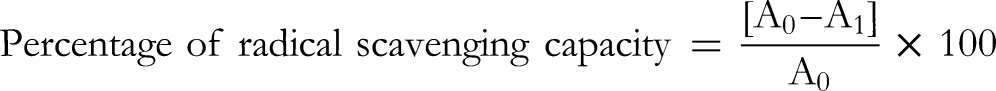Abstract
Introduction
Plants and herbs are an abundant source of medicine and a major contributor to the world's healthcare system. It has been reported that almost 80% of the global community relies on traditional medicines, phytomedicine has a special interest because of its minimal secondary side effects and toxicity. Bacopa monnieri (Brahmi), a plant native to the southern and eastern parts of India was conventionally used in Ayurvedic and Indigenous systems of medicine also known as “Medhya rasayanas” which means brain tonic.1–3 Bacosides are the major complex active compound extracted from B. monnieri. It is a predominant medicine in treating neurological disorders and cognitive enhancement.1,2 Also, it holds major properties such as elevating the mechanism of antioxidant enzyme pathways, anticancer, antiulcer, anti-inflammatories, antipyretic, antiparkinsonian, antidepressant, analgesic, nootropic, antiepileptic, and sedative agents.1,2 Danio rerio (Zebrafish) has emerged as a predominant vertebrate model in toxicological assessments. It is felicitous for ease of handling, external fertilization, transparent embryos, rapid reproduction of eggs, and faster embryonic development. Given toxicity assessments, the zebrafish embryo holds a completely functioning metabolizing system in the liver at 72 h post-fertilization (hpf).4,5 A study by Teschke and Bahre has reported that prolonged intake of Brahmi tablets which contain the extracts of B. monnieri results in chronic hepatoxicity. They have also stated that the withdrawal of Brahmi tablets led to the recovery from hepatoxicity and aids in the normal functioning of the liver.6,7 Similarly, the study reported by Muchhara and his colleagues has revealed that a total glycoside compound named Bacognize® from B. monnieri has shown no observed adverse effect level (NOAEL) up to 1000 mg/kg in the in vivo study in rats. 8 Likewise, BacoMind™, a phytochemical compound enriched from the extracts of B. monnieri has shown NOAEL up to 500 mg/kg through the in vivo study carried out in Sprague–Dawley rats. 9 Furthermore, a study examined by Srinivasan and his colleagues has revealed that a nanoparticle named Magnesium oxide synthesized from the B. monnieri plant (bMgO NPs) has shown better antioxidant and biocompatibility in zebrafish embryos. 10 Correspondingly, a study by Mudgal and his colleagues has shown that 0.2% Brahmi extract has revealed low-stress response and minimum toxicity in the zebrafish larvae. Several plant extracts were tested for toxicity in zebrafish embryos. The study by Ali et al 11 screened the toxic effect of native Australian fruit using a zebrafish embryo model. A toxicity study of Piper sarmentosum aqueous extract by Abidin et al 12 showed that 60 µg/mL of plant extract has the greatest toxicity level. Tran et al 13 colleagues have evaluated the toxic effect of twelve medicinal plants belonging to northern Vietnam in the early stages of zebrafish embryos. However, most of the studies suggest an in-depth evaluation of the toxic effect of B. monnieri in any of the non-rodent animal models also zebrafish has recently gained substantial heed in phytotherapeutics and herbal medicine, which provides ease of teratogenicity including developmental anomalies, delayed embryo development, and lethality screening because of its rapid embryo growth and optical transparency, on this basis, the current study aimed to investigate the preliminary phytochemical components of B.monnieri followed by an estimation of in vitro antioxidant assays along with possible developmental fatal and non-fatal toxic effects of the leaf extract of B. monnieri in the life genesis stages of zebrafish embryos.
Materials and Methods
Sample collection: B. monnieri fresh leaves are grown and collected in Coimbatore, India. The plants were authenticated botanically in the Botanical Survey of India, Tamil Nadu Agricultural University Campus, Coimbatore (BSI/SRC/5/23/2022/118).
