Abstract
Here we determined the phenolic compounds and antioxidant, antimicrobial, genotoxic, and anticancer potential of edible mushrooms Craterellus cornucopioides. The phytochemical analysis was evaluated using high-performance liquid chromatography. Antioxidant activity was evaluated by free radical scavenging, superoxide anion scavenging, and reducing power. Craterellus cornucopioides extract had potent antioxidant activity. Further, the antimicrobial potential was determined by a microdilution method, where minimum inhibitory concentration values ranged from 0.1 to 10 mg/mL. Genotoxic potential was determined by cytokinesis block micronucleus test. The separate treatment did not show genotoxic effect, whereas the combined treatment with mitomycin C significantly reduced the micronuclei frequency in a dose-dependent manner. The highest concentration significantly reduced nuclear division index in comparison to untreated human peripheral blood lymphocytes, whereas in combined treatment, the extract did not significantly affect this parameter. Finally, the cytotoxic activity was tested using microculture tetrazolium test where measured IC50 values ranged from 65.5 to 131.7 μg/mL.
In many parts of the world, wild mushrooms are regularly collected and used directly as a main source of food or as a condiment. Today mushrooms are valuable not only because of the appealing texture and unique taste, but also of their important chemical and nutritional characteristics. 1
Mushrooms are considered to be a good source of digestible proteins with a content of 10% to 40% on a dry weight basis, more than the vegetables but somewhat less than most meats and milk. Data relating to the studies of amino acids in mushrooms show that mushrooms contain all the essential amino acids. Fresh mushrooms contain 3% to 21% carbohydrates and 3% to 35% fiber on a dry weight basis; thus a considerable proportion of the carbohydrate of mushrooms is functioning essentially as dietary fiber; in this way the calorific value of most mushrooms is low. Mushrooms are also rich in minerals and appear to be an excellent source of vitamins. They also contain all the main classes of lipid compounds including free fatty acids, mono-, di-, and triglycerides, sterols, sterol esters, and phospholipids, but in low levels, around 2% to 8% of dry weight, which make them a low-calorie food. 2
Besides the important nutritional values, mushrooms also have valuable health benefits. Their consumption has consistently been shown to have beneficial effects on human health, because of various immunological and anticancer properties of certain mushrooms, antioxidants, antihypertensive and cholesterol-lowering properties, liver protection, as well as anti-inflammatory, antidiabetic, antiviral, and antimicrobial properties. 3
Hence, the present investigation was designed to show the potential antigenotoxic, antioxidant, antimicrobial, and anticancer activities of the important edible mushrooms Craterellus cornucopioides (L. Fr.) Pers. (Cantharellaceae). There are several papers about the chemical composition and bioactivity of the species mentioned. Guo et al 4 isolated two new illudane sesquiterpenoids, craterellins D and E, and one new menthane monoterpene, 4-hydroxy-4-isopropenyl-cyclohexanemethanol acetate, from C. cornucopioides and evaluated their cytotoxic activities. Similarly, Yang et al 5 isolated a novel polysaccharide fraction from C. cornucopioides and explored its antioxidant activity.
The developed high-performance liquid chromatography with diode-array detection (HPLC-DAD) method was applied for the analysis of phenolic compounds in tested mushroom. Peak identity was established by both the retention time compared to that of reference compounds and the characteristic UV spectrum. Calibration curves were used for the quantitative determination of phenolic compounds. The results of the contents of analytes in mushroom are presented in Table 1.
Phenolic Compounds in Acetone Extract of Craterellus cornucopioides. Results Are Expressed as Micrograms of Phenolic Per Gram of Dried Mushroom.

The results of identification of phenolics in C. cornucopioides were in agreement with the study of Palacios et al 6 which found ferulic acid (14.03 µg/g) and gallic acid (118.8 µg/g) in C. cornucopioides. Also, Liu et al 7 determined quercetin (39.6 µg/g), p-coumaric acid (8.6 µg/g), caffeic acid (16.2 µg/g), and gallic acid (0.7 µg/g) in C. cornucopioides.
