Abstract
Aim:
We added items relevant to continuous glucose monitoring (CGM) to the Diabetes Family Conflict Scale (DFC), Diabetes Family Responsibility Questionnaire (DFR), and Blood Glucose Monitoring Communication Questionnaire (GMC) and evaluated the psychometric properties of the updated surveys.
Research Design and Methods:
Youth with type 1 diabetes who recently started CGM and their parents completed the updated surveys and additional psychosocial surveys. Medical data were collected from self-reports and review of the medical record.
Results:
Youth (N = 114, 49% adolescent girls) were aged 13.3 ± 2.7 years and had mean glycated hemoglobin (HbA1c) 7.9 ± 0.9%; 87% of them used pump therapy. The updated surveys demonstrated high internal consistency (DFC youth: α = .91, parent: α = .81; DFR youth: α = .88, parent: α = .93; and GMC youth: α = .88, parent: α = .86). Higher youth and parent DFC scores (more diabetes-specific family conflict) and GMC scores (more negative affect related to glucose monitoring) were associated with more youth and parent depressive symptoms (r = 0.28-0.60, P ≤ .003), more diabetes burden (r = 0.31-0.71, P ≤ .0009), more state anxiety (r = 0.24 to r = 0.46, P ≤ .01), and lower youth quality of life (r = −0.29 to −0.50, P ≤ .002). Higher youth and parent DFR scores (more parent involvement in diabetes management) were associated with younger youth age (youth: r = −0.76, P < .0001; parent: r = −0.81, P < .0001) and more frequent blood glucose monitoring (youth: r = 0.27, P = .003; parent: r = 0.35, P = .0002).
Conclusions:
The updated DFC, DFR, and GMC surveys maintain good psychometric properties. The addition of CGM items expands the relevance of these surveys for youth with type 1 diabetes who are using CGM and other diabetes technologies.
Introduction
Treatment for type 1 diabetes (T1D) has changed rapidly in recent years. In 2016, the US Food and Drug Administration (FDA) approved the use of continuous glucose monitoring (CGM) for non-adjunctive use. As a result, youth with diabetes could use CGM values to make diabetes treatment decisions (eg, insulin dosing) without also checking finger-stick glucose values. In the same year, the FDA approved the first hybrid closed-loop insulin delivery system. Such advances have drastically changed daily diabetes management for persons with T1D. 1 These advances in diabetes technology require a corresponding update to the tools used to measure health-related outcomes, including patient-reported outcome (PRO) measures.
Patient-reported outcomes are the report of a person’s health condition from the perspective of the individual or proxy (eg, parent, caregiver). 2 Assessment of PROs is becoming an integral part of routine care and clinical research because it allows clinicians to have a deeper understanding of the youth and their family’s experience with T1D.3,4 In addition, it helps clinicians understand the impact of a new treatment, for example, CGM, on the youth, tailor treatment plans, and identify individuals who need additional support.4,5 In clinical trials, PROs can be used to measure the impact of an intervention on participants’ health status. Numerous studies have demonstrated the importance of PROs, such as diabetes-related distress, fear of hypoglycemia, negative affect around blood glucose monitoring, disordered eating behaviors, and diabetes-specific family conflict, on diabetes management behaviors and glycemic control.6 -10
As CGM uptake and use have significantly increased in recent years, 11 it is important to update PRO measures to include questions about CGM. However, many surveys assessing PROs, such as diabetes-specific family conflict, diabetes family responsibility, and negative affect around blood glucose monitoring, were developed before the approval of CGM for non-adjunctive use. CGM-specific surveys have been developed, but these do not apply to people who are not using CGM. In addition, updated diabetes-specific psychosocial measures may help identify youth who are at risk for CGM discontinuation or need additional support.
To allow measurement of the same constructs in both youth using CGM and youth not using CGM, we updated several diabetes-specific psychosocial measures for youth and parents to include questions about CGM. Specifically, we updated the Diabetes Family Conflict Scale (DFC), the Diabetes Family Responsibility Questionnaire (DFR), and the Blood Glucose Monitoring Communication Questionnaire (GMC). These surveys measure diabetes-specific family conflict, parent involvement in diabetes management, and negative affect related to glucose levels, respectively.
