Abstract
Isoflavones have attracted much notice due to their health advantages; however, a comprehensive understanding of the effects of isoflavones on endometrium biology remains undiscovered. The expression and deficiency of leukemia inhibitory factor (LIF) and LIF receptor (LIFR) has been shown to be involved in multiple implantations failures in female infertility. Mechanisms implicated in the failure of implantations require further researches, thus our aim is to investigate the effect of the Trifolium pratense L. isoflavone extract with abundant formononetin content on implantation through assessing LIF and LIFR expressions. The Ishikawa cells were cultured with 20, 30, and 40 µg/mL concentrations of Trifolium pratense L. isoflavone extracts for 24 h and detected staining intensity of LIF and LIFR by immunocytochemistry and immunofluorescence staining using image analysis software. As compared with the control and 20 µg/mL Trifolium pratense L. groups, the staining intensity of LIF and LIFR in 30 and 40 µg/mL Trifolium pratense L. groups were significantly increased (P < .0001). Our findings suggest that Trifolium pratense L. isoflavone extract may alter the endometrium expression of LIF and LIFR in the human endometrial adenocarcinoma cell line.
Introduction
Previous studies have demonstrated that controversial results concerning the effect of phytoestrogens on preimplantation. Recent studies have shown that phytoestrogens have favorable effects on the expression of implantation regulatory genes.1-4 However, some previous studies suggested that phytoestrogens were related to embryonic losses through the modification of some transcription factors.5 Previous studies demonstrated that the isoflavone supplement induces expression of leukemia inhibitory factor (LIF) and other signaling pathways in the human endometrial adenocarcinoma cell line (Ishikawa cells).1,2 The isoflavone supplement has increased the expression of LIF in human primary endometrial epithelial cells.2 These findings demonstrate that the isoflavone supplement affects the attachment of the embryo to endometrial cells. However, a comprehensive understanding of the useful effects of the isoflavone supplement on endometrium biology is lacking.1
The Trifolium pratense L. extract has been recognized as an important source of phytoestrogens.6,7 Three classes of phytoestrogens are found in the Trifolium pratense L. extract: isoflavones, coumestrol, and lignans.7,8 The isoflavones are found in physiologically significant amounts in the Trifolium pratense L. extracts.9,10 Trifolium pratense L. is a well-known source of phytoestrogenic isoflavones. It contains mainly formononetin and biochanin A, and much smaller amounts of daidzein and genistein, mostly in the glycosidic form.11,12 The formononetin has been especially listed as the most abundant isoflavone in the Trifolium pratense L. extracts. On the other hand, daidzein and genistein, represent only a small portion of the total isoflavone concentration.9,10 The Trifolium pratense L. extracts have been used in menopause as a natural and safety form of hormone replacement therapy because of their high content of estrogenic isoflavones such as daidzein, genistein, formononetin, and biochanin A.13 These isoflavones are estrogenic in vitro and in vivo, but little information exists their effects on the endometrium and inflamed decidua.14,15 In Ishikawa cells, various isoflavones have induced the expression of various genes that are associated with embryo implantation. Different isoflavone supplements lead to higher expression levels of these genes. The reasons for the differences are unknown, but the content or concentration of isoflavones and other components may enchanges the effects of isoflavones.1-3
The LIF, a major cytokine regulating endometrial receptivity. Recent studies have shown that the LIF and LIF receptor (LIFR) play an important role in embryo implantation and adhesion, and can be used as a marker of endometrial receptivity in the in vitro fertilization treatment.16 In humans, maximal LIF and LIFR secretion by the endometrium coincide with the window of implantation.17 The LIF expression deficiency has been shown to be involved in multiple implantations failures in patients with female infertility.18 Therefore, we examined the effects of the Trifolium pratense L. extracts on the expressions of LIF and LIFR in the Ishikawa human endometrial cell line.
Materials and Methods
Collection and Extraction of the Plant Material
The natural tetraploid Trifolium pratense L. originated from Erzurum-Tortum-Turkey, is used in this study.19 The Trifolium pratense L. was grown in the experimentation gardens of Zonguldak Bülent Ecevit University, Faculty of Arts and Science and Department of Biology. We used flowering herba parts in experiments.
