Abstract
Wheat germ agglutinin (WGA) is a lectin composed of 4 homologous hevein domains. It has been shown that WGA binds N-acetyl glucosamine (GlcNAc)-related oligosaccharides and has applications as commercial reagent to detect glycans containing such modified residues. Peptidoglycan (PGN), the main component of the bacterial cell wall, is a polymeric material made of repeating disaccharide units of GlcNAc-N-acetylmuramic acid cross-linked with short polypeptide fragments. Wheat germ agglutinin is able to bind bacterial cells, a phenomenon that could correlate with its plant-defense capacities, but there is no information at the molecular level about how WGA binds to the PGN. Herein, we present structural data on the binding of a short PGN fragment to WGA by means of saturation transfer difference nuclear magnetic resonance studies. The results show that the GlcNAc residue establishes the major contacts with WGA, followed by the N-acetylmuramic acid residue. In contrast, the peptide moiety displays minor contacts at the binding site.
Many plants express chitin-binding lectins that have been identified as plant-defense proteins. A family of these plant-defense proteins contains one or several chitin-binding sites of the so-called hevein domains. These domains have been named after hevein, a small protein of 43 residues and 4 disulfide bonds, which binds chitin and chitooligosaccharides.
The hevein domain has become a powerful tool in several fields of biological research and its lectin properties have been extensively studied in the field of glycosciences. 1 Its activity as a protein for plant defense has been the focus of interest in plant biology. 2 Moreover, its potential applications in biotechnology have been increasing the resistance to fungi, transforming plants with genes encoding the hevein domain, 3,4 as well as in biomedicine, as an antimicrobial agent. 5
The best characterized chitin-binding protein with multiple hevein domains is wheat germ agglutinin (WGA), which shows 4 hevein repetitions and is present in the seeds of wheat as a stable homodimer of 36 kDa. 6–8 Its antimicrobial properties have been described: WGA inhibits fungal growth through interactions with the components of the cell walls 9–11 and it is also able to agglutinate cells transformed in vitro 12 by recognizing oligosaccharides which contain N-acetyl glucosamine (GlcNAc) and N‐acetyl neuraminic acid residues at the nonreducing end. 7,13–16
Previous X-ray and nuclear magnetic resonance (NMR) structural studies have identified the aromatic residues at relative positions 21, 23, and 30 in hevein domains as the key players for sugar binding. 1,9,17,18 Moreover, hevein displays an extended binding site that is capable of accommodating several GlcNAc units, with 2 strategically placed aromatic residues establishing CH-π interactions with 2 contiguous pyranose rings. 19 This extended binding site also allows for the existence of 2 different modes of interaction for trisaccharides and longer oligosaccharides. 20,21 For instance, in the case of domain B of WGA, for the (GlcNAc)3 trisaccharide, the first binding mode shows the nonreducing terminal GlcNAc residue at the so-called subsite +1, interacting with Tyr66 (corresponding to aromatic residue at 23 in hevein) through CH-π interactions showing a hydrogen bond with Ser62 (residue 19 in hevein) and displaying both CH-π and hydrogen bond contacts with Tyr73 (residue 30 in hevein). The intermediate residue is located at subsite +2, providing a CH-π interaction with Tyr64 (residue 21 in hevein) whereas the reducing GlcNAc residue displays very minor contacts with the lectin at subsite +3 (Figure 1). 1 In the second orientation for this trisaccharide, the residue at the reducing end is now placed at subsite +2, interacting with Tyr64. The intermediate residue is located at subsite +1 showing the contacts described above, and the nonreducing terminus shows minor contacts with the lectin at the so-called subsite −1 (Figure 1). 