Abstract
Background:
DBLG1 (Diabeloop Generation 1) stands as one of the five commercially available closed-loop solution worldwide for patients with type 1 diabetes as of 2023. Our aim was to provide an overview of all data obtained with this system regarding outcomes and populations, with an emphasis on interoperability.
Methods:
This report includes all available sources of data (three randomized control trials and five surveys on real-life data). Collection ran from March 3, 2017 to April 30, 2022.
Results:
We gathered data from 6859 adult patients treated with closed-loop from three to 12 months. Overall, all sources of data showed that time in range (TIR) 70 to 180 mg/dL, starting from 47.4% to 56.6%, improved from 12.2 to 17.3 percentage points. Time in hypoglycemia was reduced by 48% in average (range: 26%-70%) and reached a level of 1.3% in the largest and most recent cohort. In patients with excessive time in hypoglycemia at baseline (≥5%), closed-loop allowed a reduction in time below range (TBR) by 59%. The comparison of days with declared physical activity versus days without physical activity did not show differences in TBR. The improvement in TIR observed with three different pump systems (Vicentra Kaleido, n = 117; Sooil Dana-I, n = 84; and Roche Insight, n = 6684) ranged from 15.4 to 17.3 percentage points.
Discussion:
These data obtained in different European countries were consistent throughout all reports, showing that this closed-loop system is efficient (high improvement in TIR), safe (remarkably low level of TBR), and interoperable (three pump settings so far).
Introduction
Automated insulin delivery has been a very important breakthrough in the one-century-old quest for improving glucose control, prognosis, and daily care burden of patients living with type 1 diabetes (T1D). 1 Several closed-loop devices, proposed by various industrial companies as well as open-source, do-it-yourself systems, have proven their ability to achieve international standards of care, in randomized studies, pivotal trials and real-life reports.2-7 One of these closed-loop systems, DBLG1 (Diabeloop Generation 1), was designed as a modular and interoperable system, thus theoretically allowing to overcome bias in analyzing the impact of the algorithmic, control module per se, compared to the glucose sensor module and the pump module, on the glucose metrics outcome. Our general aim was to provide an overview of all data obtained with DBLG1, regarding outcomes and populations. interoperability. More specifically, we wanted to study the consistency of the results across all reports, regarding glucose metrics. Finally, as this has not been addressed with other closed-loop systems so far, our specific aim was to analyze the data regarding interoperability.
Methods
DBLG1 System
DBLG1 is a hybrid, closed-loop insulin delivery system combining a continuous glucose monitoring device (so far, Dexcom G5 then G6), an insulin pump (so far, Kaleido, Dana-i, Roche Insight) and the DBLG1 software into a controller handset (so far, a dedicated smartphone). Briefly, DBLG1 system works by using personal medical parameters and adjustable algorithm action parameters to regulate insulin flows continuously every five minutes and keep the patient in the normoglycemia range (between 70 and 180 mg/dL, configurable values). It combines an algorithm based upon machine-learning within a physiological framework with an expert system and self-learning algorithms. 8 The patient is only requested to enter his or her meals in a semi-quantitative fashion (small, medium, large meal) and physical activities, preferably in advance. DBLG1 allows to customize the algorithm, through 10 different settings, in order to respond to the diversity of existing T1D physiologies (details in Benhamou et al 8 ).
Sources of Data
This report includes all available sources of data, that is, three randomized control trials (RCTs) and five surveys on real-life data.9-16 Among the three RCTs, SP7 was the pivotal trial that led to CE-marking of the product. SP7 was a randomized, crossover trial performed within 12 centers in 63 adult patients, with two periods of 12 weeks each under either open-loop or closed-loop therapy, separated with an eight-week washout period. 9 This study was initially designed to use the CellNovo pump; however, because of a fault in the safety sensor of this pump identified during the first arm of the crossover, a switch to the Kaleido patchpump had to be set for the second arm of the study. Here, we only report data on the second arm of the crossover (n = 31). The glucose sensor used in this study was the Dexcom G5. This trial was completed on August 28, 2018.
The second trial (SP8) was designed as a randomized, parallel-group trial to be performed within nine centers in 148 adults and 30 adolescents during three months, using the Dana-i pump and the Dexcom G6 sensor, with a 4:1 closed-loop:open-loop ratio. Because of the Covid crisis, this trial had to be stopped on March 14, 2020. The current report involves 84 adult patients in whom a minimum of 14 days with 75% of continuous glucose monitoring (CGM) data under closed-loop were available. 10
The third trial (SP9) included 17 children aged six to 12 that were randomized to either closed-loop treatment with the Kaleido pump and Dexcom G6 sensor or open-loop therapy, in a crossover design with two six-week periods. 11 This trial was completed on December 23, 2019.
