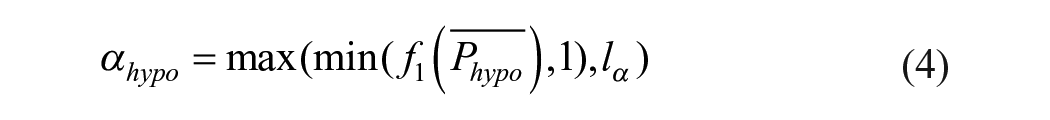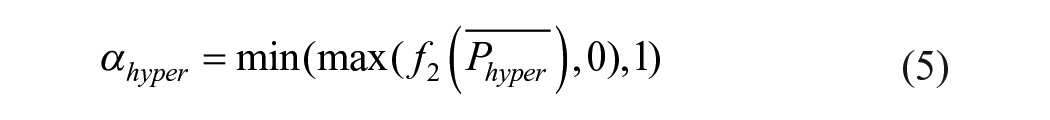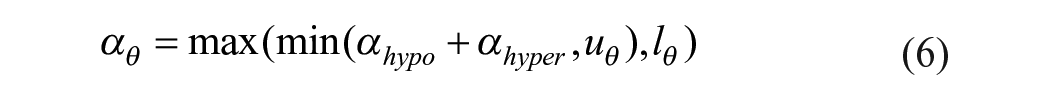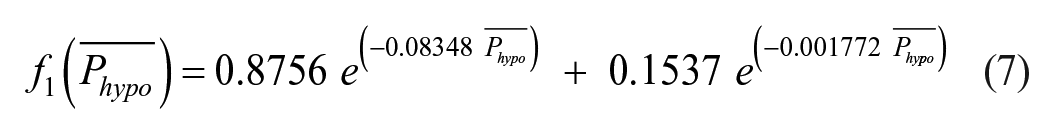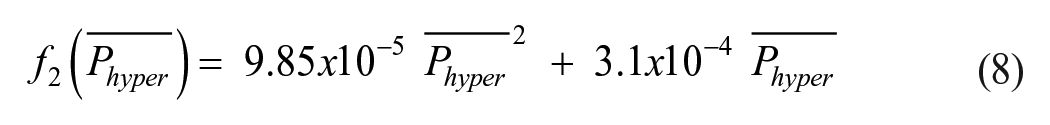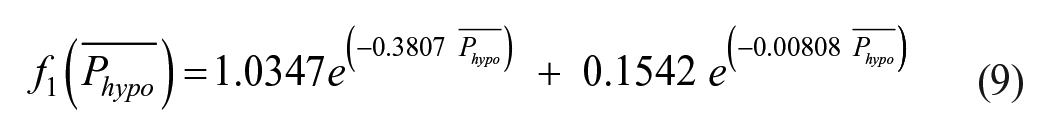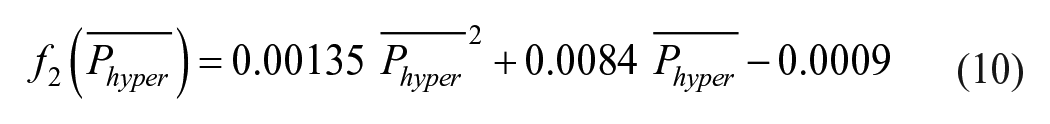Abstract
Background:
Automated insulin delivery (AID) algorithms can benefit from tuning of their aggressiveness to meet individual needs, as insulin requirements vary among and within users. We introduce the Performance-Based Adaptation Index (PAI), a tool designed to enable automatic adjustment of an AID system aggressiveness based on continuous glucose monitoring (CGM) metrics.
Methods:
PAI integrates two CGM-based metrics—one for hypoglycemia and another for hyperglycemia exposure—over a previous time window into a single index (
Results:
Negligible performance variations were observed in the baseline scenario. For the conservative scenario, adjusting
Conclusion:
In-silico results demonstrated the safety and efficacy of using PAI to automatically tune the UVA-MPC controller, achieving TIR values above 70% under fully closed-loop conditions and across various physiological states. Clinical validation of these results is warranted.
Keywords
Introduction
Automated insulin delivery (AID) systems have significantly simplified glucose management for individuals with type 1 and type 2 diabetes (T1D and T2D).1-4 However, achieving safe and effective glucose control can still pose challenges for some individuals due to the considerable variability in metabolic and psycho-behavioral factors, including circadian rhythms, physical activity, menstrual cycle, diet, and stress. Since customizing an AID system for each user is impractical, there is a clear need to develop AID algorithms that can adapt to individual insulin requirements and their temporal variations.5,6
Adaptive AID systems are those capable of modifying themselves in response to physiological and/or behavioral changes. 5 While there are specific definitions for adaptive problems, for our purposes, we can categorize AID adaptation as either indirect or direct. In the case of indirect adaptation, the parameters of the underlying design model or plant are updated, resulting in a readjustment of the control response. For example, this could involve re-identifying the glucose metabolic model in response to increased insulin sensitivity due to physical activity, ultimately leading to a more conservative control law. Typically, this adaptation requires solving an optimization problem to re-identify a set of model parameters periodically using historical observations.7-10
On the other hand, direct approaches involve adjusting the controller parameters based on specific criteria or measurements, such as hypoglycemia risk. In the literature, various approaches such as run-to-run strategies and iterative learning control have been suggested to update these parameters. These methods involve adjusting penalty weights in optimization-based control algorithms or incorporating time-varying constraints based on predefined rules for glucose regulation performance.6,11-13
In this paper, we introduce a novel Performance-Based Adaptation Index (PAI) that could be seamlessly integrated into any AID system. Performance-Based Adaptation Index consolidates glucose performance metrics into a single index, which can be then communicated across the internal logic of the AID system. While various retuning strategies can be followed, we propose a simple Performance-based Adaptation System (PAS). In this system, PAI modulates a predefined set of controller parameters multiplicatively. We test this strategy with our fully automated UVA-model predictive control (MPC) system, demonstrating how continuous adaptation with PAS can safely and effectively overcome performance plateaus.