9,17 Because chitin is the major component of fungi cell walls as well as of invertebrates exoskeleton, the implication of these peptides in plant defense and antifungal activities has been associated with their lectin character as chitin ligands. 22 However, the description of the antimicrobial activities against other microorganisms such as bacteria, which do not contain chitin, has required to find alternative mechanisms of action. 22,23 In fact, in some cases, the antimicrobial activity has been associated with the polycationic nature of these peptides. 24 Interestingly, although the saccharidic part of the bacterial cell wall structure [constituted by peptidoglycan (PGN)] is closely related to chitin, few studies have addressed the analysis of the direct interaction between oligosaccharide-derived PGNs and hevein domains. 25–27 In fact, to the best of our knowledge, no structural data have been described, although the existence of a possible interaction has been speculated. For example, Lotan et al 25 deduced that PGN-derived di- and tetra-saccharides were able to inhibit WGA-induced hemagglutination, whereas Ayouba et al 27 described evidences of similar oligosaccharides inhibiting Urtica dioica agglutinin, which gathers 2 hevein domains. Interestingly, low-molecular-weight PGN fragments are very active molecules in host-bacterial interactions acting as citotoxins or virulence factors or stimulating the innate immune response. 28 Despite this dearth of information, it is noteworthy to mention that WGA is marketed as a reagent for histochemical analysis with application to stain Gram-positive bacteria (Alexa Fluor® 488 conjugate reagent of WGA, MolecularProbes) 29 because of its acknowledged recognition capability for bacterial PGN. 30

Model representations of the 2 existing binding modes for wheat germ agglutinin (WGA) domain B and (N-acetyl glucosamine)3 complex. (a) Mode 1/2/3, (b) Mode −1/1/2. 18 Numbering corresponds to the WGA-B domain as in Protein Data Bank: 2UVO, 6 equivalent to residues Ser19 and aromatics at 21, 23, and 30 in hevein.
Herein, the binding of short chitin oligosaccharides and PGN fragments to WGA has been characterized by saturation transfer difference (STD)-NMR. 31,32 For this purpose, N-acetyl glucosaminyl-muramyl-dipeptide (GMDP) was selected as a model of the minimal disaccharide dipeptide repeating unit of PGN and compared with triacetylchitotriose used as a positive ligand control (Scheme 1).

Formulas of (a) (N-acetyl glucosamine)3 and (b) N-acetyl glucosaminyl-muramyl-dipeptide. The reducing end is free.
The existence of one unique binding mode to WGA and preference for the β-anomer isomer at the reducing end have been confirmed. The determined binding pose places the GlcNAc residue at position +1 and the muramyl moiety at position +2 (Figure 1).
The Interaction of N,N',N″-Triacetylchitotriose with WGA
First, the interaction of WGA with N,N′,N″-triacetylchitotriose (GlcNAc)3 was studied as a positive control 7 for the experimental approach based on STD previously used to study the interaction with diacetylchitobiose. 33 It is well known that STD allows to define the ligand epitope when bound to the receptor because of the efficient intermolecular transfer of magnetization from proton spins of the receptor to the nearest protons of the ligand 34,35 and it has been extensively applied to study carbohydrate-protein interactions. 36,37 The corresponding STD spectra are shown in Figure 2.

Saturation transfer difference (STD) spectra of (N-acetyl glucosamine)3 in deuterated buffer in the presence of wheat germ agglutinin (ratio 20:1). (a) Off-resonance spectrum, with the 1H NMR assignments
18
and definition of different spectral regions: a (H2,3,5-1α; H6a-3; H6a-2; H6a-1β); b (H2,6b-3; H2,3
As a preliminary control the lack of nonspecific transfer of saturation in the absence of protein was also verified by performing a reference STD experiment on a free (GlcNAc)3 sample.