The DBLG1 system was launched on the European market, following its CE marking for adult patients with type 1 diabetes, and several real-life data were reported. Two different settings of the system were made available according to the country. In France, the reimbursement from health insurance authorities was obtained for the Kaleido-Dexcom G6 setting. A commercial prelaunch was set among 25 patients in two hospital centers, in the second half of 2019, allowing a first six-month report. 12 The nationwide launch took place early 2022, allowing a second six-month report among 61 patients. 13 In other European countries, the distribution was set with the Roche Insight pump, starting March 2021, leading to several real-life reports among 62 patients in Spain after three months, 14 3706 patients in Germany after one year of use 15 and 6658 patients in Europe after one year. 16 Of note, this latter report included patients from the following countries: Belgium, Germany, Italy, Netherland, Spain, and Switzerland. When reporting the sum of the patients, we excluded the numbers in Spain and Germany in order for them not be counted twice.
Results
Patients
All the information regarding the sources of data, number of patients, age category, and pump setting are summarized in Table 1. Overall, from March 3, 2017 until April 30, 2022, we gathered data among 6859 adult patients and 17 children aged six to 12. The observation period under closed-loop therapy ranged from three months to one year in adults and was six weeks in children.
Overview of All Available Sources of Data With DBLG1 Closed-Loop System.
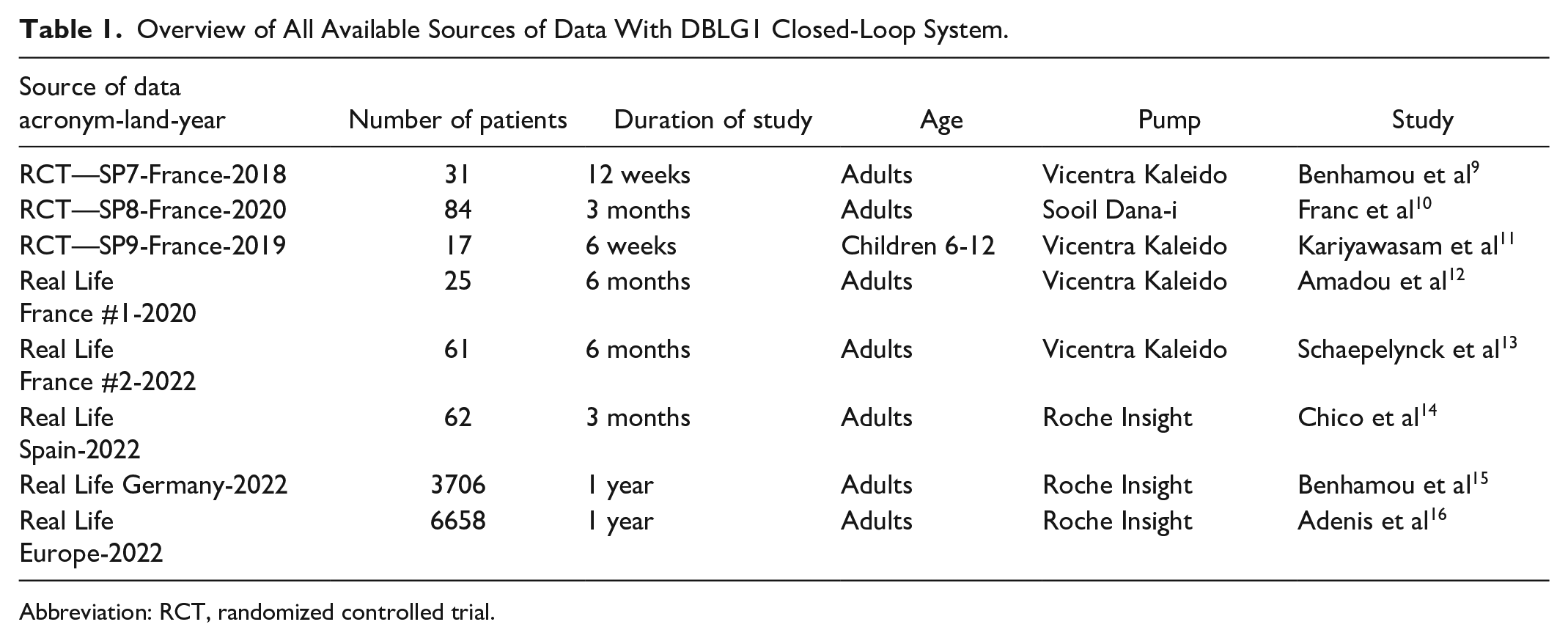
Abbreviation: RCT, randomized controlled trial.