Numerous studies have shown that higher conflict has been associated with poorer glycemic control.6,12,13 Continuous glucose monitoring has the potential to increase diabetes-specific family conflict. For example, sharing CGM readings with parents may result in more questions from the parents about the youth’s CGM readings. 14 Studies have shown that caregiver involvement has been associated with improved diabetes management and glycemic outcomes.15 -17 On the contrary, CGM can foster and increase independence. 14 As a result, there could disagreement about responsibility between caregiver and youth around certain CGM tasks. Finally, more negative affect is associated with poorer glycemic control.18,19
These three measures were included in a clinical trial in which all participants would use CGM for two years. Therefore, it was a timely opportunity to update these surveys by adding CGM-specific items. These surveys have been widely used and measure important PROs related to family functioning and diabetes management, and the addition of CGM-specific items will increase their versatility. The aim of our study is to evaluate the psychometric properties of the updated surveys.
Participants and Methods
Participants were 114 youth with T1D and their caregivers who were enrolled into a two-year randomized clinical trial that evaluated the effectiveness of a family-focused behavioral intervention for CGM use compared with standard CGM care. During the study, participants used the most recent FDA-approved DexCom (San Diego, CA, USA) CGM system, either Seven Plus or G4 Platinum. The study predated the US FDA labeling for non-adjunctive CGM use. Therefore, participants used blood glucose results for insulin dosing as well as CGM calibration.
The inclusion criteria included ages 8 to 17 years, diabetes duration ≥1 year, glycated hemoglobin (HbA1c) 6.5% to 10%, daily insulin dose ≥0.5 U/kg, blood glucose monitoring frequency ≥4 times per day, and no consistent CGM use in the 6 months prior to entering the study. For the validation of the updated surveys, we used data from the six-month study visit to capture data after all participants had at least several months of experience using CGM. The protocol was approved by the institutional review board. Parents provided consent and youth provided assent before the start of any study procedures.
The demographic and biomedical data were collected by chart review and interview. The CGM data were downloaded at the time of each visit, capturing 4 weeks of data preceding the visit. Youth and parents independently completed self-reported surveys across multiple domains described in the following paragraphs. A pediatric multidisciplinary team added CGM items to the DFC, DFR, and GMC based on clinical experience and literature review. The CGM-specific items were created by the members of the multidisciplinary team having many years of clinical and research experience in T1D. The items were reviewed by additional staff members and individuals with T1D.
Measures
Diabetes Family Conflict Scale
The validated DFC measures diabetes-specific conflict in families for youth and parents. 6 It consists of 19 items about direct and indirect diabetes management tasks. We added nine CGM-specific items to the survey (eg, inserting the CGM sensor, calibrating the CGM sensor, and charging the CGM sensor). Both parent and youth DFC surveys are rated on a three-point Likert scale (1 = never argue, 2 = sometimes argue, and 3 = always argue). The total score can range from 0 to 100 with higher scores indicating more conflict.
Diabetes Family Responsibility Questionnaire
The DFR is a previously validated survey assessing parental involvement in diabetes management. 15 The original parent and youth versions each have a total of 19 items. We added seven CGM-specific items to the original survey (eg, inserting the CGM sensor, responding to CGM alarms, and downloading CGM data). Items on the DFR are rated on a three-point Likert scale (1 = child takes responsibility for this almost all of the time, 2 = parents and child share responsibility for this about equally, and 3 = parents take responsibility for this almost all of the time). The total score can range from 0 to 100, with higher scores indicating more parental responsibility.
Glucose Monitoring Communication (GMC) Questionnaire
The original measure is a validated eight-item survey that assesses negative affect related to blood glucose monitoring for youth and parents. 9 The GMC responses are on a three-point Likert scale (1 = almost never, 2 = sometimes, 3 = almost always). We added seven CGM-specific items (eg, worry when CGM shows levels moving down quickly, upset when looking at CGM data and there are numbers above target range). Total scores can range from 0 to 100, with higher scores representing greater negative affect related to glucose monitoring.
State-Trait Anxiety Inventory for Children (STAIC) and State-Trait Anxiety Inventory (STAI)
The STAIC and STAI are previously validated surveys that assess self-reported anxiety in youth (STAIC) 20 and adults (STAI). 21 The STAIC and STAI evaluate both state anxiety (how you feel right now, 20 items) and trait anxiety (how you generally feel, 20 items). Responses on the STAIC are on a three-point Likert scale and responses on the STAI are on a four-point Likert scale. Total scores can range from 20 to 80, with higher scores indicating greater state or trait anxiety.
Center for Epidemiologic Studies Depression Scale for Children (CES-DC) and Center for Epidemiologic Studies Depression Scale (CES-D)
The CES-DC 22 was completed by youth and the CES-D 23 was completed by parents. The CES-DC and CES-D are 20-item validated measures of depressive symptoms assessing how often in the past week youth and parents experienced certain symptoms, using a four-point Likert scale. 23 Total scores can range from 0 to 60, with higher scores indicating more depressive symptoms. A total score of 15 or higher is considered a positive screen in youth and a total score of 16 or higher is considered a positive screen in adults.