The scavenging 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals, superoxide anion radicals, and reducing power of the studied extract are shown in Table 2. The IC50 value was 19.7 for DPPH radicals and superoxide anion radicals scavenging activity was 221.8 µg/ml. As shown in Table 2, reducing power was concentration dependent. Measured value of absorbance for reducing power was about 0.10. In various antioxidant activities (free radical scavenging, superoxide anion radical scavenging, and reducing power), there was a statistically significant difference (P < 0.05) between the extract and positive control which was prepared by the same procedure as the extract except replacing the mushroom extract with ascorbic acid.
DPPH Radical Scavenging Activity, Superoxide Anion Scavenging Activity, and Reducing Power of Acetone Extract of Craterellus cornucopioides.
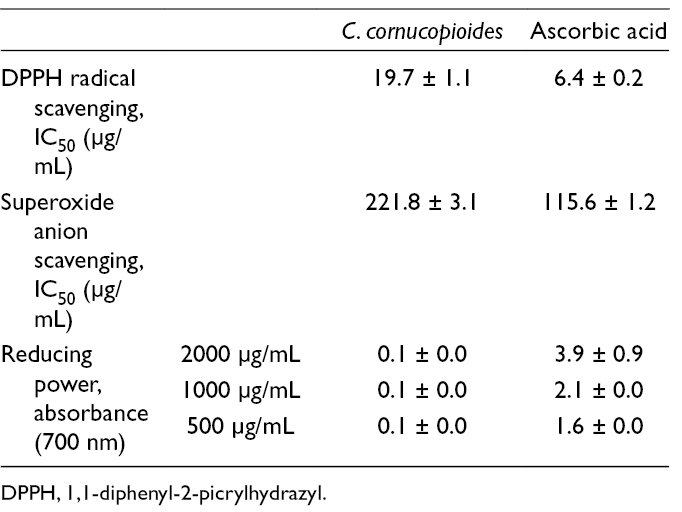
DPPH, 1,1-diphenyl-2-picrylhydrazyl.
In the literature, there are several data for the antioxidant activity of C. cornucopioides. 8,9 The studies determined the antioxidant activity for this species, but using other extraction solvents. In this study, the antioxidant activity of the selected mushroom was confirmed by acetone extract. Relatively strong antioxidant effects were previously found for polyphenolics that constitute C. cornucopioides (gallic acid, quercetin, rutin, catechin, p-coumaric acid) by many researchers 10,11 so these components are probably responsible for the detected powerful activity.
The antimicrobial activity of the mushroom extract against the test microorganisms is shown in Table 3. Extract from C. cornucopioides acted on all the microorganisms tested. The minimum inhibitory concentration (MIC) fluctuated in a range of 0.1 to 0.2 mg/mL for bacteria and 5 to 10 mg/mL for fungi. The most sensitive, among the microorganisms, were Bacillus cereus and Bacillus subtilis (MIC value was 0.1 mg/mL).
MIC of Acetone Extract of Craterellus cornucopioides.

MIC, minimum inhibitory concentration.
The antimicrobial effect of C. cornucopioides depended on the used concentration of the extract and the tested microorganisms. In these experiments, antibacterial effect was observed against both Gram-positive and Gram-negative bacteria but it should be noted that the Gram-negative bacteria were more resilient. It has been generally reported that the Gram-negative bacteria are more resistant than Gram-positive. Compared to bacteria, fungi were more resistant due to the more complex structure of the cell wall. 3 Similar to our results, numerous researchers found antimicrobial activity for C. cornucopioides. 9,12 This mushroom contains quercetin, ferulic, gallic, and p-coumaric acids, which have been shown to exhibit powerful antimicrobial activity against numerous bacteria and fungi 13,14 so such activity for C. cornucopioides was expected.
The genotoxic and antimutagenic effects of the tested extract on genomic damage in the peripheral blood lymphocytes (PBLs) of healthy donors are shown in Table 4. Results show that individual treatment of PBLs with five different concentrations of extract did not significantly affect micronuclei (MN) frequency in comparison to the untreated PBLs (P > 0.05). Pearson correlation coefficient between the extract concentrations and MN frequency was negative (r = –0.7; P = 0.0). All the tested concentrations decreased nuclear division index (NDI) values, but significantly only in the highest concentration in comparison with untreated PBLs (P < 0.03).