Main Glucose Metrics in Adult Patients
The main glucose-related outcomes in adult patients are summarized in Table 2. Overall, all RCTs data and real-life reports showed an improvement in the time in range (TIR) 70 to 180 mg/dL. This improvement ranged from 12.2 points (pivotal study finalized in 2018) to 17.3 points (commercial data published in 2023) (Figure 1). Importantly, the baseline TIR in open-loop ranged from 47.4% to 56.6%. Regarding the time below range (TBR) (<70 mg/dL), all available data showed a reduction in hypoglycemia by 48% in average (reduction ranging from 26% to 70%). The time spent in hypoglycemia ranged from 2.2% (pivotal study in 2018) to 0.9% (commercial data in 2023) (Table 2 and Figure 2). We looked at the percentage of patients achieving the international recommendations for TIR and TBR17,18 among our three largest cohorts: 47.5% of the 62 patients in the Spanish cohort, 55.9% of the 3706 patients in the German cohort and 53% of the 6658 patients in the European cohort, respectively, achieved both a TIR>70% and a TBR<4%.14-16 Finally, an important point raised in the pivotal SP7 study was a positive correlation between improvement in outcome and baseline HbA1c. Thus, improvements were observed across the whole range of baseline HbA1c values: patients with hyperglycemic concern spent a longer time in the target glucose range, and patients with lower glucose values at baseline spent a reduced proportion of time in the low glucose range. 9
Summary of Main Glucose Metrics Observed With DBLG1 Closed-Loop System.
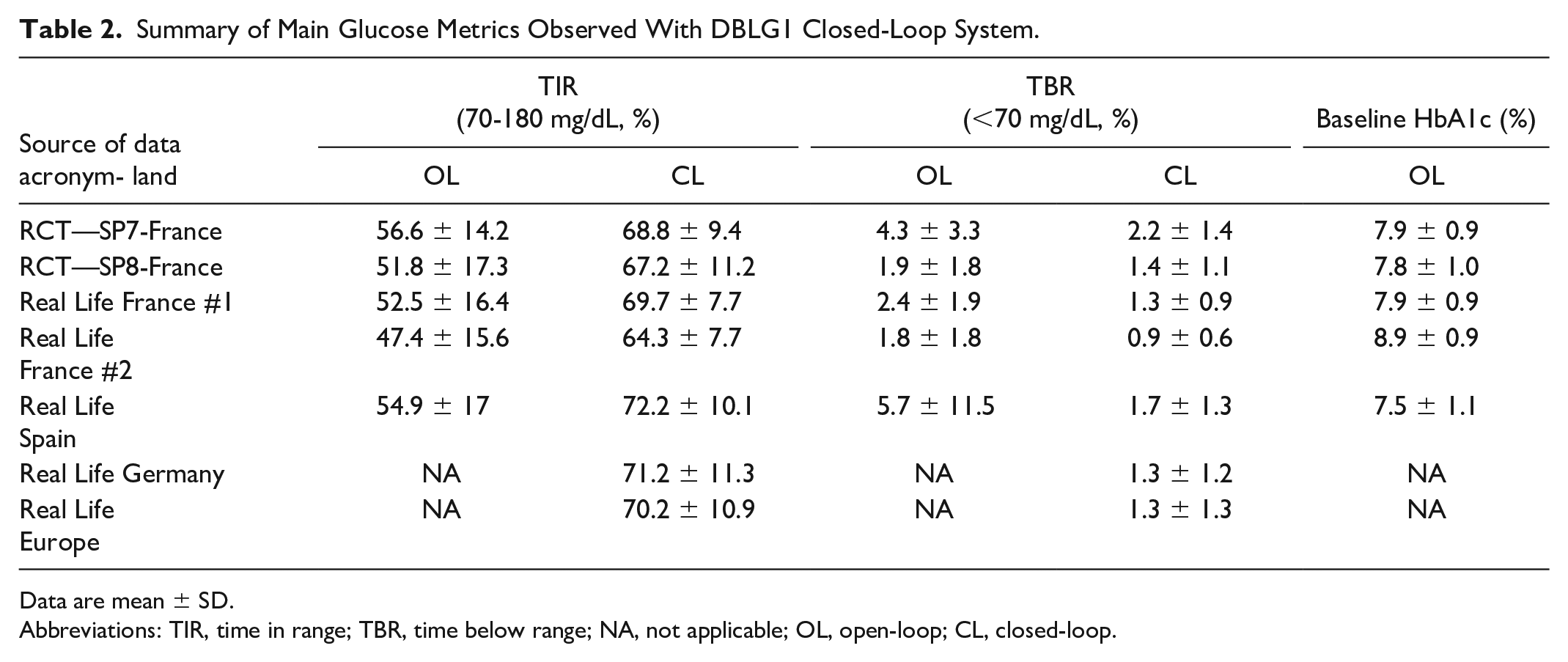
Data are mean ± SD.
