Abstract
Three known polyol menthane monoterpenoids, namely, (4R)-1-p-menthen-6,8-diol (
In recent years, a huge importance is paid to the chemical and biological investigations of indigenous medicinal plants forming a treasure trove of phytoconstituents with extraordinary diverse biological activities and constituting the best source of novel therapeutically effective drugs and biochemicals useful in pharmaceutical and agrochemical industries. The extraction and isolation of targeted nontoxic and potentially antimicrobial secondary metabolites is becoming of paramount importance to fight the microbial resistance to synthetic drugs and to combat food deterioration caused by either bacterial or fungal infections. 1 North Africa is still a promising source of unexplored medicinal plants making the subject of extensive screenings for natural drugs discovery. As an example, Tunisian steppes (Northern Africa) are rich in plants with ethnopharmacological uses; it has more than 500 species of medicinal aromatic plants. 2 However, many of them remain untapped. Species belonging to Apiaceae family are flowering aromatic plants endowed with high therapeutic benefits including estrogenic, sedative, diuretic, carminative, and antispasmodic properties. 3 Daucus is one of the most popular and economically important genera belonging to family Apiaceae. In Tunisia, this genus includes 16 taxa. 4 Common carrot, Daucus carota L. s. lat. is the richest taxon in subspecies and shows about nine intraspecific taxa. 4,5 Daucus carota is commonly famous in Tunisian traditional medicine for chilblains treatments, the seeds possess carminative and diuretic properties, and they are widely used as appetite stimulants. 6 It is reported to contain numerous chemical constituents such as flavonoids, coumarins, alkaloids, and anthocyanes. 7 Besides, terpenoids are cited as the most widespread class of secondary metabolites in D. carota and other Daucus species. 8 Due to the wide variety of their biological activities, the number of research studies focused on new terpenoids isolation has grown. Within this context and as a part of our ongoing phytochemical and biological investigations of crude extracts from Tunisian medicinal plants, our interest in the discovery of new bioactive constituents prompted us to initiate, and for the first time in this study, the phytochemical and biological investigations of the dichloromethane extract from D. carota subsp. hispidus collected in Tabarka region (Northwestern of Tunisia).
Herein, this work includes the isolation and the structure elucidation of 3 polyol menthane monoterpenoids reported from this plant for the first time,
6
well-known phenolic compounds and 1 steroid. The compounds
The chemical characterization of the chloroform-soluble portion of the aerial parts of Tunisian D. carota subsp. hispidus led to isolation and identification of 3 known menthane monoterpenoids namely (4R)-1-p-menthen-6,8-diol,
1,9,10
1-p-menthen-4,7-diol,
2,11
and (1R,2R,4R)-p-menthane-1,2,4-triol,
3,12
6 known phenolics,
4-9,13,14
and β-sitosterol 3-O-glucoside (

Isolated compounds from Daucus carota subsp. hispidus.
(4R)-1-p-Menthen-6,8-diol (

Experimental circular dichroism of compounds 1 to 3.
a J (MHz).
bMultiplicity of carbons was determined by DEPT-135 and HMQC experiments.
cs, quaternary; d, methine; t, methylene; q, methyl.
dCompounds
Compound
Compound
Antimicrobial Activity Assay
Compounds
Superoxide Dismutase-Like Activity Assay
Compounds
Cytotoxic Activity Against HeLa and HSC-2 Cancer Cells
Monoterpenoids
Experimental
General Experimental Procedures
Optical rotations were recorded on a JASCO P-2300 polarimeter (Tokyo, Japan). 1H (500 MHz), and 13C NMR (125 MHz) spectra were recorded on a Bruker 500 NMR spectrometer (United States). The chemical shifts were given in δ (ppm), and the coupling constants were reported in Hz. High resolution mass spectroscopy spectra were obtained on a JEOL JMS-700 instrument (Tokyo, Japan). Circular dichroism was displayed using CD spectra: JASCO 810 spectropolarimeter. Column chromatography (CC) was carried out on normal-phase silica gel column, Silica gel 60 (Merck, 230-400 mesh; Merck, Darmstadt, Germany). Precoated silica gel plates (Merck, Kieselgel 60 F254, 0.25 mm; Merck, Darmstadt, Germany) and precoated RP-18 F254S plates (Merck, Darmstadt, Germany) were used for thin layer chromatography analysis; and detection was achieved by spraying with (1:9) H2SO4-MeOH followed by heating. High-performance liquid chromatography (HPLC) was performed on a Jasco PU-980 pump intelligent HPLC pump equipped with a Jasco UV-970 intelligent UV/VIS detector at 210 nm. A semipreparative reversed-phase column (Supelco C18 column 250 × 10 mm, 5 µm) was used for HPLC. Isolera one flash chromatography (Biotage, Suite C Charlotte, NC, United States) was used for flash chromatography and purification.
