Abstract
Background:
Continuous glucose monitoring (CGM) devices are increasingly used in critical and non-critical care hospital units. The efficacy of CGM in assessing glucose control in adults with diabetic ketoacidosis (DKA) is unknown.
Methods:
This single-center pilot study compared glycemic control by real-time CGM (Dexcom G6), capillary point-of-care (POC), and basic metabolic panel (BMP) during intravenous (IV) insulin treatment and after the resolution of DKA. We compared the mean absolute relative difference (MARD), median absolute relative difference (ARD) glucose values, and Diabetes Technology Society (DTS) Error Grid analyses.
Results:
We recruited 52 patients (49 ± 19 years, admission glucose: 503 ± 239.4 mg/dL) with type 1 diabetes (n = 24) and type 2 diabetes (n = 28). Compared with POC testing, the MARD was 17.4% ± 13.2%, and the median ARD was 14.2% (interquartile range [IQR]: 6.4, 28) during the initial IV insulin period and 19.8% ± 18.7% and 14.3% (7, 26.2) after DKA resolution. The DTS Error Grid analysis showed that 100% of values during the IV insulin treatment and 95% after the DKA resolution were in zones A+B. Compared with BMP glucose values, the MARD and median ARD were 18.5% ± 19.1% and 12.2% (5.4, 23.8) during the IV insulin treatment and 22.5% ± 24.7% and 15.1% (6.6, 27.6) after DKA resolution.
Conclusion:
This is the first report on the use of real-time CGM in adults with DKA. Our study indicates that CGM technology is a reliable tool for hospital use during acute insulin treatment and after the resolution of DKA. Future multicentre randomized studies are needed to determine the benefits of real-time CGM in facilitating diabetes care in hospitalized patients with hyperglycemic crises.
Introduction
Diabetic ketoacidosis (DKA) is the most common hyperglycemic emergency in patients with type 1 and type 2 diabetes mellitus. 1 Most patients with DKA are treated with continuous intravenous or frequent subcutaneous insulin administration.1,2 Close monitoring of glucose levels is essential for assessing therapeutic response and preventing hypoglycemia during the initial treatment and after the resolution of DKA.3,4 Bedside capillary point-of-care (POC) glucose monitoring is the standard method for evaluating glycemic control in the hospital. 5 National guidelines recommend POC testing every one to two hours during IV insulin treatment until DKA resolves and then four times daily as patients transition to maintenance insulin therapy.2,5 However, POC testing can be uncomfortable, increases nursing workload, provides a limited evaluation of daily glycemic variations, and may miss nocturnal hypoglycemia during a hospital stay.6,7
Multiple randomized and real-world studies have shown that continuous glucose monitoring (CGM) provides a more comprehensive glycemic profile than POC testing in outpatient and inpatient settings. 8 Recent randomized controlled trials demonstrate that real-time CGM improves glycemic control and reduces the frequency and duration of hypoglycemia in hospitalized patients compared with POC glucose testing.9,10 Furthermore, real-time CGM is safe and effective for guiding insulin administration in general medicine and surgery services9,10 and has shown reliable accuracy in insulin-treated patients in critical care units. Boeder and colleagues conducted a retrospective study using Dexcom G6 CGM for patients receiving continuous insulin infusion in critically ill patients with COVID-19, reporting a median absolute relative difference (ARD) between 11.0% and 16.2%. 11 Finn et al 12 reported in a real-world study involving critically ill patients a mean absolute relative difference (MARD) of 17.4% when comparing CGM with the POC glucose testing and a MARD of 12.4% when CGM was compared with laboratory glucose levels. Recently, Davis et al 13 reported a MARD of 12.8%, and median ARD was 10.1% (interquartile range [IQR]: 4.6, 17.6). However, the efficacy of CGM in assessing glucose control in adults with DKA has not been reported.
While frequent glucose monitoring is essential for managing DKA, data on the accuracy of real-time CGM in adult patients with DKA is limited. The hesitancy to use CGM in DKA arises from the rapid changes in glucose levels during acute treatment of DKA, which may affect the accuracy of real-time CGM. However, during the COVID-19 pandemic, our hospital conducted a pilot study to assess the safety of CGM combined with remote glucose monitoring in patients with mild to moderate DKA. This approach aimed to reduce reliance on hourly POC glucose testing, minimize direct patient contact, and lower the risk of cross-infection, enabling safer and more frequent glucose monitoring.14-17 Here, we report our experience using real-time CGM to manage adult patients with DKA.