Abbreviations: TIR, time in range; TBR, time below range; NA, not applicable; OL, open-loop; CL, closed-loop.
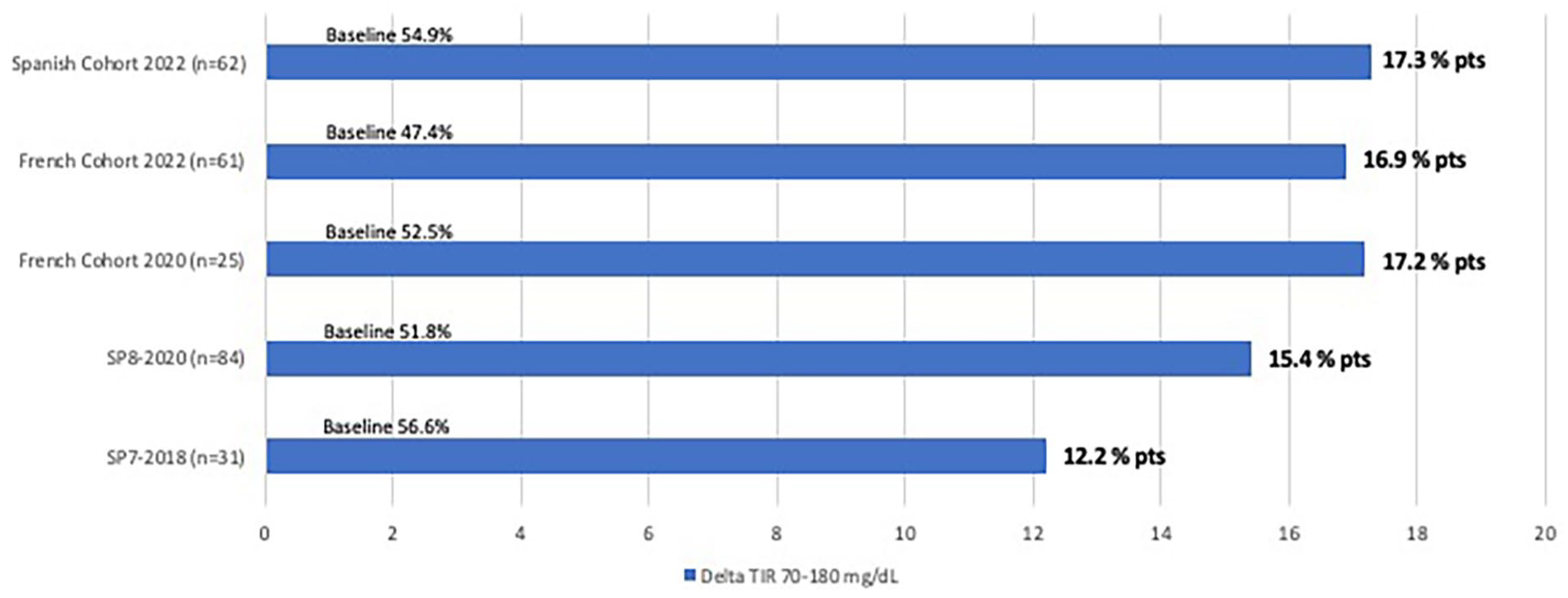
TIR (70-180 mg/dL): differences between closed-loop and open-loop according to the source of data. Data are in percentage points.