Problem Areas in Diabetes Pediatric (PAID-Peds) and Parent Revised (PAID-PR)
The 20-item PAID-Peds 24 and the 18-item PAID-PR 25 assess perceived burden related to current problems in diabetes management using a five-point Likert scale ranging from agree to disagree. Total scores can range from 0 to 100, with higher scores indicating greater burden.
Pediatric Quality of Life Inventory 4.0 Generic Core Scales (PedsQL)
The PedsQL is a previously validated survey that measures youth and parent perceptions of the youth’s health-related quality of life. 26 It comprises 23 items, including physical, emotional, social, and school functioning. Response options are on a five-point Likert scale (0 = never a problem to 4 = almost always a problem). Total scores can range from 0 to 100, with higher scores indicating higher perceived health-related quality of life.
Diabetes Management Questionnaire (DMQ)
The DMQ is a previously validated survey that measures adherence to diabetes management by youth report and parent report. 27 The survey consists of 20 items and response options are on a five-point Likert scale (1 = almost never to 5 = almost always). Total scores can range from 0 to 100, with higher scores indicating greater adherence.
Statistical Analysis
Statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, NC, USA). Preliminary data analysis was performed to assess CGM device type (Dexcom G4 vs Seven Plus) and intervention group (control vs intervention) differences. There were no significant differences in the updated survey measures by CGM device type or intervention group, allowing for the assessment of survey performance in the entire sample.
Descriptive statistics for youth and parent characteristics are presented as Mean ± SD for continuous variables and in percentages for categorical variables. The updated surveys underwent statistical analysis to evaluate their psychometric properties. Sociodemographic and diabetes-specific characteristics as well as other self-reported measures were included in the assessment of the surveys’ validity and reliability. Cronbach’s alpha provided internal consistency. In evaluation of convergent validity, each updated survey was assessed independently against appropriate measures using Pearson correlations. Predictive validity of the surveys was assessed against future HbA1c, CGM percentage time in range (TIR), and CGM wear 3 months later. P values of <.05 were deemed statistically significant.
Results
Participant Characteristics
The sample characteristics are presented in Table 1. In brief, youth represented a relatively high SES, with more than nine out of 10 youth coming from two-parent households, and most youth had a parent with a college education or higher. The overwhelming majority of youth received intensive insulin therapy. Youth had a mean ± SD diabetes duration of 6.7 ± 3.6 years with an HbA1c of 7.9% ± 0.9% (63 ± 10 mmol/mol), which is above the target recommended by the American Diabetes Association (ADA). 28 The average CGM percentage TIR (70-180 mg/dL, 3.9-10.0 mM) was 47%.
Participant Characteristics.

Abbreviations: CGM, continuous glucose monitoring; HbA1c, glycated hemoglobin.
Updated Diabetes Family Conflict Scale
We removed nine of the original 19 items due to outdated diabetes management (eg, logging blood sugar results), low response variability (eg, making doctor appointments), or low item-to-total correlation on both youth and parent surveys (eg, changes in health). We revised one of the original items to make it more applicable to current treatment. We added eight CGM items but removed three due to outdated features (eg, calibrating CGM sensors, charging CGM receiver). The final updated survey has 15 items.
The updated DFC survey demonstrated high internal consistency (youth, α = .91; parent, α = .81). In addition, item-to-total correlations were mostly in the range of 0.40 to 0.80 (Table 2). One item in the youth survey and four items in the parent survey had item-to-total correlations <0.40. These items were kept because of their clinical relevance and for symmetry between the youth and parent surveys.
Internal Consistency for Updated Diabetes Family Conflict Scale: Item-to-Total Correlations and Cronbach’s Alpha.