Materials and Methods
Performance-Based Index Computation
The goal of PAI is to consolidate CGM-based metrics that represent exposure to hypoglycemia (
If the AID was active for at least 80% of the time, and at least 80% of the CGM data was available, the following procedure is followed for calculating
1. Retrieve the previous day’s CGM and insulin records, and compute
2. Construct an array of all daily metrics estimate
3. Compute average metrics over the time window as follows:
with
4. Compute the dimensionless index as follows:
with
where
The 80% data availability requirement is a design choice intended to ensure that sufficient data is available to evaluate the AID system’s performance. The index
Functions
1. If
From equations (4) and (7),
2. If
Equations (9) and (10) have the same form as equations (7) and (8), respectively. So
PAS: Performance-Based Adaptation System
We propose a simple PAS to adjust automatically the AID system using
The AID system can then propagate
Modulating the Aggressiveness of the UVA AID System
The UVA AID system has been extensively described in previous works.15-18 It integrates four main components: a core MPC for background insulin regulation (see Appendix A-2), a Bolus Priming System (BPS)
17
to safely administer insulin when glucose levels rise rapidly, and a Hyperglycemia Mitigation System (HMS) and Safety Supervision Module (SSM) to correct sustained hyperglycemia and protect against hypoglycemia,
19
respectively. Figure 1 shows a block diagram of the UVA AID system in which
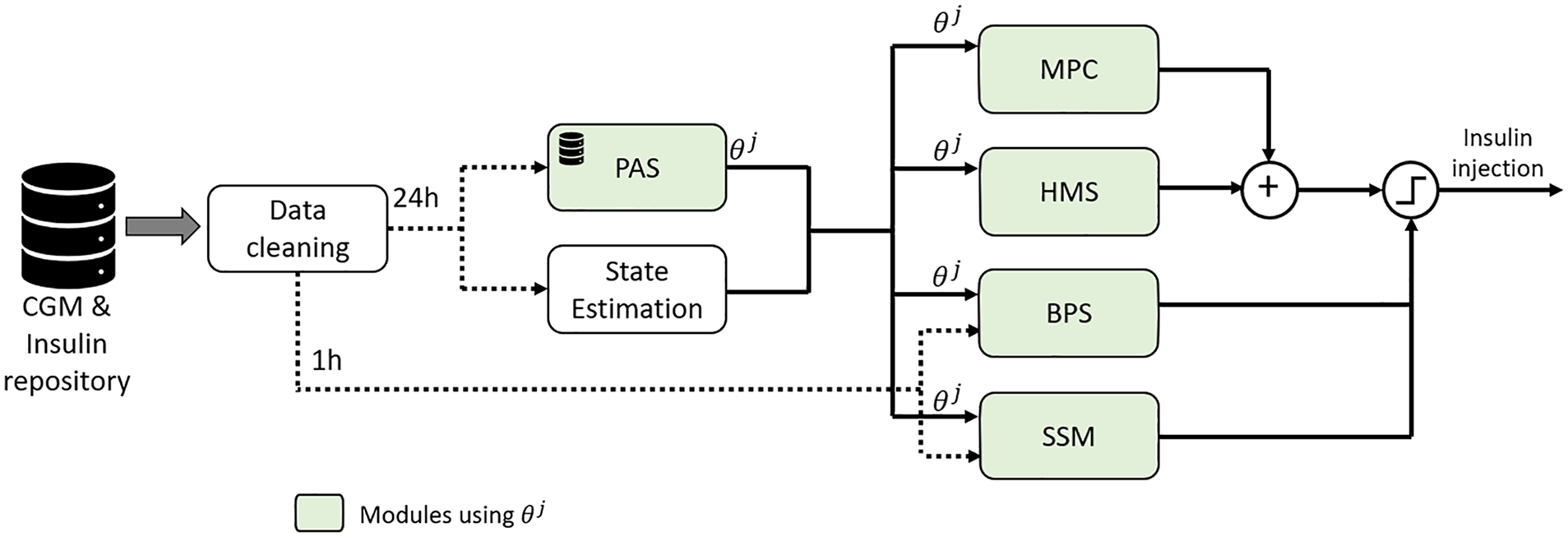
Component diagram of UVA AID system with PAS. The data cleaning block refers to the process of verifying that at least 80% of the CGM data is available and that the system was in closed-loop mode for at least 80% of the day. The green blocks show the controller modules that use
At each step
In Silico Study Designs
The 100-adult cohort of the UVA/Padova T1D Simulator was considered to evaluate the performance of the UVA AID system with and without PAS in five different scenarios:
Baseline: Defining
Conservative: Defining
Aggressive: Defining
Disturbance in insulin sensitivity (IS): Defining
Change in insulin needs: Defining
For each scenario, simulations were performed over 21 days, considering intra/inter-day variability in insulin sensitivity parameters. The eating patterns included three meals (breakfast, lunch, and dinner) and up to three snacks per day based on the body weight (BW) of each subject. Meals and snack sizes were randomly selected from uniform distributions between [0.7, 1] g/kg and [0.15, 0.4] g/kg, respectively, with the possibility to skip snacks. Mealtimes were randomly selected from uniform distributions within [7, 13, 19] ± 1.5 hours for main meals and [9, 16, 22] ± 1 hours for snacks. Hypoglycemia rescues were administered every 20 minutes when simulated blood glucose dropped below 70 mg/dL. Note that scenarios 4 and 5 were designed to evaluate the system’s performance during changes in insulin requirements due to, for instance, menses, exercise, illness, or periods of stress.
