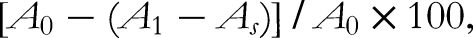Abstract
Introduction
Litsea cubeba (Lour.) Pers. is a perennial, dioecious, aromatic plant widely distributed across tropical and subtropical regions of Oceania, the Americas, and Asia.1,2 Known for its distinctive fragrance, this plant has been traditionally utilized for various purposes. Among Taiwanese indigenous people, its fruit has long been used as a condiment, underscoring its cultural and culinary significance. Over time, researchers have increasingly explored the bioactive properties of essential oils (EOs) derived from Litsea cubeba, particularly those obtained through hydrodistillation from various plant parts, including leaves, stems, roots, and fruits.1–3 These studies highlight the potential of Litsea cubeba EOs for diverse applications, driven by their unique chemical profiles and bioactivities.
The essential oils extracted from Litsea cubeba (LCEOs) exhibit a broad spectrum of biological properties, such as antibacterial, antifungal, antioxidant, anticancer, anti-inflammatory, and mosquito-repellent activities.4–6 These bioactivities are attributed to the volatile compounds that constitute the EOs, which vary in composition and concentration depending on the plant part used and the extraction method employed. Notably, fruit-derived LCEOs demonstrate superior antioxidant and antibacterial activities compared to oils obtained from other parts, such as leaves or flowers, making them highly valuable for therapeutic and industrial applications.1–3 However, optimizing these bioactivities requires a deeper understanding of how extraction methods and conditions influence the chemical composition of the EOs.
While hydrodistillation remains the primary method for extracting LCEOs, limited attention has been paid to the impact of varying extraction temperatures on the chemical and biological properties of the oils. The temperature-sensitive nature of volatile compounds means that different extraction conditions can result in significant variations in the EO profile and activity. Recent studies on volatile compounds and their applications, such as those by Thiara et al (2022), 7 Mozaniel et al (2021), 8 and Leonardo et al (2024), 9 suggest that nuanced approaches to extraction can yield more precise insights into the bioactivities of plant-derived EOs. Integrating these findings into research on Litsea cubeba could enhance the understanding and utility of its EOs in pharmaceuticals, cosmetics, and beyond.
To address this knowledge gap, our study employs a stepwise heating approach during the hydrodistillation of Litsea cubeba fruit, enabling the extraction of EOs across distinct temperature ranges. This innovative method allows us to isolate and compare EO fractions, examining their chemical compositions and corresponding biological activities. By correlating specific temperature ranges with bioactivity, we aim to optimize the extraction process for enhanced efficacy and explore the relationship between chemical composition and biological function. Through this work, we seek to contribute to the broader application and understanding of LCEOs, advancing their potential in various industries.
Methodology
Plant Material
Fruits of Litsea cubeba (Lour.) Pers., harvested between June 18 and July 1, 2023, were purchased from farmers in Naluo Village, Guanxi Township, Hsinchu, Taiwan (altitude 500-600 m, latitude 24°40′ N and longitude 121°14′ E). The species was identified and registered in the herbarium with the species number T0074609. 10
EO Isolation
After the fruits were allowed to naturally dry in a ventilated environment for 2 to 3 days at room temperature, they were refrigerated at 4 °C until extraction. To start the extraction process, 300 g of fruit was introduced into a 2-L round-bottom flask containing 3 to 4 times its weight in reverse-osmosis water and was subjected to hydrodistillation in a Clevenger-type apparatus equipped with a temperature controller (HT-720; Newlab, Taipei, Taiwan). Once the temperature reached 95 °C to 98 °C, a small amount of EO started to flow out. The flow of EO stopped after approximately 1 h of refluxed extraction, and the EO was collected and designated as LCF-1. Subsequently, the temperature was increased to 99 °C to continue the extraction process. When the flow of EO stopped after approximately 2 to 3 h of refluxed extraction, the EO was collected and designated as LCF-2. Subsequently, the temperature was increased to 100 °C to continue the extraction process. When the flow of EO stopped after refluxed extraction, the EO was collected and designated as LCF-3. These EOs were stored at 4 °C in the dark for analysis.