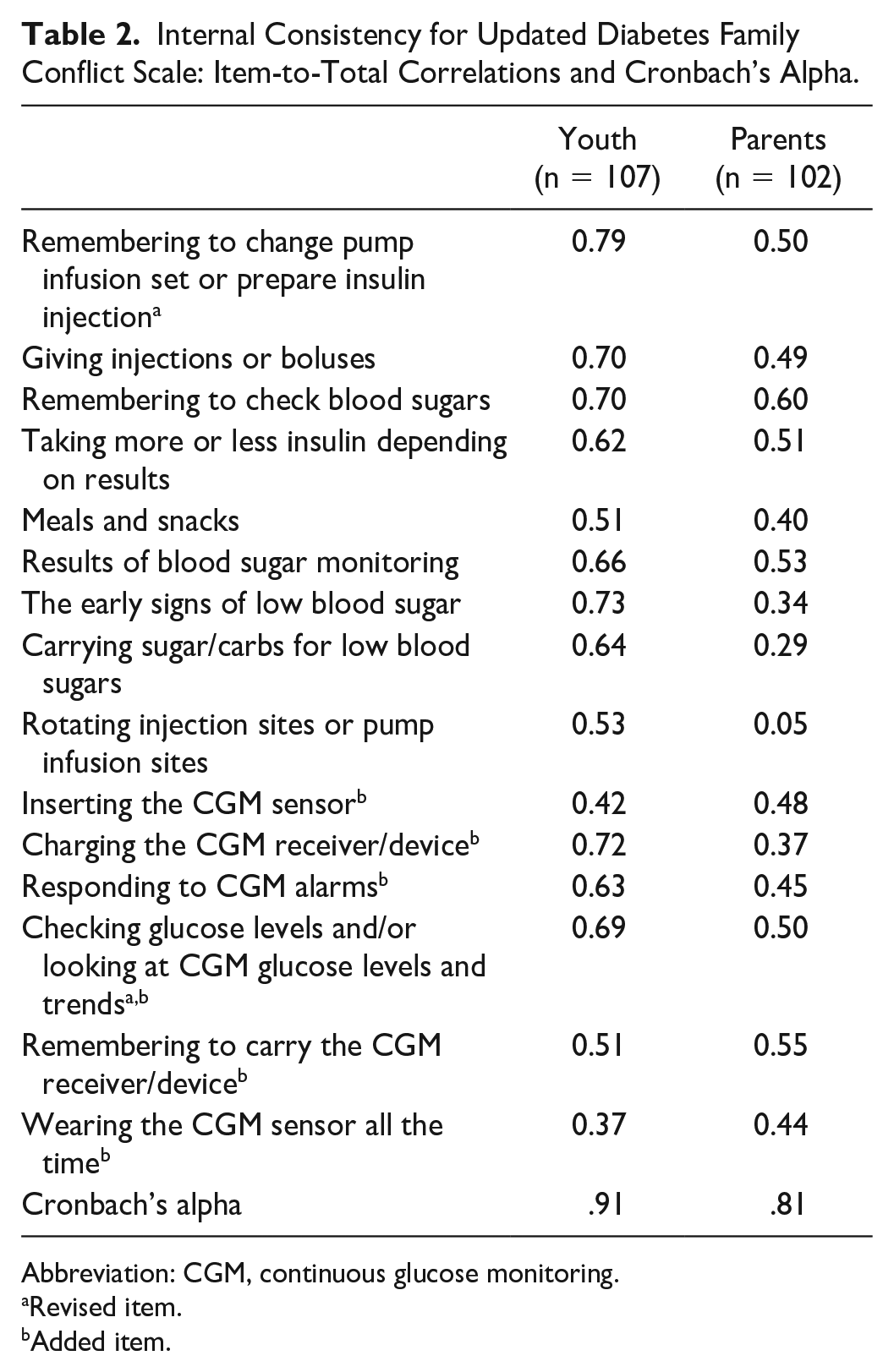
Abbreviation: CGM, continuous glucose monitoring.
Revised item.
Added item.
The youth and parent DFC scores were positively correlated (r = 0.47, P < .0001). The mean DFC score was 13.8 ± 16.9 for youth and 14.2 ± 12.6 for parents. Higher youth and parent DFC scores (more conflict) were associated with younger youth age (youth: r = −0.23, P = .02; parent: r = −0.26, P = .005) and less CGM wear (youth: r = −0.33, P = .0003; parent: r = −0.31, P = .001). Higher parent DFC scores were also associated with higher youth HbA1c (r = 0.23, P = 0.02), less CGM percentage TIR (70-180 mg/dL, 3.9-10 mM; r = −0.29, P = .005), and more CGM percentage time >180 mg/dL (r = 0.31, P = .002). Neither youth nor parent DFC scores were related to diabetes duration, blood glucose monitoring frequency, or diabetes management/adherence as assessed by the DMQ (see Supplemental Tables 1 and 2).
Convergent validity
Higher youth DFC scores were associated with more depressive symptoms (r = 0.38, P < .0001), more state anxiety (r = 0.24, P = .01), more diabetes burden (r = 0.31, P = .0009), and lower youth quality of life (r = −0.29, P = .002).