Plant Material
Aerial parts of D. carota subsp. hispidus were collected in Tabarka region (Northwestern of Tunisia) on September 2016 and identified by one of the authors (R.E.M.). A voucher specimen (AP/D-c-hsp; 00127/2016) was deposited in local herbarium at the Department of Botany and Plant Biology, Faculty of Pharmacy of Monastir, Tunisia.
Extraction and Isolation
Daucus carota subsp. hispidus aerial parts (2200 g) were air-dried under room temperature until constant weight to be coarsely powdered. The pulverized plant sample was macerated exhaustively in methanol until complete extraction to afford 140 g of crude methanol extract obtained after filtration and solvent evaporation under vacuum. The methanol extract thus obtained was suspended in water and partitioned between dichloromethane, ethyl acetate, and butanol to yield 78, 4, and 21 g of crude extracts successively. The dichloromethane extract was subjected to first chromatographic separation over a normal-phase silica gel stationary phase eluted with n-hexane gradually increased with EtOAc and MeOH to afford 20 fractions (F1-F20). Fraction F18 (1.5 g) was further chromatographed over a normal-phase silica gel CC eluted with a solvent gradient of increasing polarity (CH2Cl2/MeOH) starting from pure CH2Cl2 to yield 10 subfractions (G1-G10). The subfraction G8 (100 mg) was subjected to RP-18 HPLC (MeOH-H2O, 5.5:4.5) and afforded compounds
Antimicrobial Activity Assay
The isolated monoterpenoids
Superoxide Dismutase-Like Activity Assay
The monoterpenoids
Cytotoxicity Assay
Cell Culture
HSC-2 and HeLa were obtained from Japanese Collection of Research Bioresources Cell Bank. Both cells were cultured in minimum essential medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 g/mL streptomycin at 37°C in a humidified atmosphere of 5% CO2.
Cell Viability Assay
Cytotoxic activity of the isolated monoterpenoids
Statistical Analysis
Statistical analysis was conducted using one-way analysis of variance. P < 0.05 was considered to indicate the statistical significance.
(4R)-1-p-Menthen-6,8-Diol (1)
Rf: 0.43 (n-hexane-EtOAc, 3:2).