Preparation of Plant Extract
Dry powder extract: The distilled water is used to clean the obtained leaves completely. This was followed by thorough shade-drying them for about 12 days, making the dry homogenous powder using a grinder, and kept in an airtight container at room temperature (RT) 15-25 °C for future use. The plants’ extract was prepared using the Soxhlet extraction apparatus. The dry powder of the plant weighed 25 grams in each thimble and was placed in an extraction chamber. The solvents for extractions are chosen based on the polarity, including aqueous, ethanol, acetone, chloroform, and petroleum ether. The extraction process was carried out with each solvent at 35-50 °C. The process was accomplished until the solvent became uncolored in the siphon arm. 14
Fresh leaf extract: The preparation of the plant enzyme extract sample begins by weighing approximately 1 g of fresh leaves, then pestled with 5 mL of saline buffer (pH 7.8). The homogenates are centrifuged at 15 000 g for 20 min at 4 °C. The liquid phase (enzyme extract) is collected in a pre-chilled fresh tube. The above step is repeated to collect additional supernatants. The combined supernatants are stored on ice at 2-8 °C, and the fresh leaf extract is used for enzymatic antioxidant assays. 15
Determination of Plant Yield
The plant extract obtained from Soxhlet was air-dried and collected in the 2 mL glass tubes. The extractive value of the collected plant extracts was determined employing the following formula:
Preliminary Qualitative Phytochemical Screening
1 g of dry homogenous powder was dissolved in five varied extracts such as aqueous, ethanol, acetone, chloroform, and petroleum ether, respectively. The weighed dry powder was kept soaked for 48 h for the extractions of phytochemicals completely and strained using Whatman filter paper at RT, the centrifugation of the filtrate was performed at 3000 rpm for 5 min. The solvent portion obtained served as a sample for qualitative analysis of phytochemicals in B. monnieri. Phytochemical screening tests have been performed, such as carbohydrates, phenolic compounds, terpenoids, flavonoids, saponins, steroids, glycosides, tannins, alkaloids, carbohydrates, and proteins.15–17
Estimation of Total Phenolic Content (TPC)
The Folin-Ciocalteu (FC) protocol was employed to quantify TPC and gallic acid served as a standard.15,16 The reaction mixture was made of varied concentrations of 0.2 to 1 mL of plant extracts in different test tubes of all plant extracts, 2.5 mL of 10% FC reagent, and 7.5% sodium carbonate and incubated for 45 min at 45 °C. The findings were represented as mg of gallic acid per gram of dried extract (mgGA/g d.e.) based on the obtained absorbance at 765 nm in a UV spectrophotometer.
Estimation of Total Flavonoid Content (TFC)
The TFC was quantified using the aluminum chloride method using Quercetin.17,18 The test solution was made of varied concentrations of 0.2 to 1 mL of plant extracts in different test tubes of all plant extracts diluted to 4 mL using distilled water. After five minutes 0.3 mL of sodium nitrite and 10% aluminum chloride was included in each tube and left at RT for 6 min. The final capacity added up to 10 mL with sterile water and 2 mL of 1 M sodium hydroxide immediately. The optical density (OD) was measured using a spectrophotometer at 510 nm. The findings were represented as mg of quercetin per gram of dried extract (mgQE/g d.e.).
Estimation of Total Tannin Content (TTC)
The TTC of the sample was assessed using the FC method using Tannic acid. 19 The reaction mixture is made of different concentrations ranging from 0.2 to 1 mL of plant extracts in different test tubes of all plant extracts and added 0.5 mL and 1 mL of FC reagent and 35% sodium carbonate respectively were included and the final capacity was diluted to 10 mL using sterile water. The reagents were kept for 30 min incubation at RT after thorough shaking. The OD was read using a spectrophotometer at 700 nm. The findings were expressed in terms of mg of tannic acid for each gram of dried extract (mgTA/g d.e.).
Determination of Antioxidant Activity Using 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Free Radical Scavenging Method
The assay was carried out using butylated hydroxytoluene as a standard dissolved in methanol and 1 mL of methanol served as a blank.20,21 The sample concentration ranges from 200-1000 μg/mL of all solvent extracts, the resulting capacity diluted to 1 mL with methanol. Further, tubes were incubated for 30 min after adding 0.5 mL of 0.2 mM DPPH solution at RT. Upon incubation, the purple color of the reaction mixture changed to yellow color, it was measured for its absorbance at 517 nm utilizing a spectrophotometer. The formula below is used for determining the inhibition percentage:
Determination of Antioxidant Activity Using 2,2-azinobis[3-Ethylbenzothiazoline-6-Sulfonate] (ABTS) Radical Scavenging Method
The assay was performed with minor modifications using Trolox as a standard and 1 mL of methanol as a blank.
22
The sample concentration ranges from 200-1000 μg/mL of all solvent extracts, the final volume is made up to 1 mL with 50 mM phosphate buffer saline (PBS) pH 7.2. Furthermore, the reaction tubes were kept at RT for 30 min after the addition of 7 mM ABTS solutions. The discoloration of the solution was assessed using a UV spectrophotometer at the absorbance of 734 nm. The inhibition percentage formula below is used for calculation.
Determination of Antioxidant Activity Using the Ferric Reducing Antioxidant Power (FRAP) Method
The ferric reducing antioxidant power (FRAP) assay uses ascorbic acid as the standard, with 1 mL of demineralized water serving as the blank. 23 The sample concentrations ranged from 200 to 1000 μg/mL for all solvent extracts, with the resulting capacity adjusted to 1 mL using demineralized water. Further, the tubes were heated for 20 min at 50 °C after the addition of 2 mL of 0.2 M saline buffer and 2 mL of 1% potassium ferricyanide. After heating 2 mL of 10% trichloroacetic acid (TCA) was included and centrifuged for 10 min at 3000 rpm. Following this, take the equal volume of liquid phase obtained and sterile water, including 1 mL of 0.1% ferric chloride, and leave for 10 min at RT. A color shifts from pale yellow to green were observed and its OD was assessed at 700 nm using a UV spectrophotometer.
Determination of Total Antioxidant Capacity (TAC) Using Phosphomolybdate Assay
The total antioxidant capacity (TAC) of the dry leaf extract was quantified.