Simulation conditions for each scenario were maintained across all controller settings and the entire resulting glucose traces were used to compute the outcomes for each in-silico individual. Due to space limitations, only the results of the in-silico analysis using TBR and TAR for computing
Outcomes and Statistical Analysis
Simulations, data formatting, preparation, and analysis, and statistical analyses were performed in MATLAB R2022a and Simulink (MathWorks, Natick, MA, USA). Performance glycemic metrics were computed from the simulated BG traces and include average glucose concentration (mg/dL), standard deviation (mg/dL), coefficient of variation (%), percentages of time spent below 54 mg/dL, below 70 mg/dL (TBR), between 70 and 140 mg/dL (TTR), between 70 and 180 mg/dL (TIR), above 180 mg/dL (TAR) and above 250 mg/dL, TDI, total daily basal insulin (TDBa), total daily bolus insulin (TDBo), and number of hypo-treatments per day. All results are presented as across-subject mean value and standard deviation, unless otherwise stated. Comparison between different therapy adjustments was performed using the paired-sample t-test and a paired two-sided Wilcoxon signed rank, with significance level at a P-value <.05 if the distribution of the differences was normally or non-normally distributed, respectively. Normality was addressed using Lilliefors Test.
Results
Glycemic metrics for the baseline scenario under both controller settings (i.e., AID and AID+PAS) are reported in Table 1 for days 1, 5, 21, and overall. As shown, overall glycemic metrics show slight changes between both controller settings. Average BG was slightly reduced, while TIR increased by 1.4% and TBR by 0.1% when using PAS. In addition, insulin infusion, both basal and boluses, increased following elevated TAR, which remained higher than the desired target. Notably, after day 7 (not shown in the table), the θ values remained virtually unchanged, with the population’s mean fluctuating between 44.6 and 45.3.
Glycemic Outcomes During the “Baseline” Scenario for AID and AID With PAS.
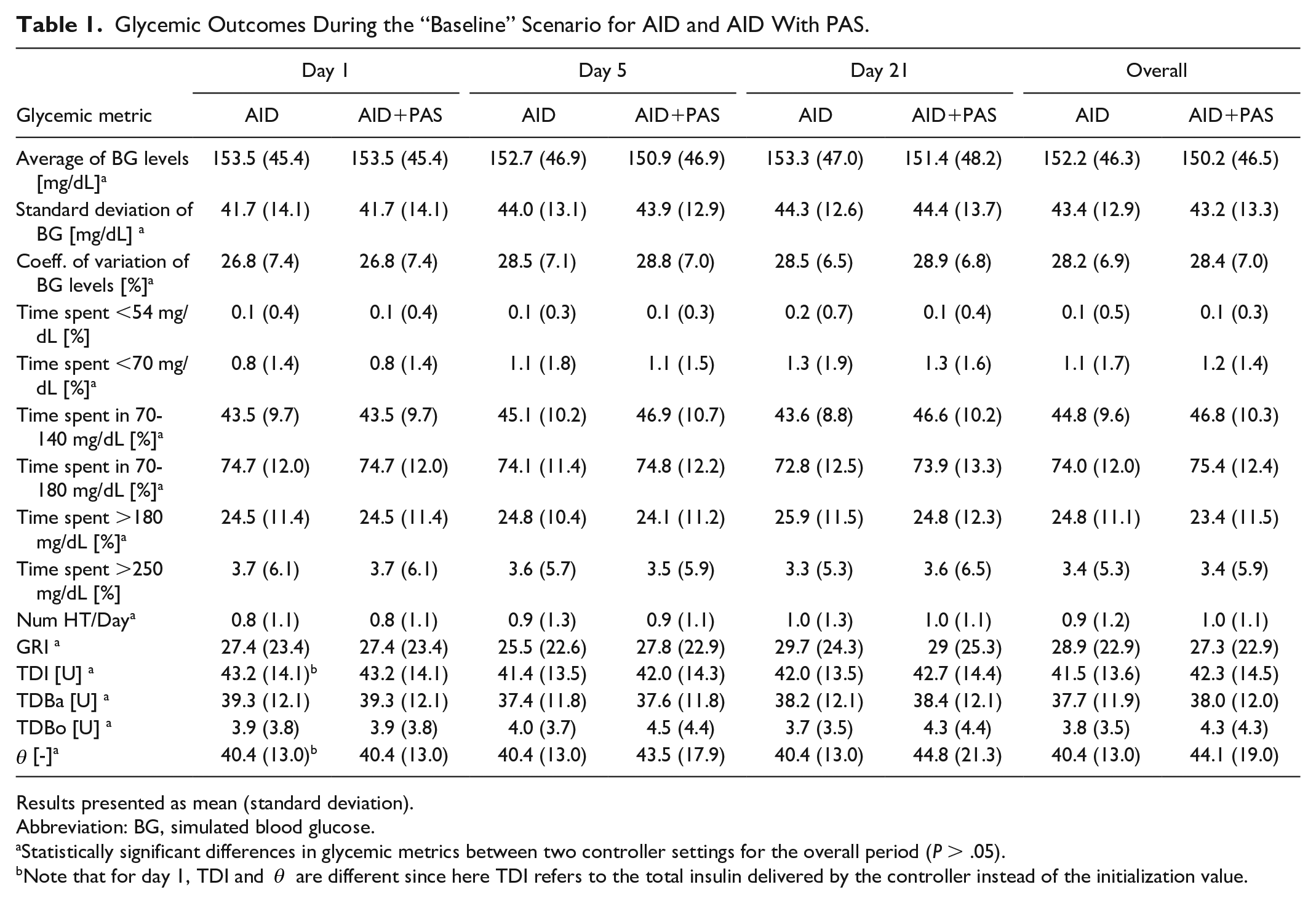
Results presented as mean (standard deviation).
Abbreviation: BG, simulated blood glucose.
Statistically significant differences in glycemic metrics between two controller settings for the overall period (P > .05).
Note that for day 1, TDI and
Simulation results for the conservative and aggressive scenarios under both controller settings are presented in Figures 2 and 3, respectively. In addition, all glycemic metrics for day 21 and overall are reported in Tables 2 and 3 for the conservative and aggressive scenarios, respectively.
