Abstract
Objective
Epidermal growth factor receptor 2 (C-erbB-2) is one of the most frequently mutated oncogenes in human tumors. We aimed to evaluate the knockout efficiency of clustered regularly interspaced short palindromic repeat (CRISPR) technology using ultrasound microbubble transfection to target
Methods
Three single guide RNAs (sgRNAs) targeting
Results
RT-PCR showed that C-erbB-2 mRNA expression was significantly lower in sgRNA1-transfected cells (0.57 ± 0.06) than in blank (1.00 ± 0.09) and negative-control groups (1.02 ± 0.12). Western blotting revealed C-erbB-2 protein expression to be significantly lower in sgRNA1-transfected cells (0.269 ± 0.033) than in blank (0.495 ± 0.059) and negative-control groups (1.243 ± 0.281). However, there was no significant difference in C-erbB-2 protein and mRNA expression in sgRNA2- and sgRNA3-transfected cells compared with controls.
Conclusion
Ultrasound microbubbles can mediate plasmid transfer into HEC-1A cells to interfere with gene expression and knockout
Introduction
Endometrial cancer (EC) is a common malignant tumor in women whose morbidity and mortality are increasing, 1 so it is becoming a serious threat to human health. Treatment modalities for EC are surgery, radiotherapy, chemotherapy, and hormone therapy. However, the prognosis in patients with recurrent or advanced EC is poor.
Gene-targeted therapy for EC has been explored.2,3 This new approach attempts to regulate gene expression in tumor cells and alter the biologic behavior of tumor cells at the genetic level to improve local efficacy and reduce systemic side effects. It is expected to improve treatment efficacy and the prognosis of patients with recurrent or advanced EC.
Epidermal growth factor receptor 2 (C-erbB-2) is a transmembrane glycoprotein with activity towards tyrosine protein kinases. It is an important regulator of cell growth, differentiation, and survival, and is one of the most frequently mutated oncogenes in human tumors. 4 Significant overexpression of C-erbB-2 protein has been observed in EC compared with virtually no expression in healthy endometrial tissue. Moreover, C-erbB-2 protein expression also increases in EC because around 20% of precancerous lesions develop into cancer. 5
Clustered regularly interspaced short palindromic repeats (CRISPRs) are found in bacteria and provide immunity against bacteriophage infections and other environmental challenges. They have been developed as a gene editing method which is more efficient than previous technologies for inducing changes or mutations in genomes. 6 For example, sequence-specific DNA editing can be achieved through CRISPR-associated (Cas) nucleases guided by single guide RNAs (sgRNAs). The CRISPR/Cas9 system is also convenient for the construction and use of vectors, has been used in various species, and, with its high specificity for recognizing sgRNA-targeting sequences, can be used to repair mutations in disease-associated genes and to knock out targeted genes. 7
Gas-filled lipid-shelled ultrasound microbubbles can improve the cavitation effect of ultrasound and the efficiency of gene transfection, and produce a good contrast effect. Moreover, biodegradable lipid shells can be used to carry or “wrap” drugs or genetic material. 8 In the present study, we aimed to transfect human endometrial cancer (HEC)-1A cells by ultrasound microbubbles using the CRISPR/Cas9-C-erbB-2 plasmid, and investigated the expression of C-erbB-2 following gene knockout.
Materials and methods
Reagents and apparatus
HEC-1A cells were obtained from Servicebio (Wuhan, China). The CRISPR/Cas9 plasmid was purchased from GenePharma (Shanghai, China). The plasmid extraction kit and reverse transcription (RT)-PCR kit were obtained from Omega Bio-tek (Norcross, GA, USA). High-glucose Dulbecco’s modified Eagle’s medium (DMEM) with serum phosphate-buffered saline (PBS) and fetal bovine serum (FBS) were purchased from HyClone (Jülich, Germany). Dipalmitoylphosphatidylethanolamine (DPPE) and dipalmitoylphosphatidylcholine (DPPC) were obtained from Lipoid (Steinhausen, Switzerland). DC-cholesterol (3ß-[N-(N′, N′-dimethylaminoethane)-carbamoyl]cholesterol) was purchased from Avanti Polar Lipids (Alabaster, AL, USA). The IX51 inverted fluorescence microscope was obtained from Olympus (Tokyo, Japan). The GCZZ ultrasound transfection system was developed by Chongqing Medical University (Chongqing, China). The 7500 Real-Time PCR System was from Applied Biosystems (Foster City, CA, USA).