The analysis of the STD data allowed to draw a qualitative map of the contact epitope (Figure 3). The data showed that most of the protons of the nonreducing end and middle residues (3 and 2) received significantly higher saturation than those at the reducing end, being the highest one on the acetate protons of residue 3. In fact, in both binding modes in Figure 1, the methyl group of the reducing end residue
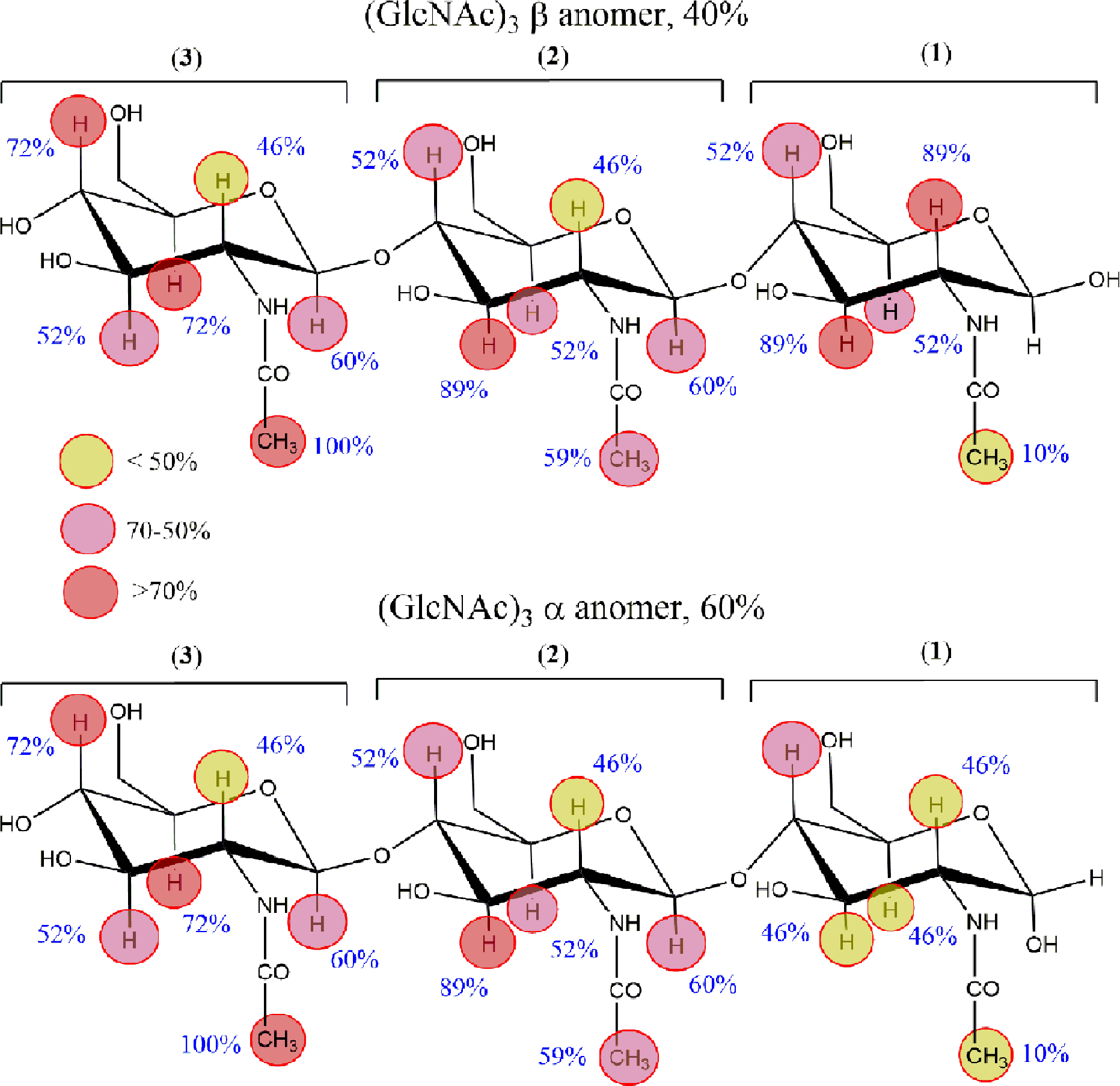
Representation of the relative saturation transfer difference (STD) intensities for each (N-acetyl glucosamine)3 proton. Top: β-anomer; bottom: α-anomer. The colors are scaled following the relative saturation from dark red colors, indicating that the STDs are equal or larger than 70%, up to yellow, corresponding to STDs below 50%.