24
The 1.1 mL of reaction mixture composition containing 0.1 mL of dry leaf extract was combined with 1 mL of reaction mixture containing 0.6 M sulfuric acid, 28 mM sodium phosphate, and 4 mM ammonium molybdate. The reaction mixture without a sample served as the blank. The test solution was capped, mixed thoroughly, and heated in a 95 °C thermal block for 90 min. Then, allow the mixture to cool at RT. The OD was obtained at 765 nm against blank utilizing a spectrophotometer. The antioxidant capacity was estimated using the below formula:
Determination of Superoxide Dismutase (SOD) Antioxidant Enzyme
The SOD enzyme activity was evaluated using the Nitroblue tetrazolium (NBT) reduction assay by preparing 1.4 mL of the reaction mixture. 25 The 100 μL of fresh plant enzyme extract was added to the reaction solution and kept for 5 min at RT then added with 50 μM riboflavin and the reaction solution was exposed for 10 min under a 200 W fluorescent lamp. After exposure, 1 mL of Greiss reagent was added. The OD of the color developed was read at 543 nm. 25
Determination of Catalase (CAT) Enzyme Activity
The CAT enzyme activity was evaluated using the method of Hydrogen peroxide (H2O2) decomposition assay.26,27 The reaction capacity is composed of 500 μL of fresh leaf extract, 2 mL of 50 mM PBS, and 500 μL of 10 mM Hydrogen peroxide (H2O2). The decrease in absorbance was assessed for 2-min intervals for every 30 s at 240 nm.
Determination of Peroxidase (Px) Activity
The Px activity was evaluated using the pyrogallol oxidation assay by preparing the 3 mL reaction solution consisting of pyrogallol, 0.1 M phosphate buffer, 1% H2O2, and 500 μL of fresh leaf extract. 27 The OD was read at 430 nm for 2 min for every 30 s 27
Determination of Ascorbate Oxidase (AO)
The AO activity was assessed using oxidation of ascorbic acid assay by preparing the reaction solution containing 2.5 mL of substrate solution consisting of ascorbic acid and 0.1 M phosphate buffer and adding 0.5 mL of enzyme source. The solution was immediately mixed. The change in absorbance was read using a UV spectrophotometer at 265 nm every 30 s for about 5 min.27,28
Determination of Polyphenol Oxidase (PPO)
The PPO activity was examined using a catechol oxidation assay.27,28 The reaction mixture consists of a 1:2 ratio of fresh leaf enzyme extract, and sterile water that was mixed thoroughly. To the above solution, 1 mL of catechol solution (0.4 mg/mL) was added. The solution was mixed immediately. The enzyme activity was evaluated at the absorbance of 490 nm for 1 min.
Animals and Treatment
The wild-strain zebrafish (>6 months) was purchased from a regional vendor. The fish were kept for acclimatization in dechlorinated water for 4 weeks. The fish were housed in 4 L of dechlorinated water containing 1 g fish per liter at 27 ± 2 °C with a standard circadian cycle of 14:10 h light-dark at pH 7-7.2 in a glass tank kept under constant aeration. Additionally, the chemical composition of the water is maintained using a commercial kit (Agappe test kit). They were fed twice a day with brine shrimp. The health and normal activity of the fish was monitored daily.29,30 All the experiments were carried out according to the CPASEA guidelines, India (Committee for the Purpose of Control and Supervision of Experiments on Animals) and previously obtained ethical approval from the “Institutional Animal Ethics Committee (IACE)” of the Chettinad Academy of Research and Education (CARE) (Approval number: IAEC3/proposal-127.Lr:98) on 03/10/2023.
Fertilized Egg Production
The wild-strain zebrafish that has a greater potential to spawn was selected and placed in the spawning tanks containing dechlorinated water (system water) and equipped with the internal partition to prevent the fish from coming in contact with lay eggs. The tank holds the fish in a ratio of 2:1 males and females respectively. The fish maintained a 10-h dark cycle and in the early morning (dawn) the partition between the male and female was released and kept undisturbed providing them a noise-free environment for their mating that occurred within 30 min in the dawn light. The eggs were collected after 30 min of mating. The eggs were kept in system water for 6 h and after that, the eggs were transferred to the E3 medium to make the 60X stock solution of E3 medium, Nacl (34.8 g), KCl (1.6 g), CaCl2.2H2O (5.8 g), MgCl2.6H2O (9.78 g), and double distilled water (ddiH2O) (1.95 L) are used. The pH is adjusted to 7.2, and the volume is adjusted to 2 L using ddiH2O. Add 100 µL 1% methylene blue (fungicide). and allowed to develop at a controlled temperature of 27 ± 2 °C. 29