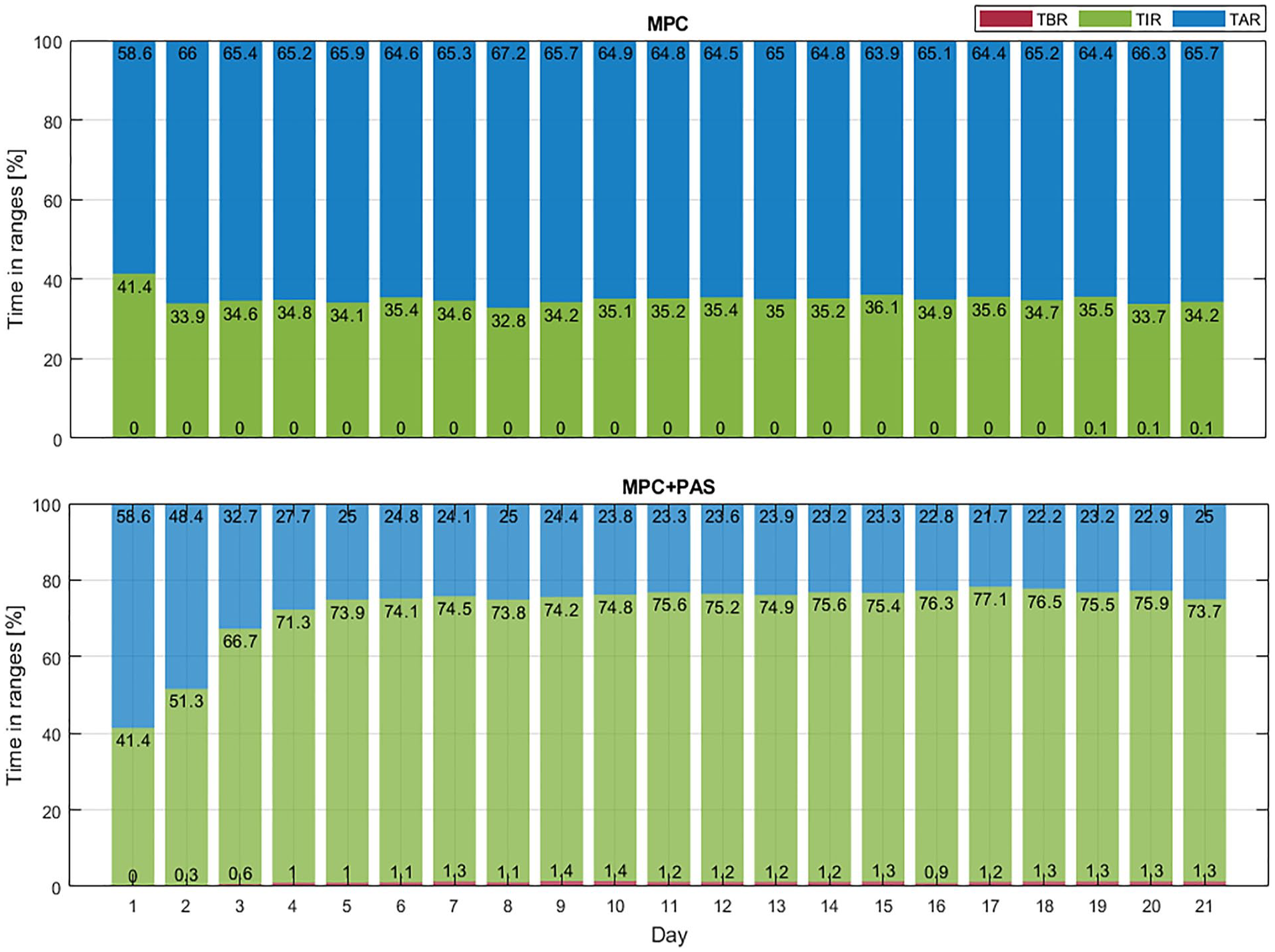
Glycemic metrics (TBR, TIR, and TAR) per day when using AID (top panel) and AID+PAS (bottom panel) in the conservative scenario.
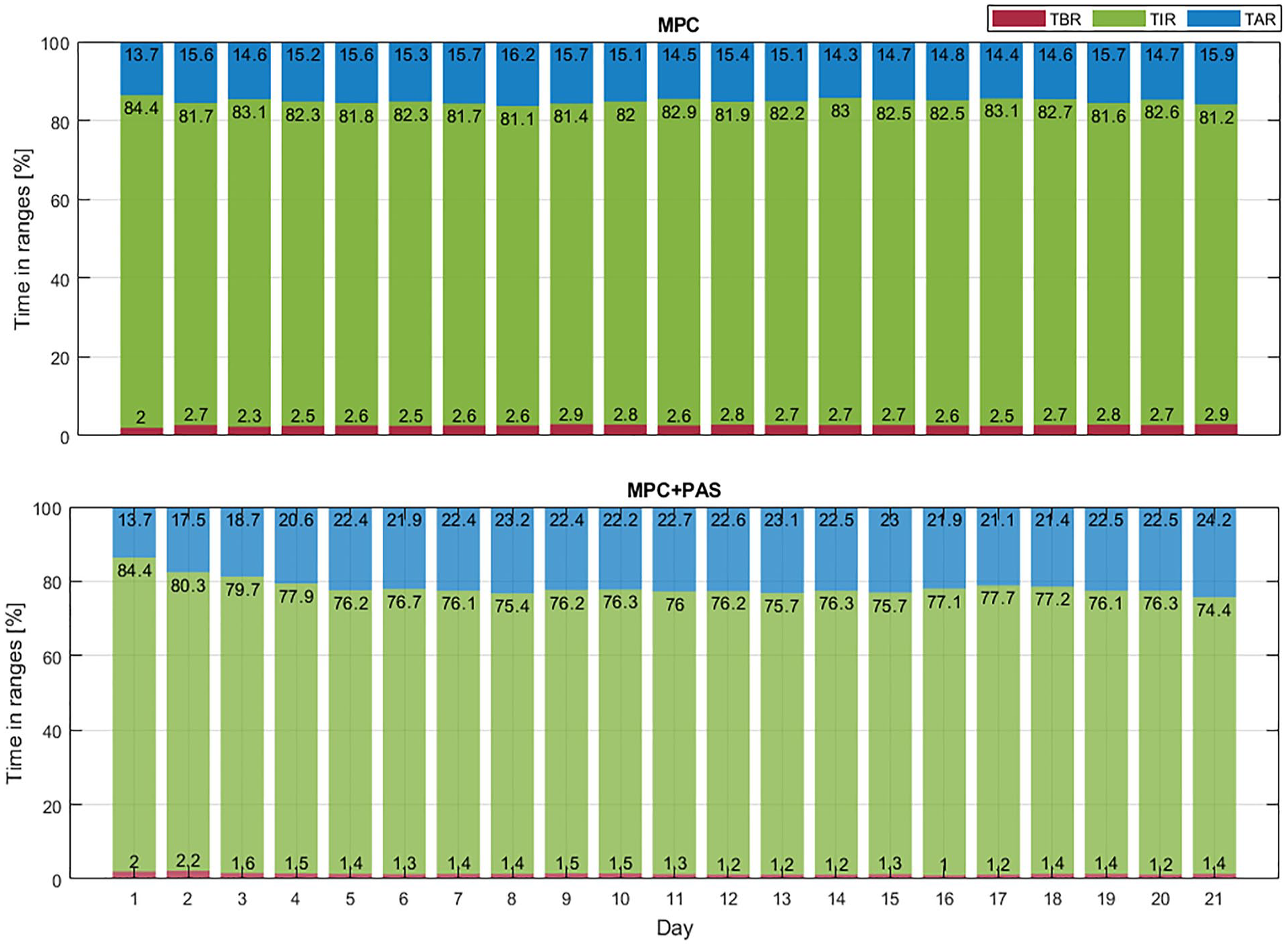
Glycemic metrics (TBR, TIR, and TAR) per day