Higher parent DFC scores were associated with more parent depressive symptoms (r = 0.34, P = .0003), more parent state anxiety (r = 0.45, P < .0001), more parent diabetes burden (r = 0.43, P < .0001), and lower proxy report of youth quality of life (r = −0.44, P < .0001).
Predictive validity
Higher parent DFC scores were associated with less CGM wear by youth 3 months later (r = −0.28, P = .004).
Updated Diabetes Family Responsibility Questionnaire
We removed nine of the original 19 items due to low response variability (eg, making doctor appointments) or low item-to-total correlation on both youth and parent surveys (eg, remembering day of clinic appointment). We revised two of the original items to make them more applicable to current treatment. We added six CGM items but removed two due to outdated features (eg, calibrating CGM sensors, downloading the CGM receiver). The final updated survey has 14 items.
The updated DFR had high internal consistency (youth, α = .88; parent, α = .93). Item-to-total correlations were mostly in the range of 0.40 to 0.80 (Table 3). One item on the youth survey had an item-to-total correlation <0.40. However, the item was retained because it had higher item-to-total correlation on the parent survey.
Internal Consistency for Updated Diabetes Family Responsibility Questionnaire: Item-to-Total Correlations and Cronbach’s Alpha.
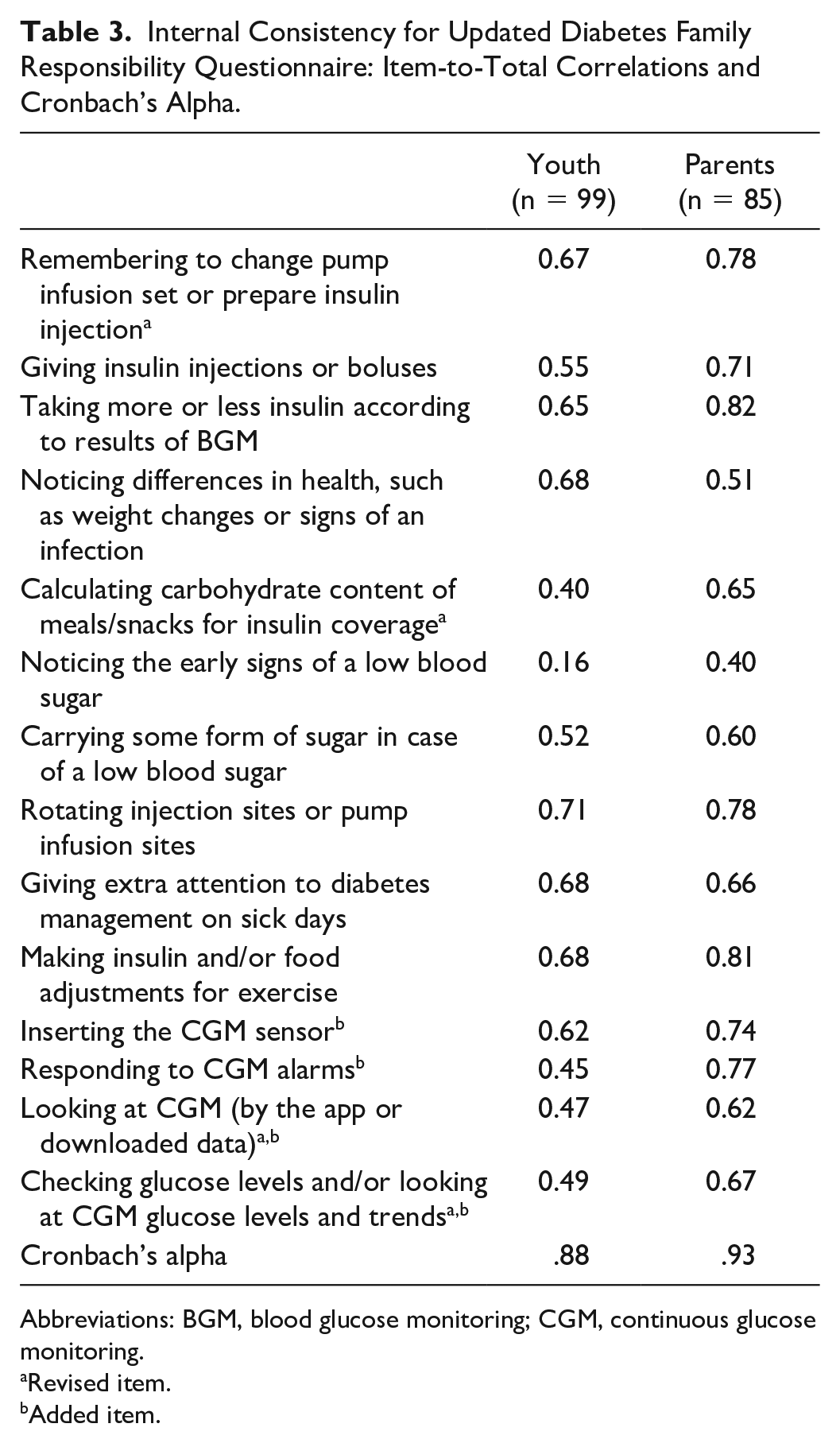
Abbreviations: BGM, blood glucose monitoring; CGM, continuous glucose monitoring.