The Interaction of GMDP with WGA
Next, the interaction between WGA and the GMDP PGN fragment was studied. Interestingly, related PGN fragments have been described as strong immunoadjuvants. 43 N-Aacetyl glucosaminyl-muramyl-dipeptide shows the N-acetylmuramyl residue as free-reducing end in a 60:40 α:β equilibrium.
First, the assignment of the 1H NMR signals of the ligand GMDP was performed using a combination of standard TOCSY, NOESY, and ROESY experiments. A combination of NOESY and TOCSY experiments performed in water (10% D2O) was used to assign differentially the N-Acetyl signals of both GlcNAc and MurNAc residues (see assignments in Table 1). Figure 4 shows the 1H NMR spectrum in the absence or presence of WGA, as well as the STD spectra. The corresponding chemical shift and saturation values are shown in Table 1.

(a) 1H nuclear magnetic resonance spectrum of N-acetyl glucosaminyl-muramyl-dipeptide (GMDP) in the absence of protein at 298K, pH 6, at 600 MHz. (b) Off-resonance spectrum for GMDP (2 mM) in the presence of wheat germ agglutinin (0.2 mM, saturation time = 2 s; off-resonance frequency: δ 100 ppm). The existence of line broadening is evident. The dashed lines connect the same peaks in the a and b spectra. (c) Saturation transfer difference spectrum (on-resonance frequency: 7 ppm). Inset amplifies the region corresponding to methyls groups.
1H Nuclear Magnetic Resonance Chemical Shifts (δ, Ppm) for N-Acetyl Glucosaminyl-Muramyl-Dipeptide Protons.

The corresponding saturation transfer difference (STD) values in the presence of wheat germ agglutinin are also shown. Values are given in ppm with reference to methyl trimethylsilylpropanoic acid (TSP) (0 ppm). In the case of broad signals and/or overlapping, the saturation values are approximate.
The comparison of 1H NMR spectra in the absence and presence of the protein (Figure 4) allowed to assess the existence of significant broadening of the ligand signals in the presence of the lectin, as also observed for fluoroacetylated chitooligosaccharides. 20,43 This phenomenon indicates the existence of binding in a nonambiguous manner. 44 Fittingly, the broadening effect is selective and remarkable only for the signals corresponding to the muramyl residue in its β-form. Thus, the β-muramic H-1 signal at δ 4.6 ppm practically disappears, whereas the corresponding signal for the α-anomer (above δ 5 ppm) is still clearly visible.
The STD spectra (Figure 4), especially in the region of the acetyl groups, shows clearly the highest saturation on the reducing end GlcNAc acetyl (22%) followed by the MurNAc acetyl of the β-anomer (6.7%) and much less intense α-anomer (1.1%, Table 1). The observed selectivity allowed defining the binding epitope of GMDP. Fittingly, discrimination favoring the β-anomer was also observed.
The methyl signal of the nonreducing GlcNAc residue received the maximum saturation (22%, Table 1). The rest of the sugar protons displayed STD values between 2% and 12%, whereas the peptide protons did not reach STD values higher than 4%. This fact indicates that the peptide region provides minor contacts with lectin (Table 1). The binding epitope of GMDP to WGA is shown in Figure 5.

Schematic perspective of the binding epitope of N-acetyl glucosaminyl-muramyl-dipeptide to wheat germ agglutinin as deduced from the saturation transfer difference (STD) analysis. The colors are scaled following the relative saturation from dark red colors, indicating that the STDs are equal or larger than 70%, up to yellow, corresponding to STDs below 30%. Signal broadening in the presence of the protein is marked with a star.
With this experimental information, a 3D model for the GMDP-WGA complex was built. As starting point, the GMDP conformation present in Protein Data Bank (PDB) code 1D0K 19 and the WGA-B geometry deposited as PDB code 2UVO 6 were used. The ligand geometry corresponds to the crystal structure of GlcNAc-MurNAc-L-Ala-D-Glu (in fact, a GMDP analogue with deamidated glutamic acid instead of the isoglutamine residue) bound to a lytic transglycosylase, whereas that of the lectin geometry is present in the crystal structure of the WGA-GlcNAc complex. The ligand was then manually docked into the binding pocket of the WGA-B domain and the corresponding complex was optimized using molecular dynamics (MD) simulations.