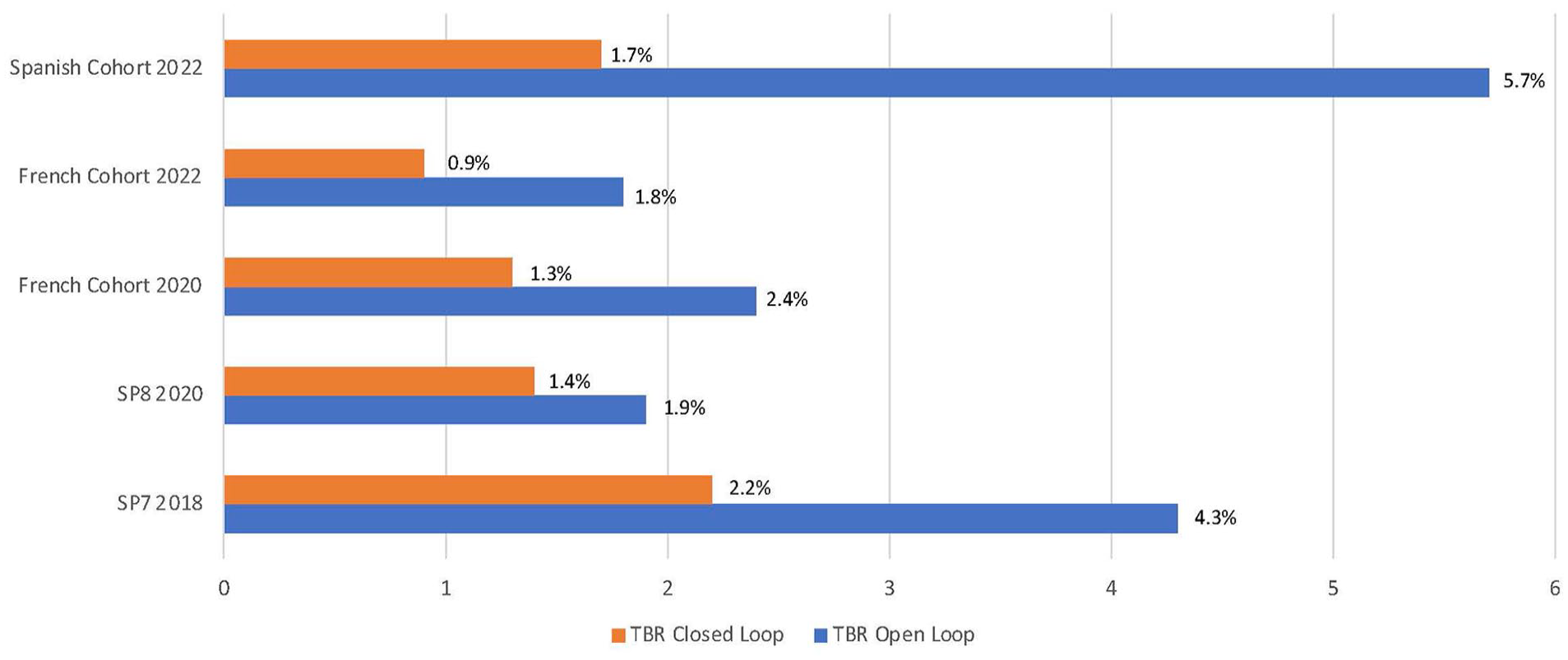
TBR (<70 mg/dL): differences between open-loop and closed-loop according to the source of data. Data are in percentage.
Interoperability
So far, data were obtained with three different pump systems in adult patients: Vicentra Kaleido (n = 117), Sooil Dana-i (n = 84), and Roche Insight (n = 62). The pooled data exposed in Table 3 showed an improvement in Time in Range with all three devices ranging from 15.4 to 17.3 percentage points.
Interoperability of DBLG1 Closed-Loop System.