The freeze-dried samples were ground to powder by mortar and pestle. The powders were weighed and extracted with 80% methanol by shaking at 180 rpm for 1 h. The samples were filtered and collected in a volumetric flask. Furthermore, 80% methanol was added to the samples and shaken 24 h. After filtered dried at 45°C with an evaporator. The dry extract powder was suspended in dimethyl sulfoxide (DMSO) for 24 h at room temperature.
Cell Culture and Treatments
The Ishikawa endometrial cancer cell line is widely used in reproductive biology research.20 The Ishikawa cells were obtained from a European collection of authenticated cell cultures (ECACC) 99040201. The Ishikawa cells were cultured for 24 h in a Dulbecco’s modified Eagle's medium-F12 with 10% FBS, 50 units/mL penicillin, and 50 µg/mL streptomycin cell culture medium containing Trifolium pratense L. extracts. The cell line became confluent, and it was counted at a Neubauer chamber in equal volumes of culture medium and trypan blue solution.21 The cell lines were seeded at a density of 20 000 cells per well into 6-well plates. The various concentrations of extracts were used 20, 30, and 40 µg/mL for 24 h incubation time.
Immunocytochemical Staining
The coverslips were washed with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde (PFA) for 15 min at room temperature. The coverslips were permeable with 0.1% Triton X-100 in PBS for 10 min at RT, washed with PBS, blocked with 0.5% bovine serum albumin (BSA) for 30 min at room temperature, and incubated with polyclonal goat anti-LIF (Santa Cruz Bio Technology; cat. no sc-1336, 1:50 dilution) and polyclonal rabbit LIFR (Santa Cruz Bio Technology; cat. sc-659, 1:50 dilution) primary antibodies overnight at 4°C. The coverslips were incubated at room temperature for 10 min with biotinylated anti-goat secondary antibody (ScyTek, UltraTek AGL125) or biotinylated anti-rabbit secondary antibody (ScyTek, UltraTek, ABK125), followed by a 10 min room temperature incubation with UltraTek streptavidin/horseradish peroxidase (HRP) (ScyTek, UltraTek, AGL250). The coverslips were visualized using 3-amino-4-ethylcarbazole (AEC) as a chromogen. The coverslips were counterstained with Harris's hematoxylin and mounted with a medium. The stained images were captured at 40 × magnification using an Olympus BX61 light microscope with the DP72 Olympus camera system. The randomly selected 5 digital images were analyzed with Aperio ImageScope v.12.40 software. The LIF positive pixel count algorithm was determined from 5 random views within the images.
Immunofluorescence Staining
For immunofluorescence staining, coverslips were fixed for 15 min by adding 4% PFA solution, and then coverslips were washed with PBS and permeable with PBS solution containing 0.1% Triton X-100 and 1% BSA for 30 min. The anti-LIF and anti-LIFR primary antibodies were added at the 1:50 dilution and incubated overnight at 40°C. The coverslips were washed twice with PBS and added anti-goat IgG-fluorescein isothiocyanate (FITC) (Santa Cruz Bio Technology; cat. no K1715, 1:50 dilution) and anti-rabbit IgG-FITC (Santa Cruz Bio Technology; cat. no K1715, 1:50 dilution) secondary antibodies for 1 h at 37°C. The coverslips were washed with PBS and then mounted by mounting medium with 4′,6′-diamidino-2-phenylindole (DAPI; ab104139, Abcam). The coverslips were visualized using the Olympus BX61 fluorescence microscope with the DP72 Olympus camera system at 40 × objective.
Statistics
Analyses were performed using KaleidaGraph 4.0 software. Data were determined as mean ± standard error of the mean (SEM) and evaluated by analysis of variance (ANOVA) for comparison among multiple groups using the Tukey post-test analysis for comparison. All P-values less than .05 were considered to be statistically significant.
Results
In the control and 20 µg/mL Trifolium pratense L. groups, the Ishikawa cells contained LIF-positive labeled small vacuoles in their cytoplasm with intermediate staining intensity (Figure 1A, B and 2A). The control and 20 µg/mL Trifolium pratense L. groups showed an intermediate LIF fluorescence intensity in their cytoplasm (Figure 3A and B).