when using AID (top panel) and AID+PAS (bottom panel) in the aggressive scenario.
Glycemic Outcomes During The “Conservative” Scenario for AID and AID With PAS.
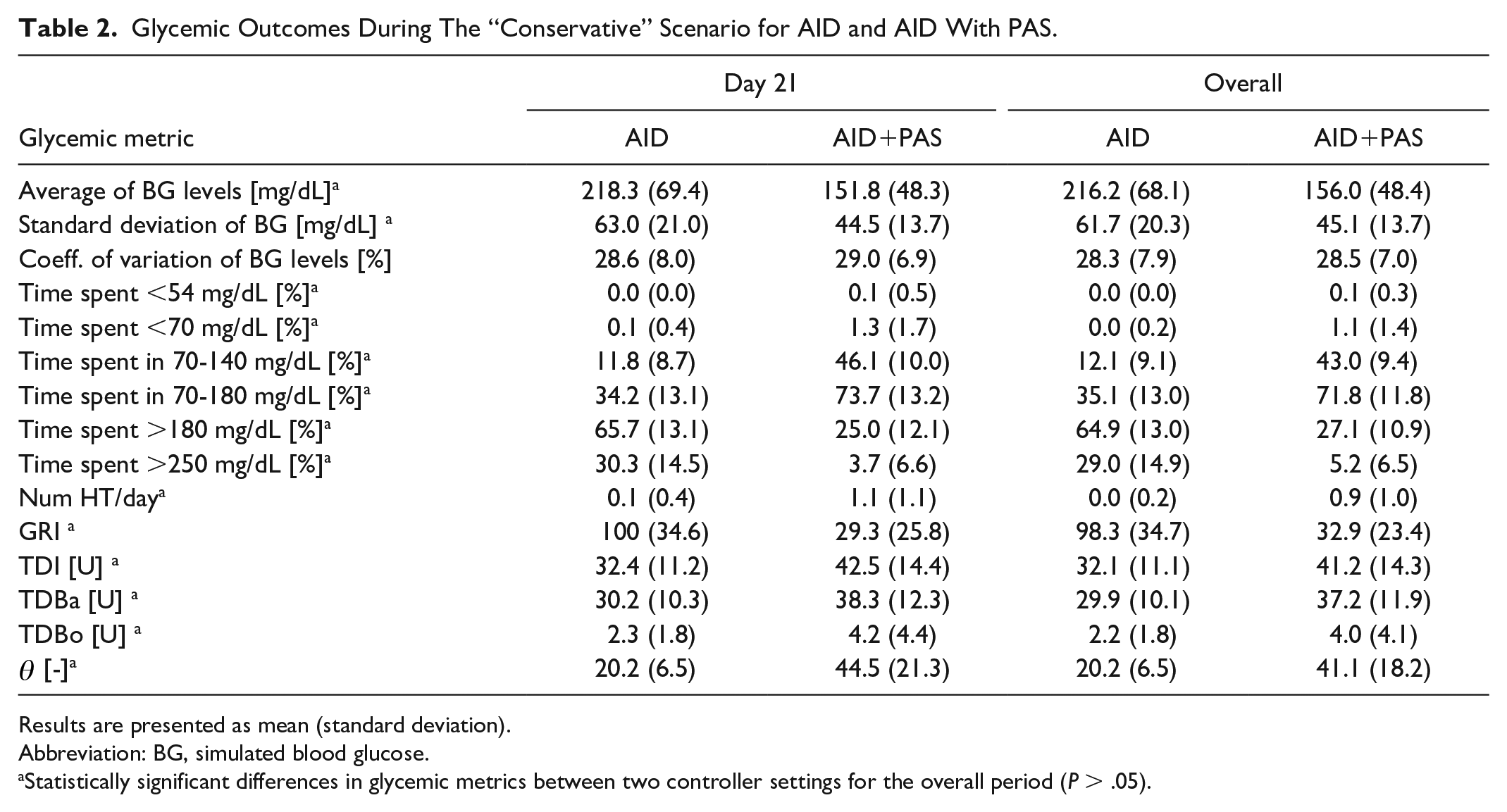
Results are presented as mean (standard deviation).
