Abstract
The incidence rate of (EC) has increased significantly in the past 2 decades. The current chemotherapy for this disease can cause intolerable side effects. Recently, both cordycepin and gold nanostars have demonstrated the ability to inhibit the growth and differentiation of cancers. This study aimed to investigate the antiEC effects of cordycepin, gold nanostars (Au NS), and the combination of the two. The gold nanostars were made by seed-mediated reduction, and their morphology was confirmed by laser particle size analysis, spectrophotometry, and electron microscopy. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide cytotoxicity assays, clonogenic assays, and flow cytometry were employed to evaluate the inhibition rate, survival fraction, and apoptosis in HEC-1A cells under different concentrations of cordycepin, Au NS, and their combination. This study revealed that both cordycepin and Au NS with 808 nm light exposure could inhibit cell proliferation, cause cell apoptosis, and inhibit the clone ability of EC cells. This study supports further investigations into Au NS-cordycepin in the development of new treatments for EC.
Endometrial cancer (EC) is one of the most common gynecological epithelial malignancies. 1 Although the actual cause of the disease is unknown, it is generally believed that estrogen plays an important role in the development of the disease. Common treatments for EC are surgical resection, radiation therapy, chemotherapy, and hormonal therapy. 2 Approximately two-thirds of diagnosed EC patients are treated with surgical resection, which helps to reduce EC complications and the use of painkillers and also prolongs life. 3 However, the consequences of surgical resection are infertility and osteoporosis. Radiotherapy combined with chemotherapy is often used as the treatment of choice for mild to moderate states of cancer. The most common physiological side effects caused by these treatments include loss of appetite, fatigue, nausea, constipation, paralysis, insomnia, pain, and hair loss. Certainly, these side effects have a great impact on patient’s quality of life. Novel treatments are thus required to replace chemotherapy or to reduce the required dose.
Herbal medicines have played a significant role in cancer treatment, as some of them can enhance the efficacy and reduce the side effects of radiotherapy and chemotherapy. 4 Some can inhibit tumor progression, reduce surgical complications, and increase the sensitivity of chemotherapeutic agents. 5 For example, cordycepin is an active component of Cordyceps sinensis, which has been found to have activity against various types of cancer, including gynecological malignancies, such as cervical and EC. 6 Cordycepin is an adenosine receptor agonist, which can inhibit the synthesis of nucleic acids and promote the induction of apoptosis or autophagy in cancer cells. 7 Studies have shown that cordycepin inhibits polyadenylation, affects G2/M phase cell cycle progression, and prevents protein synthesis. 8 However, cordycepin tends to be metabolized rapidly by adenosine deaminase in animals and humans. This leads to an unfavorable short half-life of cordycepin and limited clinical usage. 9 One of the methods to overcome this unfavorable pharmacokinetic problem is to cover cordycepin in an appropriate dosage form, such as the gold nanostar (Au-NS) system, which may help to reduce metabolism and effectively deliver cordycepin to its therapeutic site.
Au NS is a genus of particles with a structure of about 50-80 nm in size and with a “star-like” appearance. 10 It has been demonstrated that these particles have dual actions on cancer cells by producing photothermal effects and act as a carrier to deliver chemotherapeutic agents into cells. A recent study demonstrated an acceptable 17% drug loading capacity of the anticancer drug doxorubicin and highlighted its good biocompatibility. 11 Another study conjugated cyclic RGD (cRGD) and doxorubicin with Au NS to build a multifunctional photothermal and chemotherapy system. 12 The study found promising antitumor efficiency against both a breast cancer cell line and in mice. These studies inspired us to investigate the potential of conjugating Au NS with cordycepin to inhibit the growth of EC cells.
Results and Discussion
Transmission electron microscope (TEM) and laser particle size analyzer were employed to measure the size distribution of nanoparticles in solution. The highest absorption peak of Au NS was at 800-850 nm. Au NS had an average diameter of 59.3 nm and a polydispersity index (PDI) of 0.245. Au NS showed a plurality of thorn-like bulges and a star-like structure (Figure 1).

Appearance of gold nanostar by transmission electron microscopy.
