonplatinum-based chemotherapeutic drugs in treating endometrial cancer. A further in vitro study of MRS5698 on normal endometrial cancer cells should be performed to investigate its cytotoxic effects.
Experimental
Cell Culture
RPMI-1640 (Gibco, 11875, Waltham, MA) supplemented with 10% fetal bovine serum (Gibco, 16000044) was used for culturing Ishikawa (ATCC, Manassas, VA) and HEC-1A (ATCC) cell lines under the conditions of 37°C and humidified 5% carbon dioxide.
Cytotoxic Assay
MTT (tetrazolium) assay is a widely used method for assessing the cytotoxic activity of a drug and cell viability. 20 Here, 1 × 106 Ishikawa cells and HEC-1A cells cultured in a 96-well plate were taken to undergo MTT assay. The cells were divided into several experimental groups in various concentrations and 2 control groups (Table 1). Cisplatin (Sigma-Aldrich, P4394, Burlington, MA) was used as the positive control group, and a blank group was the negative control group. The experimental procedures were as follows: The cells were exposed to MRS5698 (Tocris Bioscience, China) in different concentrations and/or cisplatin for 24 hours (Table 1). The cells were then washed with phosphate buffer saline (PBS). MTT solution (100 µL) was added to the cells which were kept in an incubator at 37°C and in a humidified 5% carbon dioxide atmosphere. After removing the media from the 96-well plate, 150 µL of DMSO (Sigma-Aldrich, D4540) was added to dissolve the formazan crystals and the solution was shaken for 15 minutes. The optical density (OD) of the solution was then measured with a microplate reader (SPECTROstar Nano, Ortenberg, Allmendgrün, Germany) at 550 nm. The inhibition percentage was calculated as follows 20 :


Detection of Apoptosis
Apoptosis, also known as programmed cell death, is another essential way to detect the cytotoxicity of an anticancer drug. 21 Differentiation of early and late apoptosis is necessary in research and clinical treatment. 22 Hence, the different state of apoptosis of Ishikawa cells and HEC-1A cells after adding MRS5698 and/or cisplatin was detected in our study. The Annexin V-FITC/PI staining kit (BD Biosciences, 556547, San Diego, CA) was used for the detection of apoptosis. If the cell is in the early state of apoptosis, an essential phospholipid called phosphatidylserine (PS) is translocated to the surface of the cell membrane. Annexin V is able to bind with PS and stain the cells which are in early apoptosis. When the cell is in late apoptosis, its membrane becomes incomplete and PI is able to penetrate the incomplete membrane and stain the cell. 23 Thus, both Annexin V and PI are positive when a cell is in late apoptosis.
The protocol of the experiment was as follows: 1 × 106 Ishikawa cells and HEC-1A cells were seeded in 35 mm culture dishes for 1 night. Different concentrations of MRS5698 and cisplatin (Tables 4 and 5) were then added into culture dishes and stayed for 24 hours. The cells were harvested and resuspended before being washed with PBS 2 times. After washing with PBS, the cells were centrifuged at 200g for 3 minutes. Binding buffer (500 µL) from the apoptosis kit was used for resuspension after centrifuging. The suspension containing 100 µL of cells was stained by using 5 µL Annexin V-FITC and 5 µL PI for 15 minutes. An Applied Biosystems Attune flow cytometer (Waltham, MA) and the manufacturer’s software were utilized for apoptosis analysis following staining.
Combination Index Calculation
This study aimed to investigate whether MRS5698 may potentiate the anticancer effects of cisplatin, and, if so, whether, in patients taking MRS5698 and cisplatin together, the dose of cisplatin can be lower than the normal dose and hence the risks of side effects of cisplatin could be decreased. CI is a value to determine the interaction between 2 drugs. When the CI value is larger than 1, it represents that the combination is an antagonism. When the CI value is smaller than 1, it indicates the interaction between 2 or more drugs is synergy. If the CI value is equal to 1, it means that the combination has an addictive effect. In this study, CompuSyn software was used to calculate the CI of the combination of MRS5698 and cisplatin. CompuSyn, software developed by ComboSyn, Inc., uses the median-effect principle of the mass-action law to simulate and calculate the CI values. 24
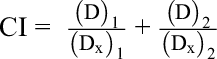
(D)1 and (D)2 are the concentrations of drugs 1 and 2 used in combination to achieve x% drug effect.
(D x)1 and (D x)2 are the concentrations for single agents to achieve the same effect.
Statistical Analysis
All results were analyzed using Microsoft Office Excel 2013 and Statistical Product and Service Solutions (SPSS) software, version 24.0, and expressed as the average ± standard deviation. The differences between 2 groups of the cytotoxic assay and cellular apoptosis assay were calculated using a 2-tailed t-test while the differences between 3 or more groups were analyzed by one-way analysis of variance multiple comparisons (one-way ANOVA). If the P-value is smaller than 0.05, it is regarded as statistically significant.
Footnotes
Acknowledgments
The authors thank Ms Kosca Leung, Mr Cheng N. Ao, and Ms Chi C. Cheong for providing technical support. The financial support of the Macao Polytechnic Institute Research Fund (Project No:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is financially supported by Macao Polytechnic Institute Research Fund (Project No.