Methods
Study Design
This single-center pilot study included 52 adults over 18 admitted for DKA at Columbia University Irving Medical Center—New York Presbyterian-Columbia University Irving Medical Center and The Allen Pavilion Hospital. During the COVID-19 pandemic, from May 2020 to January 2021, patients were treated in the intensive care unit (ICU) at The Allen Pavilion Hospital; after that, they were treated in the step-down unit at Columbia University Irving Medical Center. The primary team diagnosed DKA per the American Diabetes Association diagnostic criteria. 18 Participants were referred by primary providers or identified through electronic medical records. Once a potential candidate was identified in the emergency room, the primary provider and the individual were approached to request permission for inclusion in the study. We excluded hemodynamically unstable patients, those requiring vasopressors or mechanical ventilation, those with impaired mental status, those pregnant, those with end-stage kidney disease, or those undergoing dialysis.
This project originated from an urgent clinical need to reduce the frequency of fingerstick POC glucose testing, minimize nurses’ exposure to SARS-CoV-2, conserve personal protective equipment (PPE), and save ICU beds, which were a scarce resource during the peak of the COVID-19 pandemic in 2020-2021. In addition, the use of CGM aimed to decrease POC testing frequency, allowing patients to be treated in the step-down units and preventing ICU admissions. Our hospital’s DKA protocol mandates hourly POC testing during IV insulin infusions, and patients requiring hourly glucose testing need to be treated in the ICU. We explored whether the use of CGM and hourly POC glucose testing could be avoided in hemodynamically stable DKA patients, allowing patients to be managed in step-down units. This approach allowed studying the accuracy of CGM in real-world settings during intravenous insulin treatment and after the resolution of DKA.
Continuous glucose monitoring devices were placed on the upper arm or abdomen based on patient preference. Verbal consent was obtained during the pandemic, while written consent was secured after 2023. The Columbia University Institutional Review Board (AAAT7802) approved and registered the study protocol at clinicaltrials.gov (NCT05439928).
Patients with DKA were treated with an intravenous (IV) insulin infusion following our hospital protocol. The primary care team ordered intravenous insulin, fluids, and electrolyte therapy while managing the underlying cause of DKA. After DKA resolution, patients were transitioned to a maintenance basal-bolus insulin regimen. Insulin adjustments were based on CGM values, with POC glucose testing performed at the provider’s discretion and if values were below 100 mg/dL or above 350 mg/dL. Real-time DEXCOM CGM does not require routine calibrations in the outpatient setting. Therefore, we did not perform calibrations to monitor glucose levels in our cohort.
The diabetes consultation team trained the nursing staff on CGM. The training covered how CGM works, how to access glucose data on smartphones, and the relevant workflow processes. The team included nurse practitioners from the diabetes service and charge nurses trained in sensor insertion, data transmission, and setting up smartphone applications to view glucose levels remotely. Glucose data was displayed on smartphones acting as data receivers. All smartphones were designated study devices to protect and ensure patient privacy. One of the phones, referred as the “patient phone,” acted as a receiver and had the Dexcom G6 application to receive and display glucose data. Two additional phones, known as the “provider phone” and “nurse phone,” had the Dexcom Follow application installed and were carried by the provider and nurses taking care of the patient. The information on the application was de-identified to safeguard the patient’s privacy. Audible alerts were triggered on all smartphone devices when CGM glucose levels fell below 100 mg/dL or exceeded 350 mg/dL in addition to DEXCOM’s built-in Urgent Low alert, which notifies users when the glucose level drops below 55 mg/dL. Individual patient accounts were created on the manufacturer’s cloud-based platform for de-identified information and retrospective data review. The hospital’s Wi-Fi was used for data transmission. Glucose data was collected by providers, nurses, and study team members.
Primary demographic and inpatient clinical data were obtained from the electronic health record, and CGM sensors were placed on the abdomen or arm. POC BG values were measured using the Roche ACCU-CHEK Inform II glucose meter. We calculated the mean and median ARD by matching CGM and POC glucose tests with plasma glucose from the basic metabolic panel (BMP) during the acute phase when DKA was treated with IV insulin infusion and after DKA resolution. A total of 311 matched pairs of CGM-POC glucose values and BMP glucose were analyzed. The CGM-POC glucose pairs were matched by time, using the closest sensor glucose value within a five-minute window of the POC glucose measurement. Matched pair values were excluded outside the CGM reading range (BG <40 mg/dL or >400 mg/dL). Mean absolute relative difference and median ARD were analyzed during the first 12 hours of hospitalization, between 12 and 24 hours, and beyond 24 hours.