Abbreviation: BG, simulated blood glucose.
Statistically significant differences in glycemic metrics between two controller settings for the overall period (P > .05).
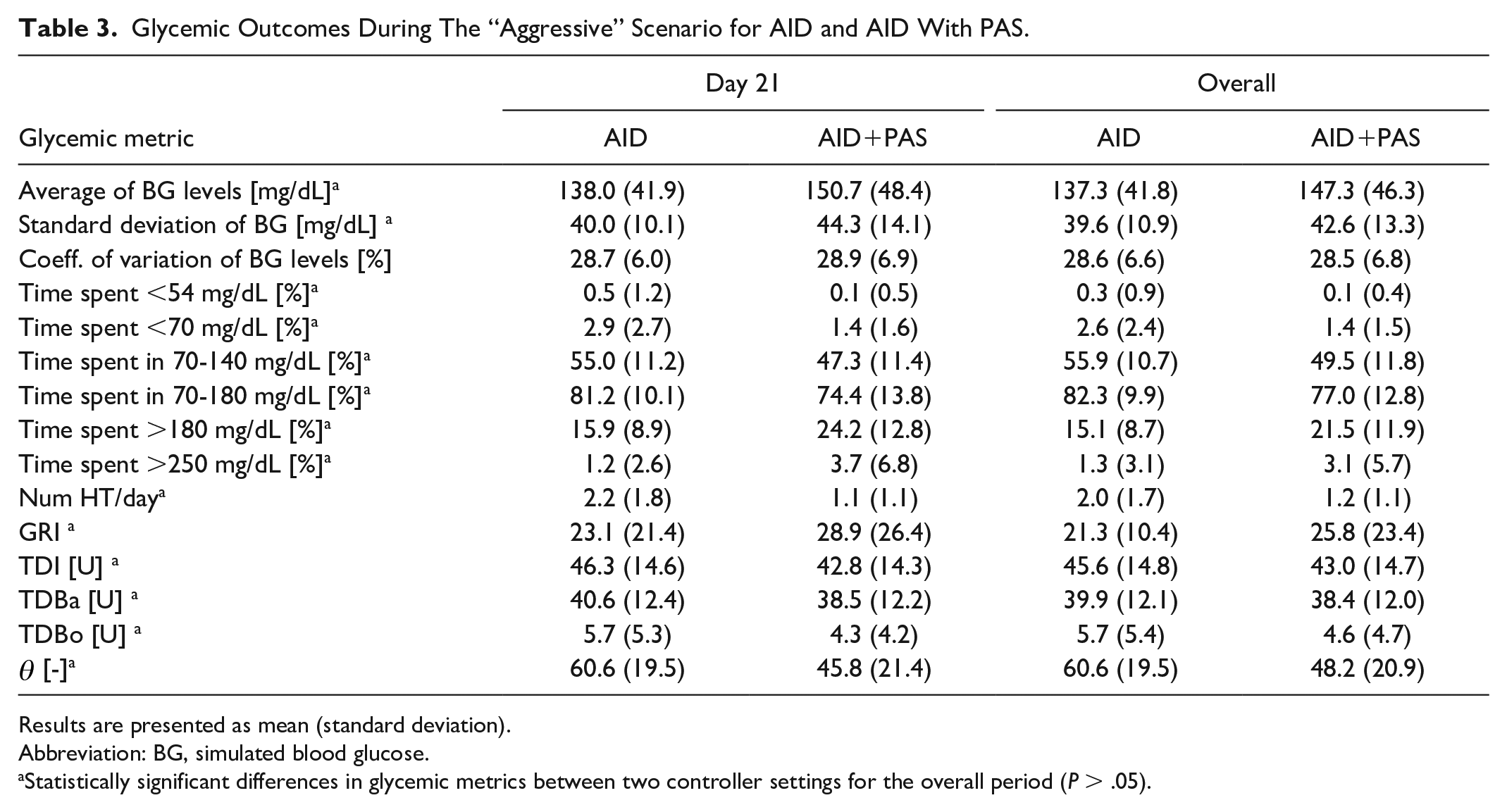
Glycemic Outcomes During The “Aggressive” Scenario for AID and AID With PAS.
Results are presented as mean (standard deviation).
Abbreviation: BG, simulated blood glucose.
Statistically significant differences in glycemic metrics between two controller settings for the overall period (P > .05).
For the conservative scenario, it can be observed how the performance of the AID system is affected when the initialization of
However, when the AID is integrated with PAS, after 5 days, the glycemic control is improved, reaching TIR values (mean: 73.7% at day 21) similar to those of the nominal controller performance (i.e., observed in the baseline scenario), and is maintained throughout the days. As shown in Figure 2, TIR increased by about 10% from day 1 to 2, 15% from day 2 to 3, and 5% from day 3 to 4, with a slight increase in TBR but remaining around 1%. The increase in TIR can be attributed to reductions in TAR across days.
