Abstract
Background:
Use of real-time continuous glucose monitoring (rtCGM) in ambulatory settings improves overall glycemic control and reduces the incidence of diabetic ketoacidosis (DKA) in adults and children/adolescents with type 1 diabetes (T1D). However, the use of rtCGM in children with DKA has not been well studied.
Method:
This prospective, single-arm, single-center study assessed the accuracy, reliability, and feasibility of a commercially available rtCGM device compared with point-of-care (POC) capillary and serum glucose values in pediatric patients admitted to the pediatric intensive care unit for DKA. The primary outcome was the accuracy of rtCGM glucose values compared with POC capillary and serum glucose values during standard treatment of DKA as assessed by Clarke Error Grid (CEG) analysis. Secondary outcomes were assessment of the relationship between rtCGM readings and degree of acidosis and mean length of hospital stay (LOS).
Results:
Data from 35 hospitalized children (mean ± SD age, 11.9 ± 4.1 years) with DKA were included in our analysis. Five hundred twenty-four time-matched glucose values between serum glucose and rtCGM and 91 time-matched glucose values between POC capillary glucose and rtCGM were obtained. The effect of acidosis on accuracy CEG analysis showed 95.4% of the 524 matched CGM/POC pairs and 95.6% of the 91 matched CGM/serum glucose pairs in the clinically acceptable A + B zones. The average LOS was 1.32 ± 0.73 days. Serum bicarbonate level did not appear to affect the accuracy of rtCGM in the setting of DKA.
Conclusions:
Continuous glucose monitoring use in inpatient pediatric DKA treatment was found to be feasible and reliable.
Introduction
Diabetic ketoacidosis (DKA) is the leading cause of hospitalizations, morbidity, and mortality in children and adolescents with type 1 diabetes (T1D).1,2 Morbidity and mortality associated with DKA are most often a result of cerebral edema and have been estimated to be responsible for 50% to 60% of diabetes-related deaths in children, while the remaining etiologies are attributed to sepsis, shock, and renal failure. 3
Although glucose monitoring in the setting of DKA has historically relied on point-of-care (POC) capillary/venous blood testing to guide the medical management of DKA, there is growing interest in the use of real-time continuous glucose monitoring (rtCGM) technologies for ongoing glycemic assessment.
Unlike traditional POC testing methods, which provide only a point-in-time glucose value, rtCGM devices use glucose oxidase–coated microelectrodes to continuously measure interstitial glucose levels in the subcutaneous space. Glucose data are transmitted to a hand-held reader or smartphone app and presented in numerical and graphical formats that display the current glucose level, glucose trends, and trend arrows that indicate the direction and velocity of changing glucose in real time. These data facilitate prevention and mitigation of abnormal glucose concentrations.
Large registry studies have shown that the use of rtCGM in ambulatory settings improves overall glycemic control and reduces the incidence of DKA and related hospitalizations in adults and children/adolescents with T1D.4 -6 However, to our knowledge, there have been no prospective studies examining the use of rtCGM in children with DKA. We report findings from a prospective, single-center study that assessed the use of a commercially available rtCGM device in pediatric patients hospitalized for severe DKA.
Methods
Study Design
This prospective, single-arm, single-center study assessed the accuracy, reliability, and feasibility of a commercially available rtCGM device in pediatric patients admitted to the pediatric intensive care unit (PICU) for DKA. The study was conducted in line with Good Clinical Practice (GCP), in accordance with the US Code of Federal Regulations, Title 21, Part 50 (21CFR50). The study protocol was approved by a central institutional review board (Spectrum Health, Grand Rapids, MI, USA). All participants provided written informed consent prior to enrollment in the study.
All patients were treated in concordance with the standard of care for DKA management and followed the institutional DKA guidelines. Modifications to the treatment course were done at the discretion of the PICU Physician or Critical Care Fellow when deemed necessary, without the knowledge of the research team. Bedside nursing staff and the critical care team were aware of enrollment, but rtCGM values were not used to make treatment decisions.