The temperature of different concentrations of Au NS solutions, Au NS + endometrial cancer cell line (HEC-1A) solutions, and Au NS + cordycepin + HEC-1A solutions were detected after 10 minutes of 808 nm light exposure using an infrared temperature detector (Figure 2). The temperature of the Au NS solution increased significantly at all concentrations; the 1.2 × 1011 particles/mL treatment showed the most significant effect. Compared with the control group, the temperature increased from 29.2 °C to 78.1 °C. This result indicates that the Au NS prepared in this study demonstrated the localized surface plasmon resonance (LSPR) phenomenon and released energy under excitation at a specific wavelength. A similar pattern of results was observed with the Au NS + HEC-1A solutions, as the temperature increased with higher concentrations of Au NS (Figure 2). The temperatures with the corresponding concentration of Au NS +HEC-1A + cordycepin solutions were lower than those of the Au NS + HEC-1A solutions and Au NS solutions (without HEC-1A) (Figure 2). This may simply be due to the increased amount of material (HEC-1A and cordycepin) in the solutions, which absorb the thermal energy from the An NS particles.

Temperature of different concentrations of Au NS solutions: Au NS + HEC-1A solutions and Au NS + cordycepin + HEC-1A solutions after 10 minutes of 808 nm light exposure. Concentrations of Au NS and cordycepin are 1010 particles/mL and mg/mL, respectively. AU NS, gold nanostar; HEC-1A, endometrial cancer cell line.
Regarding the Au NS + cordycepin + HEC-1A solutions, the temperature of each group increased by different degrees. The temperature increased when the concentration of cordycepin increased from 0.05 to 0.1 mg/mL but decreased when the concentration of cordycepin increased from 0.1 to 0.2 mg/mL. The early and total apoptosis rate of 0.2 mg/mL of cordycepin solution was much higher than that of the 0.05 mg/mL and 0.1 mg/mL cordycepin solutions (Figure 3). This enhanced apoptosis may suggest that cordycepin induced more damage to the HEC-1A cells and thus allowed more uptake of the Au NS particles in a relatively earlier stage. Thus, more thermal energy was used to destroy the HEC-1A cells, and the overall temperature in the solution decreased.

Total rate of cellular apoptosis of HEC-1A cells caused by (A) 0.05 mg/mL of cordycepin—20.5% ± 0.7%; (B) 0.1 mg/mL of cordycepin—19.0% ± 0.8%; (C) 0.2 mg/mL of cordycepin—33.1% ± 5.0%; (D) 3.0 × 1010 particles/mL of Au NS—12.8% ± 0.7%; (E) 6.0 × 1010 particles/mL of Au NS—21.6% ± 0.7%; and (F) 1.2 × 1011 particles/mL of Au NS—27.5 ± 1.4%. AU NS, gold nanostar; HEC-1A, endometrial cancer cell line.
The uptake of Au NS by HEC-1A cells was detected by ultraviolet-visible (UV-Vis) spectrophotometry. The optical density (OD) values of the HEC-1A mixture with Au NS at concentrations of 3 × 1010, 6 × 1010, and 1.2 × 1011 particles/mL were 0.036, 0.280, and 0.310, respectively. This indicates that the uptake was proportional to the concentration of Au NS. Figure 4 shows an image of HEC-1A cells following the uptake of Au NS visualized using an inverted microscope with 10 × 10 magnification. The OD values of 3 × 1010 particles/mL Au NS mixture with cordycepin concentrations of 0.05, 0.1, and 0.2 mg/mL were 0.103, 0.261, and 0.191, respectively. The values at 0.05 and 0.1 mg/mL are higher than those of the corresponding Au NS alone groups, which indicates that cordycepin may enhance the uptake of Au NS by HEC-1A cells at these 2 concentrations. Similar to the photothermal effects of Au NS and cordycepin described above, the OD value of the 0.2 mg/mL group was lower than that of the 0.1 mg/mL group. This indicates that there was a higher uptake of Au NS by HEC-1A cells in the 0.1 mg/mL group. This may be due to the enhanced rapid apoptosis of the HEC-1A cells (Figure 3) caused by the higher concentration of cordycepin, thus releasing the Au NS particles from the ruptured HEC-1A cells into the solution. However, further experiments are required to confirm this explanation.

Uptake of Au NS by HEC-1A cells captured by an inverted microscope with 10 × 10 magnification. The concentration of Au NS at A, B, and C was 3 × 1010, 6 × 1010, and 1.2 × 1011, respectively. The red circles are examples of HEC-1A cells that have uptaken Au NS. Abbreviations: Au NS, gold nanostar; HEC-1A, endometrial cancer cell line.