The effect of retuning the controller’s aggressiveness using PAS can also be seen in the differences in TDI and
In the aggressive scenario, TBR is around 2% on day 1 due to an overestimation of
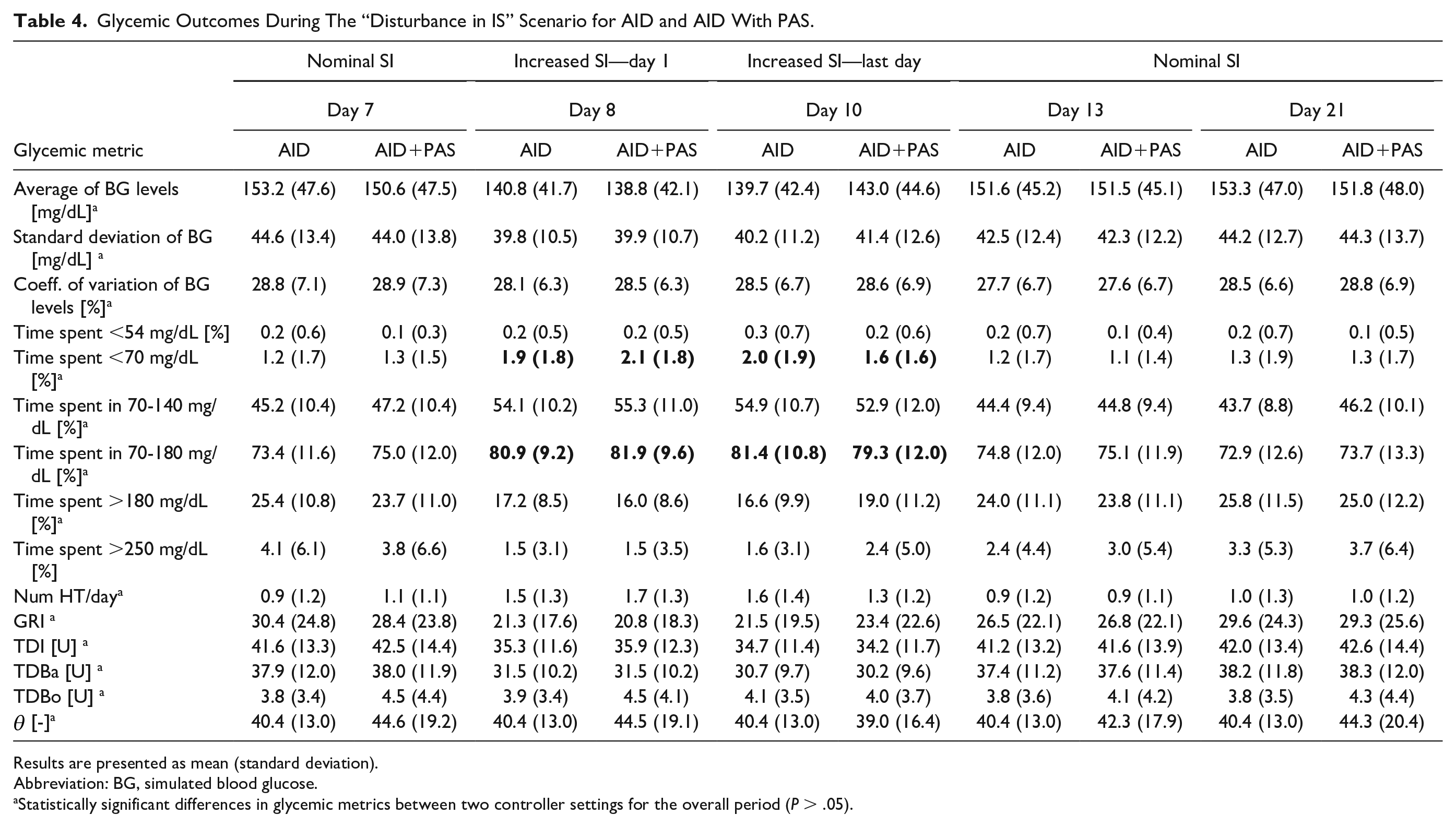
Finally, simulation results for scenarios 4 (Disturbance in insulin sensitivity) and 5 (Change in insulin needs) are presented in Tables 4 and 5, respectively, for the representative days of the modifications in the IS parameters. Table 4 shows that during the disturbance period (day 8 to 10) of increased IS parameters, PAS compensates for the rise in TBR (2.1% at day 8 and 1.6% at day 10) by reducing TDI (35.9 U at day 8 and 34.2 U at day 10) through adjustments in
Glycemic Outcomes During The “Disturbance in IS” Scenario for AID and AID With PAS.
Results are presented as mean (standard deviation).
Abbreviation: BG, simulated blood glucose.
Statistically significant differences in glycemic metrics between two controller settings for the overall period (P > .05).
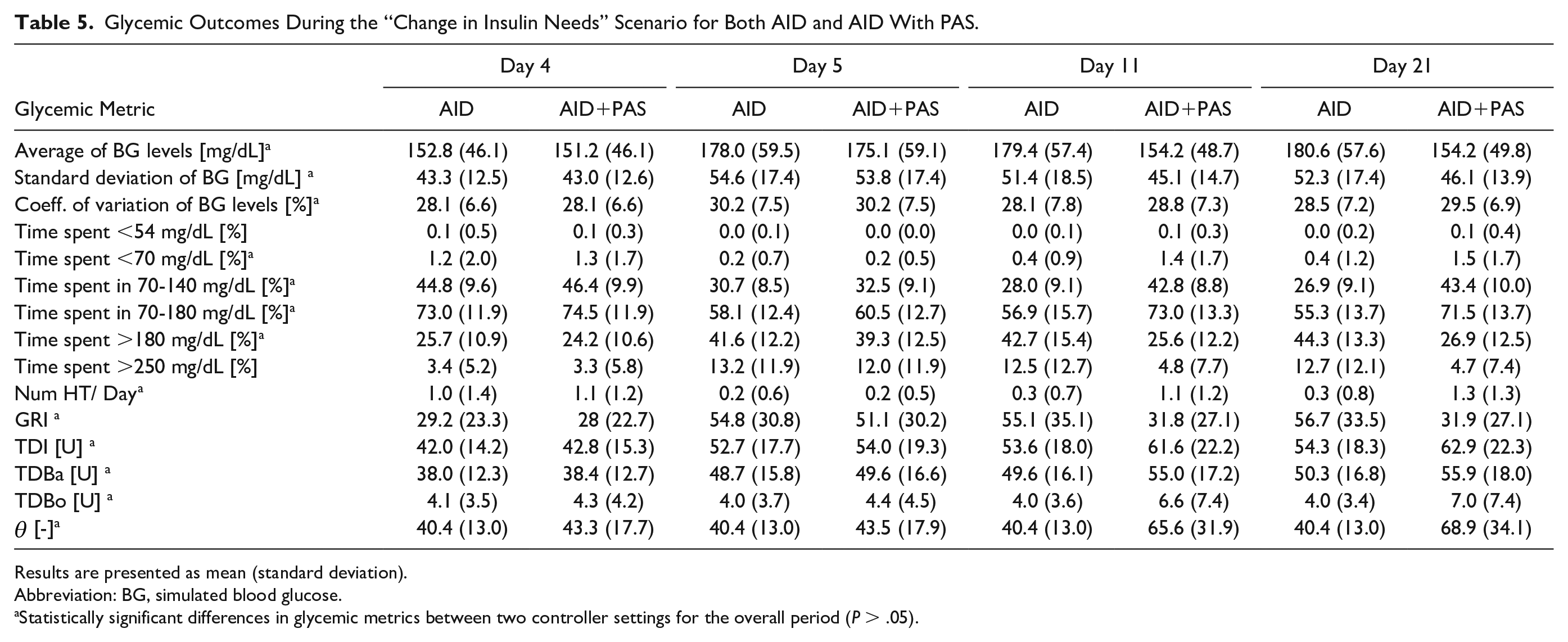
Glycemic Outcomes During the “Change in Insulin Needs” Scenario for Both AID and AID With PAS.