The study was conducted at Helen DeVos Children’s Hospital, Grand Rapids, Michigan, in line with GCP, in accordance with 21CFR50. 7 The study protocol was approved by a central institutional review board (Spectrum Health, Grand Rapids, MI, USA). All participants (parents/care givers) provided written informed consent prior to enrollment in the study.
Patients
Eligible pediatric patients admitted to the PICU at Helen DeVos Children’s Hospital were offered enrollment. The inclusion criteria at the time of presentation to the emergency department included the following: age 2 to 18 years, known or new-onset type 1 diabetes, presence of metabolic acidosis (venous pH <7.3 or serum bicarbonate <15 mEq/L), and serum glucose >300 mg/dL. Patients were excluded if they were pregnant, currently receiving dialysis, had skin changes or skin disease that precluded application of the rtCGM, or did not have a legal guardian to provide consent.
Study Devices
The Dexcom G6 rtCGM device (Dexcom, Inc., San Diego, CA, USA) was used in this assessment. The device measures glucose levels at five-minute intervals and transmits the glucose data to a receiver located at the patient’s bedside. The device is factory-calibrated; no POC blood glucose testing is required for calibration per Food and Drug Administration (FDA) approval; however, a two-hour warm-up period is needed before glucose values are made available. Alarm/alert notifications (high and low glucose, Urgent Low Soon, Rise Rate, Fall Rate, Signal Loss, No Reading) were turned off for the duration of the study.
Point-of-care capillary blood glucose values were obtained via the Stat Strip GLU® meter (Nova Biomedical, Waltham, MA, USA). The meter uses a glucose oxidase method. Serum glucose levels were measured by a perchloric acid hexokinase (PCA-HK) method (Roche Diagnostics, Indianapolis, IN, USA) at the Spectrum Health laboratories (Grand Rapids, MI, USA).
Procedures
After written informed consent was obtained from legal guardians, the rtCGM device was applied to the patient’s periumbilical region upon arrival to the PICU. During and after the two-hour rtCGM warm-up period, blood glucose levels were obtained hourly using POC testing in addition to basic metabolic panel (BMP) every six hours. The rtCGM data were matched to hourly blood glucose after the two-hour warm-up and only when the blood glucose was ≤400 mg/dL. Upon closure of subject’s anion gap, normalization of bicarbonate level to >18 mEq/L, the rtCGM was removed, and the clinical aspect of the study was completed. Treatment decisions were not based on rtCGM glucose concentrations.
Retrospective data collection included all laboratory tests obtained during the PICU admission, laboratory studies obtained in the emergency department, duration of insulin infusion, modifications to insulin infusion rates, total intravenous fluid volume, and duration of rtCGM monitoring. The rtCGM glucose levels were matched ± three minutes of the POC glucose levels and BMP. Study data were collected and managed using REDCap electronic data capture tools hosted at Spectrum Health.
Outcome Measures
The primary outcome was the accuracy of rtCGM glucose values compared with POC capillary and serum glucose values during standard treatment of DKA as assessed by the Clarke Error Grid (CEG) analysis. Secondary outcomes were assessment of the relationship between rtCGM readings and degree of acidosis and mean length of hospital stay (LOS).
Statistical Analysis
Categorical data were expressed as frequency (percent) and analyzed using a χ2 analysis. Numerical data that were normally distributed were summarized as mean ± standard deviation and analyzed using a two-sample independent
Results
Among the 46 patients assessed for eligibility, 39 met inclusion criteria, 36 were enrolled in the study, and data from 35 patients are included in our analyses. Data from one patient were not available for analysis due to a sensor malfunction. The cohort was well balanced between new-onset T1D and known T1D (Table 1). Patients with known T1D were older, had a higher body mass index (BMI), and had higher levels of creatinine, serum potassium, and serum phosphorus at the time of DKA diagnosis. Patients also had more frequent decreases in their insulin infusion rates when compared with patients with new-onset T1D. Thirteen (37.1%) patients with known T1D had experienced at least one prior hospitalization for DKA. There were no adverse events associated with the use of the rtCGM, nor were any sensors removed early. No episodes of hypoglycemia or cerebral edema were observed. A total of three patients were treated with an osmotic agent.