Experimental methods
Preparation of ultrasound cationic microbubbles
DPPE, DPPC, and DC-cholesterol were dissolved in a tube containing 10% glycerol/PBS solution (2:5:1) and placed in a 50°C water bath for 1 hour. The air in the tube was replaced with perfluoropropane. The tube was agitated with a mechanical shaker for 70 s. Finally, the mixture was diluted with PBS to produce cationic microbubbles.
Design and synthesis of sgRNA for C-erbB-2
The coding sequence of C-erbB-2 was retrieved from the National Center for Biotechnology Information database. The sgRNA sequence required for the CRISPR/Cas9 experiment was designed using an online tool (http://crispr.mit.edu). Highly specific sgRNA1, sgRNA2, and sgRNA3 were selected after screening and evaluation using the same tool.
Plasmid extraction
A 1 ml LB culture containing the CRISPR/Cas9 plasmid was agitated overnight at 37°C. The plasmid was then extracted using a plasmid extraction kit according to the manufacturer’s instructions (Omega Biotek). The extracted plasmid (>1.0 μg/μL) was used for the next step.
Cell culture
HEC-1A cells were cultured in DMEM containing 10% FBS in a 5% CO2 incubator at 37°C. Upon reaching 90% confluence, they were digested with 1 mL of 0.25% trypsin for 1 minute and then centrifuged (500 ×
Microbubble transfection
Ten µg of CRISPR/Cas9-C-erbB-2 plasmid carrying the green fluorescent protein gene (
Measurement of protein expression using western blotting
Forty-eight hours after transfection, total protein was extracted in an ice bath at 4°C using the ProteoPrep Sample Extraction Kit (Sigma-Aldrich, St Louis, MO, USA). Protein levels were quantified by a nucleic acid/protein analyzer. Forty µg of protein was separated by 12% sodium dodecyl polyacrylamide gel electrophoresis, transferred to nitrocellulose membranes, blocked with bovine serum albumin (Solarbio, Beijing, China) for 2 hours at room temperature, and incubated for 2 hours at 37°C. Nitrocellulose membranes were washed with PBS-Tween, incubated for 2 hours at 37°C, and incubated with a chemiluminescence reagent from the DAB Substrate Kit (US Everbright Inc., Suzhou, China) for ∼2 minutes. Proteins were visualized using a luminescence imager, and the absorbance of each band was measured using ImageProPlus software (Media Cybernetics Inc., Shanghai, China). Three replicates were used for each group. The relative expression of the target protein was defined as the ratio of the absorbance of the target band to that of the internal control band.
RT-PCR
Total RNA was extracted from each group of HEC-1A cells 48 hours after transfection using the RNeasy Plus Mini Kit (Qiagen Inc., Valencia, CA, USA). RT-PCR was performed as described by the manufacturer (Omega Bio-tek) at 37°C for 15 minutes and 85°C for 5 s. PCR amplification of the obtained cDNAs used primers C-erbB-2 5′-CACCCAGCACAATGAAGATCAAGAT-3′ and 5′-CACCCAGCACAATGAAGATCAAGAT-3′; the internal control β-actin was amplified by primers 5′-CCAGTTTTTAAATCCTGAGTCAAGC-3′ and 5′-ATCTTTGGGAGCCTGGCATTT-3′. Amplification conditions were an initial denaturation at 94°C for 2 minutes followed by 10 cycles of 94°C for 15 s, 60°C for 10 s, and 72°C for 30 s, then 25 cycles of 94°C for 15 s, 50°C for 10 s, and 72°C for 30 s, with a final extension at 72°C for 5 minutes, holding at 4°C. Three replicates were used for each group. The relative mRNA expression of
Statistical analyses
Data were analyzed using SPSS software version 18.0 (SPSS Inc., Chicago, IL, USA). Measurement data were expressed as (X ± s). Student’s
Results
Sequencing and transfection
Sequencing of plasmids containing the three sgRNAs is shown in Figure 1. Ultrasound microbubble-mediated transfection of HEC-1A cells with CRISPR/Cas9-C-erbB-2 plasmids expressing

Sequences of sgRNA1, sgRNA2, and sgRNA3 targeting

Transfected HEC-1A cells under an inverted fluorescence microscope.