The inhibition rates of cordycepin, Au NS, and the combinations on HEC-1A were assessed using the 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay (Table 1). The inhibition rates caused by cordycepin were dose-dependent. The 0.4 mg/mL cordycepin solution produced inhibition of over 90%. Regarding Au NS, Table 1 illustrates the effects of light exposure; Au NS significantly increased the inhibition rates (P < 0.01). For instance, the inhibition rates at 1.2 × 1011 particles/mL with and without light exposure were 96.9% and 9.0%, respectively. The inhibition rates of both 0.4 mg/mL cordycepin and 1.2 × 1011 Au NS particles/mL were over 90%. Comparing the results of either Au NS or cordycepin alone with their combinations, the inhibitory ability of the mixture was higher (Table 1). For example, the inhibition rates of light-exposed 3.0 × 1010 particles/mL Au NS, 0.1 mg/mL cordycepin, and Au NS-cordycepin (3.0 × 1010 particles/mL + 0.1 mg/mL) were 2.3%, 46.1%, and 68.7%, respectively. This indicates that the combined use of Au NS and cordycepin inhibited HEC-1A cells more effectively than the individual treatments. As with the Au NS solutions alone, the inhibition rates of the light-exposed Au NS-cordycepin solutions were significantly higher than those without light exposure (P < 0.01) (Table 1). The inhibition rates of all the light-exposed Au NS-cordycepin solutions with an Au NS concentration of 6.0 × 1010 particles/mL or higher were at least 90%. Again, this value is much higher than that with Au NS alone.
Inhibition Rates of Au NS, Cordycepin, and Their Combinations on HEC-1A Cells.
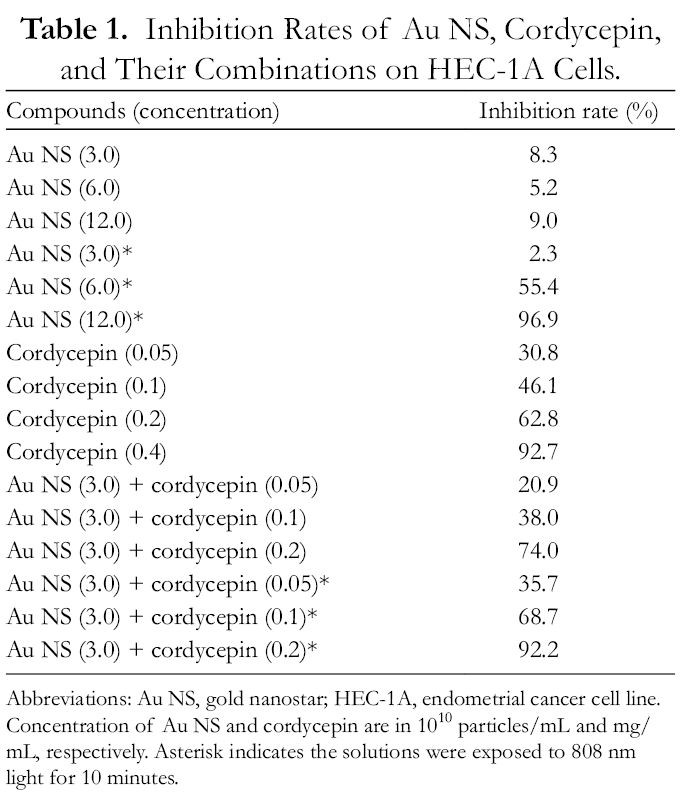
Abbreviations: Abbreviations: Au NS, gold nanostar; HEC-1A, endometrial cancer cell line.
Concentration of Au NS and cordycepin are in 1010 particles/mL and mg/mL, respectively. Asterisk indicates the solutions were exposed to 808 nm light for 10 minutes.
The total apoptosis rate of all the different concentrations of Au NS, cordycepin, and their combinations was significantly higher than that of the control samples (P < 0.01) (Figures 3, 5 and 6). The rates of the experimental groups were generally dose-dependent. The apoptotic rate of the control was 5.1% ± 0.4%, whereas the rates for 0.05, 0.1, and 0.2 mg/mL of cordycepin were 20.5%, 19.0%, and 33.1%, respectively. Au NS at 3.0 × 1010, 6.0 × 1010, and 1.2 × 1011 particles/mL provided an apoptotic rate of 12.8%, 21.6%, and 27.5%, respectively. Combined Au NS-cordycepin with light exposure achieved a higher apoptotic rate than the individual treatments (Figures 3, 5 and 6). For instance, the rate with the Au NS-cordycepin (6.0 × 1010 particles/mL + 0.05 mg/mL) combination was 71.0%, which was a much higher rate than that of the individual treatments at the same concentrations. The rate of Au NS at 6.0 × 1010 particles/mL and cordycepin at 0.05 mg/mL were 21.6% and 20.5%, respectively.