Study Endpoints
The primary endpoints were the MARD and the ARD during the acute insulin infusion treatment phase and after the resolution of DKA. We also conducted Diabetes Technology Society (DTS) Error Grid analysis during acute treatment and after the resolution of DKA.
Statistical Analysis
Mean absolute relative difference and ARD were calculated as the average relative difference between CGM and POC. The CGM and BMP glucose-matched pairs were expressed as a percentage. Statistical methods for analyzing CGM performance followed recommended approach by the DTS. 19 We evaluated MARD and median ARD during the acute IV insulin infusion phase until DKA resolution and after transitioning to subcutaneous (SQ) insulin therapy. Data are also segmented into the first 12 hours of hospitalization, 12 to 24, and beyond 24 hours. Continuous variables are presented as mean (± SD), and categorical variables are shown as count (percentage). The DTS error grid accuracy matrix was utilized for error grid analysis. Additional statistical analyses were conducted using SAS 9.4 statistical software (SAS Institute, Cary, North Carolina).
Results
The clinical characteristics of the study population are summarized in Table 1. We recruited 52 participants: 24 with type 1 diabetes and 28 with type 2 diabetes. The mean age of the patients was 49 ± 19 years, with an average body mass index of 27.0 ± 6.8 kg/m2. Blood glucose levels averaged 503 ± 239.4 mg/dL, while HbA1c was 11.57% ± 2.8%. Most patients were admitted with mild to moderate DKA with a mean bicarbonate level of 13.2 ± 4.4 mmol/L, an anion gap of 27.1 ± 5.5 mmol/L, and a beta-hydroxybutyrate level of 6.0 ± 2.3 mmol/L. Patient characteristics did not significantly differ between those with type 1 and type 2 diabetes, except that patients with type 2 diabetes were older. The average hospital length of stay was 7.3 ± 9.3 days, and the average time to resolution of DKA was 17.2 ± 13.2 hours (Table 1).
Characteristics of Study Population and Laboratory Values on Admission and After Resolution of DKA, Categorized by Diabetes Type.

Variables are presented as mean ± standard deviation (SD).
Abbreviations: BMI, body mass index; DKA, diabetic ketoacidosis.
During the acute IV insulin treatment, the calculated MARD was 17.4% ± 13.2%, and the median absolute relative difference (ARD) was 14.2% (6.4, 28.0) for CGM values matched with POC glucose levels. After the resolution of DKA, the MARD was 19.8% ± 18.7%, and the median ARD was 14.3% (7, 26.2) (Table 2). When CGM values were compared with BMP glucose, the MARD and median ARD were 18.5% ± 19.1% and 12.2% (5.4, 23.8) during IV insulin treatment and 22.5% ± 24.7% and 15.1% (6.6, 27.6) after DKA resolution. The MARD and median ARD were higher for patients with type 1 diabetes than those with type 2 diabetes when CGM glucose levels were matched with POC glucose during the acute treatment of DKA and after DKA resolution. Individuals with type 1 diabetes had a MARD of 19.3% ± 12.8% and a median ARD of 18.3% (9.1, 27.8). Those with type 2 diabetes had a MARD of 15.4% ± 13.4% and a median ARD of 12.0% (3.7, 28.5). A similar trend was observed when comparing MARD and median ARDs with CGM glucose levels matched to plasma glucose from the BMP (Table 2).
Comparison of CGM With Laboratory BMP and POC During IV Treatment and After Resolution of DKA, Categorized by Type of Diabetes.

Abbreviations: CGM, continuous glucose monitor; POC, point-of-care; ARD, absolute relative difference; IV, intravenous; IQR, interquartile range; DKA, diabetic ketoacidosis; BMP, basal metabolic panel.
The DTS Error Grid analysis showed high clinical reliability, with 100% of all matched CGM/POC pairs within zones A and B during IV insulin treatment (Figure 1a) and 95% following DKA resolution (Figure 1b). For CGM/BMP-matched data, 96% of pairs remained within zones A and B during IV insulin treatment and 94% after DKA resolution (Supplemental Figure 1a, Supplemental Figure 1b).