Expression of C-erbB-2 protein
Western blotting revealed that C-erbB-2 protein expression in the sgRNA1 group (0.269 ± 0.033) was significantly lower than in the blank group (0.495 ± 0.059) and the NC group (1.243 ± 0.281) (
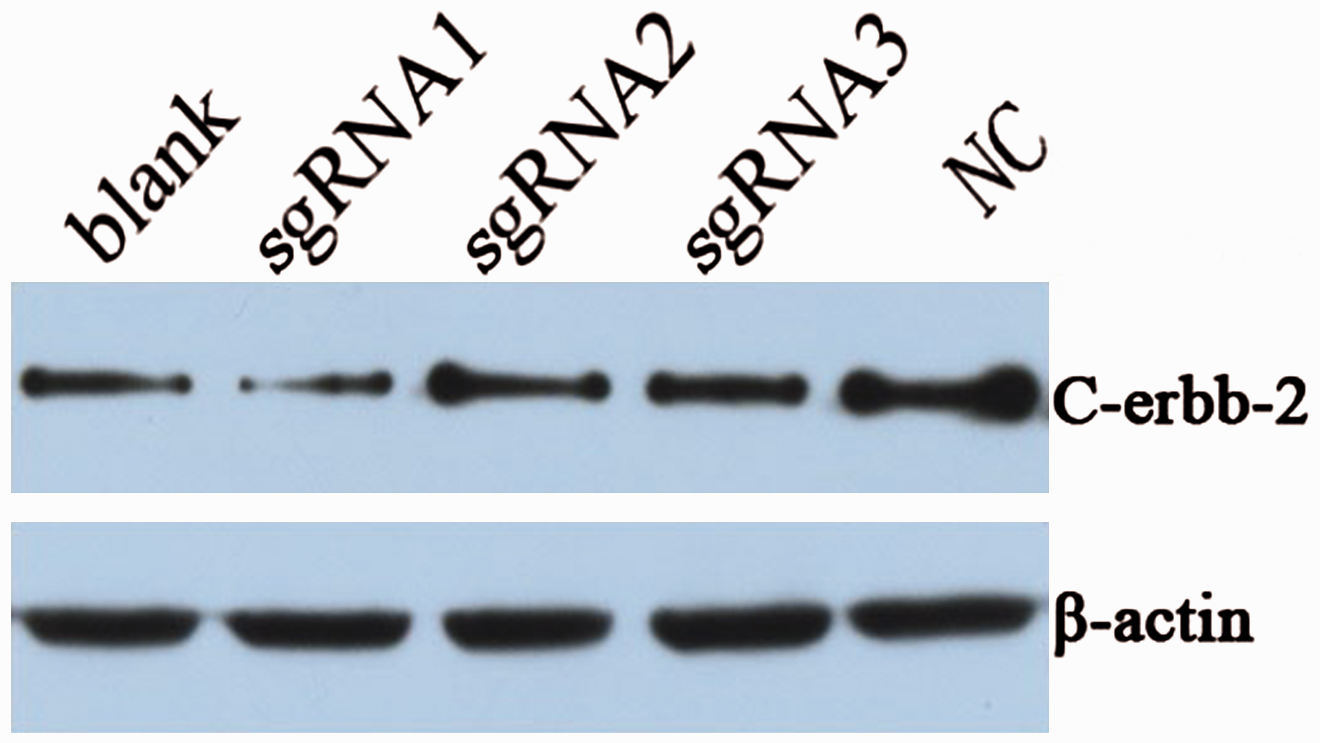
Expression of C-erbB-2 protein as determined by western blotting.