Total rate of cellular apoptosis of HEC-1A cells caused by light exposure (A) Au NS-cordycepin (3.0 × 1010 particles/mL + 0.05 mg/mL)—13.9% ± 1.9%; (B) Au NS-cordycepin (3.0 × 1010 particles/mL + 0.1 mg/mL)—28.4% ± 1.9%; and (C) Au NS-cordycepin (6.0 × 1010 particles/mL + 0.05 mg/mL)—71.0% ± 1.3%. AU NS, gold nanostar; HEC-1A, endometrial cancer cell line.

(A) Total rate of cellular apoptosis of HEC-1A cells caused by the control. (B) Bar graph presentation of apoptosis rate in HEC-1A cells caused by the control and different concentrations of cordycepin and Au NS solutions. AU NS, gold nanostar; HEC-1A, endometrial cancer cell line.
Clonogenic survival assays were performed to investigate the effects of Au NS, cordycepin, and the combinations on the clonogenic ability of HEC-1A cells. Both cordycepin and Au NS with light exposure inhibited clone formation in a dose-dependent manner (Table 2). The combination increased the SF of Au NS and cordycepin alone. For example, the SF of Au NS (3.0 × 1010 particles/mL), cordycepin (0.1 mg/mL), and Au NS-cordycepin (3.0 × 1010 particles/mL, 0.1 mg/mL) were 0.93, 0.61, and 0.01, respectively. This indicates that the combinations induced greater clonogenic inhibition than either treatment alone. Similar to the results of the cytotoxic assay, Au NS with no light exposure did not induce significant clonogenic inhibitory effects.
Survival Fraction of HEC-1A Cells of Cordycepin, Au NS, and Their Combinations.

Abbreviations: Abbreviations: HEC-1A, endometrial cancer cell line; AU NS, gold standard.
Concentration of Au NS and cordycepin are in 1010 particles/mL and mg/mL, respectively. The values of all survival fractions are P < 0.01, except Au NS (3.0), Au NS (6.0), and Au NS (12.0). Asterisk indicates the solutions were exposed to 808 nm light for 10 minutes.
Many studies have shown the anticancer properties of cordycepin against various cancers, that is, liver cancer, 13 tongue cancer, 14 and breast cancer. 15 The pharmacological mechanism responsible for these properties of cordycepin is thought to be activation of adenosine A3R, which can inhibit cell growth. Cordycepin can integrate with ribonucleic acid (RNA) and prevent transcription, elongation, and RNA synthesis. 16 These effects of cordycepin can effectively induce cancer cell apoptosis and reduce the potential for cancer metastasis. 17 Although cordycepin has anticancer properties, its clinical usage is limited. This is because of its short duration of action in the body due to metabolism by adenosine deaminase (ADA) enzymes. 9 One way to improve the bioavailability of cordycepin is by co-administration with ADA inhibitors. Studies have shown that potent ADA inhibitors, such as naringin, can significantly reduce the deamination of cordycepin. 9 However, ADA inhibitors can cause unacceptable side effects to patients. 8 The nanoscale of Au NS means it can be safely eliminated through the urinary system, thus reducing the risk of accumulation and causing adverse effects.
Au NS is a star-shaped nanomaterial with a coarse surface and high surface-to-volume ratio, which allows it to act as a drug carrier for anticancer drug delivery. 18 Au NS has shown its ability to facilitate and localize drug molecules into tumor cells by the enhanced permeation retention effect. 18 This property may protect cordycepin from ADA metabolism and increase the amount that can reach tumors. Au NS produces LSPR when exposed to infrared light at 808 nm. These nanoparticles are very stable and produce efficient photothermal effects after the absorption of laser energy. 19 This generates heat energy and kills cancer cells. Recently, researchers have investigated the use of Au NS in antitumor research and found that LPRS can be changed by altering the shape and size of Au NS. 20 This means that it is possible to adjust the photothermal properties of Au NS for different types of cancer treatments.