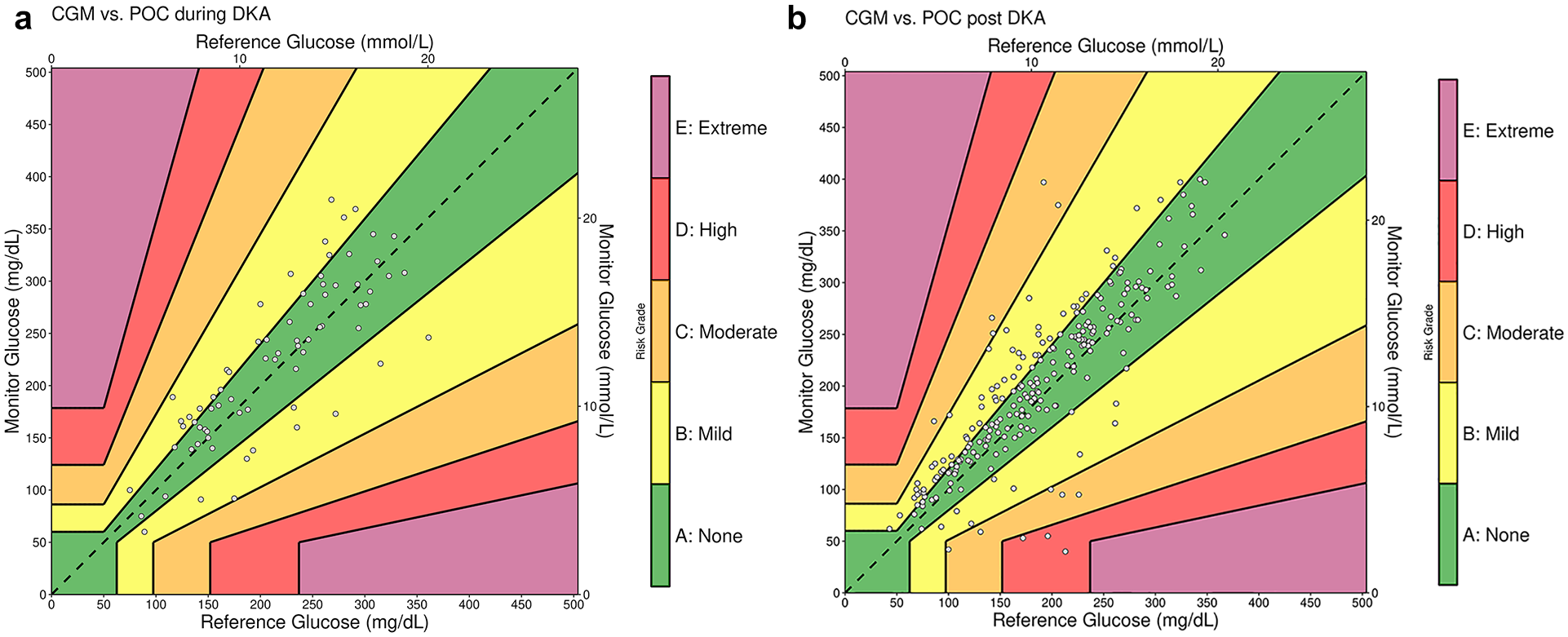
DTS error grid of CGM and POC glucose-matched pairs during intravenous insulin therapy (a) and after resolution of DKA (b).
During entire hospitalization, 96% of CGM/POC-matched data fell in DTS error grid zones A and B (zone A: 63%, zone B: 33%, zone C: 3.2%, zone D: 1%, zone E: 0%) (Supplemental Figure 2a) and, 95% of CGM/BMP-matched pairs remained within zones A and B (zone A: 63.5%, zone B: 31.1%, zone C: 4.5.2%, zone D: 0.8%, zone E: 0%) (Supplemental Figure 2b).
Discussion
This is the first report on the use of real-time (RT) CGM in adult patients admitted with DKA. Our analysis shows that Dexcom G6 CGM has overall good accuracy, with a MARD of 17.4% and a median ARD of 14.2% (6.4, 28.0). In addition, 100% of CGM-matched values were within the DTS Error Grid zones A and B compared with standard POC glucose monitoring during DKA treatment. After DKA resolution, the MARD was 19.8%, and the median ARD was 14.3% (7, 26.2). A similar accuracy was observed when comparing MARD and median ARDs with CGM glucose levels matched to plasma glucose from the BMP (Table 2). Moreover, the DTS Error Grid analysis demonstrated high clinical reliability, with 96% of all matched CGM/BMP-matched pairs within zones A and B during intravenous insulin treatment and 94% after DKA resolution. These results indicate that CGM technology is a reliable tool for hospital use during acute insulin treatment and after the resolution of DKA.