Zebrafish Fish Embryo Toxicity (FET) Bioassay
The Aqueous extract of B. monnieri was employed for the zebrafish embryo toxicity assay (a modified protocol of OECD-FET assay). Initially, the eggs were placed in the system water, after 6 h post fertilization (6 hpf), the healthy eggs were inspected by microscopic examination and transferred to E3 media. Only the fertilized healthy eggs were employed in subsequent procedures. The Fish Embryo Toxicity (FET) assay was carried out in 6 well plates containing E3 media with 0.1% DMSO and five varied concentrations ranging from 2-10 mg/L of B. monnieri leaf aqueous extract in different wells. The assay was performed in three independent wells for each concentration. About 20 fertilized eggs were placed in each well. The exposure medium was renewed every 24 h. The developmental endpoints both lethal and non-lethal such as hatch rate, heart rate, mortality, and phenotypic abnormalities were recorded using an inverted microscope.29,30
Determination of Embryonic and Developmental Toxicity
After placing zebrafish fertilized eggs in E3 media containing varied concentration ranges from 2 mg/L to 10 mg/L of plant extract, the embryos were observed every 24 h extending up to 96 h. The embryos and larvae were regularly monitored for embryonic and teratogenic effects under an inverted microscope including toxic effects such as egg coagulation and hatching, the hatch rate percentage means the abundance of eggs hatched and the number of eggs unhatched when comparing the control group versus each exposure test group of B. monnieri. The survival rate percentage means the number of embryos alive versus the number of dead embryos including before 24 h, and after 24 h extended till the endpoint of the study. The heart rate was estimated at 72 hpf for control versus all plant dose groups. As from 48-120 hpf, the developmental deformities include swollen yolk (sy), body pigmentation (bp), spinal cord curvature (sc), somite formation (s), blood circulation (bc), Motility (m) were observed. 30
Estimation of Embryos’ Hatchability
The hatch rate of zebrafish embryos treated with 2-10 mg/L of B. monnieri leaf extract was determined by monitoring the eggs from mid-48 h to the beginning of 72 h. The hatched eggs were noticed by rupture and release of the chorion membrane using an inverted microscope. 29
Estimation of Larvae’ Cardiac Rate
The cardiac rates of zebrafish larvae were observed at 72 h by observing the cardiac ventricles under an inverted microscope, the heartbeat count was recorded using a computer fitted with camera devices and counted for 1 min. 29
Statistical Analyses
Statistical analysis and graphics were developed using GraphPad Prism 8.0 and Microsoft Excel. The findings were expressed as mean ± standard deviation (SD). Two-way ANOVA analysis has been performed to identify the significant differences among varied test groups in toxicological assessments.
Results
Identification of Plant Yield
The comparative yield of different extracts of B. monnieri leaf is tabulated (Table 1). The aqueous extract provided a higher yield percentage of 42.62%, and the petroleum ether extract provided the lowest yield percentage of 6%. Table 1 depicts the plant extract yield from varied solvents.
Comparative Yield of B. monnieri Leaf Extracts.

Preliminary Qualitative Phytochemical Screening
The preliminary phytochemical screening test assessed the phytocompound in the B. monnieri leaf extract. The tests have revealed the presence of carbohydrates, alkaloids, flavonoids, phenols, saponins, tannins, terpenoids, steroids, glycosides, coumarin, polysterol, protein, and quinones in given five extracts (aqueous, ethanol, acetone, chloroform, and petroleum ether). Among the five extracts, the aqueous extract exhibited the major presence of phytochemicals followed by ethanol, acetone, chloroform, and petroleum ether. Coumarin was absent in all the extracts. The results were tabulated in Table 2.
Preliminary Qualitative Phytochemical Screening.
Estimation of TPC
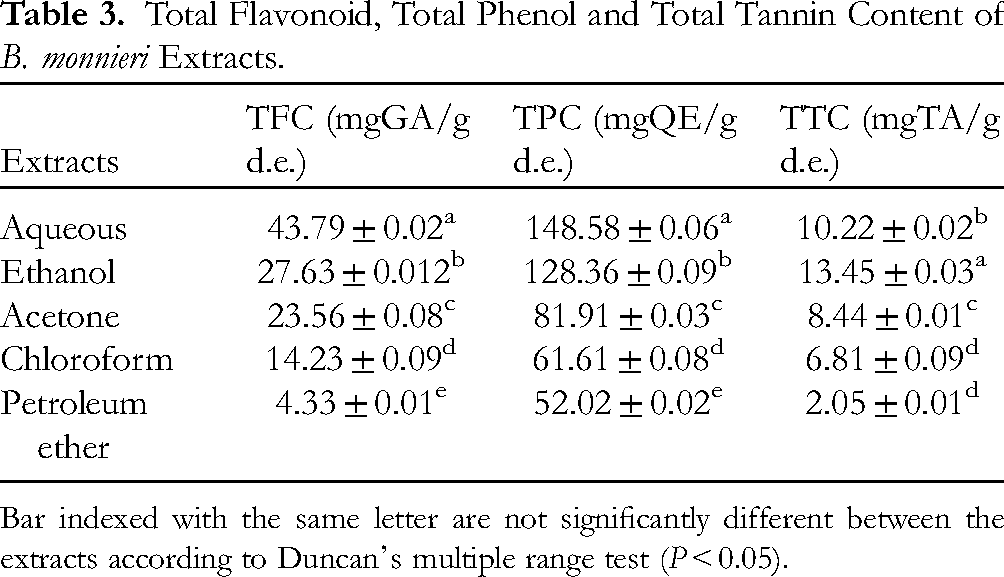
The phenolic content of B. monnieri leaf in five different extracts was analyzed using the FC method. The aqueous extract exhibited a greater amount of TPC with a value of 148.58 ± 0.06 mg QE/g. d.e. The aqueous extract had 28.9%, 41.37%, 7.30%, and 48.13% higher concentrations of TPC than acetone, chloroform, ethanol, and petroleum ether extracts respectively. The specific quantities of TPC are presented in Table 3.