EO Analysis
Gas chromatography mass spectrometry (GC-MS) was used to analyze the 3 EO fractions on a Clarus 690/SQ8S GC-MS system (PerkinElmer, Waltham, MA, USA). 4 Analysis was conducted using a DB-5MS capillary fused silica column (30 m, with an internal diameter of 0.25 mm and a film thickness of 0.25 μm; Agilent Technologies, Santa Clara, CA, USA). Subsequently, 1 μL of a diluted sample (2 μL/mL in ethyl acetate) was injected into the column at a flow rate of 1 mL/min. The temperature program was run as follows: 40 °C for 2 min, rising at a rate of 4 °C/min to 100 °C and then at a rate of 10 °C/min to 250 °C and held for 5 min. The other parameters were set as follows: injection temperature, 260 °C; ion source temperature, 280 °C; electron ionization, 70 eV; carrier gas, He 1 mL/min; and mass scan range, 40 to 600 m/z. All components were identified using the Wiley/NBS Registry of Mass Spectral Data (v.8.0) and National Institute of Standards and Technology (v.2.0) GC-MS libraries. Retention indices (Kovats’ RI) were calculated for all volatile components with a homologous series of n-alkanes (C9-C24) (49451-U, Supelco, Mo. U.S.A). 11 These components were identified by using co-injection with standards whenever possible.
Antibacterial Activity
Bacterial Strains and Growth Conditions
Two foodborne bacterial strains, namely Gram-positive Staphylococcus aureus (BCRC 10451) and Gram-negative Escherichia coli (BCRC 11634), were used in this study. These strains were grown on nutrient agar and incubated overnight at 35 °C. Bacterial suspensions were prepared in sterilized water to achieve a McFarland standard of 0.5, with 108 colony-forming units (CFUs) per milliliter.
Disc Diffusion Assay
An agar disc diffusion assay was conducted to measure the antibacterial activity of the 3 EO fractions. 5 Briefly, agar plates were prepared as 90-mm Petri dishes containing 22 mL of nutrient agar (Difco Laboratories, Becton Dickinson, Franklin Lakes, NJ, USA) to achieve a final depth of 3 mm. Subsequently, 100 μL of bacterial suspension (108 CFUs/mL) was spread on the surface of these plates. After the agar plates were inoculated with the test microorganisms, filter discs with a diameter of 6 mm were individually impregnated with 25 μL of LCF-1, LCF-2, and LCF-3 and placed on the plates. After the plates were maintained at 4 °C for 2 h to enable diffusion of EOs, they were incubated at 35 °C for 18 to 24 h. Kanamycin (1.0 mg/mL) was used as a positive control. Negative controls were prepared using blank discs. Finally, the diameters of the resulting zones of inhibition were measured, including the diameters of the paper discs. The tests were conducted in triplicate.
Determination of Minimum Inhibitory Concentrations
Minimum inhibitory concentrations (MICs) were determined using a microdilution broth assay. 6 EOs were dissolved in dimethyl sulfoxide, and a serial 2-fold dilution of EOs in nutrient broth was prepared to obtain the following concentrations of EOs: 900, 450, 225, 112.5, 56.2, 28.1, 14.0, 7.0, 3.5, and 1.8 μg/mL. After shaking, 20 μL of each solution was added to each well on a 96-well microtiter plate. Subsequently, 20 μL of microbial suspension, adjusted to 107 CFUs/mL, and 160 μL of nutrient broth containing 0.5% (v/v) Tween 80 were added to each well. Finally, the plates were incubated at 35°C for 24 h. The MIC of the sample was determined as the lowest concentration that did not allow for any visible growth of the test microorganism after incubation. The tests were conducted in triplicate.
Antioxidant Activity Evaluation
The antioxidant activity of LCF-1, LCF-2, and LCF-3 was measured using a 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging method.
12
Briefly, 20 μL of EOs at concentrations ranging from 125 to 2000 μg/mL was mixed with 80 μL of methanol and added to 100 μL of DPPH radicals in methanol (0.25 mM). After the mixture was vigorously shaken, it was left to stand for 60 min at room temperature in the dark. Absorbance was measured at 517 nm. The percentage of radical scavenging activity was calculated using the following formula:
Statistical Analysis
The results are presented as the means of at least 3 replicates ± standard errors. The data were subjected to one-way analysis of variance, and differences between means were determined at a 5% probability level by using Fisher's least significant difference test (CoHort Software, Monterey, CA, USA).
Results
As presented in Table 1, the total yield of EO obtained through the hydrodistillation of fresh L. cubeba fruit was 4.95% (v/w), with 8.60% obtained at 95°C to 98°C (LCF-1), 84.83% obtained at 98°C to 99°C (LCF-2), and 6.53% obtained at 99°C to 100°C (LCF-3). These results indicate that the majority of the EO from L. cubeba fruit was extracted at 98°C to 99°C.
Yield and Percentage of Essential Oils Extracted from Litsea cubeba (Lour.) Pers. Fruit Through Hydrodistillation at Different Temperatures.