Patient Baseline Characteristics
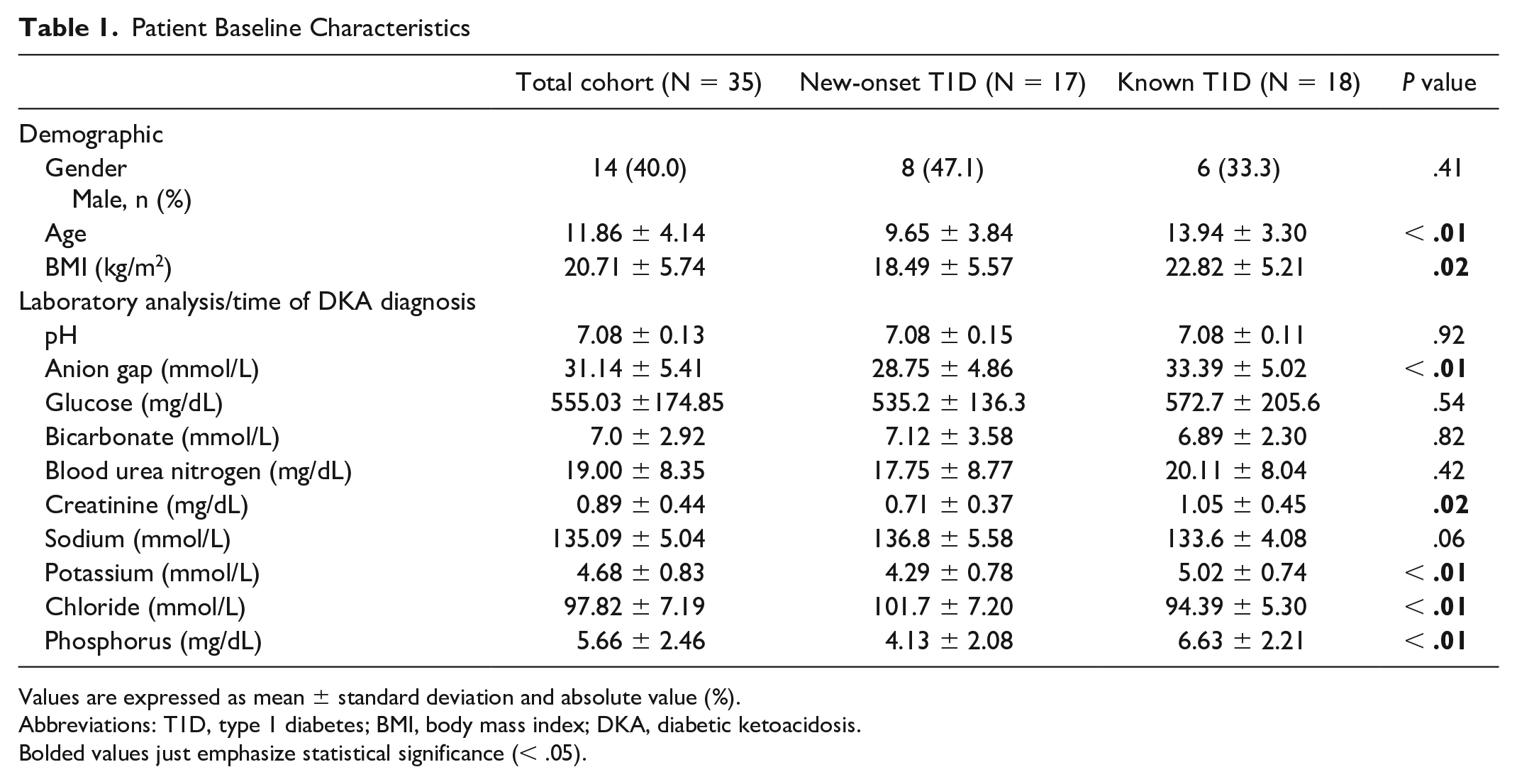
Values are expressed as mean ± standard deviation and absolute value (%).