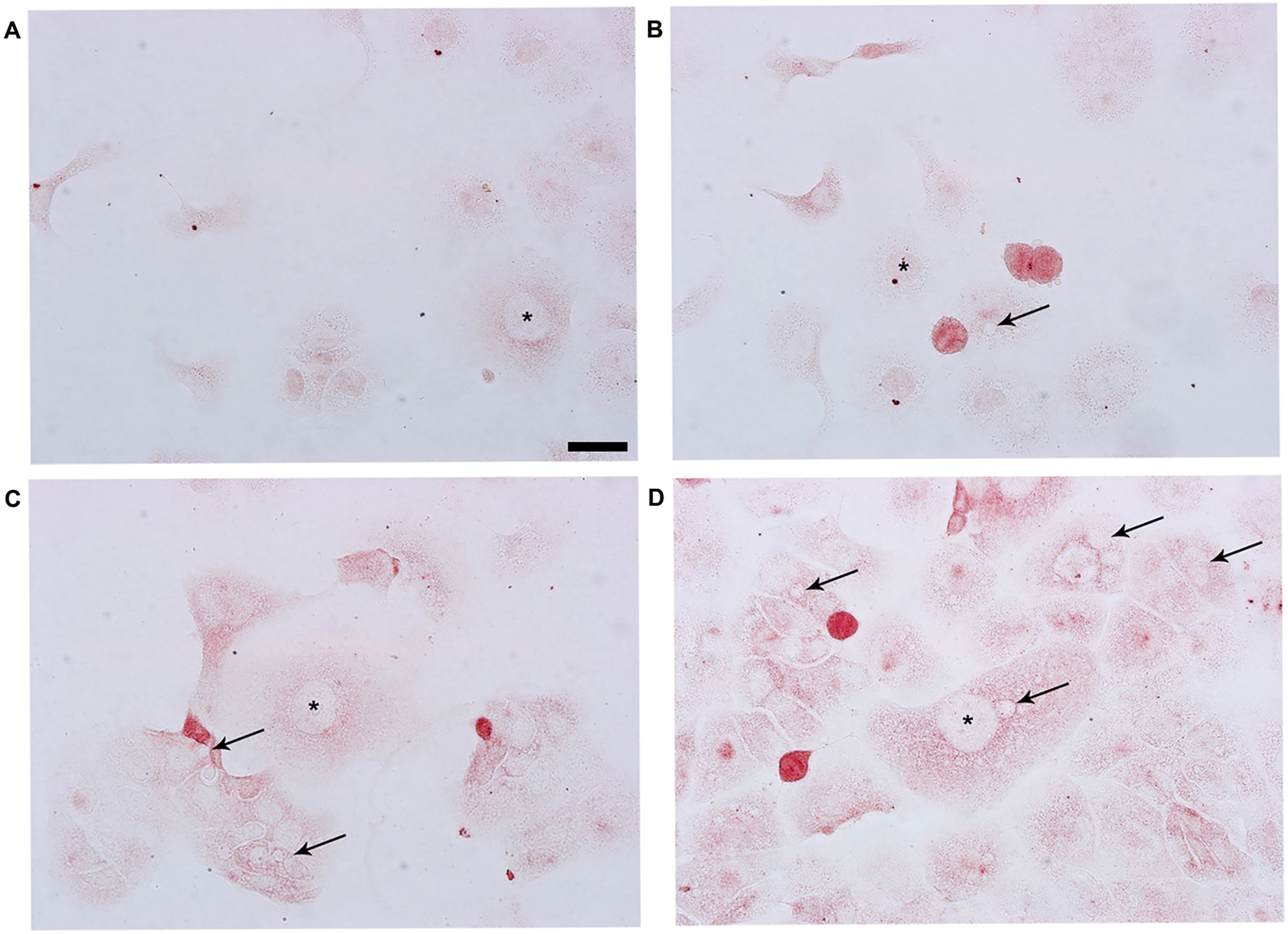
The leukemia inhibitory factor (LIF) staining of the groups in cultured Ishikawa cell line. In the control (A), 20 μg/mL (B), 30 μg/mL (C), and 40 μg/mL (D) Trifolium pratense L. groups. The asterisks and arrowheads indicate nuclei and vacuoles, respectively.