Total Flavonoid, Total Phenol and Total Tannin Content of B. monnieri Extracts.
Bar indexed with the same letter are not significantly different between the extracts according to Duncan's multiple range test (P < 0.05).
Estimation of TFC
The TFC of B. monnieri leaf in five different extracts was analyzed by employing the aluminum chloride method. A higher amount of TFC was found in the aqueous extract a value of 43.79 ± 0.02 mg QE/g. d.e. The aqueous extract has 30.034%, 50.94%, 22.62%, and 81.99% higher concentrations of TFC than acetone, chloroform, ethanol, and petroleum ether respectively. The specific quantities of TFC are presented in Table 3.
Estimation of TTC
The TTC of B. monnieri leaves was assessed by Folin-Ciocalteu's method. The ethanol extract produced a higher concentration of TTC with the value of 10.22 ± 0.02 mg TA/g. d.e. The ethanol extract has 13.64%, 22.88%, 32.77%, and 73.54% higher concentrations than aqueous, acetone, chloroform, and petroleum ether respectively. The specific quantities of TTC are presented in Table 3.
Determination of Antioxidant Activity Using the DPPH Free Radical Scavenging Method
The DPPH radical scavenging activity of five different extracts of B.monnieri was found to be decreased in the order of aqueous > ethanol > acetone > chloroform > petroleum ether. Since, the inhibitory concentration (IC50) value is inversely proportional to the antioxidant effect, among the five extracts, the aqueous extract showed the highest antioxidant effect with an of 312.78 ± 0.13 μg/mL followed by ethanol, acetone, chloroform, and petroleum ether which showed an IC50 value of 345 ± 0.09 μg/mL, 376 ± 0.14 μg/mL, 486.31 ± 0.14 μg/mL, 676.04 ± 0.17 μg/mL, and 835.35 ± 0.21 μg/mL respectively whereas IC50 value of standard ascorbic acid is 170.51 ± 0.03 μg/mL. Furthermore, at 200 μg aqueous, ethanol, acetone, chloroform, and petroleum ether extracts exhibited 34.23 ± 1.15, 39.52 ± 2.69, 37.96 ± 0.38, 25.69 ± 0.20 and, 24.67 ± 0.19 inhibition percent and at 1000 μg aqueous, ethanol, acetone, chloroform, and petroleum ether extracts exhibited 90.37 ± 0.55, 83.74 ± 0.70, 69.98 ± 0.40, 68.48 ± 0.76 and, 55.23 ± 0.21 inhibition percent. The graphical representation of the DPPH assay results is given in Figure 1.

Illustrates the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay of five different extracts such as aqueous, ethanol, acetone, chloroform, and petroleum ether of B. monnieri leaf.
Determination of Antioxidant Activity Using ABTS Radical Scavenging Method
The ABTS radical scavenging activity of five different extracts of B.monnieri was found to be decreased in the order of acetone > ethanol > chloroform > aqueous > petroleum ether. Since, the IC50 value is inversely proportional to the antioxidant effect, among the five extracts, the acetone extract showed the highest antioxidant effect with an IC50 of 280.02 ± 0.45 μg/mL followed by ethanol, chloroform, aqueous, and petroleum which showed an IC50 of 292.5 ± 0.31 μg/mL, 309.01 ± 0.48 μg/mL, 323.37 ± 0.56 μg/mL, and 324.3 ± 0.28 μg/mL respectively whereas IC50 value of standard ascorbic acid is 108.9 ± 0.92 μg/mL. Almost all five extracts showed the same range of antioxidant activity. Furthermore, at 200 μg aqueous, ethanol, acetone, chloroform, and petroleum ether extracts exhibited 43.21 ± 2.73, 45.54 ± 0.20, 48.79 ± 0.28, 41.96 ± 0.16 and, 40.01 ± 1.38 inhibition percent and at 1000 μg aqueous, ethanol, acetone, chloroform, and petroleum ether extracts exhibited 89.77 ± 1.36, 80.05 ± 1.08, 86.94 ± 3.92, 85.78 ± 1.42 and, 84.73 ± 1.39 inhibition percent. The graphical representation of the ABTS assay results is given in Figure 2.

Illustrates the 2,2'-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid). (ABTS) assay of five different extracts such as aqueous, ethanol, acetone, chloroform, and petroleum ether of B. monnieri leaf.
Determination of FRAP Assay
The reducing power of the plant extract is detected by the formation of blue colored by the conversion of ferric to ferrous ions. The color intensity indicated the ferric reduction. The aqueous extract showed a higher reduction of ferric ions. Based on the ferric-reducing power, the extracts were found to decrease in this order aqueous > acetone > ethanol > chloroform > petroleum ether. The graphical representation of the FRAP assay results is given in Figure 3.

Illustrates the ferric reducing antioxidant power (FRAP) assay of five different extracts such as aqueous, ethanol, acetone, chloroform, and petroleum ether of B. monnieri leaf.