Abbreviations: T1D, type 1 diabetes; BMI, body mass index; DKA, diabetic ketoacidosis.
Bolded values just emphasize statistical significance (< .05).
After receiving fluid resuscitation (21.89 ± 11.19 mL/kg) and initiation of intravenous insulin, comparison of laboratory values at the time of diagnosis with those obtained ±3 hours of the first rtCGM reading showed reductions in average anion gap and mean glucose with increased serum bicarbonate levels within the full cohort. The improvements were most notable among new-onset T1D patients versus known T1D but with no between-group differences.
Accuracy Assessment
We obtained 524 time-matched glucose values between serum glucose and rtCGM and 91 time-matched glucose values between POC capillary glucose and rtCGM. Clarke error proportions for each zone comparing each modality of glucose monitoring to rtCGM are provided in Figure 1.
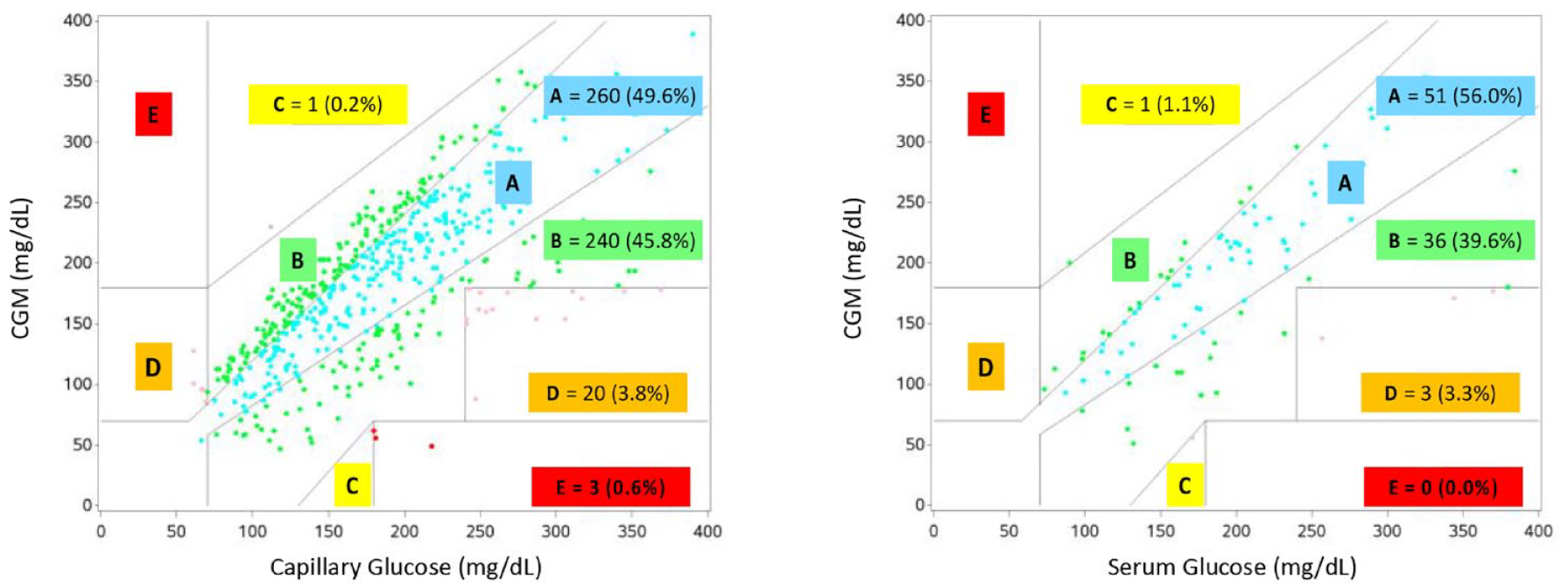
Clarke Error Grid analysis for rtCGM versus capillary and serum glucose. Abbreviation: rtCGM, real-time continuous glucose monitoring.