The leukemia inhibitory factor receptor (LIFR) staining of the groups in cultured Ishikawa cell line. The control (A), 20 μg/mL (B), 30 μg/mL (C), and 40 μg/mL (D) Trifolium pratense L. groups. The asterisks and arrowheads indicate nuclei and vacuoles, respectively.
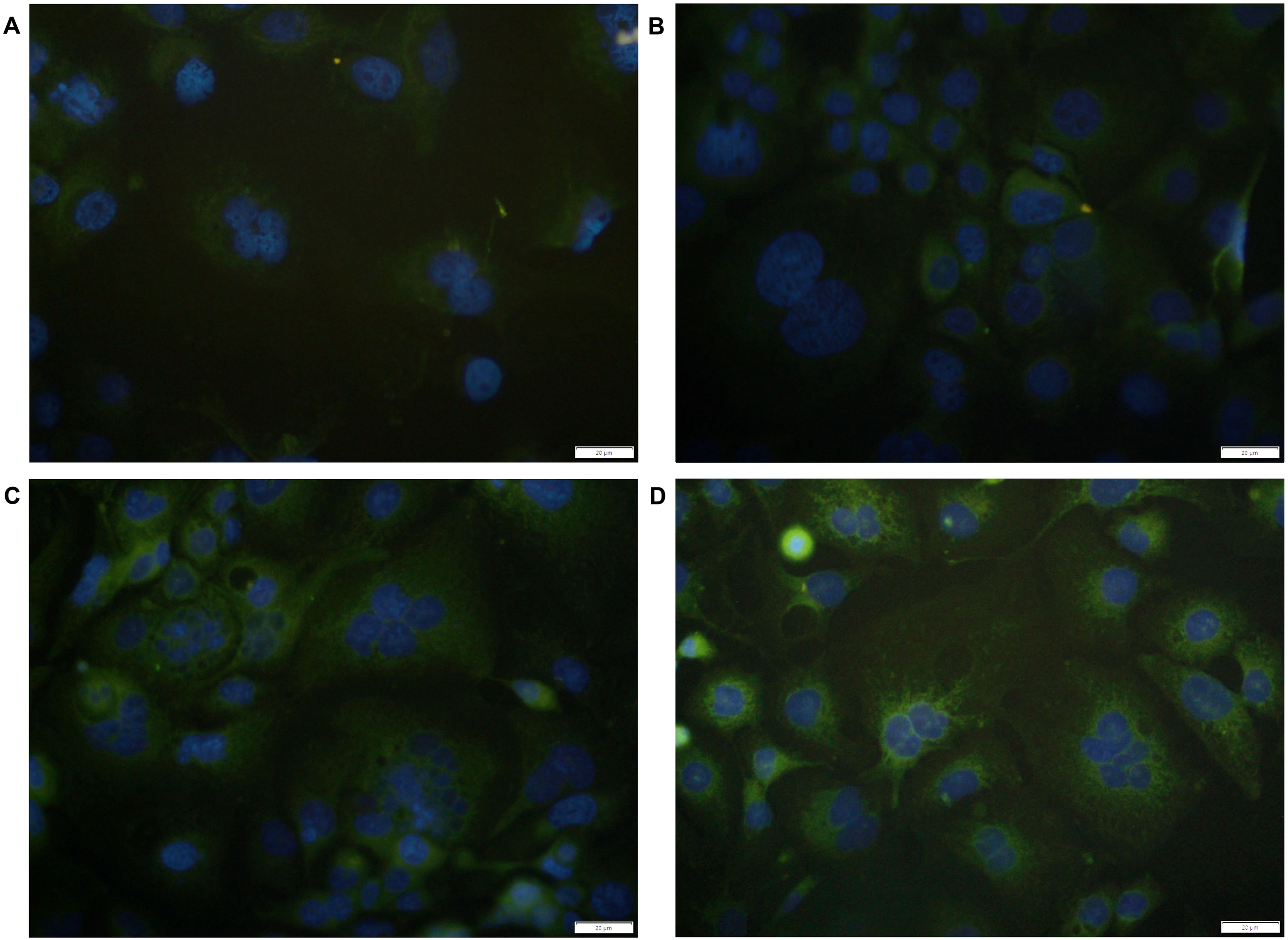
Representative images of Ishikawa cells on the coverslips with DAPI-stained nuclei (blue) and LIF staining (green). The control (A), 20 μg/mL Trifolium pratense L. (B), 30 μg/mL (C), and 40 μg/mL (D) Trifolium pratense L. groups.