Determination of TAC
The extracts derived from B. monnieri leaf exhibited various degrees of antioxidant capacity based on the dose. The TAC of the extract decreases in the following order ethanol > aqueous > acetone > chloroform > petroleum ether. The ethanol extract produced the highest antioxidant ability with the IC50 value of 158 ± 0.49 μg/mL which is statistically equivalent to standard ascorbic acid followed by 250 ± 0.86 μg/mL, 76 ± 1.28 μg/mL, 39 ± 1.37 μg/mL, and 36 ± 0.85 μg/mL of the aqueous, acetone, chloroform, and petroleum ether respectively. The graphical representation of the TAC assay results is given in Figure 4.

Illustrates the total antioxidant capacity (TAC) of five different extracts such as aqueous, ethanol, acetone, chloroform, and petroleum ether of B. monnieri leaf.
Determination of Enzymatic Antioxidant Assays
The quantity of antioxidant enzymes was assessed widely in many plant sources. This study examined the level of antioxidant enzymes in various extracts of B. monnieri, which is represented in Table 4. SOD molecules were generally defined as the primary defense mechanism of aerobic cells, extracellular matrix, and the major source of peroxides. The molecules of SOD convert the SOD anion into hydrogen peroxide and molecular oxygen. The SOD activity found in fresh leaf extract of B. monnieri was 12 ± 0.05-unit mg−1 protein min−1. Catalase is one of the principal antioxidant enzymes, that plays a protective role by transforming hydrogen peroxide into water and oxygen. A gradual decline in catalase activity was observed in the given reaction time, and the catalase activity in the fresh leaf extract of B. monnieri was 72 ± 0.19-unit mg−1 protein min−1. Peroxidase activity denotes the amount of oxidation of pyrogallol into purpurogallin. A steady increase in the peroxidase activity was observed in the given reaction time and the peroxidase activity in the fresh leaf extract of B. monnieri was 7.088 ± 1.94-unit mg−1 protein min−1. AO is a multicopper oxidase enzyme that oxidizes the ascorbic acid into monodehydroascorbate Since a decrease in AO is correlated with an increase in ascorbic acid, this study also observed a steady reduction in the AO and the AO activity in the fresh leaf extract of B. monnieri was 62.23 ± 0.24-unit mg−1 protein min−1. PPO catalyzes monophenolase activity upon hydroxylation and diphenolase activity upon oxidation. When catechol served as a substrate, the predominant reduction in PPO activity was observed in the given reaction time, and the PPO activity in the fresh leaf extract of B. monnieri was 68.44 ± 0.28-unit mg−1 protein min−1. The enzyme activity is tabulated in Table 5.
Assessment of Developmental Endpoints of Aqueous Extract Effect on Zebrafish Embryos.

✓ Observation of normal developmental endpoints.
Х No observation of developmental endpoints.
The Quantification of Enzymatic Antioxidants.

Developmental Toxicity
The phenotypic alterations induced by aqueous extract on each individual presented dose-dependent alterations. The control group displayed a normal appearance of growth and no developmental deformities throughout the test period. At lower concentrations (2 mg/mL) exhibited delayed hatching and in higher concentrations (4 to 10 mg/L) exhibited developmental deformities such as egg coagulation, delayed hatching, hatched dead larvae, pericardial edema (pe), sy, and hypopigmentation (hyp) are represented in Figure 5. Especially, at 8 and 10 mg/L a pronounced representation of sy, pe, and hypopigmentation was noticed. all the developmental endpoints evaluated are represented in Table 4.

Illustrates the developmental endpoints of zebrafish embryos exposed to aqueous extract of B. monnieri across the time interval of 24 hpf to 96 hpf. (SY, Swollen yolk; PE, Pericardial edema; hyp, Hypopigmentation).
The Effect of B. monnieri Aqueous Extract on the Embryo's Survival Rate
The zebrafish embryos’ survival rate was subjected to various proportions of aqueous extract at different time intervals were represented as a graph in Figure 6. At lower concentrations, the survival rate showed minor changes, but at higher concentrations, drastic changes in survival rate were observed when compared to the control group. At 72 hpf, the 10 mg/mL group displayed 100% mortality, and at 96 hpf, the 8 mg/mL group displayed 100% mortality.

Illustrates the survival rate of zebrafish embryos exposed to aqueous extract of B. monnieri across the time interval of 24 hpf to 96 hpf. The survival rate was significant at **P < .01, ****P < .0001. The p values were determined using two-way ANOVA.
The Effect of B.monnieri Aqueous Extract on the Embryo's Hatch Rate
The zebrafish embryo's hatching rate subjected to various proportions exhibited concentration-dependent delayed hatching at 48 hpf about 85.3 ± 2.16%, 66.6 ± 3.49%, 53 ± 1.26%, 49.7 ± 1.97%, and 22 ± 2.16% of hatching was observed in 2 to 10 mg/L of aqueous extract respectively, whereas the control group displayed 100% hatching. Following the observation at 72 hpf, almost all the embryos hatched but at 10 mg/mL 100% hatched death was observed, represented as a graph in Figure 7.

Illustrates the hatch rate of zebrafish embryos exposed to aqueous extract of B. monnieri across the time interval of 48 hpf and 72 hpf. The hatch rate was significant at ****P < .0001. The P values were determined using two-way ANOVA.