A representative structure is shown in Figure 6. The GMDP fits well into the binding site and establishes the characteristic hevein-sugar interactions: Ser62 provides a hydrogen bond to the carbonyl group of the GlcNAc acetamide; Tyr73 establishes a van der Waals interaction with the methyl group of the same residue. This fact is consistent with the epitope described above (Figure 5), wherein the methyl N-Ac group received the maximum saturation. Tyr66 and Tyr64 establish stacking interactions with the two consecutive GlcNAc and MurNAc moieties. This model also justifies the observed line broadenings and STDs for the protons of the pyranose rings. The peptide region is essentially exposed to the solvent, whereas the methyl group of the lactic acid at position 3 of muramic acid points toward Tyr64, thereby justifying its significant STD. The switch of the configuration of the muramic acid moiety from β to α makes the axial hydroxyl group point toward Tyr64. This orientation is highly unfavorable for binding and provides the explanation for the preferred binding for the β-anomer.

Representative structure from the explicit solvent Desmond Molecular Dynamics simulation consistent with the observed STD.
In conclusion, the STD analysis of the interaction of a PGN fragment with WGA has been carried out. The results indicate that this lectin indeed interacts with the ligand in a specific manner. The binding epitope has been deduced, providing the structural basis for exploring the possible use of WGA for recognition and binding immunoactive PGN fragments as well as antimicrobial agent directed against bacterial cell wall.
Experimental
Materials
Saccharides and lectins were obtained from commercial sources. N,N ',N ″-triacetylchitotriose was from Toronto Research Chemical, WGA from Sigma, and N-acetylglucosaminyl-N-acetylmuramyl-l-alanyl-d-isoglutamine (GMDP) from Calbiochem.
NMR Experiments
All experiments were conducted with Bruker Avance AV 600 MHz equipped with cryoprobe or AV 500MHz spectrometers. For each saccharide, 2 sets of proton 1D and 2D homonuclear experiments TOCSY (20 and 60 ms mixing time), NOESY (0.5 s mixing time), and ROESY were acquired, one in D2O and another in 90:10 H2O:D2O, for signal assignments.
Saturation transfer difference experiments were recorded by irradiating for 2 s at 2 frequencies: 100 ppm (off-resonance frequency) and 7 ppm (on-resonance frequency). The target samples were prepared using (GlcNAc)3 (1 mM) in the presence of WGA, with a ligand:lectin 100:1 molar ratio and GMDP 0.5 mM in the presence of WGA 12.5 mM, ligand:lectin 40:1 molar ratio. The specific STD spectra were calculated by subtracting the intensities of the signals in the on-resonance spectrum from the reference spectrum off-resonance (Figure 2). Every signal in the STD spectrum was referenced to its intensity in the off-resonance spectrum and normalized. A 100% value was assigned to the signal that received the maximum saturation.
Molecular Modeling
Molecular dynamics simulations were performed by using the Explicit Solvent Desmond Molecular Dynamics Simulation 45 provided with the Schrödinger Suite Program. In a first step, an orthorombic box of TIP3P water molecules was created. The system protein/ligand/explicit water molecules were then subjected to a short minimization to avoid wrong clashes and used as starting point for a short 4 ns MD. A set of 834 frames were generated and analyzed.
Footnotes
Acknowledgments
We thank AgenciaEstatal de Investigacion of Spain (projects CTQ2015-64597-C2-1P, CTQ2015-64597-C2-2P, and SEV-2016-0644 Severo Ochoa Excellence Acreditation). K. el B. acknowledges a fellowship from Spanish Agency for International Development Cooperation (AECID).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support from AgenciaEstatal de Investigacion of Spain (projects CTQ2015-64597-C2-1P and CTQ2015-64597-C2-2P).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