This study reports the effects of cordycepin and Au NS on the HEC-1A cell line. Certainly, performing our experiments on different types of EC cell lines can further enhance the significance of this study. Our previous study 8 had reported the inhibition effects of cordycepin on Ishikawa cells, and we believe similar inhibition effects on HEC-1A cells would also be discovered on other endometrial cell lines. Thus, we are hoping that this study will lead to future investigations of Au NS as an EC treatment.
Conclusions
Through the use of cytotoxic assays, clonogenic survival assays, and apoptosis assessments, this study showed that both cordycepin and Au NS (photothermally excited) can effectively inhibit the proliferation of endometrial HEC-1A cancer cells in a dose-dependent manner. When cordycepin was combined with Au NS, cell proliferation was inhibited more effectively, and the inhibition rate was enhanced when compared with the individual treatments. Furthermore, the apoptosis rate of the Au NS-cordycepin combination at a concentration of 6.0 × 1010 particles/mL and 0.05 mg/mL reached 71%, while the apoptosis rates of the individuals’ treatments were only 17.8% and 21.6%. Hence, the combined use of these treatments greatly increased the apoptosis rate and may potentially be used in treating endometrial cancer. We believe that this study provides a fundamental basis for further in vivo studies on the anticancer effectiveness of Au NS-cordycepin.
Experimental
Au NS particles were prepared by a seed-mediated reduction method. The morphology and size distribution of the particles were analyzed by a laser particle size analyzer, a spectrophotometer, and a TEM. The uptake of Au NS by the endometrial cancer cell line (HEC-1A) at different temperatures was observed by spectrophotometry and using an inverted microscope. Cytotoxicity tests and apoptosis experiments were then carried out by MTT assays and flow cytometry, respectively, to analyze the effects of Au NS, cordycepin, and their combination on HEC-1A cells.
Preparation of Au NS
Au NS was prepared by seed-mediated reduction. 21 A tetrachloroauric acid solution with a concentration of 1 mmol was prepared using 0.08494 g of tetrachloroauric acid (HAuCl4) and 250 mL of deionized water. The solution was heated for 40 minutes. Then, 4.5 mL of freshly prepared 1% sodium citrate solution was added to 30 mL of the tetrachloroauric acid solution, followed by another 15 minutes of heating and vigorous stirring. The solution was allowed to cool and filtered through a 0.22 µm filter membrane, which was used as the seed. The hydrodynamic size and polydispersity of the particles in the seed solution were measured using a laser particle size analyzer (ZETASIZER Nano series, Malvern).
Stock solutions of 1 mmol hydrochloric acid solution, 4 mmol silver nitrate solution, 100 mmol ascorbic acid solution, and 0.25 mmol tetrachloroauric acid solution were prepared. Next, 10 µL of 1 mmol hydrochloric acid solution was mixed with 10 µl of 1 mmol hydrochloric acid solution at 700 rpm. After 30 seconds, 100 µL of the seed solutions was added to the mixture. After another 30 seconds, 100 µL of a 4 mmol silver nitrate solution and 50 µL of a 100 mmol ascorbic acid solution were also added. After centrifugation at 5000 rpm for 15 minutes, the precipitate was removed and resuspended in 1 mL of deionized water. Finally, the concentration of the Au NS solution was determined by the extinction coefficient and the Beer-Lambert law.
Morphology and Size
The morphology of the Au-NS particles was observed by TEM (FEI Tale, 200 keV), and the LSPR was measured by UV-Vis spectrophotometry (SPECTROstar Nano). The highest absorption peak of LSPR in this study was 800-850 nm. 22 The laser particle size analyzer (ZETASIZER Nano series) was used to measure the particle sizes, aiming for a diameter between 50 and 80 nm 10 ; the AU NS morphology was observed by TEM.
The Photothermal Effect of Au NS and Its Conjugates
Different concentrations of Au NS alone and Au NS-cordycepin solutions were cultured with HEC-1A cells for 24 hours. The supernatant was removed, and the cells were washed 3 times with phosphate-buffered saline (PBS). The temperature change induced by each concentration (Figure 2) was detected after 10 minutes of 808 nm light exposure using an infrared temperature detector (Fluke Ti400). The experiments were repeated 3 times, and the temperature changes of the cell culture medium were used as the control.