The Effect of B. monnieri Aqueous Extract on the Embryo's Heart Rate
The zebrafish embryo's heart rate was examined concerning hpf and concentration. The control groups revealed that heart rate increases with the ontogeny, the mean heart rate at 48 hpf was 142 ± 2.44 bpm, and at 72 hpf was 146 ± 3.13 bpm. In the exposed groups, the aqueous extract of B. monnieri influences the heart rate causing bradycardia based on the concentration and cardiac rate is represented as a graph in Figure 8.

Illustrates the heart rate of zebrafish embryos exposed to aqueous extract of B. monnieri across the time interval of 48 hpf and 72 hpf. The heart rate was significant at **P < .01, ****P < .0001. The p values were determined using two-way ANOVA.
Discussion
Therapeutic plants are gaining adoration in the health sectors. Evaluating the toxicity profile of herbal plants is essential and it is important for the search for plants with exceptional antioxidant actions with minimal toxic effects for performing various pharmacological activities, thorough investigation provided insight into the exceptional antioxidant action and zebrafish embryotoxicity of B. monnieri. The bioactive phytochemicals are considered as the high donors of electrons and can neutralize the free radicals thus ultimately reducing the oxidative stresses. In the present study, the screening of preliminary phytochemicals in B.monnieri has revealed the presence of carbohydrates, alkaloids, flavonoids, phenols, saponins, tannins, terpenoids, steroids, glycosides, polysterol, protein, and quinones in all the five extracts such as aqueous, ethanol, acetone, chloroform, and petroleum ether. The extracts are arranged in the order based on their Phyto constitutes availability aqueous > ethanol > acetone > chloroform > petroleum ether respectively. Parveen et al 30 and Indoliya et al 31 reported an equal number of phytochemicals present in aqueous, methanol, and ethanol extracts the present study also reports similar results as aqueous and ethanol extracts of B. monnieri exhibited the equivalent presence of phytochemicals. The study by Indoliya et al 31 reported a higher yield percent of ethanol and aqueous. The yield percentage obtained in the present study was also found to be similar, where aqueous and ethanol extracts have nearly similar yields and the order of other solvents is given in the order as aqueous > ethanol > acetone > chloroform > petroleum ether.
In this present study, the aqueous extract has the greater amount of TFC and TPC with values of 43.79 ± 0.02 mg/g and 148.58 ± 0.06 mg/g respectively. Vats and Tiwari, 32 have reported the 0.5 mL sample volume of methanol extract of Brahmi has 0.4% ± 0.1 and 1.59% ± 0.06 of TFC and TPC respectively. Furthermore, the report by Volluri et al 33 and Jain et al 34 showed that TPC in methanol extract has 27 mg/g and 24.75 mg/g of gallic acid equivalent (GAE) respectively which is less than the present study. The quantity of TFC is reported as 29.66 mg/g of GAE which is lesser when compared to the present study. In the present study, the ethanol extract has the greater amount of TTC with a value of 13.4 ± 0.03 mg/g. The study by Hossain et al, 35 reported the TTC of methanolic extract as 41.07 ± 2.87 mg/g which is slightly greater than the TTC value of the present study. The study by Jain et al, 34 has reported an almost similar quantity of TTC to the present study with a value of 12.5 mg/g. The significance of P < .05 was observed across the extracts in this study.
Parveen et al 30 have stated that aqueous and methanol extracts exhibited concentration-dependent DPPH radical scavenging action where the highest inhibition percent of aqueous and methanol extract was reported as 94.1 ± 0.004% and 94.7 ± 0.005% respectively and the lowest inhibition percent of aqueous and methanol extract was reported as 17.1 ± 0.005%. and 24.47 ± 0.0052% respectively. This present study also exhibited a similar concentration-dependent antioxidant action and neutralized the free radicals. The highest and lowest inhibition percent of the aqueous extract are 90.3 ± 0.55 and 34.2 ± 1.15 which is closer to the inhibition percent reported by Parveen et al 30 The study by volluri et al, 33 has revealed that the IC50 value of methanolic extract is 104.8 ± 1.9 µg/mL which is lesser than the present study. This might be due to the concentration variance among the studies. Jain et al 34 reported the inhibition percent of methanolic extract was 94% which is almost equal to the inhibition percent of ethanolic extract of the present study. The study by Joshi et al, 36 has reported that the ABTS scavenging activity of the methanolic extract is 54.13% at the concentration of 275 µg/mL with the IC50 value of 385 µg/mL which is almost similar to the present study where the ethanolic extract of B. monnieri exhibited 292 µg/mL of IC50 value at the similar concentration range. In the ferric ion-reducing assay, the present study showed that the antioxidant activity is dose-dependent, which is similarly reported in Parveen et al 30 Preethi et al 37 reported that ethanol extract has greater ferric ion-reducing power than aqueous extract which has been similarly reported in the present study. Antioxidant enzymes present in the cellular environment as protein clusters play a crucial role in alleviating total antioxidant scavenging activity. As the report stated by Joshi et al, 36 the ethanol extract at 475 µg/mL has 60.08% scavenging activity which is found a greater total antioxidant capacity which similarly resulted in this present study. Since most of the earlier reports provided the examination in highly polar solvents such as aqueous, ethanol, and methanol conversely there were no or least reports on low polar solvents such as acetone, chloroform, and petroleum ether.