Revised item.
Added item.
The youth DFR scores were highly correlated with parent DFR scores (r = 0.82, P < .0001). The mean youth DFR score was 42.2 ± 20.5 and the mean parent DFR score was 51.4 ± 22.6. Higher youth and parent DFR scores (more parent involvement) were correlated with younger youth age (youth: r = −0.76, P < .0001; parent: r = −0.81, P < .0001) and shorter diabetes duration (youth: r = −0.24, P = .01; parent: r = −0.34, P = .0002). Neither youth nor parent scores were related to HbA1c, CGM wear, or percentage TIR (see Supplemental Tables 1 and 2).
Convergent validity
Higher DFR scores were correlated with more frequent blood glucose monitoring (youth: r = 0.27, P = .003; parent: r = 0.35, p = .0002). Higher parent DFR scores were also associated with better diabetes management adherence as measured by the Diabetes Management Questionnaire (r = 0.49, P < .0001) and more diabetes burden (r = 0.22, P = .02). There were no significant correlations between youth and parent DFR scores and depressive symptoms, youth quality of life, or state anxiety.
Updated Glucose Monitoring Communication Questionnaire
We added six CGM items but removed one item (“I feel scared when the CGM alarms for high glucose levels”) due to its low endorsement among youth and parents. The final updated survey has 14 items.
The updated GMC survey demonstrated high internal consistency (youth, α = .88; parent, α = .86). The item-to-total correlations were in the range of 0.31 to 0.73 for youth report and 0.24 to 0.68 for parent report (Table 4). One item on the youth survey and three items on the parent survey had item-to-total correlations less than 0.40; these items were retained due to the clinical meaning.
Internal Consistency for Updated Glucose Monitoring Communication Questionnaire: Item-to-Total Correlations and Cronbach’s Alpha.
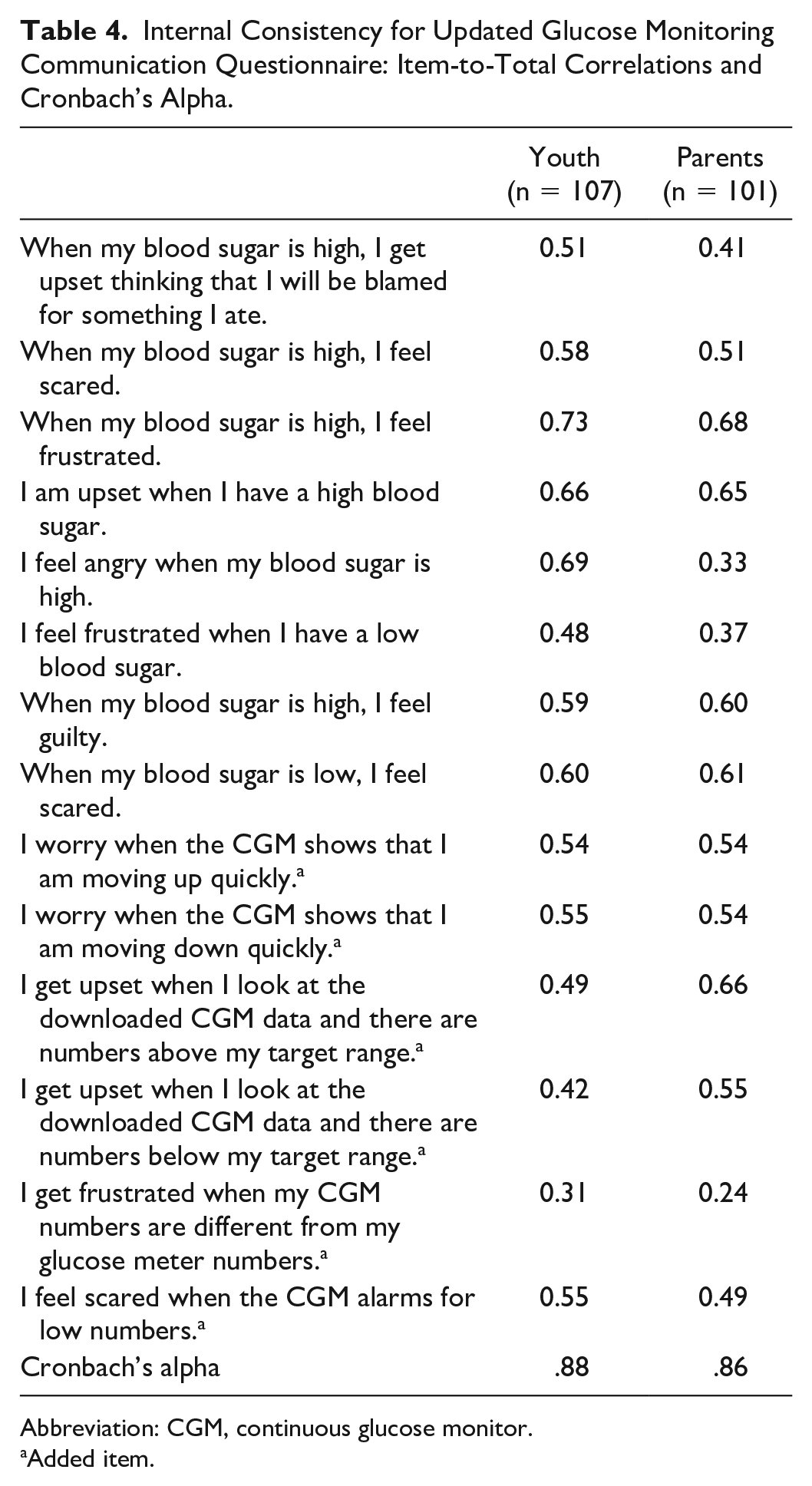
Abbreviation: CGM, continuous glucose monitor.
Added item.
The youth and parent GMC scores were positively correlated (r = 0.21, P = .03). The mean GMC score was 20.5 ± 18.8 for youth and 26.1 ± 18.3 for parents. Higher youth GMC scores (more negative affect) were associated with older youth age (r = 0.23, p = 0.01), whereas higher parent GMC scores were associated with younger youth age (r = −0.19, P = .047). Neither youth nor parent GMC scores were associated with youth diabetes duration or blood glucose monitoring frequency (see Supplemental Tables 1 and 2).