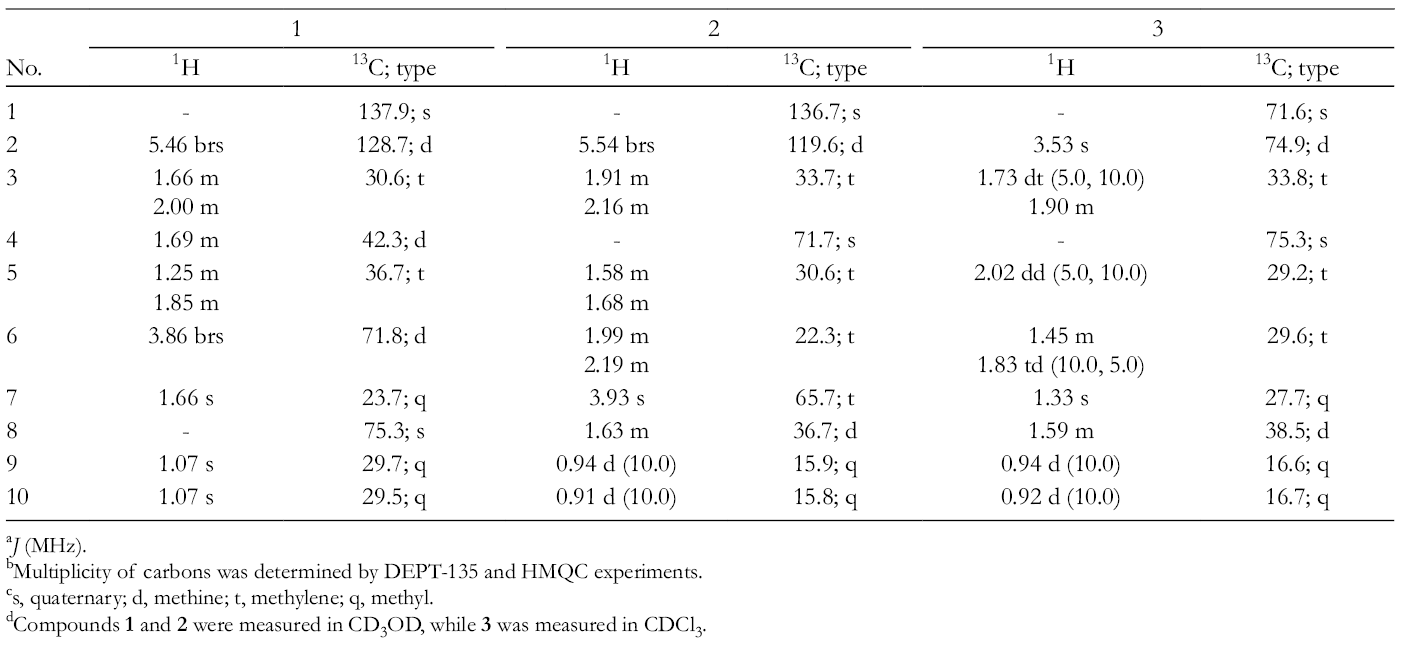
1H NMR (500 MHz) and 13C NMR (125 MHz, CD3OD): Table 1
CI-MS (EI): m/z (%) =170 [M] (10), 152 (85), 109 (100), 79 (75), 59 (93), 43 (38).
HRCI-MS: m/z 170.1316 [M], [calcd for C10H18O2: 170.1307].
4R-1-p-Menthen-4,7-Diol (2)
Rf: 0.46 (n-hexane-EtOAc, 3:2).
1H NMR (500 MHz) and 13C NMR (125 MHz, CD3OD): Table 1
HRCI-MS (CI, positive mode): m/z (%) =169 [M-1] (15), 152 (80), 135 (88), 127 (57), 109 (52), 81 (70), 79 (100), 43 (28), HRCI-MS (CI, positive mode): m/z 170.1306 [M], (calcd for C10H18O2: 170.1307).
(1R,2R,4R)-p-Menthane-1,2,4-Triol (3)
Rf: 0.58 (n-hexane-EtOAc, 3:2).
1H NMR (500 MHz) and 13C NMR (125 MHz, CD3OD): Table 1
CI-MS (CI, positive mode): m/z (%) =188 [M] (11), 171 (50), 153 (100), 135 (100), 127 (100), 111 (100), 99 (100), 81 (82), 71(92), 43 (100).
HRCI-MS: m/z 188.1411[M], [calcd for C10H20O3: 188.1412]
Supplemental Material
Supplementary material - Supplemental material for Chemical Constituents of the Aerial Parts of Daucus carota subsp. hispidus Growing in Tunisia
Supplemental material, Supplementary material, for Chemical Constituents of the Aerial Parts of Daucus carota subsp. hispidus Growing in Tunisia by Saoussen Hammami, Abdelsamed I. Elshamy, Ridha El Mokni, Ali Snene, Kanako Iseki, Hatem Dhaouadi, Yasuko Okamoto, Midori Suenaga, Masaaki Noji, Akemi Umeyama, and Yoshinori Asakawa in Natural Product Communications
Footnotes
Acknowledgments
Dr Elshamy gratefully acknowledges the Takeda Science Foundation, Japan for the financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was also supported by Tokushima Bunri University, Japan, University of Monastir, Tunisia, and National Research Centre Centre, Egypt.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.