Results are presented as mean (standard deviation).
Abbreviation: BG, simulated blood glucose.
Statistically significant differences in glycemic metrics between two controller settings for the overall period (P > .05).
Discussion
Designing individualized or personalized control strategies for glycemic control remains a challenge in diabetes technology. Automated insulin delivery systems are typically designed at a population level, which could prove impractical for each patient due to individual fluctuations in insulin needs over time. While AID systems are primarily tuned considering in-silico populations that represent the variability of the real T1D population, edge cases can still result in high hyperglycemia or hypoglycemia exposure, regardless of whether more aggressive or more conservative control algorithms are used.
To overcome the challenges of designing a control system for each individual, various adaptation and personalization strategies have been proposed in the literature. However, most of these strategies require highly demanding computational implementations for real-time optimizations to adjust the controller.
In this work, we presented a strategy to automatically adapt or retune the controller’s aggressiveness on a daily basis using CGM-based hypo- and hyperglycemic metrics (PAI). In our proposal, two different options to compute PAI were considered, one based on time in ranges and another based on risks (LBGI and HBGI) over the previous two weeks. Both the glycemic metrics and the length of the time window can be modified as needed. Modifications to account for other relevant factors to insulin therapy, such as hypoglycemia treatments, could also be included. For instance, combining hypoglycemia rescues with TBR or LBGI as a new metric of hypoglycemia could help account for unreliable TBR resulting from consecutive hypoglycemia rescues, which may mainly affect in-silico testing where hypoglycemia treatments are automated.
Simulation results showed that the proposed PAS is safe in all the scenarios, as it keeps the controller settings consistent with the baseline scenario and recovers to approximately the desired glycemic outcomes (TBR and TAR) in the conservative and aggressive scenarios. For the baseline scenario, no differences between AID and AID+PAS were expected as the controller was tuned for the same in-silico population and with the correct TDI to initialize
Although it is not presented here, a similar analysis was performed using LBGI and HBGI to compute PAI. Simulation results show similar results to those computed using TBR and TAR. There was a slight improvement in TAR and a larger improvement on the time above 250 mg/dL, while maintaining similar levels of TBR. However, the
Scenarios 4 and 5 demonstrated the performance of the proposed PAS using PAI when insulin requirements change temporarily or permanently. In the first case, PAS safely attempted to mitigate the increase in TBR by reducing insulin infusion. This showed how the proposed strategy can handle such disturbances without compromising overall glycemic control. In addition, when a permanent change in insulin requirements was tested, the results showed that using AID+PAS the baseline performance of the controller could recover progressively and without oscillations that would put the patient at risk of hyperglycemia or hypoglycemia. A limitation of these simulations is the absence of a CGM error model, which may slightly affect the results.
Although the proposed index and strategy were evaluated only with our UVA AID, PAI, and PAS can be used in any AID system, as PAI is designed based exclusively on controller-agnostic metrics. In this regard, simulations with other control systems could be performed straightforwardly. Finally, the use of PAS is currently undergoing a clinical trial with humans and results are expected to be presented in 2025 (NCT06041971).
Conclusions
Individual variations in insulin needs present obstacles to optimal control in T1D, even to AID systems. Our work introduces a novel strategy for adapting the controller’s aggressiveness by using CGM-based metrics (PAI), offering a promising solution to this challenge. Simulations demonstrated that the proposed strategy effectively maintained safe control across various scenarios, adjusting insulin delivery accordingly. Furthermore, PAS showed resilience in addressing temporary and permanent changes in insulin requirements. Importantly, our approach is not limited to specific control algorithms, offering potential applicability across various AID systems. Clinical trials with the UVA AID + PAS are underway, promising further insights into its efficacy in real-world settings.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251315499 – Supplemental material for A Performance-Based Adaptation Index for Automated Insulin Delivery Systems
Supplemental material, sj-docx-1-dst-10.1177_19322968251315499 for A Performance-Based Adaptation Index for Automated Insulin Delivery Systems by Jenny L. Diaz C., Patricio Colmegna, Elliot Pryor and Marc D. Breton in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AID, automated insulin delivery; T1D, type 1 diabetes; TIR, time in range; TBR, time below range; TDI, total daily insulin, therapy adaptation; FCL, fully automated closed loop; MPC, model predictive control.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JD is at present an employee of Insulet Co. and this work was performed during her postdoctoral program at UVA during which she received research support and royalties from Dexcom handled by the University of Virginia’s Licensing and Ventures Group. P.C. is at present an employee of Dexcom, Inc. The work presented in this article was performed as part of his UVA appointment and is independent of his employment with Dexcom, Inc. MDB has received research support from Tandem Diabetes Care, Dexcom, and Novo Nordisk through his institution. MDB has received honoraria, travel expenses, and consulting fees from Roche Diagnostics, Dexcom, Vertex, Tandem, Sanofi, BoydSense, and Portal Insulin.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.