The CEG analysis of the 524 time-matched POC capillary glucose versus rtCGM revealed 95.4% of readings within A and B zones, signifying clinically accurate values leading to correct or benign treatment decisions. Analysis of the 91 time-matched serum glucose values versus rtCGM revealed 95.6% of readings within zones A and B, signifying clinically accurate values leading to correct or benign treatment decisions. The McNemar test revealed no evidence to suggest a statistical difference between rtCGM and serum glucose values, χ2(1, N = 91) = 0.52,
Relationship Between Glucose Values and Serum Bicarbonate Levels
There was a negative correlation (−0.27) between serum glucose and rtCGM delta when compared with serum bicarbonate level (
Treatment Duration
The average LOS was 1.32 ± 0.73 days. Patients with new-onset T1D tended to have longer hospital stays compared with those with known T1D; however, the difference was not statistically significant: 1.04 days [1.0, 1.7] versus 0.90 days [0.79, 1.3], respectively,
Longer durations of insulin infusion and rtCGM use were also observed in the new-onset T1D groups versus known T1D groups but with no significant between-group differences: 22.82 ± 14.51 hours versus 21.00 ± 7.10 hours, respectively,
Discussion
To our knowledge, ours is one of the first prospective studies examining the use of rtCGM in pediatric DKA patients. Our study used rtCGM in addition to standard DKA management to determine its accuracy, compared with POC capillary and serum glucose testing, in 35 pediatric patients treated for DKA in the PICU.
Results from the McNemar test assessment demonstrated that rtCGM values were comparable with POC capillary glucose and serum glucose measures. The clinical relevance of these findings was confirmed through the CEG analysis, which showed that >95% of the rtCGM results would facilitate acceptable medical decision-making. Analyses of serum bicarbonate levels demonstrated that the degree of acidosis and impaired perfusion during an episode of DKA does not impact the accuracy or reliability of rtCGM values. In addition, our study reported a mean duration of rtCGM use of 18.6 ± 9.7 hours, which correlates to >18 fingerstick POC tests that could have been avoided if rtCGM data had been used for treatment decisions. Importantly, there were no reported adverse events, which further demonstrates that rtCGM use is both safe and reliable in managing pediatric DKA.
A key limitation of the study is the time delay between diagnosis of DKA and the time at which the rtCGM device provided glucose readings. Because the study did not allow for delay of management, treatment of DKA was initiated prior to the initiation of rtCGM. The rtCGM device warm-up period also increased the time to first rtCGM reading. During this period, patients were treated with insulin and intravenous fluids to correct the degree of acidosis and hyperglycemia. In addition, many of the enrolled patients did not present with extreme acidosis or hyperglycemia. These extremes are the factors that are of most clinical interest because the functionality and accuracy of rtCGM are potentially most affected within these extreme ranges. A final limitation was the inability to provide a blinded evaluation; however, because the study protocol prohibited nursing staff and physicians to act upon the results of the rtCGM, we do not feel this impacted our findings.
Conclusions
Our study suggests that the use of rtCGM in the management of DKA is both feasible and accurate as evidenced by greater than 95% of values in the clinically acceptable range based on the CEG analysis. Serum bicarbonate level did not appear to impact the accuracy of rtCGM in the setting of DKA. Additional studies examining the use and cost-effectiveness of rtCGM use in treatment decisions are needed.
Footnotes
Acknowledgements
TP, JJ-V, and RF designed the study and wrote the manuscript. JP conducted the statistical analysis. All authors had access to the data and reviewed the final manuscript. TP is the guarantor of this study findings and takes responsibility for the article. The authors wish to thank Christopher G. Parkin, MS, CGParkin Communications, Inc., for his editorial assistance in preparing the manuscript.
Abbreviations
CEG, Clarke Error Grid; DKA, diabetic ketoacidosis; GCP, Good Clinical Practice; LOC, length of hospital stay; PCA-HK, perchloric acid hexokinase; PICU, pediatric intensive care unit; POC, point of care; rtCGM, real-time continuous glucose monitoring; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dexcom, Inc. provided the rtCGM devices as an unrestricted research grant and was not involved in the study design, coordination, results, or statistical analysis.