Previous reports suggested only antioxidant enzyme activities such as SOD, CAT, AO, Px, and PPO on experimental models or under any treatment conditions. Still, there were least reports in fresh leaf extracts of B. monnieri.38,39 Parveen et al 30 have reported the quantity of SOD in aqueous and methanol extract as 0.006- and 0.023-units mg/protein. As there were no defined earlier reports, this study has reported the presence of SOD of 12 ± 0.05 units mg−1 protein min−1, catalase of 72 ± 0.19-unit mg−1 protein min−1, AO of 62.2 ± 0.24-unit mg−1 protein min−1, peroxidase of 7.08 ± 1.94-unit mg−1 protein min−1 and PPO of 68.4 ± 0.28-unit mg−1 protein min−1. Fluctuations in enzyme activity may exist depending on the environment, mineral source, stressors, and physiochemical conditions. Furthermore, a prominent decrease in the activity of polyphenol oxidase in B. monnieri helps in combating oxidative damage at the cellular level and improves the defense system against the Reactive oxygen species (ROS) similarly reported by Preethi et al. 37
The embryotoxicity of the aqueous extract of B. monnieri has been chosen because it has shown better antioxidant capacity than other extracts in the majority of the in vitro antioxidant assays performed in this study and the toxic effect of crude aqueous extract of B. monnieri has not been reported so far. The embryotoxicity study of aqueous seed extract of Croton tiglium L by Yumnamcha et al, 39 exposed to varied concentrations ranging from 125-250 µg/mL has shown a dose-dependent lethality rate in zebrafish embryos and has observed developmental deformities such as pe, sy, hyp, delayed hatching, and sc. Similarly, the present study also observed varied survival rates of embryos across concentrations ranging from 2-10 mg/L providing dose-dependent toxic effects. The lower doses exhibited a lower mortality rate than higher concentrations (8 and 10 mg/L) where survival count substantially reduced at higher concentrations (P < .0001). The increased mortality may be due to the disruption of metabolic pathways by the phytocompounds in the aqueous extract. In contrast, no mortality count was observed in control groups. Furthermore, most embryos displayed delayed hatching based on the concentration, though the test groups exhibited delayed hatching but at the end of 72 h time point, the test groups eventually showed 100% hatching. The control groups showed optimal hatching patterns throughout the experiment. The experimental groups showed a heart rate ranging from 70-148 ± 2 beats per minute (bpm). Also, no major structural deformities were noticed at 2 and 4 mg/L. However, sub-lethal toxic effects such as hyp, sy, and pe were noticed in 6 to 10 mg/L in this present study similar results were noticed in the experiments by Yumnamcha et al 39 in aqueous seed extract of C. tiglium. Similarly, dose-dependent toxic effects in zebrafish embryos were noticed in plant extracts such as native Australian fruits, Piper sarmentosum, and twelve different plants belonging to northern Vietnam by Ali et al, 11 Abidin et al, 12 and Tran et al 13 respectively.
Limitations
This study opened diverse avenues for future research. The crude plant extract is often composed of a diverse range of bioactive phytochemicals which complicates the understanding of which component is truly responsible for the observed effects. Complete identification and investigation of bioactive compounds in extracts and their role in the stimulation of metabolic and enzymatic pathways causing toxic effects, safety profiles, and environmental concerns may further harness the antioxidant potential of B. monnieri leaf extract.
Conclusion
From this study, it is concluded that the zebrafish embryotoxicity assessments of aqueous extract of B. monnieri leaf reveal that the plant has the potential least toxic effects on developmental stages noticeably delayed hatching and hypopigmentation in zebrafish embryos were observed in the majority groups irrespective of the dosage and also a substantial reduction in the survivability of larvae was observed in higher doses (8 mg/L and 10 mg/L). Further research is needed to examine the mechanism of toxic/pharmacological constituents of this plant that can also help emphasize caution in higher concentrations which may be associated with potential risks. Though the plant has a sublethal effect at lower concentrations (2-6 mg/L) it has a potent antioxidant capacity and can serve as a promising ROS scavenger and may provide prophylactic action against various ROS interfering diseases.
Footnotes
Acknowledgments
We are deeply grateful to our co-authors for their continuous expertise, collaboration, insightful guidance, and encouragement for the successful completion of this work.
Author Contributions
Conceptualization, A.V.A., and R.S.; Methodology, A.B. and R.S.; Software and validation, P.G. and P.P.; Investigation and formal analysis, A.G. and K.G., and N.S.; Original draft preparation, R.S.; Supervision and resources, A.V.A. All authors have contributed and agreed to the publication of the manuscript.
Consent to Participate
Not applicable.
Consent to Publication
Not applicable.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
The IACE of CARE (Approval number: IAEC3/proposal-127.Lr:98) on 03/10/2023 has approved the experiments used in this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Statement of Human and Animal Rights
This article does not contain any studies on human subjects.
Statement of Informed Consent Form
This article consists of no study on human subjects and an informed consent form is not applicable.