Patients with type 1 diabetes exhibited a higher MARD compared with those with type 2 diabetes. This finding is expected because individuals with type 1 diabetes often experience significant glucose variability. In addition, both MARD and the median ARD were higher during the first 12 hours of sensor initiation than during more extended usage periods (Supplemental Table S1, Supplemental Table S2), influenced by the rapid rate of change in glycemia. 20 Although MARD was higher with rapid change in glycemia and during the first few hours following CGM initiation, it remained within a safe range for insulin adjustments.
Previous studies evaluating RT-CGM systems in the ICU, general medicine, and surgery services have reported MARD ranging from 11.1% to 17.4% when compared with POC glucose values and higher MARD observed in critically ill patients. 11 -13,21 Most CGM studies have reported a MARD below 10% in ambulatory settings. In simulation studies, a MARD above 10% has been associated with spending more time outside of the target range, while a MARD below 10% does not appear to affect the time within the target. This finding has resulted in the non-adjunctive indication for CGMs, allowing insulin dosing decisions to be made solely based on CGM glucose levels, including insulin dosing via automated insulin delivery systems. 22 However, the most recent consensus statement for CGM metrics for inpatient use reports that additional studies are needed to determine a realistic and acceptable target MARD in the inpatient setting. 23
Close monitoring of glucose levels is essential for achieving glycemic control and preventing adverse outcomes in patients admitted with DKA. The standard recommendation is to monitor glucose through POC testing every one to two hours. This approach can be time-consuming, creates a high workload for nurses, and causes patient discomfort. The use of CGM overcomes these limitations. Our study provides new information on the acute management of patients with DKA undergoing IV treatment after transitioning to a maintenance insulin regimen. The CGM glucose limit is 400 mg/dL; thus, monitoring via RT-CGM when the glucose level is <400 mg/dL is a promising approach for inpatient glucose monitoring, helping to reduce the care burden associated with frequent bedside POC glucose testing.
Our study has limitations. This was a real-world study aimed at reducing the frequency of POC glucose testing during the peak of the COVID-19 pandemic. Primary care providers and nurses conducted POC glucose testing at their discretion, allowing us to examine the accuracy of RT-CGM compared with POC testing. However, this resulted in limited POC/CGM glucose pairs and BMP glucose/CGM pairs. The POC testing was performed only when providers suspected sensor inaccuracy. Therefore, the MARD and median ARD could have been higher in this cohort than in previous CGM studies. Future studies evaluating RT-CGM accuracy should include more frequent POC testing to compare POC and CGM glucose levels and incorporate novel features of RT-CGM technology, including trend arrows and information on the speed of glucose levels. Furthermore, dual glucose and ketone continuous monitoring devices should be studied during the treatment of DKA when they become available.
Conclusions
Our study indicates that CGM technology is a reliable and promising tool for monitoring glucose levels in hospitalized patients during acute insulin treatment and after the resolution of DKA. Real-time CGM with remote monitoring significantly reduces the need for frequent glucose testing, allowing DKA management outside the ICU and easing the burden on health care resources. Future multicenter, randomized studies are needed to explore RT-CGM’s role in improving diabetes care for hospitalized patients with hyperglycemic crises.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251316887 – Supplemental material for Continuous Glucose Monitoring in Hospitalized Adults With Diabetic Ketoacidosis: A Prospective Open-Label Pilot Study
Supplemental material, sj-docx-1-dst-10.1177_19322968251316887 for Continuous Glucose Monitoring in Hospitalized Adults With Diabetic Ketoacidosis: A Prospective Open-Label Pilot Study by Magdalena M. Bogun, Chunhui Wang, Paul A. Kurlansky, Nur Bedeir and Guillermo E. Umpierrez in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
ARD, absolute relative difference; BMI, body mass index; BMP, basic metabolic panel; CGM, continuous glucose monitoring; DKA, diabetic ketoacidosis; DTS, Diabetes Technology Society; IV, intravenous; MARD, mean absolute relative difference; POC, point of care; PPE, personal protective equipment.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.M.B. has received research funding and has been a paid consultant for Dexcom, Inc. G.E.U. has received research funding (to Emory University) from Dexcom, Inc., Abbot, and Bayer; and participated on advisory board/consultant for Dexcom and GlyCare. C.W., N.B., and P.A.K. have no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Dexcom Inc. and NIH, NIDDK P30DK063608.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