Detection of Au NS Uptake by UV-Vis Spectrophotometer
Different concentrations of Au NS alone and Au NS-cordycepin solutions were cultured with HEC-1A cells for 24 hours. Two hundred grams of the cell suspension was centrifuged for 3 minutes, and the cells were washed 3 times. The supernatant was collected for absorbance measurements by spectrophotometry at 808 nm. The experiments were repeated 3 times, and the absorbance of the cell culture medium was used as the control. The OD was calculated using the following formula:

Cell Culture
The HEC-1A cell line (ATCC, Manassas, VA, USA) was cultured in Roswell Park Memorial Institute-1640 (Gibco, 11 875, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Gibco, 16 000 044, Waltham, MA, USA). Cells were incubated at 37 °C in a humidified 5% carbon dioxide (CO2) atmosphere.
Cytotoxic Assay
MTT assays were used to evaluate quantitatively the effect of Au NS, cordycepin, and their combination on HEC-1A cells. The concentrations of Au NS were 3 × 1010 particles/mL, 6 × 1010 particles/mL, and 1.2 × 1011 particles/mL; the concentrations of cordycepin were 0.05 mg/mL, 0.1 mg/mL, 0.2 mg/mL, and 0.4 mg/mL. The concentrations of the Au NS-cordycepin mixtures were 6 × 1010 particles/mL (AU Ns) + 0.05 mg/mL (cordycepin) and 3 × 1010 particles/mL (AU Ns) + 0.1 mg/mL (cordycepin). The HEC-1A cells were cultured in a 96-well plate, and the exposure time for all samples was 24 hours. PBS was used to wash the cells, and 120 µL of MTT solution was added. The mixtures were incubated at 37 °C in a humidified 5% CO2 atmosphere. After another 4 hours of incubation, 150 µL of dimethyl sulfoxide (Sigma-Aldrich, D4540, Burlington, MA, USA) was added to each well to dissolve the formazan crystals. The plates were gently shaken for 15 minutes at room temperature. The OD of the solution was then measured using a microplate reader (SPECTROstar Nano, Ortenberg, Allmendgrün, Germany) at 550 nm. The inhibition percentage was calculated as follows 23 :

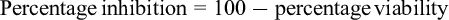
Clonogenic Survival Assay
Clonogenic assays (CA) were performed to determine the division ability of HEC-1A cells, and hence, to determine the ability of Au NS and cordycepin to inhibit the growth of HEC-1A cells. Two hundred HEC-1A cells were seeded in a 48-well plate for 24 hours in a humidified 5% CO2 atmosphere at 37 °C. Cordycepin, Au NS, and the combinations were added to the cells and left for 24 hours. The culture medium was then substituted with complete medium, and cells were incubated for 7 days. Wright-Giemsa staining (Sigma, WG16) was used to stain the cell colonies, and those with 50 cells or more were counted. The plating efficiency (PE) and survival fraction (SF) were used to determine the ability of the HEC-1A cells to grow into colonies. This was calculated by the following equations 24 :
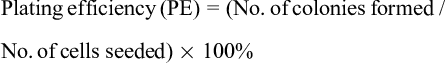

Apoptosis Assessment
Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) staining was performed to detect apoptosis. Two milliliters of 1.0 × 105 HEC-1A cells/mL were inoculated in 35 mm culture dishes and incubated for 24 hours at 37 °C, with 5% CO2 and 95% humidity. The cell supernatants were then collected, and 1 mL of each experimental solution was added to the dishes and incubated at 37 °C, with 5% CO2 and 95% humidity. After 24 hours, the cells were harvested and resuspended using 100 µL of the binding buffer provided with the apoptosis kit (BD Biosciences, 556 547, San Diego, CA, USA). The suspension was stained by adding 5 µL of Annexin V-FITC and 5 µL of PI (Sigma-Aldrich, P4170, Burlington, MA, USA) for 15 minutes in the dark, followed by adding 400 µL of binding buffer for the analysis of apoptosis using an Applied Biosystems Attune flow cytometer (Waltham, MA, USA) and the manufacturer’s software.
Statistical Analysis
Microsoft Office Excel 2013 and Statistical Product and Service Solutions (SPSS) software, version 24.0 were used to analyze all the results. Data are presented as the mean ± SD. A 2-tailed t-test was used to calculate the P values of the differences between 2 groups of the cytotoxic assay and apoptosis assay, while 1-way analysis of variance multiple comparisons was used for the differences between 3 or more groups. If the P-value was smaller than 0.01, it was considered to be statistically significant.
Footnotes
Acknowledgments
The authors thank Ms Kosca Leung for providing technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study is financially supported by Macao Polytechnic Institute Research Fund (Project No: RP/ESS-04/2017).