GC-MS was used to identify the chemical compositions of the 3 EO fractions (Table 2). A total of 14, 17, and 19 components were identified, accounting for 98.17%, 98.69%, and 97.52% of LCF-1, LCF-2, and LCF-3, respectively.
Chemical Compositions of Essential Oils Extracted from Litsea cubeba (Lour.) Pers. Fruit Through Hydrodistillation at Different Temperatures.

Both limonene (LCF-1, 23.41%; LCF-2, 11.46%; and LCF-3, 31.57%) and the isomers of citral, neral (LCF-1, 26.75%; LCF-2, 33.57%; and LCF-3, 19.75%), and geranial (LCF-1, 28.99%; LCF-2, 40.38%; and LCF-3, 24.63%), were identified as predominant components, accounting for 79.15%, 85.41%, and 75.95% of LCF-1, LCF-2, and LCF-3, respectively (Table 2). The concentrations of neral and geranial were higher in LCF-2 than in LCF-1 and LCF-3, and therefore, LCF-2 contained the highest proportion of oxygenated monoterpenes. By contrast, the concentration of limonene was higher in LCF-3 than in LCF-1 and LCF-2, and therefore, LCF-3 contained the highest proportion of monoterpene hydrocarbons. Other major components were detected among the 3 EO fractions, including α-pinene (LCF-1, 1.82%; LCF-2, 0.66%; and LCF-3, 2.35%), β-myrcene (LCF-1, 2.89%; LCF-2, 1.40%; and LCF-3, 4.47%), verbenol (LCF-1, 1.53%; LCF-2, 1.17%; and LCF-3, 0.49%), isogeranial (LCF-1, 2.42%; LCF-2, 2.02%; and LCF-3, 1.19%), and methyl heptenone (LCF-1, 2.16%; LCF-2, 1.75%; and LCF-3, 2.21%). Some differences were observed between the 3 EO fractions; these differences were attributable to nonmajor components, such as sabinene (0.40%), which was identified in LCF-1 only; nerol (0.55% and 0.49%, respectively), geraniol (1.24% and 1.29%, respectively), copaene (0.30% and 1.06%, respectively), and β-caryophyllene (0.34% and 1.88%, respectively), which were identified in LCF-2 and LCF-3; and bicyclogermacrene (0.75%) and α-terpinyl acetate (1.39%), which were identified in LCF-3 only.
A filter paper disc diffusion test was conducted to examine the antibacterial activity of the 3 EO fractions (Table 3). The results indicated that the inhibition zones of LCF-1, LCF-2, and LCF-3 against S. aureus were 90 mm. After 3-fold dilution with dimethyl sulfoxide, the inhibition zone of LCF-2 remained at 90 mm, whereas those of LCF-1 and LCF-3 became 72.8 and 77.7 mm, respectively. At 9-fold dilution, the inhibition zone of LCF-2 was 19.2 mm, which was significantly larger than the inhibition zones of LCF-1 and LCF-3 (15.9 and 15.3 mm, respectively, P < .05) and the inhibition zone obtained for the positive control, kanamycin (16.2 mm). In addition, LCF-2 exerted a strong antibacterial effect on S. aureus, as indicated by an MIC of 5.24 μg/mL (data not shown).
Antibacterial Activity of Essential Oils Extracted from Litsea cubeba (Lour.) Pers. Fruit Through Hydrodistillation at Different Temperatures.