The 30 and 40 µg/mL Trifolium pratense L. groups were contained intensely LIF-positive labeled positive big and small sizes vacuoles in the cytoplasm (Figure 1C and D). The 30 and 40 µg/mL Trifolium pratense L. groups have larger size cells than the control and 20 µg/mL Trifolium pratense L. groups. The LIF staining intensity of the 30 µg/mL Trifolium pratense L. group was not significantly compared to the 40 µg/mL Trifolium pratense L. group (P = .0155, Figures 1C and 2A). The LIF staining intensity of 40 µg/mL Trifolium pratense L. group was significantly higher than in the control and 20 µg/mL groups (P < .0001, Figures 1D and 2A). The 40 µg/mL Trifolium pratense L. group showed a high LIF fluorescence intensity in their cytoplasm than the 30 µg/mL Trifolium pratense L. group (Figure 3C and D). The 40 µg/mL Trifolium pratense L. group only showed fluorescence nuclear LIF-positive spots.
The 20, 30, and 40 µg/mL Trifolium pratense L. groups were significantly increased the LIFR staining intensity than in the control group (P < .0001, Figures 4 and 2B). The immunofluorescent positive staining for LIFR was detected throughout the cytoplasm and specially intensely in the perinuclear area of Ishikawa cells (Figure 5).

Representative images of Ishikawa cells on the coverslips with DAPI-stained nuclei (blue) and LIFR staining (green). The control (A), 20 μg/mL Trifolium pratense L. (B), 30 μg/mL (C), and 40 μg/mL (D) Trifolium pratense L. groups.

The positive pixel count algorithms of the groups. The positive pixel count algorithms were used to measure the intensity of LIF (A) and LIFR (B) markers. Graphs represent the mean (bars) + SEM (error bars) in each group; differences between means were compared using a one-way ANOVA followed by the Tukey post-hoc test for LIF and LIFR markers. Compared with control group, ***P < .001; compared with 20 μg/mL Trifolium pratense L. group, #P < .001.
Discussion
The phytoestrogens binding to the estrogen receptors may lead to an estrogen-like effect.22 Therefore, phytoestrogens may alter estrogen responsiveness23 and have an effect on the implantation. 24 Intake of phytoestrogen isoflavone-rich soybean was found to significantly increase the follicular phase length and elevate plasma estradiol levels in premenopausal women.25 The dietary isoflavones regulate estrogen-regulated uterine expression of progesterone receptors in mature rats.26 A high phytoestrogen intake also regulates the expression of estrogen-responsive genes in adult female mice.27 In addition, phytoestrogens were found to regulate endometrial functions28 and to improve pregnancy rates in vitro.29 The formononetin is one of the 4 main isoflavones in the Trifolium pratense L. extract.4 The formononetin has been especially listed as the most abundant isoflavone in the Trifolium pratense L. extracts Therefore, we used formononetin isoflavone-rich extract of the Trifolium pratense L. in this study. The isoflavones in the Trifolium pratense L. have been shown to improve some symptoms associated with menopause and cardiovascular health.30,31 This reason, we think the isoflavone formononetin as a functional component may modulate the expression of LIF. However, a comprehensive understanding of the useful effects of phytoestrogens on the endometrium is lacking.