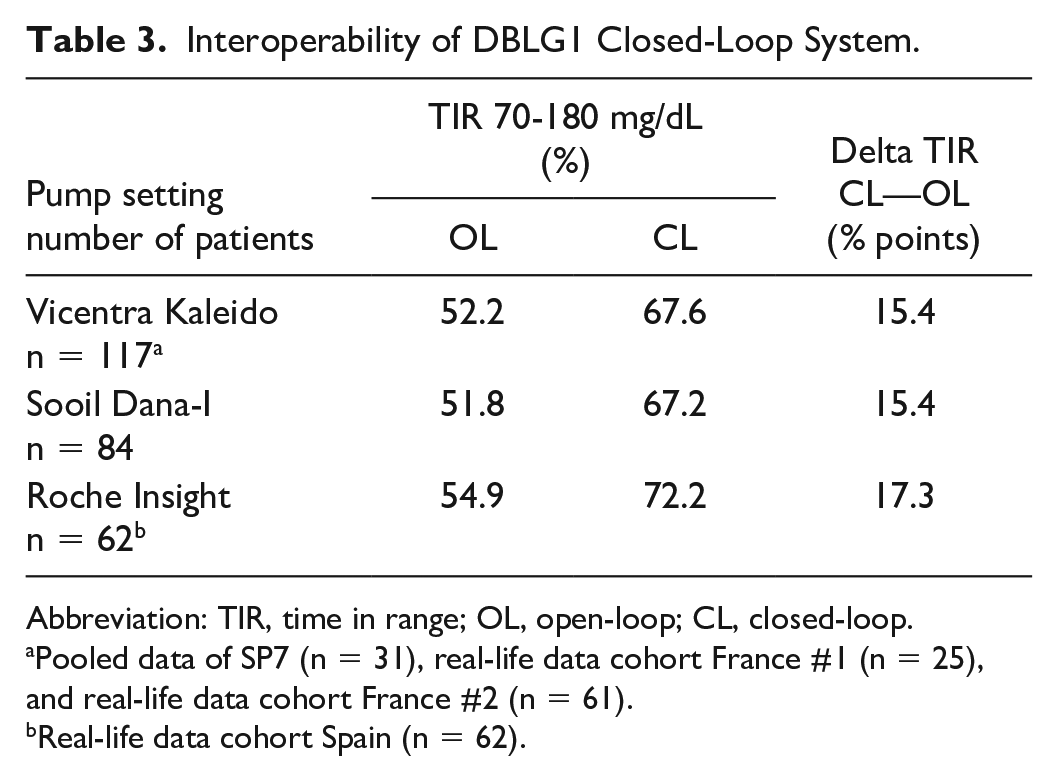
Abbreviation: TIR, time in range; OL, open-loop; CL, closed-loop.
Pooled data of SP7 (n = 31), real-life data cohort France #1 (n = 25), and real-life data cohort France #2 (n = 61).
Real-life data cohort Spain (n = 62).
Focus on Particular Populations
We had a specific look on three different populations that yielded an original publication and two post hoc reports. The first population was our pediatric study, showing after six weeks in closed-loop an improvement in TIR by 7.5 percentage points, and a reduction by 50% in time in hypoglycemic range. 11 The second population was a post hoc analysis of our pivotal SP7 trial, based on data from the closed-loop period only, where we compared 938 days with a declared physical activity and 4260 days without any declared physical activity. This analysis revealed no significant impact of physical activity on time spent in hypoglycemia, and a significant by slight reduction in TIR on the days with activity. 19 The third analysis focused on the subsets of patients included in the three RCTs (SP7, SP8, and SP9) that had excessive (≥5%) baseline time in hypoglycemia and showed that closed-loop therapy induced a significant 59% reduction in hypoglycemia while also improving TIR. 20 These data are summarized in Table 4.
Data Observed in Specific Populations Using DBLG1 Closed-Loop System.
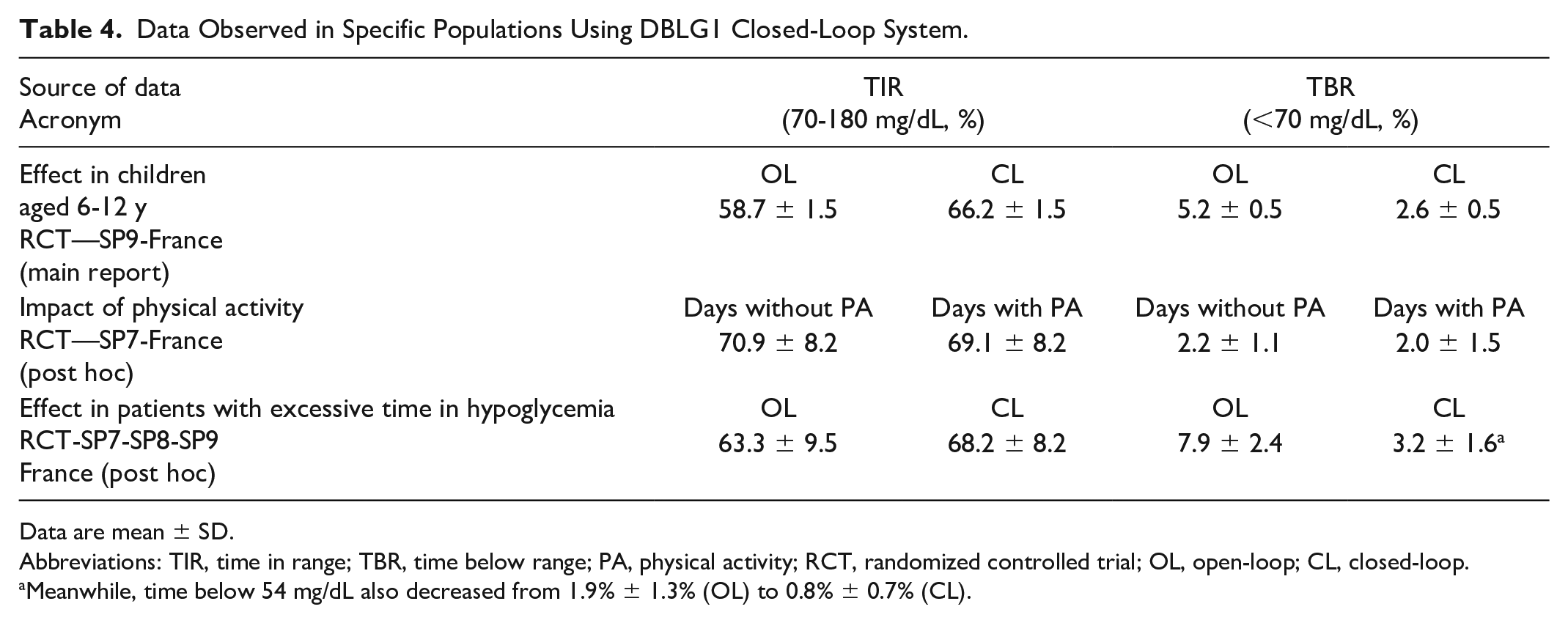
Data are mean ± SD.
Abbreviations: TIR, time in range; TBR, time below range; PA, physical activity; RCT, randomized controlled trial; OL, open-loop; CL, closed-loop.
Meanwhile, time below 54 mg/dL also decreased from 1.9% ± 1.3% (OL) to 0.8% ± 0.7% (CL).
Evolution of Algorithm
The data collected in this article were obtained with a controller that was marginally modified throughout these five years; namely, some modifications were implemented that addressed the management of the immediate post-meal period, the nocturnal glucose target, the hypoglycemia minimizer, and some various bug fixes.
Discussion