Convergent validity
Higher youth and parent GMC scores were associated with more depressive symptoms (youth: r = 0.60, P < .0001; parent: r = 0.28, P = .003), more diabetes burden (youth: r = 0.71, P < .0001; parent: r = 0.62, P < .0001), and more state anxiety (youth: r = 0.46, P < .0001; parent: r = 0.38, P < .0001). Higher GMC scores were also associated with lower youth QOL by self-report (r = −0.50, P < .0001) and parent proxy-report (r = −0.42, P < .0001). Higher parent GMC scores were associated with less youth CGM wear (r = −0.20, P = .04).
Additional Psychometric Assessment
To assess the psychometric properties of the updated surveys as they would be completed by individuals who are not using CGM, we repeated the analyses described above after omitting the CGM-specific items. The surveys demonstrated very similar internal consistency and relationships with other variables (eg, convergent validity) as when the CGM-specific items were included. Thus, the updated surveys demonstrate similar psychometric properties both with and without the CGM-specific items, supporting their versatility and applicability for use in both individuals using CGM and individuals not using CGM. The CGM-specific items can be omitted if someone is not using CGM and a total survey score can still be calculated.
Discussion
This study evaluated the psychometric properties of updated diabetes-specific psychosocial surveys to include CGM specific items. Our data demonstrate that the updated DFC, DFR, and GMC surveys have good psychometric properties. These surveys have adequate item-to-total correlations and high internal consistency for both youth and parent reports. The updated measures showed good convergent validity with other diabetes-specific psychosocial measures. Even with CGM-specific updated items, the surveys were able to retain validity and psychometric integrity. This adds value, given the widespread use of CGM in the pediatric population, with the ADA recommending starting CGM in children aged as young as 2 years. 29 Yet few available surveys measure psychosocial aspects of technology use and even fewer surveys have been updated to include technology use. 30 Thus, in the current era of rapid technology adoption among pediatric patients with T1D, 11 it is most opportune to allow for the assessment of the impact of CGM use on family involvement, family conflict, and negative affect related to glucose.