Several molecules expressed by endometrial epithelial cells help prevent first-trimester pregnancy loss. For example, the LIF is essential for embryo implantation, as defects in LIF production can induce implantation failure.32 The endometrium of infertile women produces significantly less LIF and LIFR during the period of receptivity.33 The role of LIF gene mutations in unexplained infertility and implantation failures in IVF patients is not clear yet. Recombinant human LIF might help to ameliorate the implantation rate in women with unexplained infertility.32,33 Previous reports demonstrated that the isoflavone supplement induces expression of LIF in the Ishikawa human endometrial epithelial cell line.2 These findings indicate that this isoflavone supplement influences attachment of the embryo to endometrial cells. The isoflavone supplements induced the expression of various genes, which may suggest that these supplements could promote successful implantation.
Expressions of LIF and other genes reduce in inflamed decidua by IL-1b treatment in the presence of estrogen and progesterone, and this reduction could be reversed by isoflavone supplement.1,34 Together these data indicate that the isoflavone supplement may be a useful supplement to modulate the IL-1b effects on inflamed decidua during implantation. The isoflavone supplement might restore the IL-1b-mediated changes in endometrial epithelial cells that can affect embryo implantation.1 The LIF also plays an important role in trophoblast invasion. Evidences have shown that LIF is involved in the situations during implantation which include embryo-endometrial interaction,35 trophoblast invasion,36 blastocyst growth, and development.32,37 Due to its critical role, LIF has been an important target for a nonhormonal contraception.32,38,39
On the other hand, higher concentrations of isoflavone supplement were needed to induce strong expression of progesterone-regulated glycoprotein, and the differences may be due to the different chemical compositions of these compounds. The reason for this difference is unknown but appears to be attributed to the different compositions of the isoflavone supplements. The isoflavone supplement to the addition of other components was shown to induce various effects3 that may strengthen the effects of isoflavone.1,2 The reason for this difference is unknown; however, the difference in the compositions of the isoflavone supplements may have an influence.1 Therefore, we used formononetin isoflavone-rich extract of Trifolium pratense L. in this study. Similarly, we demonstrated that Trifolium pratense L. extracts significantly (P < .05) induced expression of LIF and LIFR in Ishikawa cells. In particular, the 40 µg/mL and Trifolium pratense L. group induced significantly (P < .05) high expression of LIF and LIFR compared with control and 20 µg/mL Trifolium pratense L. groups.
The isoflavones might help to prevent the onset of carcinogenesis.40,41 Epidemiological data have shown that a diet high in isoflavones, such as those in the Trifolium pratense L., may reduce the risk of breast and endometrial cancer.30,42 In previous studies, using high dose isoflavones, inhibition of cell growth and migration was observed in the cancer cells. Therefore, the anti-cancer and anti-invasion properties observed in the previous studies with the Trifolium pratense L. might be due to the dose that was used.43 We administered the low dose of the Trifolium pratense L. and not determined the toxic effects on the cell line.
In conclusion, the Trifolium pratense L. isoflavone extract with abundant formononetin may exchanges expressions of the LIF and LIFR in the human endometrial adenocarcinoma cell line. Here, we demonstrated that 20 (but not the expression of LIF), 30 and 40 µg/mL Trifolium pratense L. extracts induced expressions of LIF and LIFR, which suggests that these isoflavones may enhance cell adhesion and promote blastocyst implantation in endometrial epithelial cells. The therapeutic use of different Trifolium species is significantly limited; thus further studies are needed.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211045467 - Supplemental material for The Effects of the Trifolium Pratense L. Extract on the Leukemia Inhibitory Factor and its Receptor in The Endometrial Epithelial Cell Line
Supplemental material, sj-docx-1-npx-10.1177_1934578X211045467 for The Effects of the Trifolium Pratense L. Extract on the Leukemia Inhibitory Factor and its Receptor in The Endometrial Epithelial Cell Line by Sibel Kokturk, Fatma Kaya Dagistanli, Sibel Dogan, Emel Usta, Hatice Colgecen, Gamze Tanriverdi, Havva Atar and Feride Ozdemir in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the scientific research projects coordination unit of Istanbul University, (grant no. TSA-2019-34998).
Ethical Approval
Ethical approval is not applicable to this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