The clinical adoption of automated insulin delivery systems remains in its early stages. There is room for assessing the pros and cons of the various available devices and their real impact on the life of patients living with T1D. In this review, we have focused on the first generation of a modular, hybrid, and interoperable system made commercially available in various European countries by a French software company.
One of the main criteria recently proposed by an international expert consensus for the proper assessment of glucose control is the time-in-range (70-180 mg/dL) measured by continuous glucose monitoring. 17 The recommendation is to achieve a TIR beyond 70%. 18 In this respect, the fact that about half of the patients in the commercial setting (Spanish, German, and European cohort) could achieve a TIR >70% combined with a TBR<4% is excellent news. The fourth 2022 report, issued from French data using a different pump setting, mentioned a lower TIR of 64%; however, baseline TIR was much lower (47% vs 54%-55%) and HbA1c higher (8.9% vs 7.5%). Indeed, the impact of a given therapy on the absolute level of this indicator is influenced by the baseline level of TIR. 21 Therefore, we chose to emphasize the baseline TIR levels and the delta TIR between open-loop and closed-loop in this report. We observed that TIR improved from 12.2 to 17.3 percentage points, whereas baseline TIR ranged from 47.4% to 56.6%.
Real-life outcomes have been reported with two other commercial closed-loop systems. When using Control-IQ technology, an improvement of TIR from 63.2% at baseline to 73.5% after one year was observed. 22 The use of the MiniMedTM 780G System was associated with a 12.1 percentage point improvement of TIR from 63.4% to 75.5%. 23 Real-life data with the two other commercial closed-loop systems (CamAPS-FX and Omnipod 5) are not yet available, but data from pivotal trials had shown similar TIR improvement (10.8 and 9.3 percentage points in adult patients, respectively).2,4 From this information, it is likely that, when adjusting for baseline glucose control, the differences in TIR outcomes between these five commercial AID systems should not be clinically meaningful, but this question has to be addressed by head-to-head trials.