In our sample, all updated surveys were associated with important diabetes outcomes, but only DFC scores were associated with CGM wear at 3 months follow-up. Although on the surface this may seem problematic, CGM wear may be independent of some psychosocial constructs, and so a careful understanding of the more complex relationships between these psychosocial constructs, CGM wear, and health outcomes that work together will be important for future studies to develop targets for interventions. In addition, data show that CGM wear slightly improves biomedical outcomes but still many adolescents and young adults wearing CGM do not meet glycemic targets,11,31 so perhaps enhancing understanding about how these psychosocial constructs contribute to CGM data utilization and effective diabetes management are preferable than CGM wear alone.
Given that parental involvement continues to be fundamental for diabetes management even in the current era of diabetes technology, we updated both youth and parent surveys. Our updated surveys allow for dual reporters; the assessment of the person with diabetes as well as the caregiver’s perspective. In our sample, we observed a moderate correlation between youth and parent scores on the DFC, and a strong correlation between youth and parent scores on the DFR, suggesting that youth and parents generally agree, but that there is still utility in dual report assessments. For example, youth may be likely to endorse diabetes-specific conflict, whereas parents may be less likely to perceive such conflict for multiple reasons, including need for parental supervision as well as attention to social desirability in their responses.
We also observed correlations between youth and parent GMC survey scores. This finding is consistent with the original validation of the GMC survey. 9 For both the original and updated versions, parents endorsed greater negative affect related to glucose monitoring than youth, likely given the greater parental awareness and worry about out of range glucose levels and potential adverse effects on future health.
Several limitations should be considered when interpreting our findings. First, our study sample was relatively small, with the majority treated with pump therapy, and all participants had recently started using CGM. Therefore, our findings represent youth and parents who were eager to start CGM within a study context and this enthusiasm may not represent all youth with T1D. On the other hand, inclusion of a cohort intending CGM use adds value to CGM items on these surveys. Second, this study included earlier CGM generations, and the updated CGM items were created before newer CGM generations and other devices, such as automated insulin devliery systems, were available. Future studies should confirm the psychometric properties of these updated psychosocial surveys in samples of youth using more recent generations of diabetes technologies. Our findings may be an overestimate of potential burdens and stress related to CGM use as more recent CGM devices have demonstrated improvements in psychosocial outcomes. 32 The study sample was also primarily white and of higher socioeconomic status and therefore further investigation of these surveys is needed in more diverse samples. Finally, data were not available to assess test-retest reliability.
Conclusion
In summary, the updated DFC, DFR, and GMC surveys with the inclusion of CGM items have strong psychometric properties and can be used in research and clinical settings to assess diabetes-specific family conflict, family diabetes responsibility-sharing, and negative affect related to monitoring glucose. These surveys can also identify targets for intervention to improve self-care and glycemic control in youth with T1D. Further research is warranted in more diverse samples of youth with T1D and in those using later generations of CGM devices and automated insulin delivery systems. Regardless, our updated surveys are the only measures that assess diabetes-specific family conflict, family diabetes responsibility-sharing, and negative affect related to monitoring glucose in this current era of diabetes technology.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231159411 – Supplemental material for Updated Psychosocial Surveys With Continuous Glucose Monitoring Items for Youth With Type 1 Diabetes and Their Caregivers
Supplemental material, sj-docx-1-dst-10.1177_19322968231159411 for Updated Psychosocial Surveys With Continuous Glucose Monitoring Items for Youth With Type 1 Diabetes and Their Caregivers by Amit Shapira, Charlotte W. Chen, Lisa K. Volkening and Lori M. Laffel in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We acknowledge the support of the research teams from the Joslin Diabetes Center and all the participating adolescents and families.
Abbreviations
ADA, American Diabetes Association; CGM, continuous glucose monitoring; CES-D, Center for Epidemiologic Studies Depression Scale; CES-DC, Center for Epidemiologic Studies Depression Scale for Children; DFC, Diabetes Family Conflict Scale; DFR, Diabetes Family Responsibility Questionnaire; DMQ, Diabetes Management Questionnaire; GMC, Blood Glucose Monitoring Communication Questionnaire; PAID-Peds, Problem Areas in Diabetes Pediatric; PAID-PR, Problem Areas in Diabetes-Parent Revised; PedsQL, Pediatric Quality of Life Inventory; PRO, patient-reported outcome; STAI, State-Trait Anxiety Inventory; STAIC, State-Trait Anxiety Inventory for Children; T1D, type 1 diabetes; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Institutes of Health grants R01DK089349, P30DK036836, T32DK007260, K12DK094721, and K12DK133995; JDRF International; the Katherine Adler Astrove Youth Education Fund; the Maria Griffin Drury Pediatric Fund; and the Eleanor Chesterman Beatson Fund. The content is solely the responsibility of the authors and does not necessarily represent the official views of these organizations.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