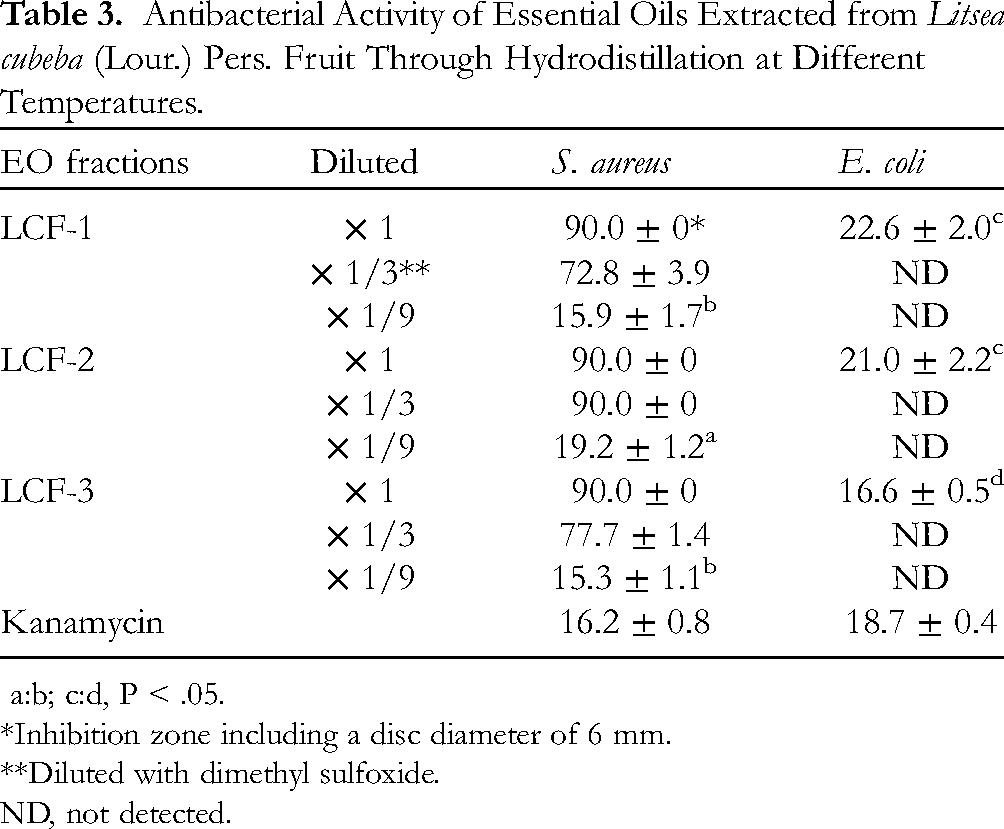
a:b; c:d, P < .05.
*Inhibition zone including a disc diameter of 6 mm.
**Diluted with dimethyl sulfoxide.
ND, not detected.
Compared with the aforementioned inhibitory effects, the inhibitory effects of the 3 EO fractions against E. coli were less significant. All inhibition zones were smaller than 25 mm, with LCF-3 having the smallest inhibition zone (16.6 mm, P < .05, Table 3).
The antioxidant activity of LCF-1, LCF-2, and LCF-3 was evaluated using a DPPH radical scavenging assay through comparison with ascorbic acid as a standard antioxidant. Figure 1 presents the results of this DPPH radical scavenging assay. At test concentrations of 125 to 2000 μg/mL, all 3 EO fractions exhibited concentration-dependent scavenging activity against the DPPH radical. Among the 3 fractions, LCF-3 exhibited the highest quenching activity, with a low IC50 value of 872.0 μg/mL. This value was lower than those of LCF-1 and LCF-2 (1625.9 and 1720.1 μg/mL, respectively) and significantly weaker than that of ascorbic acid (3.5 μg/mL, data not shown).

Antioxidant activity of essential oils extracted from Litsea cubeba fruits through hydrodistillation at different temperatures. White: LCF-1; gray: LCF-2; black: LCF-3.
Discussion
Several factors affect the yield and composition of EOs, including plant variety, 13 plant organ, 1 plant developmental stage, 14 fertilizer, 15 seasonal variation, 16 harvest time, 17 phenological stage, 18 geographical region, 19 and extraction technique. 20 During hydrodistillation, the qualitative and quantitative chemical compositions of EOs are affected by the collection period. 21 To the best of our knowledge, few studies have explored the differences between the compositions and characteristics of EOs extracted through hydrodistillation from different temperature ranges.
Multiple studies have indicated that limonene, neral, and geranial are the main components of L. cubeba fruits.17,22,23 In this study, although the highest proportion of EO was observed in LCF-2 (Table 1), limonene, neral, and geranial were predominant in the 3 fractions. Notably, limonene is widely present in the EOs extracted from the peels of citrus species, 24 and neral and geranial are the only major components in the EOs extracted from Backhousia citriodora, 25 Cymbopogon flexuosus, 26 and Ocimum basilicum. 27 These 3 components provide these EOs with their biological activity.
Among all fractions, LCF-3 had the highest proportion of limonene (31.57%), followed by LCF-1 (23.41%) and LCF-2 (11.46%, Table 2). This finding indicates that limonene was responsible for the high antioxidant activity of LCF-3 (Figure 1). Moreover, LCF-2 had the highest proportion of neral and geranial isomers (73.85%), followed by LCF-1 (55.74%) and LCF-3 (44.38%, Table 2). Given that LCF-2 was significantly more effective than LCF-1 and LCF-3 in inhibiting the growth of S. aureus (Table 3), both neral and geranial presumably played a key role. Su and Ho 1 mentioned that citral in the fruit LCEO is the main cause of the antimicrobial activity. The main components of the fruit LCEO from North-east India are methyl heptenone (30.9%) and limonene (23.1%), and its MIC value for inhibiting S. aureus is 80 μg/mL3, which is significantly lower than the 5.24 μg/mL of LCF-2 (data not shown).