Another important evaluation criteria is the time-below-range (<70 mg/dL). DBLG1 has proven to be a remarkably safe algorithm, as we observed an average reduction in time spent in hypoglycemia by ~50% and a residual TBR way below 2%. Additional data from post hoc analysis showed that the use of this algorithm was associated with an important decrease in TBR also in populations prone to hypoglycemia, 18 whereas it could prevent the increase of hypoglycemia risk related to physical activities. 17 Of note, this algorithm can prompt a text message on the controller handset calling for a rescue and calibrated carbohydrate intake when a hypoglycemic event is predicted despite the modulation of insulin delivery. A reduction in TBR has also been reported with Control-IQ technology, 22 whereas the reduction was less pronounced when examining Minimed 780G real-life data. 23 Importantly, the collection of glucose-related data in the real-world setting in the German and European cohort was done remotely through the DBLG1 handset that is connected to a web-based platform. Therefore, clinical data were lacking, such as the side-effects of the treatment (severe hypoglycemic episodes or diabetic ketoacidosis events). However, the rate of drop-outs from treatment did not exceed 1.5% as reported in the work by Benhamou et al. 15 Moreover, no severe hypoglycemia, ketoacidosis, or serious adverse events were observed in the reports from the French and Spanish cohorts.12-14
The question of interoperability is not trivial. To the best of our knowledge, there has been no published report on commercial closed-loop systems so far regarding interoperability. Our data suggest that there are no major differences in glucose-related outcomes when using the same algorithm with three different pump settings. So far, CamAPS is the only other software company to claim and show successful interoperability. On one hand, the right of patients to combine the hardware and the software of their choice is restricted by regulatory and proprietary considerations. On the other hand, the integration of all components of an AID system is a complex task, regarding connectivity, and usability issues. So far, the real impact of AID on the burden and distress of patients living with T1D has been insufficiently established. Currently, we do not have enough data to comment on this aspect, 24 besides the fact that the rate of dropouts from treatment in the German cohort did not exceed 1.5%, which is low. 15 It is likely that the ease of use of patch pumps is counterbalanced by the modularity of some AID systems that require a handset. Noteworthy, the implementation of the algorithmic controller into a smartwatch, combined with additional sensors (accelerometry, impedancemetry) potentially useful for the detection of physical activity is being currently developed and could provide an answer to some usability issues.
The perspectives for moving on with this AID system are on multiple directions. Regarding hardware, the porting of the controller into a smartwatch, as well into a dedicated smartphone app is under development. Regarding indications, the system is currently proposed to adult patients with T1D, and extensions to other populations (children, teens, pregnant women, patients with type 2 diabetes) is currently seeked through ongoing protocols, although some data have already been published.11,25 Regarding software, several improvements are being considered. The use of fast-acting insulin analogs such as ultrarapid lispro was shown, in an in silico simulator, to improve TIR, TBR, and glucose variability, 26 and confirmed clinically with case reports. A module for the management of unannounced meals is currently being tested in two RCTs in teens and in patients with type 2 diabetes. A future version of the algorithm using neural network and a deep learning approach has been validated in silico. Finally, a dedicated version of the controller, called DBLhu, was developed for the specific needs of patients with a history of hypoglycemia unawareness and grade 3, severe hypoglycemia, validated in a RCT and used in routine care for this population.27,28
In conclusion, we have collected a large amount of clinical data, both in RCTs and real-life, obtained in different European countries and various pump setting, that were consistent throughout all reports. Overall data show that the first generation of this French closed-loop system is efficient (high improvement in TIR), safe (remarkably low level of TBR), and interoperable, allowing half of the users to achieve the international CGM standards. Various areas of further improvement are being developed to improve both glucose metrics and user-experience.
Footnotes
Abbreviations
AGP, ambulatory glucose profile; CGM, continuous glucose monitoring; DBLG1, Diabeloop generation 1; GRI, glycemia risk index; T1D, type 1 diabetes; TBR, time below range; TIR, time in range.
Author Contributions
PYB: manuscript writing. AA: data management and statistical analysis. SL, SF, CA, AP, DK, and JB: patient management, manuscript editing. GC: relation with regulation authorities, manuscript editing. PYB is the guarantor of this work and, as such, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PYB, SF, and GC are consultants for Diabeloop SA. AA is employee from Diabeloop SA. No author has been paid to write this article, and the findings and conclusions in this study are those of the authors and do not necessarily represent the views of the sponsors. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