RT-PCR
RT-PCR showed that the relative expression of C-erbB-2 mRNA in the sgRNA1 group (0.57 ± 0.06) was significantly lower than in the blank group (1.00 ± 0.09) and NC group (1.02 ± 0.12) (

Relative expression of C-erbB-2 mRNA as determined by RT-PCR.
Discussion
C-erbB-2 is involved in the growth of tumor vessels and local metastasis, as well as the destruction of anti-invasion barriers. 4 Monoclonal antibodies against this receptor protein have been developed for clinical cancer treatment, but they have not been shown to significantly inhibit C-erbB-2 expression. 9 Liu et al. reported that C-erbB-2 protein expression in EC was positively correlated with pathologic stage and tissue grading, with higher expression associated with a higher tumor grade, and a greatly reduced 5-year survival. 10
Therefore, exploring new methods for inhibiting the expression of C-erbB-2 gene/protein in EC is important. We attempted to develop a new, safe, and non-toxic transfection method using the ultrasound microbubble-mediated CRISPR/Cas9 system to undertake targeted knockout of
The CRISPR/Cas9 system is a novel gene editing tool that offers the advantages of easy operation, low cost, short preparation time, and the simultaneous silencing of any number of individual genes. 11 It can be used to carry out the simple cloning and editing of multiple genomes, and provides a new approach to gene therapy. CRISPR/Cas9 has been used in lung cancer, 12 colorectal cancer, 13 and myeloid leukemia. 14
The key to gene-targeted therapy is to find a suitable gene vector that permits the safe and effective transfection of the target gene and stable long-term expression. Currently, viral transfection and electrotransfection are limited to animal experiments because of biosafety concerns. 8 By contrast, microbubbles have been widely used in clinical diagnoses as contrast-imaging agents for ultrasound. As carriers for genetic material, they can protect drugs or genes from degradation by serum enzymes. They also have biodegradable lipid shells, and are safe, highly efficient, and non-toxic. 15 Moreover, ultrasound cavitation improves the efficiency of gene transfection.15,16 Ultrasound microbubbles have been conjugated with plasmid shBirc5 for glioma treatment, 17 while cationic microbubbles have been developed to improve the transfection efficiency of neutral microbubbles. 18 Wang et al. 19 reported that the efficiency of gene expression was 20-times higher with cationic microbubbles than with neutral microbubbles.
In the present study, positively charged DC-cholesterol was added to film-forming material to make the surface charge of microbubbles positive to increase the binding efficiency. The pX459 backbone vector used in our study can express Cas9 and the inserted sgRNA simultaneously, which simplifies their co-transfection. Moreover, GFP expression allows for the visual detection of transfected cells. We used the CRISPR/Cas9 system to knock out the highly expressed
Conclusions
The ultrasound microbubble-mediated CRISPR/Cas9-C-erbB-2 plasmid was shown to significantly reduce expression of C-erbB-2 protein in HEC-1A cells. Ultrasound microbubble-mediated transfection could provide a new target for the gene therapy and prognosis of EC.
Footnotes
Author contributions
Shan Bao and Junhong Cai conceived and designed the experiments. Sizhe Huang carried out the experiments. Junhong Cai analyzed the data. Sizhe Huang and Junhong Cai contributed to the writing of the manuscript. Yuping Yi undertook other important work in the study.
Declaration of conflicting interest
The authors declare there is no conflict of interest.
Ethical approval
This study was approved by Major Science and Technology Program of Hainan Province (No. ZDKJ2017007) and Natural Science Foundation of Hainan (No. 817313).
Funding
The study was funded by the Hainan Intensified Program of Research and Development (ZDYF2017088) and Hainan Major Program of Science and Technology (ZDKJ2017007).