Multiple studies have indicated that Gram-negative bacteria are more resistant than Gram-positive bacteria are because they have an outer membrane that can prevent the penetration and action of EOs.6,25,28 In this study, the inhibition zones achieved with the 3 undiluted EO fractions for S. aureus reached 90 mm, which is significantly larger than that obtained for E. coli (<25 mm, Table 3). However, no significant difference was observed between the inhibition zones of LCF-1 and LCF-2 for E. coli, with LCF-1 and LCF-2 exhibiting a greater inhibitory effect than that of LCF-3 (P < .05). Taken together, these findings suggest that limonene, neral, and geranial are not the main components involved in the inhibition of E. coli growth.
In addition to the three main components aforementioned, the fruit LCEOs extracted from different temperature fractions still have some compositional differences and probably affect their biological activities. Asili et al 29 compared the EOs extracted from the flowering branches of 7 Artemisia species and discovered that the main component of Artemisia santolina is verbenol, which serves as an active compound inhibiting the growth of E. coli. In this study, we discovered that the proportion of verbenol was higher in LCF-1 (1.53%) and LCF-2 (1.17%) than in LCF-3 (0.49%, Table 2). Taken together, these findings suggest that verbenol is the main component responsible for the higher effectiveness of LCF-1 and LCF-2 compared with that of LCF-3 in inhibiting the growth of E. coli.
Senani et al 13 compared the free radical scavenging abilities of the EOs of 2 Teucrium species. They reported that Teucrium flavum primarily contains α-pinene, limonene, and β-caryophyllene, which explains its higher activity compared with Teucrium polium, which does not contain these components. In the present study, we discovered that LCF-3 had higher antioxidant activity than that of LCF-1 and LCF-2 (Figure 1), which is presumably attributable to the synergistic properties associated with limonene, α-pinene, and β-caryophyllene.
In this study, all 3 fractions of LCEO exhibited high Gram-positive inhibitory activity, particularly LCF-2, which had an MIC of 5.24 μg/mL for S. aureus (data not shown), and had a moderate free radical scavenging ability, with an IC50 of 872 μg/mL for DPPH (LCF-3). She et al 17 compared the activity of LCEOs extracted from fruits collected in June, July, and August. They discovered that the EOs collected in July had the highest antibacterial activity, whereas those collected in August had the highest antioxidant capacity. The sampling experiment was from June 18 to July 1, 2023 in this study. Therefore, future studies should sample LC fruits in August and extract EOs from different temperature ranges to further analyze the components and biological activity of these EOs. These studies can further identify the main components that influence the antibacterial and antioxidant properties of EOs.
In addition to the above-mentioned antibacterial and antioxidant activities, the fruit LCEO also has anti-inflammatory and anti-cancer properties, and these activities are mainly due to the presence of citral.30,31 Therefore, it is reasonable to speculate that the EO collected in the appropriate temperature range that contains higher concentrations of citral, such as LCF-2 in this study, they can have higher anti-cancer and anti-inflammatory activities than that of the traditional continuous-heating extracted EO.
Certainly, the active ingredients of EOs from various plants are different, the suitable extraction temperature range required for the special biological activity of each plant still needs to be carefully explored.
In conclusion, we discovered that the fruit LCEOs extracted from different temperature ranges using a stepwise-heating methodology exhibited distinct antioxidative and antibacterial activities, and these differences were owing to the differences in ingredients. There are limited published reports on the differences between the compositions and biological activities of EOs extracted through hydrodistillation from different temperature ranges. Therefore, this study provides an innovative extraction strategy for EOs, namely, to recover the EO in specific temperature range according to the biological activity required.
Footnotes
Acknowledgements
The authors would like to thank the Forest Products Inspection Laboratory at the College of Agriculture and Natural Resources, National Chung Hsing University, for their assistance with the GC-MS analysis.
Authors’ Contribution
C.J.D. and C.C.L. provided important intellectual contribution to the design and interpretation. K.F.X., G.J.Y., H.Y.P. conducted most experiments. W.H.L. and Pai Hsiu-Hua envisioned the project and analysed the data. P.H.H. wrote the paper and funded the experiment.
Data Availability
All the data included in the study are supplied with the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
Ethical Approval is not applicable to this article.
Funding
Toxic and Chemical Substances Bureau Environmental Protection Administration Executive Yuan, R.O.C. (Taiwan) provided support for the development this work.