The Frequency of MN and NDI Values in PBLs of Healthy Donors After the Separate Treatment of Acetone Extract Obtained From Craterellus cornucopioides and Its Combined Treatments and with MMC (=0.50 µg/mL) in Vitro.

BN, binucleated;MMC, mitomycin C;MN, micronuclei;NDI, nuclear division index;PBL, peripheral blood lymphocyte.
aStatistically significant difference in NDI values between untreated cells and PBL treated with 200 µg/mL C. cornucopioides acetone extract.
bStatistically significant difference in MN frequency between the cells treated with MMC alone (positive control) and cells treated with MMC and acetone extract of C. cornucopioides in cotreatment.
Table 4 shows the results of MN frequencies and NDI values in combined treatments of extract with mitomycin C (MMC). The MMC-induced MN frequency decreased but significantly only in the highest tested concentrations (100 and 200 µg/ml). Pearson correlation coefficient showed that all tested concentrations in combined treatments with MMC reduced the MN frequency significantly in a dose-dependent manner (r = –0.9; P = 0.0). All tested concentrations of C. cornucopioides administered in combination with MMC did not change NDI values in comparison with the same in positive control cells (MMC alone) (P > 0.05).
The analysis of the distribution of MN revealed that the binucleated (BN) cells with 1MN were mostly present in all treatments (the extract separately and combined with MMC), followed by cells with 2MN and 3MN. BN cells with 4MN were present only in combined treatment with the lowest concentration (12.5 µg/mL).
In our study, the acetone extract of C. cornucopioides in the tested concentrations did not modify the frequency of MN, suggesting that the mushroom extract had no genotoxic potential on PBLs in vitro. Concurrently, the highest tested concentration (200 mg/mL) of extract significantly reduced NDI values in comparison with untreated PBLs (P < 0.05). This suggests that the extract slowed the cell kinetics. In the combined treatment with MMC, the high tested concentrations of extract (100 and 200 µg/mL) show antimutagenic effects. The antimutagenic properties of the acetone extract of C. cornucopioides can be explained by phytochemical contents. Phytochemical analysis showed that the tested extract was rich in polyphenolics such as ferulic acid and quercetin. 15,16
The data obtained by microculture tetrazolium test (MTT) are shown in Table 5. The IC50 values against HeLa A549, and LS174 cell lines were 65.5, 108.2, and 131.7 µg/mL respectively.
Growth Inhibitory Effect of Craterellus cornucopioides on HeLa, A549, LS174, and MRC5 Cell Survival.

IC50 values are expressed as mean ± SD determined from the results of MTT assay in two independent experiments.
The tested mushroom extract expressed moderate cytotoxic effect on the used cancer cells, among which HeLa cell was most sensitive. There has been very little information available about the anticancer potential of C. cornucopioides species. 4 In our study, the cytotoxic effect of C. cornucopioides on normal cell was not detected, so it is important to emphasize that this mushroom acted selectively regarding cytotoxic potential.
The present study represents the selected mushroom as a very interesting source of bioactive compounds which provide unlimited opportunities for new antioxidant, antimicrobial, antigenotoxic, and anticancer agents.
Experimental
Fungal Materials
Fungal sample of C. cornucopioides (L. ex Fr.) Pers was collected from Kragujevac, Serbia, with the following geographical coordinates: 44°00′40″ N 20°54′40″ E in September 2016. The demonstration sample is preserved in facilities of the Department of Biology and Ecology of Kragujevac, Faculty of Science.
Extraction
Finely dry ground thalli of the test mushrooms were extracted using a Soxhlet extractor. The extracts were filtered and then concentrated under reduced pressure in a rotary evaporator. The dry extracts were stored at −180°C until they were used in the tests. The extracts were dissolved in 5% dimethyl sulfoxide for the experiments. 3
HPLC Analysis
The HPLC system (Shimadzu, Kyoto, Japan) consisted of degasser DGU-20A3, analytical pumps LC-20AT, 7125 injector, and SPD-M20A diode array detector, and CBM-20A system controller. The chromatographic data were processed using LC Solution computer software (Shimadzu). Acetonitrile was used for separation. The chromatographic separations were performed using reversed-phase Luna C18 Phenomenex (Torrance, CA, USA) (250 × 4.6 mm, particle size 5 µm) column. The mobile phase consisted of acetonitrile (A) and 0.1% formic acid aqueous solution (B) using the following gradient elution program for separation: 0 to 5 min, 5% (A); 5 to 30 min, 5% to 60% (A); 30 to 32 min, 60% to 90% (A); 32 to 35 min, 90% (A), 35 to 36 min, 90% to 5% (A); 36 to 40 min, 5% (A). The column temperature was maintained at 30°C, and the flow rate was 1.0 mL/min. The samples were monitored at 254, 290, and 360 nm. A 20 µL volume of sample was injected into HPLC system.
Antioxidant Activity
Antioxidant activity was evaluated by free radical scavenging, superoxide anion radical scavenging, and reducing power. The free radical scavenging activity of lichen extracts was measured using DPPH radical according to Dorman et al’s method. 17 The Oyaizu method 18 was used to determine the reducing power. The superoxide anion radical scavenging activity was detected according to Nishikimi et al’s method. 19
Antimicrobial Activity
Antimicrobial activity was evaluated against 10 microorganisms, including Staphylococcus aureus (ATCC 25923), B. subtilis (ATCC 6633), B. cereus (ATCC 10987), Escherichia coli (ATCC 25922), Proteus mirabilis (ATCC 12453), Aspergillus niger (ATCC 16888), Candida albicans (ATCC 10259), Penicillium italicum (ATCC 10454), Mucor mucedo (ATCC 20094), and Trichoderma viride (ATCC 13233) obtained from the American Type Culture Collection (ATCC). The 96-well microtiter assay using resazurin as the indicator of cell growth 3 was employed for the determination of MIC for the active extract.
In Vitro Cytokinesis-Block Micronucleus Test
Cell culture PBLs from three healthy donors, aged between 30 and 35 years, who had not been exposed to known mutagenic agents were used in the investigation of the in vitro effect of acetone extract of C. cornucopioides by cytokinesis-block micronucleus test. This test was carried out using the method described by Fenech and Morley. 20 Whole heparinized blood (0.5 mL) was cultured in 5 mL of the complete medium for lymphocyte culture PBMax Karyotyping (Invitrogen, CA, USA) and incubated at 37°C for 72 hr. After 24 hours of incubation period, cultures were treated with 12.5, 25, 50, 100, and 200 µg/mL of mushroom extract separately to determine the genotoxic effect and in combination with MMC to determine the antimutagenic effect. Cytochalasin B (Sigma, St Louis, MO, USA) was added 44 hours after the beginning of incubation in the final concentration of 4 µg/mL. At the end of the incubation period, the cultures were centrifuged, treated with cold (+4°C) hypotonic solution (0.6% KCl), and fixed with glacial acetic acid and methanol (1:3). The fixation procedure was repeated three times. The cell suspensions were dropped onto cold and clean slides, lamp–dried, and stained with 2% Giemsa (Alfapanon, Novi Sad, Serbia). For MN frequency, 3000 BN per concentration from each donor (1000 cells) were scored. NDI is calculated using the formula NDI = (M1 +2M2 + 3M3 + 4 M4)/N, where M1 to M4 represent the number of cells with 1 to 4 nuclei and N is the total number of viable cells scored. 21
Cytotoxic Activity
Human epithelial carcinoma HeLa cells, human lung carcinoma A549 cells, and human colon carcinoma LS174 cells were obtained from ATCC (Manassas, VA, USA). All cancer cell lines were cultured as a monolayer in the RPMI 1640 nutrient medium, with 10% (inactivated at 56°C) fetal bovine serum, 3 mM of
Data Analyses
Data analyses were performed using the EXCEL and SPSS software package. All values are expressed as mean ± SD of three parallel measurements.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Grant Nos. III41010, 173032, 175011).
